Abstract
It was hypothesized that acute postnatal Bovine viral diarrhea virus 1 (BVDV-1) infection leads to leukopenia and lymphoid depletion of gut-associated lymphoid tissues similar to acute disease in calves. The objectives of the current study were to characterize the pathologic effects, viremia, viral shedding, and viral antigen deposition in 6–24-month-old, acutely infected alpacas following experimental infection with noncytopathic BVDV-1 subgenotype 1b (BVDV C0-6). The BVDV-1 isolate was obtained from a cria with naturally occurring persistent infection. Lymphocytopenia occurred 3–7 days postinfection, with a 50% reduction in peripheral lymphocytes in infected alpacas. Depletion of B-cell populations in gut-associated lymphoid tissues was evident microscopically. Populations of T cells in parafollicular zones and in nodular aggregates along the superficial submucosa remained intact. The BVDV antigen was deposited most consistently in submucosal gastrointestinal aggregated lymphoid tissues of ileum, proximal colon, and stomach compartment three. Viral antigen was more variably evident in other lymphoid tissues. Antigen distribution correlated well with histologic lesions in gastrointestinal aggregated lymphoid tissues, confirming the role of virus in lymphoid depletion. Nasal shedding was detected in all challenged alpacas on day 6 and in 4 out of 12 challenged alpacas on day 9. Viremia was present as early as day 3, and present in all challenged alpacas on days 5, 6, 7, and 9 postchallenge. Lymphocytopenia and depletion of gastrointestinal aggregated lymphoid tissues associated with acute BVDV-1 infection likely results in immune compromise and is expected to exacerbate concurrent infections even though uncomplicated BVDV-1 infection was clinically unapparent.
Introduction
Early studies of Bovine viral diarrhea virus (BVDV; family Flaviviridae, genus Pestivirus) infection suggested that camelids were resistant to disease or that acute postnatal infections were subclinical and of limited significance. 25 Seroprevalence estimates for BVDV antibody in New World camelids range from 0.9% in North America to 4.6% in Switzerland to as high as 10% or 11% in South American camelids.1,9,18,22,23,25,26 In surveys of the U.S. alpaca population, up to 25% of herds contained seropositive animals. 25 Persistently infected (PI) alpacas were reported in 6.3% of North American herds. 25 Most seropositive herds had unrecognized BVDV-related clinical disease and thus the role of BVDV infection in herd disease was unproven and the value of control programs unknown. Subsequently, a pattern of BVDV infection and clinical disease similar to that recognized in naive cattle has been observed in infected alpaca populations. 7 Clinical syndromes of abortion, stillbirth, diarrhea, respiratory disease, and failure to thrive were reported in immunocompetent alpacas housed with PI alpacas. Descriptions of clinical signs and lesions are published for both PI alpacas and for alpacas at necropsy where one-time sampling made it impossible to determine if infection was transient or persistent.2,3,5,7,10,12,20
Bovine viral diarrhea virus 1 subgenotype 1b strains appear predominant in the North America alpaca population, and type 1b strains have been identified as a common strain worldwide.15,25 Type 1b is common in feedlot cattle with respiratory infections, and cattle are a potential origin for alpaca infection. 11 Sequence similarity of alpaca strains suggests the alpaca 1b strains might have adapted to the species; or alternatively, that these BVDV-1b strains are epidemiologically associated.15,25 While infections from cattle might serve as the origin of individual BVDV outbreaks in alpacas, repeated exposures across species seem less likely than infection with host-adapted strains. Persistently infected cria shed virus via multiple bodily discharges and can expose cohorts.3,5 Cohorts are potentially susceptible to disease resulting from acute postnatal infection. The pathologic effects and antigen distribution during acute postnatal BVDV infections have not been experimentally investigated in camelids. Given that type 1b strains are prevalent to a varying degree in alpaca populations, it is important to understand the pathogenesis of BVDV-1b in acute postnatal infection. The clinical significance and pathogenesis of acute postnatal BVDV infection is an important consideration in evaluating the benefit of vaccination and eradication programs in the species. 4
Materials and methods
Sixteen clinically normal alpacas, 6–24 months of age and negative for BVDV antibody by serum neutralization and BVDV by virus isolation, were donated from private farms. The animals were housed indoors in a biosecurity level 2 animal research facility under Institutional Animal Care and Use Committee–approved conditions. The animals were housed in age-matched groups of 2 and acclimated for 7 days prior to inoculation. Twelve alpacas 6–24 months old were experimentally challenged (BVDchal) on day 0 with virulent BVDV-1b isolated from a previously clinically recognized PI cria (BVDV C0-6) and grown on bovine turbinate cells. The infected cells were grown as a monolayer in Dulbecco modified Eagle medium with 2% equine serum in a 37°C humidified incubator with 5% CO2. The inocula of 106.87 TCID50 was given intranasally by nebulizer. 19 Four alpacas, 6–24 months of age, served as sham (cell culture media) inoculated controls (BVDcon). All animals were examined twice daily for evidence of clinical disease and to ensure patient welfare. Clinical signs, respiratory rate, dyspnea, nasal secretion, cough, feces, and general condition were observed during the study. Jugular venipuncture was performed daily to obtain blood for complete blood cell counts and to collect buffy coats for BVDV detection on days 3–9. Blood counts were performed by a local Clinical Laboratory Improvement Amendments–accredited reference laboratory (Physicians Laboratory, Lincoln, Nebraska).
