Abstract
Bead-based multiplex assays (BBMAs) are applicable for high throughput, simultaneous detection of multiple analytes in solution (from several to 50–500 analytes within a single, small sample volume). Currently, few assays are commercially available for veterinary applications, but they are available to identify and measure various cytokines, growth factors and their receptors, inflammatory proteins, kinases and inhibitors, neurobiology proteins, and pathogens and antibodies in human beings, nonhuman primates, and rodent species. In veterinary medicine, various nucleic acid and protein-coupled beads can be used in, or for the development of, antigen and antibody BBMAs, with the advantage that more data can be collected using approximately the same amount of labor as used for other antigen and antibody assays. Veterinary-related BBMAs could be used for detection of pathogens, genotyping, measurement of hormone levels, and in disease surveillance and vaccine assessment. It will be important to evaluate whether BBMAs are “fit for purpose,” how costs and efficiencies compare between assays, which assays are published or commercially available for specific veterinary applications, and what procedures are involved in the development of the assays. It is expected that many veterinary-related BBMAs will be published and/or become commercially available in the next few years. The current review summarizes the BBMA technology and some of the currently available BBMAs developed for veterinary settings. Some of the human diagnostic BBMAs are also described, providing an example of possible templates for future development of new veterinary-related BBMAs.
Introduction
Bead-based multiplex assays (BBMAs) are referred to as multianalyte profiling, a recognized under the trade names xMAP Technology or Luminex assays, a or cytometric bead array (CBA) b assays. The BBMAs are applicable for high throughput, simultaneous detection and quantification of multiple analytes in solution. Typically, for veterinary applications, the analytes can be host immune proteins (i.e., antibody, immunoglobulin [Ig], or immune cytokine), or infectious agents such as viruses, bacteria, fungi, or protozoa. The BBMA method significantly reduces time and sample volume requirements and allows the testing of many samples simultaneously for multiple targets compared to other methods, such as traditional enzyme-linked immunosorbent assays (ELISAs), microarrays, Western blot techniques, or polymerase chain reaction (PCR; Fig. 1). Because only small volumes of sample are needed to measure multiple analytes simultaneously, BBMAs are advantageous for use with limited specimens such as sera from very small avian and animal species, cerebral spinal fluid, or synovial fluid. The assay acquires results on several, up to hundreds, of analytes (50–500) per sample, with the number of targets dependent on the specific assay and instrument.

Comparison of approximate maximum number of samples and targets that can be analyzed between microarrays, enzyme-linked immunosorbent assays (ELISAs), bead-based multiplex assays (BBMAs), and polymerase chain reaction (PCR) assays. Hatched bars = maximum number of targets; solid bars = maximum number of samples. It should be noted that newer technologies with integrated electrodes and arrays are being implemented for alternate ELISA-type assays (http://www.mesoscale.com/CatalogSystemWeb/WebRoot/technology/multiarrays.htm).
To date, there are few commercially available BBMAs for veterinary applications. However, in human diagnostic medicine, numerous commercial assays are available, such as a respiratory viral panel (RVP) c that simultaneously detecst and quantitates 8 viruses and subtypes, and another assay using a gastrointestinal pathogen panel (GPP), d simultaneously detecting 14 bacteria or bacterial toxins, 3 viruses, and 3 parasites. Other commercial variations of the RVP are available.e–g Three commercial assaysc,f,g detect Influenza A virus, Influenza B virus, Parainfluenza 1–3, Respiratory syncytial virus (RSV) types A and B, Human metapneumovirus (hMVP), human Rhinovirus A, B, and C, and some adenoviruses. However, one of the assays f also detects 4 coronavirus variants, Coxsackie/Echo virus, Bocavirus, and differentiates some adenoviruses. One of these assays g detects 3 of the 4 coronavirus variants, differentiates Human parainfluenza 4 (subtypes 4a and 4b), and some adenoviruses (while another c additionally subtypes Influenza A virus as seasonal H1 and H3). Numerous assays are also available to detect and quantitate immune cytokines, growth factors and their receptors, inflammatory proteins, kinases and inhibitors, pathogens, antibodies (i.e., for isotyping or antigen-specific antibodies), neurobiology proteins, transcription factors, genotypes, metabolic, cancer and cardiac biomarkers, allergens, hormones, and tissue antigens (i.e., human leukocyte antigens [HLAs]), 49 but these are developed for use with human, nonhuman primate, and rodent specimens. For livestock species or other animals (other than rodents and nonhuman primates), BBMAs have been developed, but few are currently commercially available. For future use of BBMAs for livestock and other underrepresented animal species, it will be important to evaluate whether these assays are “fit for purpose” prior to development for specific veterinary applications. It is expected that many such assays with veterinary application will be published and/or become commercially available for routine use in veterinary diagnostic laboratories in the next few years. The current review summarizes some of the currently available and/or published protocols (although, not an exhaustive list) and discusses some of the considerations and procedures involved in the development of new assays. A summary of advantages (Table 1) and considerations (Table 2) for using the BBMA technology in veterinary diagnostic laboratories is provided.
