Abstract
An intraocular mass in the left eye causing chronic severe exophthalmia in an adult female goldfish (Carassius auratus) is described. The fish shared an aquarium with another goldfish found dead with gross and microscopic lesions consistent with mycobacteriosis. Histological examination of the left eye, histochemical (periodic acid–Schiff [PAS], Alcian blue, Ziehl–Neelsen) and immunohistochemical tests (glial fibrillary acidic protein, human neuronal protein, vimentin, and cytokeratin AE1/AE3) were carried out on the intraocular mass. Neoplastic cells forming an unencapsulated highly cellular proliferation partially covered by an intact corneal epithelium were stained with Alcian blue, which demonstrated an abundant hyaluronic acid–rich extracellular matrix. Multifocally, there were cyst-like dilatations bordered by neuroepithelial cells, which were PAS-positive. The complex neoplastic proliferation was composed of glial-like cells, neuronal-like cells (immunoreactive to glial fibrillary acidic protein and human neuronal protein, respectively) and neuroepithelium, which suggested a retinal origin.
Keywords
Spontaneous ocular tumors arising from the retina are rare in fish. Reported lesions include adenocarcinoma of the retinal pigment epithelium in a guppy (Poecilia reticulata) and retinoblastoma in a spring cavefish (Forbesichthys agassizii; syn. Chologaster agassizi), porkfish (Anisotremus virginicus), and brown bullhead (Ameiurus nebulosus; syn. Ictalurus nebulosus).9,10,27 Two medulloepithelioma have been described in a goldfish (Carassius auratus) 17 and in a tinfoil barb (Barbonymus schwanenfeldii; syn. Barbodes subwanefeldi). 13 Among other primitive ocular neoplasms with an ectodermic origin, a primitive neuroectodermal tumor was reported in a telescope goldfish. 3
In veterinary medicine, there are no reports of glioneuroma to the authors’ knowledge. This rare congenital embryonic tumor is reported in pediatric or young human patients.1,14,19,31
Medulloepithelioma is a very rare tumor in dogs2,8,15,26 and horses, 18 and is extremely rare in cats. 12 Isolated cases have been reported in other species, including birds 5 and llamas. 30 The present study describes the histomorphological and immunohistochemical features of a spontaneous intraocular tumor suggestive of a glioneuroma with medulloepitheliomatous differentiation in an adult goldfish (Carassius auratus).
Glioneuroma and medulloepithelioma may arise from either the outer or the inner layer of the optic cup and from primitive medulloepithelium. 24 It is generally thought that medulloepithelioma is a true neoplasm originating from developing embryonal retinal cells, while glioneuroma is regarded as a choristomatous malformation consisting of glial and neuronal cells without obvious neoplastic potential. 14
An adult female goldfish living in a 50-liter hobbyist aquarium was presented with unilateral progressive exophthalmos of the left eye. While the animal’s body condition deteriorated and the left eye continually enlarged, the fish maintained an appropriate body position in the water column for the several months prior to death. Only a few days before death, the fish listed to the left and its head dragged along the bottom. The right eye was normal in size and had a red pigmentation of the limbus. Neither medical treatment of the eye nor disinfection of the aquarium was carried out. The fish shared the aquarium with another goldfish found dead with gross and microscopic lesions of mycobacteriosis.
The moribund animal was euthanized with a lethal dose of anesthetic (100 mg/l of tricaine methanesulfonate). a Eyes, gills, liver, kidney, spleen, intestine, and brain were sampled, fixed in 10% buffered formalin for 24 hr, processed using routine histologic processing techniques, embedded in paraffin, and cut at 4-µm thickness. Histologic sections were stained with hematoxylin and eosin; sections from the left eye were also stained with periodic acid–Schiff (PAS) and Alcian blue (pH 2.5).