Nasal swabs were taken on days 0, 3, 6, and 9 for BVDV detection by polymerase chain reaction (PCR). Nasal swabs and buffy coats collected from each alpaca were stored at −80°C until used for reverse transcription (RT)-PCR. 17 One BVDchal alpaca was euthanized on day 7 (animal 5) postinfection and the remaining 11 on day 9 postinfection. The BVDcon alpacas were necropsied 9 days following sham inoculation. Euthanasia was performed by injection with a sedative (xylazine, 2 mg/kg, intramuscularly) followed by overdose of a barbiturate a (10 ml/pound, intravenous to effect). Postmortem examinations included evaluation of gross and histologic changes and collection of lymphoid tissue specimens, from 20 sites as described below, for immunohistochemistry determination of BVDV antigen distribution and B- and T-lymphocyte differentiation.
Real-time RT-PCR was used to detect BVDV in nasal swabs and buffy coats from each alpaca. RNA from nasal swabs and buffy coats was extracted using a commercial viral RNA kit b according to the manufacturer’s directions. The extracted RNA was stored at −80°C. Real-time RT-PCR was completed using a commercial RT-PCR kit b according to manufacturer’s directions using primers and probe previously described. 17
Gross lesions were recorded at necropsy. Samples of skin, lung, liver, kidney, spleen, heart, gonad, trachea, esophagus, stomach (C1, C2, and C3), duodenum, jejunum, ileum, proximal colon, spiral colon, descending colon, anus, thymus, lingual tonsil, and nasopharyngeal tonsil were taken for histology. Multiple lymph nodes, including tracheobronchial, prescapular, mesenteric, mediastinal, retropharyngeal, and parotid were sampled. Thymus and lymph nodes were subjectively evaluated for size during necropsy. Effort was made to preferentially select identifiable segments containing gut-associated lymphoid tissue (GALT) in sampling of C1 through anus whenever possible. Oval patches of lymphoid tissue were consistently identifiable and sampled in distal ileum and proximal colon. A large area of GALT present in proximal C3 was also easily identified and sampled consistently. Tissues were fixed in 10% neutral buffered formalin, processed routinely, sectioned at 5 µm, and stained with hematoxylin and eosin (HE). Immunohistochemical staining (IHC) utilized monoclonal antibody (mAb) 15c5, targeting the conserved Erns protein, in an immunoperoxidase reaction as previously described and was performed on all tissue submitted. 13 Slides were examined by a pathologist blinded to treatment, and the slides were scored for necrosis, inflammation, and BVDV antigen (present or absent), and for lymphoid changes (none, lymphocytolysis, depletion, and/or cellular hyperplasia). Lymphoid changes in GALT were only evaluated for lesions when 3 or more follicles were represented in the sections. Lymph node changes were scored on HE-stained sections as normal, lymphocytolysis, depletion, and/or dendritic cell hypertrophy, and each of these was reported as present or absent. Enteric tissues having submucosal lymphoid aggregates and at least 1 section from 1 animal IHC positive (10 sites) were included as GALT