Advantages of using bead-based multiplex assays (BBMAs) in veterinary diagnostic laboratories.

Considerations for using bead-based multiplex assays (BBMAs) in veterinary diagnostic laboratories.

Assay designs
The BBMA technology involves obtaining or developing ligands (i.e., peptides, nucleotides, proteins, antibodies, receptors, polysaccharides, or lipids) that are attached to fluorescent beads (Fig. 2). The ligand can be precoupled h or custom coupled; alternately, a kit i can be used to couple the ligand to the beads. The ligands will bind to a specific analyte in the sample (i.e., PCR products, antigens, antibodies). A secondary fluorescent label is then used for detection and quantification.73,74 Each bead, either fluorescent polystyrene beads (5.6 µm in diameter) a or magnetic beads (6.2 µm in diameter), a is distinguished from the others by varied amounts of 1–3 fluorescent dyes in the bead. After binding of the analyte to the bead ligand, another fluorescent marker, typically phycoerythrin (PE), attached to streptavidin (SA) is added (SAPE). The SAPE then attaches to a biotinylated secondary antibody or is incorporated into a PCR reaction product by binding to a biotinylated deoxyribonucleotide triphosphate (dNTP) or biotinylated PCR primer (see http://www.luminexcorp.com/TechnologiesScience/xMAPTechnology/; Fig. 2). Once bound, a semiquantitative measurement of median fluorescent intensity (MFI) is used to determine whether the sample is “positive” or “negative” for the analyte, and a standard curve is used for quantitative measurement. Examples of common BBMA formats most likely to be used in veterinary diagnostics laboratories are described below.