Immunohistochemical staining was performed with antibodies against cytokeratin b (diluted 1:50) and vimentin c (prediluted) only on neoplastic tissue, and human neuronal protein HuC/D d (diluted 1:400) and glial fibrillary acidic protein (GFAP e ; diluted 1:4,000) on neoplastic tissue and normal goldfish eye. The HuC/D and GFAP antibodies were double labeled using the indirect fluorescent antibody method. After deparaffinization and hydration, tissue sections were treated for antigen retrieval in citrate buffer at pH 6.0 in a microwave oven for 10 min. Subsequently, to reduce background staining and enhance antibody penetration into tissues, 10% normal goat serum and 0.5% Triton-X100 in phosphate buffered saline (PBS) were used in the preincubation stage (1 hr at room temperature [RT]) and in all antibody solutions. The sections were then incubated with a mixture of the 2 primary antibodies and incubated overnight in a humid chamber at RT. After washing in PBS (3 times for 10 min each), the next day the sections were incubated for 1 hr in a humid chamber at RT with the secondary antibodies. f The sections were then washed in PBS (3 times for 10 min each) and mounted in buffered glycerol at pH 8.6. For vimentin, an automated staining system g was used; for cytokeratin AE1/AE3, after deparaffinization and hydration, endogenous peroxidase activity was blocked with 0.3% hydrogen peroxide for 30 min. After washing once in PBS for 5 min, tissue sections were treated for antigen retrieval in citrate buffer at pH 6.0 in a microwave oven for 10 min. Sections were then incubated overnight at +4°C with primary antibody in a humid chamber. After washing in PBS (3 times for 10 min each), the next day the sections were stained with the streptavidin–biotin–peroxidase detection system kit, h according to manufacturer’s instructions. Antibody localization was subsequently visualized using 3,3′-diaminobenzidine, i and sections were counterstained with Papanicolaou stain. Finally, sections were dehydrated, and mounted with dibutyl phthalate xylene. j Brain from a dog and a normal goldfish eye were used as positive control tissues. For negative control purposes, the primary antibody was replaced by a monoclonal antibody of irrelevant specificity. k Immunohistochemical staining was performed also in visceral organ sections with the primary antibody anti–Mycobacterium bovis according to the standardized methods.[“ell”] l,28
Gross observation revealed a bulging rounded mass protruding from the left eye; it was about 2 cm in diameter, intensely pink, glistening, and highly vascularized. A corneal remnant was recognizable on the external surface, opposite the nasal margin of the eye (Fig. 1A, 1B). Sclera, iris, and pupil were not grossly visible as the mass largely had replaced the globe.
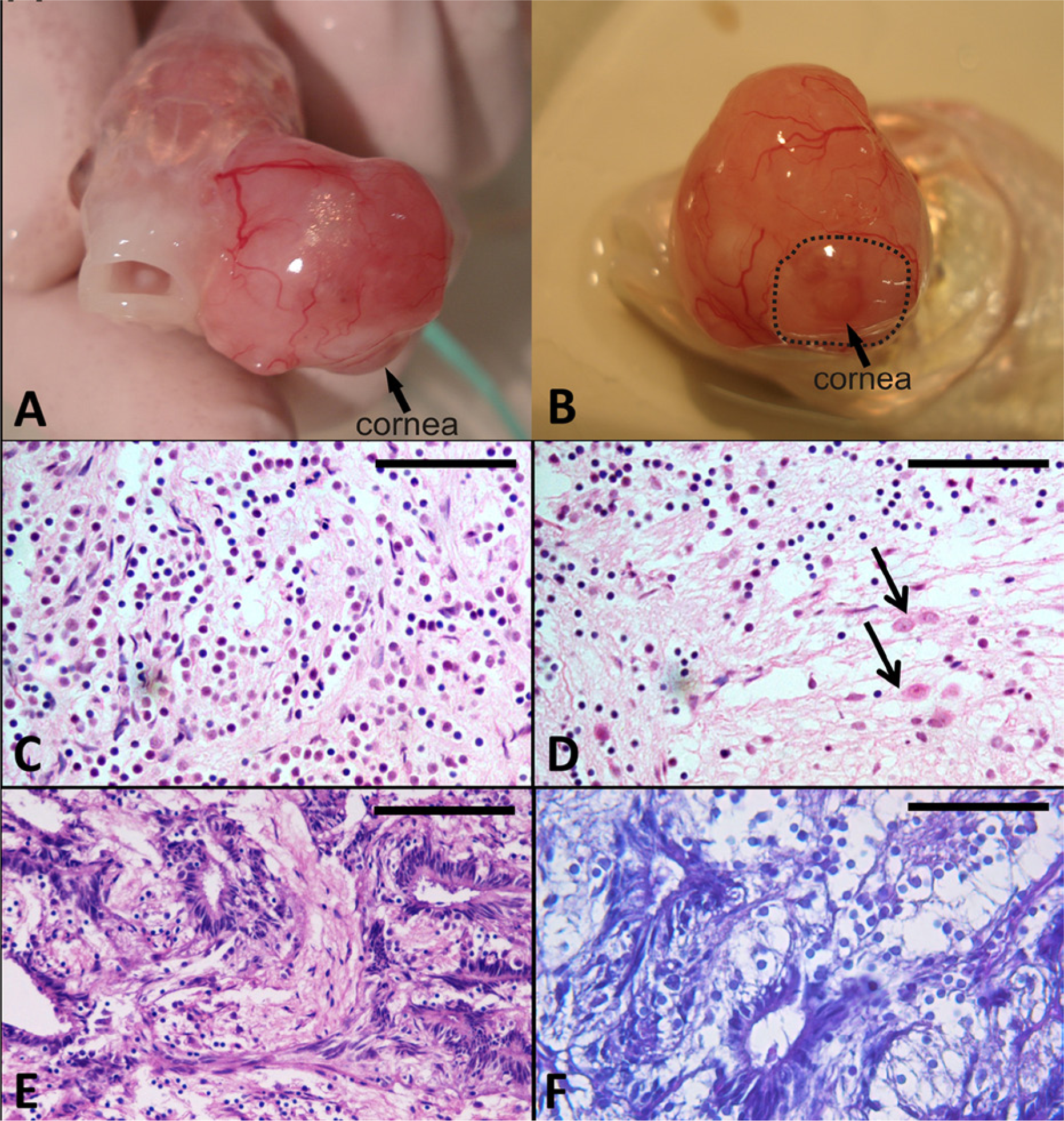
Goldfish (Carassius auratus); gross and histological findings.