sites. The sites included C1, C2, C3, duodenum, jejunum, ileum, proximal colon, distal colon, rectum, and anus. A similar approach was used for analysis of lymphoid tissues (7 sites) with thymus, oral tonsil, nasopharyngeal tonsil, parotid, prescapular, mediastinal, and mesenteric lymph nodes included in the analysis. Tissues producing slides with most frequent BVDV staining by IHC (C3, ileum, and proximal colon) were further sectioned and stained with IHC methods to: distinguish B- and T-lymphocyte populations using primary mAbs directed toward cluster of differentiation (CD)3 (T-cell specific) and Pax5 (B-cell specific), to better elucidate lymphoid follicular changes in tissue.6,8 These antibodies (anti-Pax5 and anti-CD3) have proven useful to the study of lymphocyte distribution of B and T cells, respectively, in a variety of species and were adaptable for use in the alpaca.6,8
Statistical analysis was performed using analysis of variance for repeated measures with a completely randomized design and an autoregressive error correlation structure of the residuals on the following hematological and clinical variables: basophils, eosinophils, lymphocytes, monocytes, neutrophils, platelets, rectal temperature, and respiration. Paired comparisons of least squares means were conducted to identify differences between the BVDchal and BVDcon treatment groups for all the above dependent variables. Fisher exact test was used to compare treatment groups for histologic lymphoid depletion. A value of P < 0.05 was considered significant. Commercial software was used for all computations. c
Results
Clinical and hematological data
The BVDchal alpacas remained bright and alert but demonstrated a slight, less than 0.5°C temperature elevation. No statistically significant febrile response was recorded on days 3–8 postinfection (Fig. 1). No respiratory distress or increased respiratory rates were detected, and feces remained normal. The BVDcon alpacas remained clinically normal. A leukopenia in BVDchal alpacas was evident for days 3–7 postchallenge (P < 0.05). Values were reduced to 8.08–9.73 × 103 cells/µl in BVDchal alpacas compared with 13.38–15.05 × 103 cells/µl in BVDcon alpacas. Monocytes were lower for BVDchal alpacas on days 3 and 4 (0.20–0.37 × 103 cells/µl) but higher on day 9 (0.70 × 103 cells/µl) compared with days 3 and 4 controls (0.53–.085 × 103 cells/µl) or day 9 controls (0.32 × 103 cells/µl; P < 0.05). Eosinophils were significantly lower for BVDchal on days 4 (0.57 × 103 cells/µl) and 8 (0.44 × 103 cells/µl) compared with BVDcon alpacas on days 4 (1.58 × 103 cells/µl) and 8 (1.45 × 103 cells/µl). Lymphocytopenia in BVDchal alpacas occurred on days 3–6 (P < 0.001) and day 7 (P < 0.05; Fig. 2). Lymphocyte numbers in BVDcon alpacas ranged from 4.37–5.25 × 103 cells/µl compared with 1.78–2.50 × 103 cells/µl in BVDchal alpacas between 2 and 7 days postinfection. Other cell counts including neutrophils, erythrocytes, and platelets did not differ between the treatment groups.
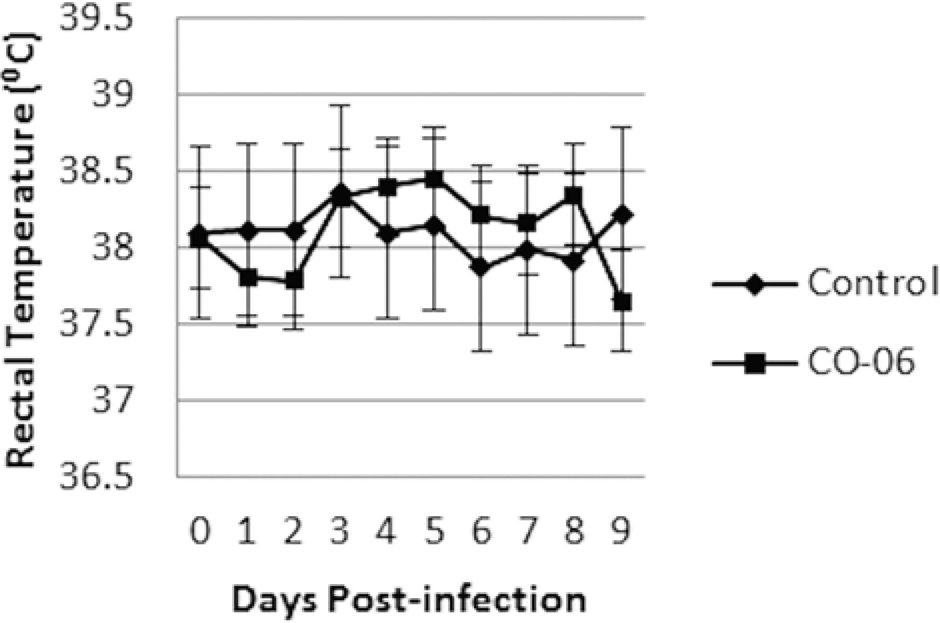
Fever response in Bovine viral diarrhea virus 1 subtype 1b (C0-6)- and sham-inoculated alpacas.