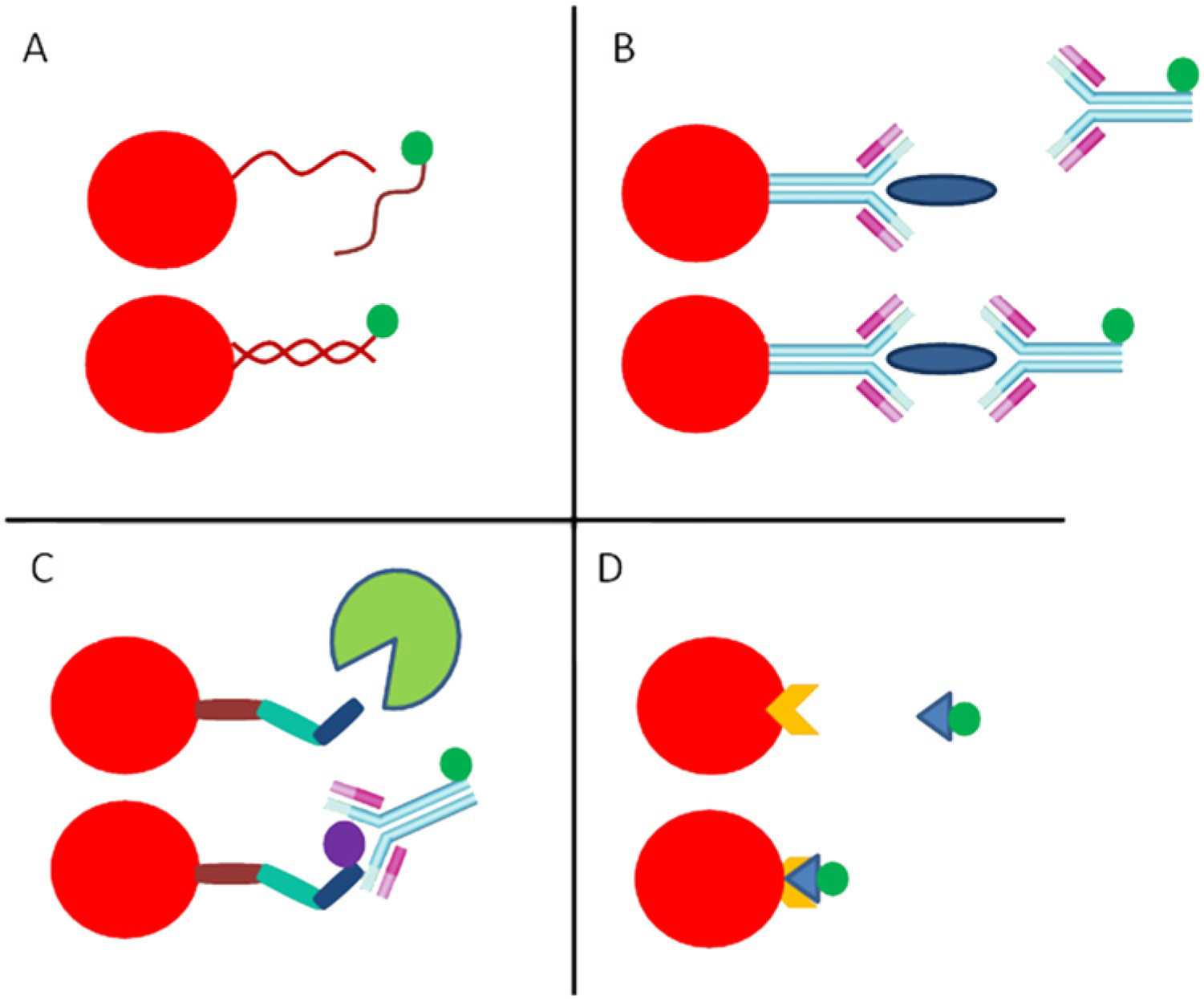
Common bead-based multiplex assay (BBMA) formats.
Bead-based multiplex immunoassays
Many veterinary-related BBMAs are “sandwich” immunoassays whereby fluorescent beads are labeled with purified proteins (such as recombinant expressed proteins) and then sera, saliva,6,61 oral fluids, 83 or other sample types that contain antibodies, are added. Next, a secondary biotinylated antibody is added for detection by the SAPE fluorescent marker. For antigen or immune protein detection and quantitation, capture monoclonal antibodies (mAbs) are coupled to the beads, the sample is added, and detection is performed with a secondary biotinylated mAb (or biotinylated polyclonal antibody), directed at a different epitope than the capture antibody; signal is subsequently identified with the SAPE fluorescent marker (Fig. 2B). In addition to BBMA “sandwich” capture immunoassays (and similar to ELISA formats), “competitive” antibody assays with a single antibody and a labeled positive target, 114 or an “indirect” assay 125 with both a target protein and anti-antibody can be designed (Angeloni S, Cordes R, Dunbar S, et al.: 2013, xMAP® cookbook: a collection of methods and protocols for developing multiplex assays with xMAP technology. Available at: http://www.luminexcorp.com).