Histological examination revealed an unencapsulated highly cellular neoplasm partially covered by an intact corneal epithelium. The neoplastic tissue was characterized by different cellular populations. Most of the mass was composed of sheets of pleomorphic cells with indistinct cell borders. These pleomorphic cells had a moderate amount of clear cytoplasm, a small round central to eccentric hyperchromatic nucleus with no visible nucleoli, that is, glial-like cells (Fig. 1C). There were larger round to oval neoplastic cells with distinct cell borders, a moderate amount of eosinophilic cytoplasm and large round central vesicular nucleus with single prominent nucleolus, that is, neuronal-like cells (Fig. 1D). Neoplastic cells were surrounded and supported by an abundant hyaluronic acid–rich extracellular matrix, which was interpreted to be vitreous body–derived material. The core of the mass contained cyst-like spaces bordered by pseudostratified neuroepithelium, composed of columnar cells palisaded along the luminal margin of the cystic spaces (Fig. 1E, 1F); these cells had oval hyperchromatic nuclei, and no evident nucleoli. Multifocal areas of hyaline scleral cartilage and scattered smooth muscle cells deriving from ciliary muscles were infiltrated by the neoplastic tissue. Serial sections of the eye also disclosed bilateral symmetric groups of skeletal myofibers interpreted as orbicular muscles. Mitotic figures averaged 2 per high-power field. The luminal border of the columnar neuroepithelial cells delimitating the cystic spaces was PAS-positive. Alcian blue staining highlighted a hyaluronic acid–rich cellular matrix.
Histological observations of the right eye showed moderate hyperplasia of the corneal epithelium associated with a mild chronic keratitis. Choroidal tissues of the iridociliary angle were hypertrophic. Metastases were not detected in the other organs sampled. Multifocal hemorrhages and hemosiderosis were evident in the liver and spleen, as were multifocal areas of necrosis with numerous intralesional basophilic bacterial aggregates; bacteria in these aggregates were Gram-positive and Ziehl–Neelsen-negative. A small granuloma, which did not have Ziehl–Neelsen-positive organisms, was present in the intestinal submucosa. There was mild branchial lamellar hyperplasia.
Immunoreactivity in normal goldfish eye showed HuC/D-positive neurons (rod and cone cells) in the outer nuclear layer, bipolar cells in the inner nuclear layer, ganglion cells in the ganglion cell layer (Fig. 2G, 2H) and GFAP positivity of glial cells (Fig. 2I, 2L). Neoplastic cells were multifocally immunoreactive to HuC/D and GFAP (Fig. 2M) and vimentin (Fig. 2N) but did not react with anti–cytokeratin AE1/AE3 antibodies (not shown). Immunohistochemical staining with antibody anti–M. bovis on normal and neoplastic eye, intestine, and spleen was negative.