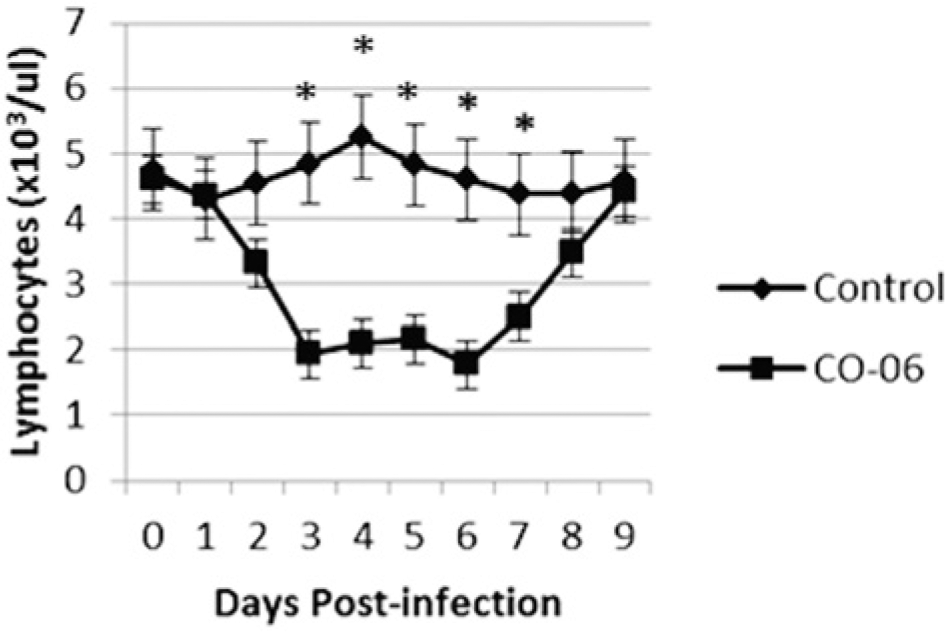
Lymphocyte counts in Bovine viral diarrhea virus 1 subtype 1b (C0-6)- and sham-inoculated alpacas. Asterisks indicate days of statistical different lymphocyte counts.
Viremia and nasal shedding
Viremia was established in each of the BVDchal alpacas. Two out of 12 were viremic on day 3; 5 out of 12 on day 4; and all were viremic on days 5–7 and on day 9. A single challenged animal tested negative on day 8 but again positive on day 9 (Table 1). Nasal shedding of BVDV was detected by PCR on day 3 in 8 out of 12 BVDchal alpacas, and all 12 were shedding virus on day 6 postinoculation. Only 4 out of 12 were still shedding detectable virus on day 9 at the end of the study (Table 2). The BVDcon alpacas remained BVDV negative by all assays throughout the study.
Viremia detected by buffy coat polymerase chain reaction in Bovine viral diarrhea virus 1 subtype 1b challenged (Chal) and control (Con) alpacas.*
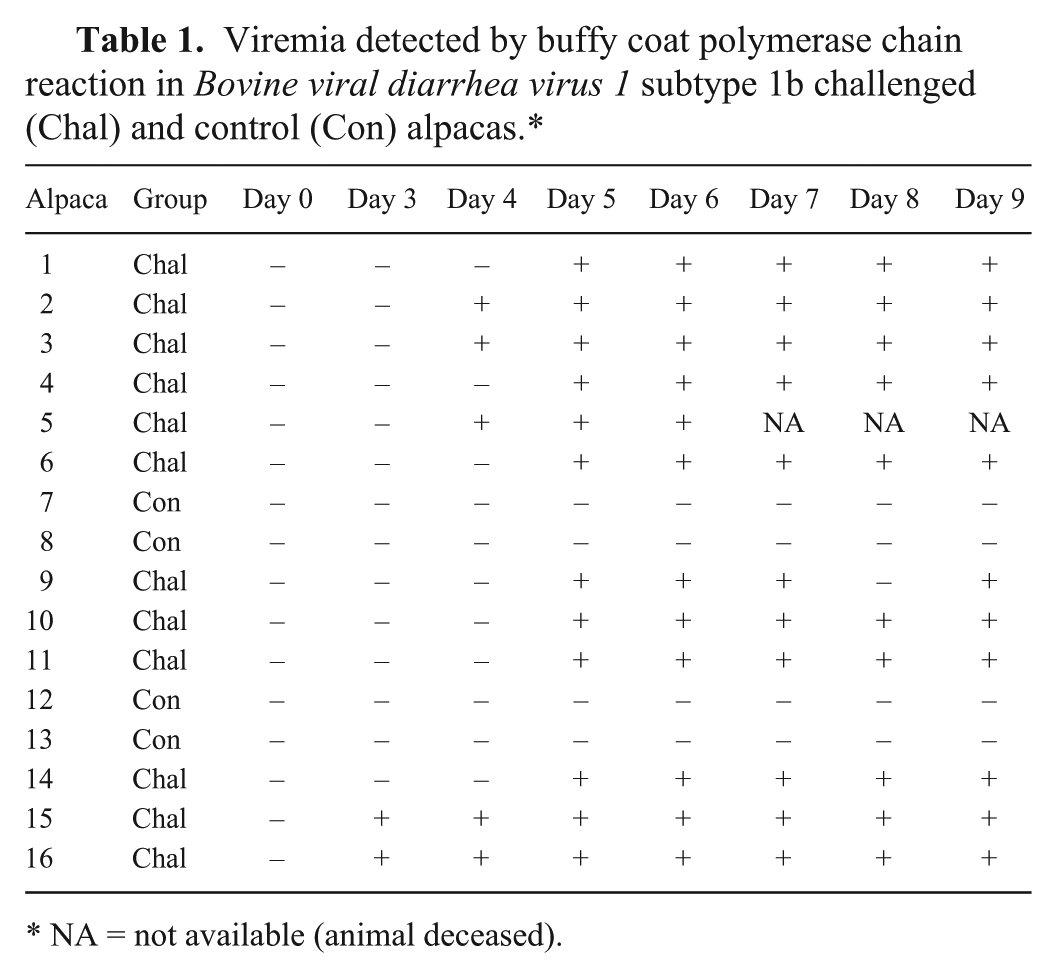
NA = not available (animal deceased).