Polymerase chain reaction–based BBMAs
A variety of PCR-based BBMAs can also be used for organism detection by coupling beads (or obtaining already coupled beads) with a nucleic acid probe (Fig. 3A–D). Nucleic acid extraction and PCR amplification is followed by hybridizing the resultant PCR product with the beads containing the specific probes. These probe tags h coupled to beads may be “ordered” and come with a corresponding anti-tag nucleic acid sequence that is added to a primer sequence. The second primer has a biotinylated moiety on the 5′-end that is detected with SAPE if the PCR product specifically binds to the “tagged” bead (Fig. 3B–D). Alternatively, a “custom” probe (“direct” nucleic acid detection) can be coupled to the fluorescent bead and a forward and reverse primer designed, one of which would be biotinylated and binds to the SAPE for detection purposes as described for identification of various Mycoplasma species. 129 This “signature” methodology was also used for viral genotyping of respiratory pathogens, 92 whereby two 20–base pair (bp) primers (one of which was biotinylated) were used for PCR and a 30-bp pathogen-specific probe was then hybridized to the PCR product to produce a 90–200-bp amplicon. 92 In another viral genotyping assay, 46 viral papilloma strains were identified using 46 probes (each 30 bp in length) that targeted the glycoprotein (gp)5 and gp6 region of the major capsid region. 169 This type-specific BBMA was developed to determine the distributions of various papillomavirus types, to monitor vaccine efficacy over time and to detect shifts in papillomavirus strains not covered by vaccines. Polymerase chain reaction–based BBMAs using custom probes have also been used to detect gene expression from messenger RNA in circulating tumor cells. 105 The intent was to use this information for prognostic and treatment evaluations, 105 but similar multiplex “biomarker” assays could be developed in veterinary medicine to determine disease resistance and predict nonclinical to clinical states. 109 Gene expression signatures have been obtained using PCR-based BBMAs. 118
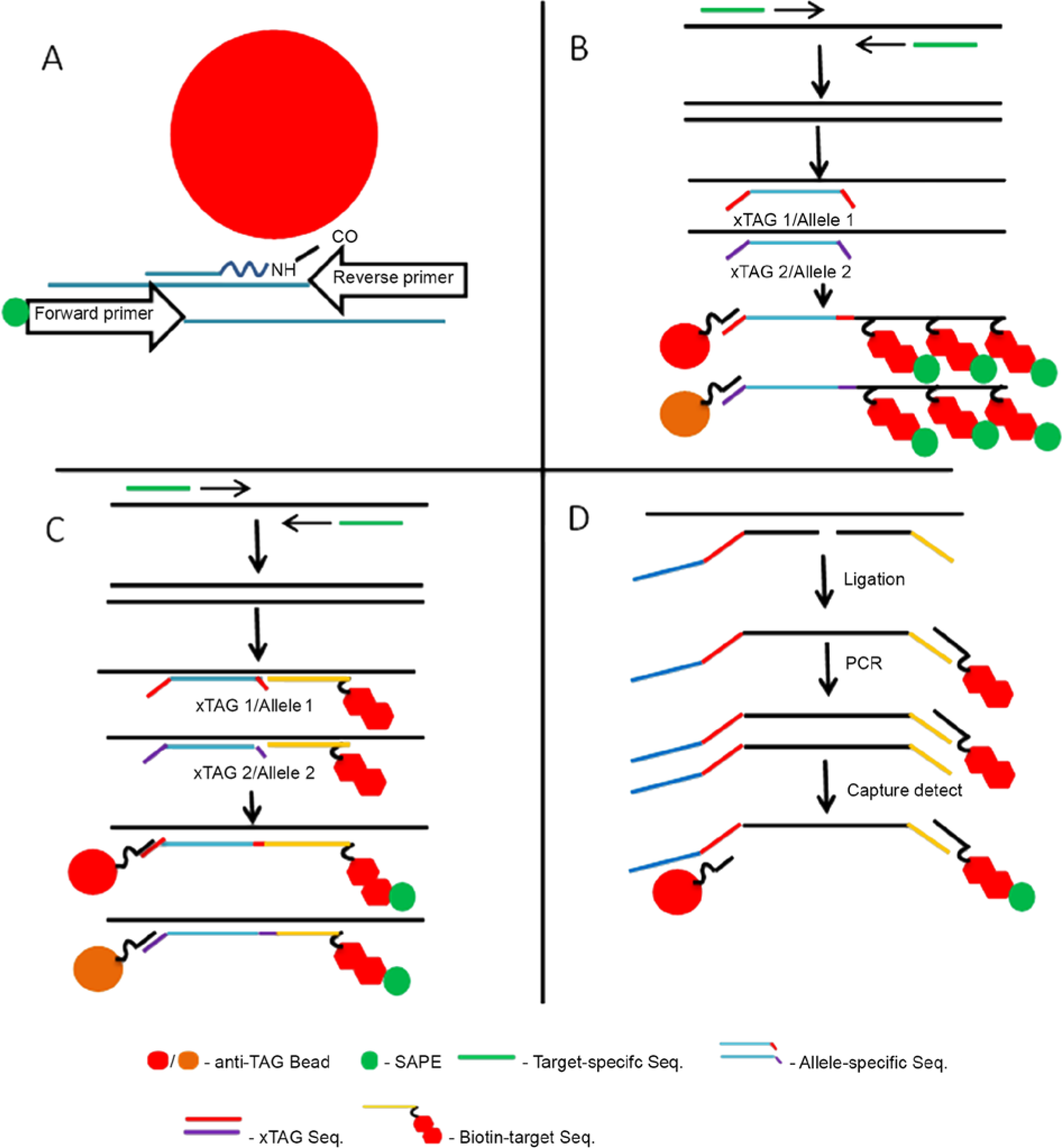
Polymerase chain reaction (PCR) bead-based multiplex assay (BBMA).