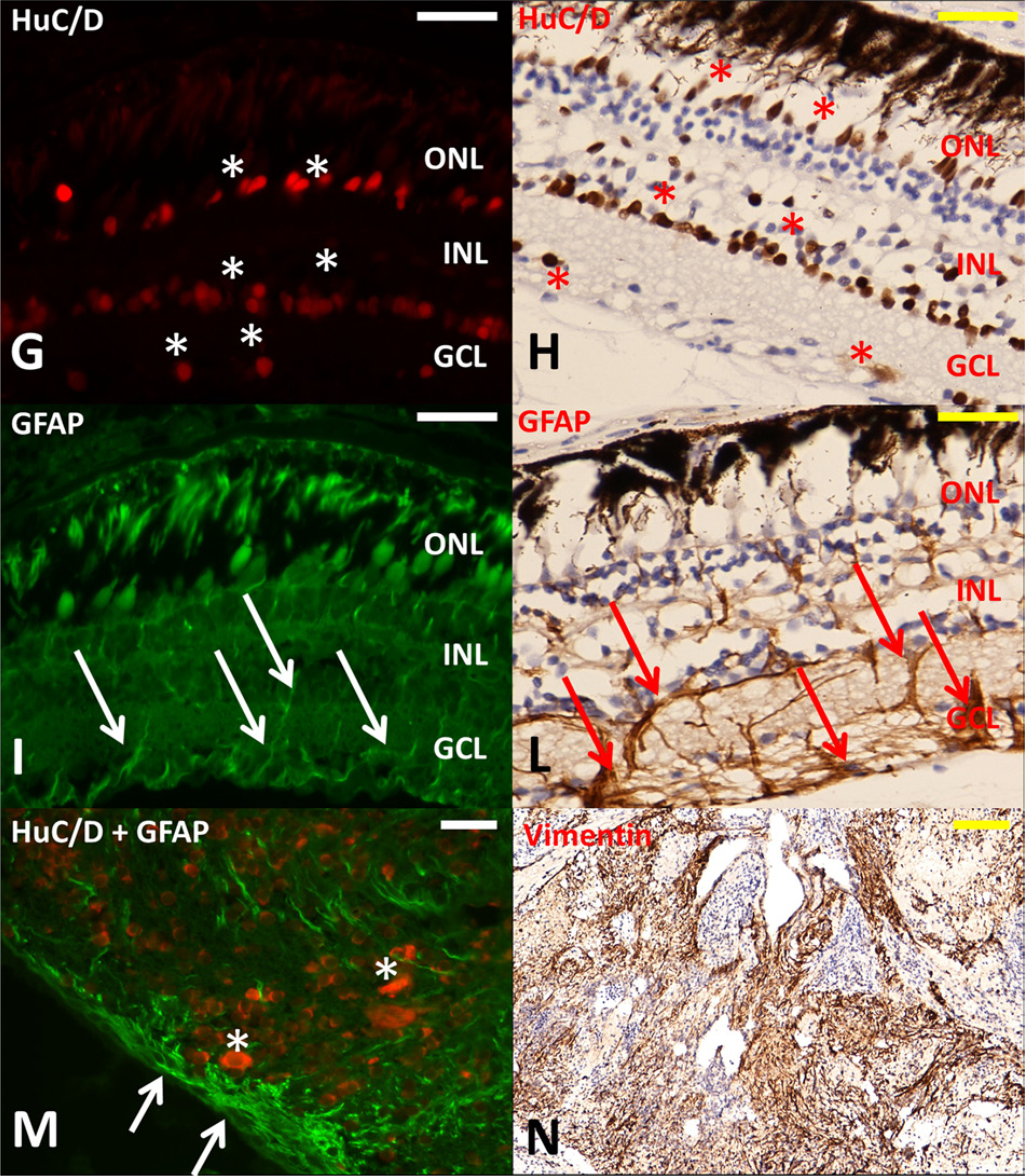
Goldfish (Carassius auratus). Immunohistochemical staining on normal goldfish retina (
The complex morphology of this neoplasm hampered a direct interpretation of its origin. Data obtained from serial sections and a panel of antibodies suggested an intraocular tumor of retinal origin, consistent with a glioneuroma with medulloepitheliomatous differentiation as described in the literature.14,19 Some authors favor the term “glioneuronal hamartoma” on the basis of the clinicopathological behavior, 16 while for the same reasons the current authors lean toward the interpretation of a tumor.
The histological features of glioneuroma include mature glial cells and neuronal cells with axonal processes; a few large retinal ganglion cells may be present. 14 In the present case, the immunohistochemical results highlighted mature glial cells, neuronal cells, and retinal ganglion cells.