Bovine viral diarrhea virus 1 subtype 1b (BVDV-1b) antigen distribution, lesions, and nasal shedding in BVDV-1b challenged alpacas.*
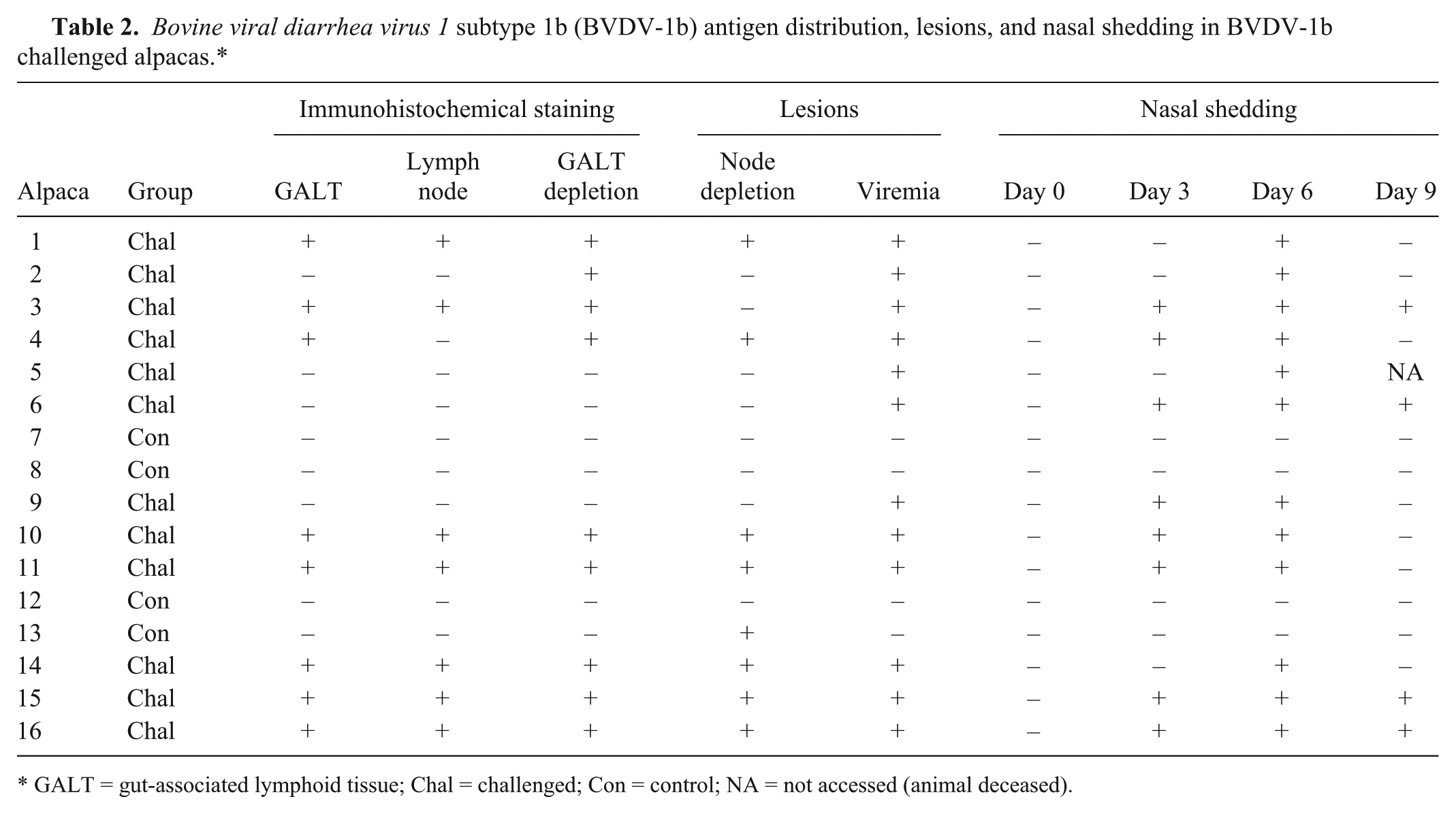
GALT = gut-associated lymphoid tissue; Chal = challenged; Con = control; NA = not accessed (animal deceased).