Genotyping BBMAs are also designed to detect single nucleotide polymorphisms (SNPs). Several methods can be used, such as oligonucleotide ligation assay (OLA; Fig. 3C), allele-specific primer extension (ASPE; Fig. 3B), 140 and single base chain extension (SBCE or xTAG/SBCE). 29 The ASPE reaction has been used to detect 17 different Gram-positive and -negative bacteria using the 16S ribosomal DNA sequence for discrimination purposes. 167 The SBCE was used to analyze 55 SNPs and evaluate 181 genotypes. 29 A target-specific primer extension (TSPE) reaction is similar to the ASPE and OLA, which use a chimeric capture primer containing an anti-tag sequence (complementary to the “tag” on the bead) h and an oligonucleotide complementary to the amplified PCR product (Fig. 3C). 155 If the target is present in the ASPE and TSPE format, the primer can be extended by Taq polymerase and a biotinylated deoxynucleotide triphosphate is incorporated that binds the SAPE for detection purposes. A TSPE with 5 degenerate primers amplified the hemagglutinin and neuraminidase genes for Influenza A virus, and 8 “tagged” primers were used in the extension reaction, which then hybridized to the fluorescent beads. 103 A direct hybridization and ASPE BBMA were compared for the detection and differentiation of multiple Candida spp. 117 In that comparison, both assays could be completed in 1 day. 117 The ASPE was somewhat more time consuming than the direct hybridization (8 hr vs. 5 hr), but was more “discriminatory” because it could detect SNPs, rather than a larger region of DNA sequence. Both assays were designed to differentiate within a 600-bp ribosomal RNA (rRNA) region. 117 Target-specific PCRs (TS-PCR), also referred to as “FAST” assays, are similar to ASPE and have been used in the commercial RVP c and GPP. d The use of TS-PCR involves amplification of extracted nucleic acid and generates a “tagged” double-stranded PCR product. It uses a tagged forward primer and a downstream primer labeled with biotin rather than incorporation of the biotinylated dNTP as in the ASPE. 116 Unlike the ASPE, the biotinylated primer and tagged sequences are introduced in the initial PCR step so there are many reactions occurring simultaneously and the assay can be performed more quickly.
For OLA, if the target is present, a ligase reaction is required, and a biotinylated probe homologous to the target DNA is present. Another type of ligation-dependent amplification reaction used for multiplexing involves ligation of specific probes first and then performing PCR with universal primers, one of which is biotinylated (Fig. 3D). This multiplex oligonucleotide ligation–PCR (MOL-PCR) was used for detection of 13 signatures of the biothreat agents Bacillus anthracis, Yersinia pestis, and Francisella tularensis. 40
Such PCR-based BBMAs have been previously described and diagrammed in more detail elsewhere.68,74,82,102,107 All the aforementioned genotyping BBMAs can be used for host SNP genotyping. As an example, an algorithm for SNP calling using R software was used to assist in SNP association for childhood leukemia patients. 21
It is important to note that PCR-based BBMAs are still relatively new platforms, and novel formats of BBMA for detection of viral or bacterial nucleic acids are being actively developed and are evolving. These new formats may streamline the multiplex assay, prevent false-positive or -negative reactions by incorporating nonstandard nucleotides on biotinylated primers, 166 allow for quantitation, and allow for “closed tube” assays in which the PCR is performed with the microspheres in the reaction tube, thereby reducing the risks of contamination and false-positive reactions inherent in multistep assays. 94
Cytometric bead array and variations of BBMA
An alternate flow cytometric-based multiplex bead method, the CBA b method, has been developed for quantifying multiple proteins simultaneously. 