Rosettes and tubules are thought to represent dysplastic neuroepithelium. In 3 human cases, the glioneuromatous mass was directly connected to the dysplastic retina; in these cases, the proliferating neuroepithelial cells imitated medulloepithelioma, 14 very similar to the mass observed in the fish in the current study.
Intraocular medulloepitheliomas are characterized by cords of primitive neuroepithelial cells, resembling the embryonic retina or neural tube, surrounded by a loose mesenchymal tissue rich in hyaluronic acid. Many medulloepitheliomas present with a pseudostratified epithelium and a hypocellular stroma. Under high magnification, the cords of the primitive neuroepithelium may display features of the developing neurosensory retina. When the medullary epithelium folds so that the vitreous surface faces inward, it creates Alcian blue–stained cyst-like dilations rich in hyaluronic acid 29 ; this feature was also present in the current case.
In the present case, histological sections revealed some hypercellular areas with pseudostratified epithelium which helped to identify the tumor origin. These areas were interpreted as medullary epithelium arising from the neuroepithelial tube. Areas rich in loose-extracellular matrix were interpreted as vitreous-like. 29 The external limiting membrane was identified with PAS stain. 21
Immunohistochemical markers of human medulloepithelioma have different reactivity patterns depending on whether the neoplasm is neuroepithelial or heteroplastic. 29 Vimentin is positive in the nonteratoid component, whereas limited and conflicting results have been reported for other markers like chromogranin, synaptophysin, GFAP, S100 protein, pancytokeratins, and cytokeratins 18, 7, 20, and epithelial membrane antigen. 29 In the present case, the positive results obtained for vimentin and GFAP are in accordance with the results reported in a human case, 14 and in domestic and nondomestic animal cases of medulloepithelioma.5,30
The Hu proteins (especially HuD) are recognized as one of the earliest markers of the neuronal phenotype, 25 as such proteins suppress neuroblast proliferation and promote neuronal differentiation. 22 Antibodies to HuC/D have been extensively used to label all enteric neurons in mammals, 7 birds, 20 and also fish.4,23 HuC/D positivity of most of the neoplasm confirmed a neuronal and glial origin leading to the diagnosis of glioneuroma.
In fish, medulloepithelioma have been experimentally induced in medaka (or, Japanese rice fish; Oryzias latipes) using methylazoxymethanol acetate. 11 In the current case, the influence of environmental factors (i.e., aquatic contaminants) cannot be evaluated, but the environment could play a potential role. The water used in the aquarium where the affected goldfish was kept came from the local water supply, and chemical treatments were never carried out. In this case, a genetic basis must also be considered. Other similar cases are needed to understand the occurrence of this tumor.
Because a goldfish living in the same aquarium died with lesions of mycobacteriosis, a common event in ornamental species in a confined environment, 32 and Mycobacterium marinum has been implicated as tumor promoter in medaka, 6 testing was performed to determine if Mycobacterium sp. infection was present in the goldfish with ocular glioneuroma. No evidence of mycobacterial infection was obtained after Ziehl–Neelsen staining and immunohistochemical testing of tissues from this goldfish.
Footnotes
Acknowledgements
The authors thank Anne Collins for English editing. The authors have contributed equally to the drafting of the paper.
a.
Argent Chemical Laboratories Inc., Redmond, WA.
b.
Mouse (clone AE1/AE3), Dako North America Inc., Carpinteria, CA.
c.
Mouse (clone V9), Novocastra Laboratories Ltd., Newcastle upon Tyne, United Kingdom.
d.
Mouse (clone 16A11), Molecular Probes, Life Technologies, Grand Island, NY.
e.
Rabbit (polyclonal), Dako North America Inc., Carpinteria, CA.
f.
Goat anti-mouse IgG Alexa Fluor 594 (dilution 1:200), Molecular Probes, Life Technologies, Grand Island, NY; goat anti-rabbit IgG fluorescein isothiocyanate (dilution 1:300), Calbiochem, EMD Millipore Corp., Billerica, MA.
g.
BenchMark, Ventana Medical Systems Inc., Tucson, AZ; Bond maX, A. Menarini Diagnostics Srl, Florence, Italy.
h.
LSAB, Dako Denmark A/S, Glostrup, Denmark.
i.
DAB, Sigma-Aldrich, St. Louis, MO.
j.
Fluka, Sigma-Aldrich, St. Louis, MO.
k.
Mouse anti-IgG1 (DAKO X0931), Dako Denmark A/S, Glostrup, Denmark.
l.
Rabbit (polyclonal), Dako Denmark A/S, Glostrup, Denmark.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