Necropsy
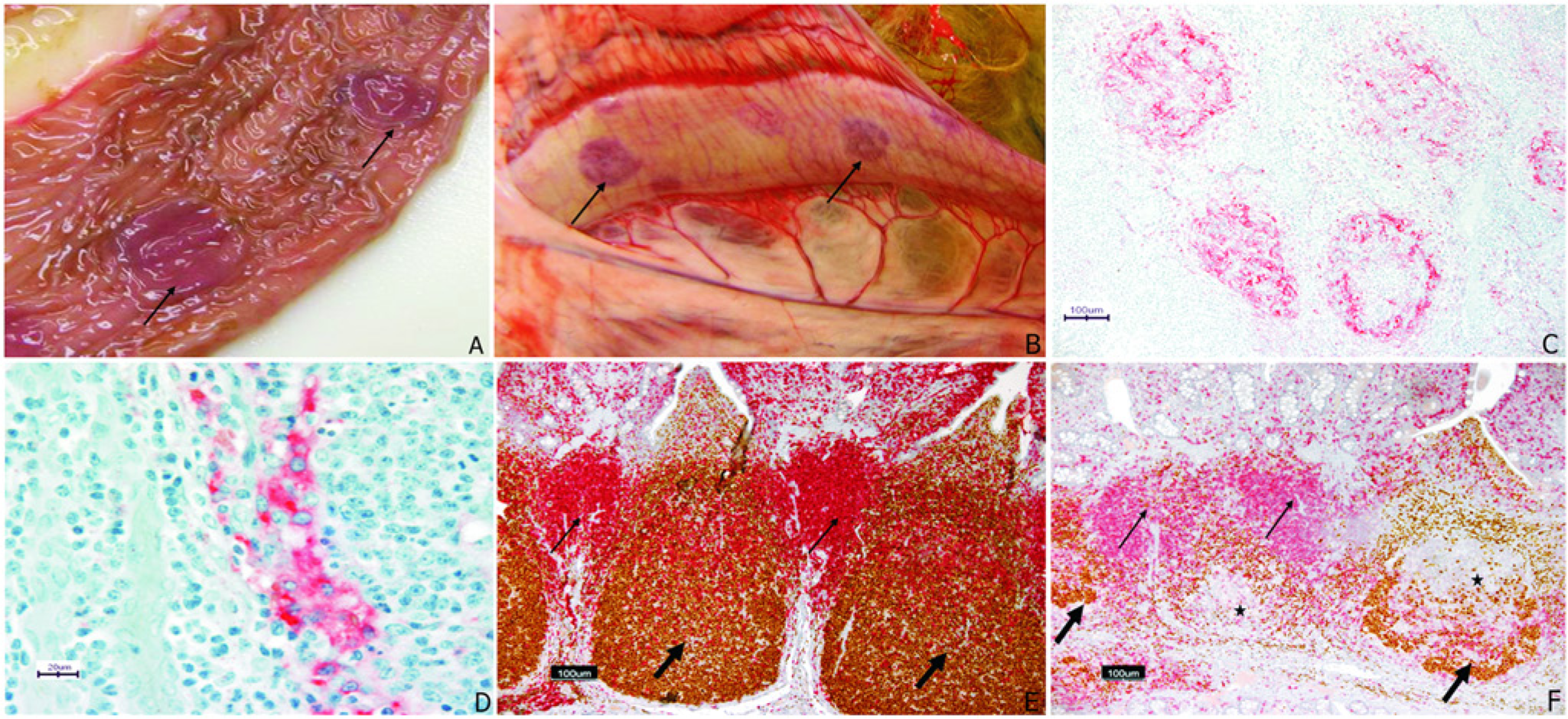
Eleven of the 12 BVDchal alpacas had normal body condition while 1 was noticeably thin as it was before challenge. Six out of 12 BVDchal alpacas had reduced thymus size, as did 1 of the 4 BVDcon alpacas. Enlarged lymph nodes were observed in 7 BVDchal and 1 BVDcon alpacas. The mesenteric nodes were enlarged in 1 BVDcon alpaca while lymph node enlargement in BVDchal animals was as follows: mesenteric (3), cervical (2), prescapular (2), submandibular (1), and mediastinal (1). The ileal GALT was variably prominent in control alpacas and presented as ovals of tissue with slightly raised borders. Subjectively, GALT was less prominent in 3 BVDchal alpacas and 1 BVDcon. Prominent mucosal hyperemia of ileal GALT was visible in 3 BVDchal alpacas (Fig. 3A). Serosal petechiation or hyperemia over GALT was present in 2 BVDchal alpacas (Fig. 3B). Serosal petechiation was present with fine fibrinous exudate overlying the colon in 1 BVDchal alpaca.