111 The CBA uses antibody-coated beads to capture analytes, and the capture beads contain unique amounts of a single red dye. The unique spectral properties of this dye enable analysis of samples on flow cytometers that have a single 488-nm laser or dual lasers (488 nm or 532 nm and 633 nm). With CBAs and other BBMAs, unconjugated beads are available so researchers can conjugate their own antibody or protein of interest using sulfosuccinimidyl-4-(N-maleimidomethyl)cyclohexane-1-carboxylate (sulfo-SMCC) chemistry. Each bead has a unique fluorescence intensity so that beads can be mixed and run simultaneously in a single tube, although the CBA is limited to 30 targets per sample. The detection mAbs are labeled with unique fluorochromes or with biotin (followed by fluorochrome-labeled streptavidin). As with CBAs and other BBMAs, researchers select and evaluate antibody pairs for dynamic range and sensitivity, and compare titration curves with native biological samples. Assay diluents and wash buffers have been formulated to reduce detrimental effects of serum and plasma proteins on assay performance. A consideration for these assays is the flow cytometer and number of lasers needed, so commercial product selection depends on whether the CBA is compatible with the local instrument. However, if a laboratory has an existing flow cytometer, these instruments can typically be used for the CBA. Other variations of BBMA have been previously addressed (Fee L: 2003, Assays galore. The Scientist. Available at: http://www.the-scientist.com/?articles.view/articleNo/15083/title/Assays-Galore/).
Instrumentation, costs, optimization, analysis, and interpretation
Instrumentation
Bead-based multiplex assayj,k instrumentation uses 2 light sources, one that excites the 2 or 3 fluorescent dyes that identify each bead and another that excites the reporter dye (typically PE) captured during the assay. The instruments can detect up to 100 j or 500 k analytes simultaneously per sample in a 96- or 384-well format, respectively, with few adjustments for an increasing number of targets. In 2010, a smaller-sized platform instrument l was introduced that is based on the principles of fluorescent imaging but uses a more cost efficient reader with light emitting diodes (LEDs) instead of lasers. This smaller instrument requires the use of magnetic beads because the instrument is a “fluorescent imager” rather than a flow cytometer type instrument. Lasers and photo multiplier tubes are replaced with LEDs and a charge-coupled device camera, which saves on costs and is more compact and portable, with the capability of detecting up to 50 analytes simultaneously. Prior to reading on the BBMA instruments, efficient automated systems for incubation and washing steps of magnetic beads are very useful, most of which can be performed on an ELISA washer m with a magnetic plate.
For CBA assays, b users do not necessarily have to buy a new instrument if there is access to a flow cytometer. However, reference to the company fluorochrome specifications chart, instrument, fluorochrome guide, and fluorochrome/laser reference chart is needed to determine the instrument configuration and corresponding kits that can be used. However, some of these CBA tests have limited commercial assays for use only on human and laboratory animal specimens. It would also be important to document reproducibility, precision, and accuracy in standard curves and samples, within and between kits for whichever BBMA is used in the diagnostic or research laboratory. 128
Costs
Costs for the BBMA are kept low because of the ability to assay large numbers of samples and increasing numbers of analytes or targets per sample. For example, when examining distributor prices, it is noted that the cost per test per analyte for a 30-plex panel would be less than the cost per test per analyte for a 5-plex panel (i.e., in the latter case, almost double the cost per test per analyte with less multiplexing). One genotyping study using 2,000 samples and measuring 15 SNPs in a BBMA showed minimal reagent costs that decreased with increasing multiplexing and increasing sample numbers. It should be noted that the costs given in that study also reflect a lower number of beads per assay than recommended in the BBMA.a,23
As previously described, various instruments may detect up to 50, l 100, j or 500 k analytes simultaneously per sample in a 96- or 384-well format with few adjustments for an increased number of targets. The current maximum number of analytes that the CBA assay can measure is up to 30 analytes simultaneously. The 50-, 100-, and 500-plex BBMAs are performed in a 96- or 384-well format with approximate “read times” for each instrument being 60 min per 96-well plate, l 40 min per 96-well plate, j 20 min per 96-well plate, k or 75 min per 384-well plate. k The BBMA instrument l that tests up to 50 analytes simultaneously is approximately half the cost of the other instruments and uses less reservoir space and fluid. Currently, real-time PCR may be able to distinguish up to 5 analytes using 5 different fluorophores, but with some difficulty, as buffers, enzymes, and other reagents may need adjustments to provide the same sensitivity and specificity when more targets are added.