Histology
Very mild submucosal or interstitial and perivascular lymphocytic or lymphoplasmacytic infiltrates were present in esophagus, kidney, liver, lung, and gonad of multiple animals. These cellular infiltrates were present in control and challenged alpacas, not typical of those seen with BVDV in other species, and not associated with antigen deposition. Prominent perivascular and peribronchiolar lymphocytic cuffs were present in the lungs of 2 BVDchal and 2 BVDcon alpacas, and similar lesions of less severity were present in 2 additional BVDchal alpacas; there was mild suppurative bronchopneumonia in 1 BVDchal alpaca; there was mild interstitial lymphocytic myocarditis in 2 BVDchal alpacas, one of which also contained several sarcocysts located distant to the inflamed area; 1 BVDcon alpaca and 6 BVDchal alpacas had mild to moderate lymphocytic interstitial nephritis; small and widely scattered foci of lymphocytes and histiocytes with occasional neutrophils were present in the liver of 3 BVDcon and 4 BVDchal alpacas; and finally, intestinal and gastric tissues from all alpacas had low numbers of lymphocytes in the lamina propria. Histologic lesions were not observed in skin, trachea, or brain. These nonlymphoid lesions, with the exception of myocarditis and pneumonia, were present in both groups and thus did not appear treatment related. The lesions were chronic (lymphohistiocytic) and likely reflect residual lesions not unexpected in random sourced animals of undocumented medical history prior to study entry.
Immunohistochemical staining
In 8 of the 12 BVDchal alpacas, antigen was detected by IHC in at least 1 GALT site, and 7 of these had 1 or more IHC-positive lymph nodes (Fig. 3C; Table 2). Four of the BVDchal alpacas were IHC negative for BVDV antigen (IHC negative) in all tissues, as were all of the sham-inoculated controls. BVDV antigen was frequent within follicular dendritic cells, occasional in lymphocytes and rare in endothelial cells (Fig. 3D).
In IHC-positive animals, BVDV antigen was detected in an average of 5.3 of the 10 GALT sites and 2.6 of the 7 lymph nodes tested. Frequency of BVDV antigen detection in organs of the 8 IHC-positive alpacas was ileal GALT (7), proximal colon GALT (6), C3 GALT (5), and tonsils (5). The GALT of C2, duodenum, and jejunum, lymphoid nodules of the anal junction, and parotid lymph nodes were each IHC positive in 4 animals. One animal was IHC positive in C3 GALT and negative in ileal GALT. No antigen deposition was demonstrated in other tissues including the lymphocytic inflammatory lesions in nonlymphoid organs and skin.
Within the IHC-positive group there was lymphoid depletion in an average of 8.9 of the 20 lymphoid tissue specimens from each animal, compared to a mean of 0.8 tissues with lymphoid depletion in specimens from the 4 IHC-negative BVDchal alpacas. Distinctive, treatment-specific changes were limited to GALT and lymph nodes; where the most common features were B-lymphocyte depletion and dendritic cell proliferation within follicles (Fig. 3E, 3F). This depletion and the dendritic cell proliferation were present in 10 of the 12 BVDchal alpacas, including all 8 that were IHC positive (Table 2), which was statistically different than the BVDcon group where 1 alpaca had follicular lymphoid depletion (P < 0.01). The observed follicular depletion was most consistent in C3 and ileum. Follicular depletion was present in 7 of the 8 IHC-positive alpacas, and depletion was associated with the detection of viral antigen in C3 and other GALTs (P < 0.05). Outside of the GALT, the lymphoid depletion and dendritic cell proliferative lesions were most consistent in prescapular and mediastinal lymph nodes, occurring in 6 and 5, respectively, of the 12 challenged alpacas. The GALT of the proximal colon was also commonly affected with 6 out of 12 challenged alpacas having follicular lesions. All BVDchal alpacas had follicular B-cell depletion or dendritic cell proliferative lesions somewhere in non-GALT lymphoid tissues but they were inconsistent compared with those in the aggregated GALT of C3 or the ileum. The control alpaca that had follicular depletion was the one alpaca with marked peribronchiolar infiltrates in lung.