Analysis and interpretation
In the BBMA, the PE fluorescent signal is proportional to the amount of bound analyte. Analysis software identifies each bead population, generates a standard curve, and calculates the concentration of unknown samples. The concentration of unknown samples is based on an MFI with a corresponding quantity (i.e., picogram/ml, sample-to-positive ratio value) obtained from a standard curve. A 5-parameter logistic regression has been recommended for sandwich immunoassays to obtain a broader range of concentrations (compared to linear regression) for unknown samples from a single set of standards for multiple analytes (http://www.biocompare.com/Application-Notes/42701-Principles-Of-Curve-Fitting-For-Multiplex-Sandwich-Immunoassays-Rev-B/). 42
Most instruments provide appropriate software to interpolate results. A “positive” MFI is typically described as 2 standard deviations above the background MFI or negative control (Angeloni S, et al.: 2013, xMAP® cookbook). Interpretation of results could be an issue when multiple analytes are detected, as with any multianalyte assay. For example, it may be difficult to determine if 1 or more of the pathogens in a GPP d was the cause of the clinical problem or whether one of the organisms was shed for prolonged periods of time and was just detected when the BBMA was used. Therefore, before using the assay, it is important to determine the multiple scenarios that could result and how to interpret them. Clinical and contextual knowledge are important in these interpretations, to determine if the presence of the organism is or is not indicative of disease. However, the multianalyte tests can also be used as a tool for the discovery of unexpected infectious agents, which may or may not be a cause of disease. It has been suggested that changes in the global environment involving changing vectors, animal and human behaviors and movements have influenced and will continue to influence whether disease occurs. 10 Therefore, it will be important that “modern-generation diagnostic tests be readily adaptable to, and capable of, detecting and identifying both known and unknown, even variant or novel, viruses in field samples.” 10 Multiplex testing will be important in this regard.
Sample and assay optimization
There are many considerations and steps required for sample and assay optimization. 48 For example, with multiplex immunoassays, steps include finding and selecting specific mAbs or polyclonal antibodies for capture and detection, determining the coupling efficiency of the primary mAb, optimizing incubation times and concentrations of capture and detection mAbs, comparing singleplex and multiplex assays, checking for cross reactivity of reagents, determining precision (inter- and intralaboratory variability), performing comparisons to “gold standard” assays, determining the dynamic range, limits of detection (analytical and diagnostic sensitivity), and specificity, and performing the assay using “known status” clinical samples.48,87
Both mAbs and polyclonal antibodies can be used for capture and detection in a pairwise format, but if mAbs are used, the capture and detection mAbs should detect different epitopes (Angeloni S, et al.: 2013, xMAP® cookbook). It is generally accepted that a mAb should be used for capturing the analyte for the best sensitivity and specificity, but if a polyclonal antibody is used, it should be monospecific and affinity purified. Confirmation of primary coupling is critical as an initial first step and is usually performed by combining coupled beads with an anti-species IgG coupled to PE (i.e., anti-mouse IgG-PE).36,55 For oligonucleotide, PCR-based BBMAs, coupling confirmation is performed using a biotinylated target oligonucleotide labeled with SAPE.
Cross-reactivity evaluation can be performed using 3 tests for 1) analyzing whether analytes cross-react with nontarget beads, 2) to determine if reporter antibodies cross-react with nontarget analytes, and 3) to determine if there is cross-reactivity in the complete assay using multiplexed analytes and multiplexed detection antibodies (Angeloni S, et al.: 2013, xMAP® cookbook).