Discussion
The effects of postnatal BVDV infections in immune-competent alpacas with BVDV-1b strains were similar to those reported in cattle challenged with BVDV-1.16,19,21,24,27 Clinical signs in the alpaca were unapparent, which is similar to previous studies.3,25 Consistent and statistically significant leukopenia was documented. The leukopenia lasted from days 3 to 7 postinfection in the BVDchal alpacas, which is similar to descriptions of infected cattle. The lack of significant leukopenia in prior reports of naturally exposed alpacas was unexplained but may relate to sample timing, dose, or viral strain. 3 The leukopenia was due to a dramatic drop in lymphocyte numbers. Statistical leukopenia appeared to be temporally associated with the onset of viremia in BVDchal alpacas. Studies in cattle suggested that the most severe pathologic effects can be expected 9 days postinfection. A similar temporal response was anticipated in alpacas as lesions were consistently found in the day 9 postchallenge alpacas. Myocarditis was the only nonlymphoid lesions unique to the BVDchal alpaca group. This change was not associated with antigen deposition and, not being typical of pestivirus infection, was presumed preexistent and unrelated to challenge. Additional studies with clean source animals for challenge would need to be performed to determine if any relationship to BVDV infection might exist. The gross size variability of thymus, lymph nodes, and GALT were nonspecific. Thymus atrophy can occur with age and chronic disease, lymph node enlargement can occur with any antigen stimulation, and GALT patch visibility varies among unchallenged alpacas studied by the authors (Kammerman, Steffen, unpublished observations). The serosal hyperemia, while rare in the treatment group, was related to challenge status and corresponded to histologic lesions of depletion and congestion.
Histologically, the depletion of lymphocytes in GALT was less in the challenged alpaca lymphoid follicles on day 9 compared with the depletion described in cattle.14,19 Many of the follicles contained proliferated dendritic cells rather than cytolytic debris, suggesting alpacas exhibited a response in dendritic cells slightly different than cattle or perhaps that regeneration of the dendritic cells occurred earlier. The submucosal lymphoid follicles of the alpaca were consistently the preferred location for localization of lesions and antigen deposition in this acute BVDV infection model. The ileal GALT of camelid was discontinuous, and lymphoid patches occurred as small ovoid structures 1–3 cm long with raised edges. The consistent and readily recognizable lymphoid patch of C3 was easily identified in the alpacas, and the alpacas had BVDV antigen deposition of similar frequency to ileal GALT. This additional sampling site was advantageous as ileal GALT was not easily distinguished in a few of the alpacas. Sampling sites for BVDV detection in deceased camelids suspected of acute BVDV infection should include GALT of C3, ileum, and proximal colon, as these sites were most consistently BVDV positive. The distribution of antigen in acute cases was not as dispersed as described in PI cria and thus acute infections may be less effective in transmission of infection to cohorts.6,13 Four cria had depletion suggestive of BVDV, and were viremic and had demonstrable nasal shedding but were IHC negative. The IHC-positive animals only had 50% of GALT sites, and 37% of lymph node sites were positive for BVDV antigen. These 2 facts suggest that IHC is likely less sensitive at viral detection than the PCR assay used for nasal swab and buffy coats.
The transient nasal shedding demonstrated in each of the BVDchal alpacas demonstrates the potential for acute transmission through nasal droplets. The lymphoid follicle depletion of B lymphocytes (Pax3 positive) was spatially associated with establishment of demonstrable infection as evidenced by BVDV antigen localization in depleted follicles (Table 2). A longitudinal study would be necessary to fully elucidate the temporal aspects and development of lymphoid lesions in acute BVDV infection of this species. The lymphoid depletion seen in BVDcon alpacas demonstrates that the lesion, while prominent in BVDV infections, is not specific for BVDV particularly if outside the GALT and not widely distributed. The current study clearly demonstrated that noncytopathic BVDV-1b was capable of causing subclinical disease in the alpaca with viremia, nasal shedding, and a statistically significant lymphocytopenia. Systemic infection of lymphoid tissues and pathology of GALT was similar to that occurring in cattle. Leukopenia and lymphoid depletion may result in immune suppression and secondary infections, which have been commonly reported in BVDV-related deaths of crias. 7
Footnotes
Acknowledgements
The authors acknowledge the cooperation of individual breeders that contributed materially to the project and to the Alpaca Registry for their continued cooperation.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the University of Nebraska Agricultural Research Division (grant NEB 39-152) from funds provided through Animal Health, U.S. Department of Agriculture.
a.
Euthasol, Virbac AH Inc., Fort Worth, TX.
b.
QIAamp Viral RNA mini kit, QuantiTect Probe RT-PCR kit; Qiagen Inc., Valencia, CA.
c.
SAS 9.2, SAS Institute, Cary, NC.