Sample type is also a consideration in assay optimization. Blocking agents, assay diluents, sample dilution, and buffers may need to be changed depending on the sample type. For example, serum concentration can alter the standard curve, and some studies revealed that human sera may contain heterophilic antibodies that directly bind to beads causing immunoassay interference.49,106,156 Methods including dilution of sera and inactivation of complement in a HLA BBMA prevented false-negative reactions caused through a “prozone” effect. 158 Nonspecific binding of antibodies to microspheres causing false-positive reactions was also noted in another study. 124 Removing a protein wash step and adding a proprietary blocking agent n prevented the problem. Microspheres have 100–2000-Å pores on their surfaces, and the pores can trap proteins, thus blocking agents are needed (Angeloni S, et al.: 2013, xMAP® cookbook). Assay diluents can also be a source of interference; therefore, diluents and assay buffers must be optimized, and all reagents and proteins must be included in a BBMA under common assay conditions. 123 It is generally acceptable to dilute a sample 1:5 to prevent interference or microsphere agglutination. However, some samples such as urine, cerebral spinal fluid, and synovial fluid may not require dilution (Angeloni S, et al.: 2013). More specific protocols for optimizing immunoassay or PCR-based BBMAs are available (Angeloni S, et al.: 2013).
Comparison of BBMA with ELISA
It would be ideal if the antibody pairs used in ELISA and BBMA kits were identical so that the absolute antigen values could be compared in both types of assays. However, because of assay parameters, there may be differences in antibody pairs and sample diluents, and these are likely causes of observed differences between the 2 types of assays and among various BBMA kits. 44 Additionally, differences among purified recombinant proteins used to generate standard curves and in manufacturer-supplied assay buffers can cause variations in cytokine and chemokine quantification. 108 Several reports30,49,76 compared BBMA kits from different vendors and, while each kit resulted in different absolute concentration of cytokines, each revealed comparable relative trends in cytokine concentrations.
Comparison of BBMA with real-time PCR
Because many veterinary diagnostic laboratories use real-time PCR routinely for fast, sensitive, and efficient diagnostics, a comparison of the nucleic acid detection BBMAs is important before deciding whether they can take the place of real-time PCR. A previous article 115 describing the human RVP c assay used a kappa score analysis to assess the level of agreement between real-time PCR and the BBMA where a kappa score of 1.0 indicates almost perfect agreement. 143 Of the 6 viral targets that were compared by real-time PCR and BBMA, the kappa scores were 0.97–1.0 for 4 targets but were lower for 2 viral targets (i.e., adenoviruses, kappa score = 0.72; RSV, kappa score = 0.91). The discordant samples were positive by PCR and negative by BBMA. The discordant samples generally had higher threshold cycle (Ct) levels (37.7 Ct mean for RSV) or a higher Ct with more than 1 target in the sample (i.e., 34.8 Ct mean for adenoviruses). These results indicate that further BBMA optimization is needed to detect samples with high Ct levels indicative of low levels of viral nucleic acid.
For rapid detection of newly emerging diseases, it is particularly important to have highly sensitive tests. The real-time PCR for detection of Foot-and-mouth disease virus (FMDV) was more sensitive than a 7-plex BBMA that detected the viruses that cause FMD, swine vesicular disease, vesicular stomatitis, vesicular exanthema of swine, bluetongue, bovine viral diarrhea, and malignant catarrhal fever. 64 However, if further BBMA optimization could be done, this test could be used as stated, “to facilitate embedded foreign animal disease surveillance while conducting routine testing for animal diseases viruses that are endemic to the area.” 64
“Fit for purpose” assay evaluations
As with any diagnostic assay, the first question that needs to be addressed is whether the assay is “fit for purpose” as described in the validation workflow from the World Organization for Animal Health (OIE) Terrestrial Manual, 2012 (http://www.oie.int/fileadmin/Home/fr/Health_standards/tahm/1.01.05_VALIDATION.pdf). Formulating the question that the test will answer is essential and should be decided prior to validation procedures. For example, if a test is to be used for early detection of an infectious agent, the diagnostic sensitivity should be equal to or better than that of an individual assay currently in use. Various technologies may be more advantageous than others in answering the question and are dependent on many factors including the number of samples to be tested and the number of targets being evaluated (Fig. 1). A BBMA is flexible and can be used for multiple purposes, for early diagnostics and later for pathogen response detection (i.e., for vaccine differentiation from infection assays [DIVAs]). Some assays may also be used in combination, such as broad screening of multiple targets in a sample via a microarray and then, for lower expense and high throughput testing of multiple samples, refining the number of consistently found targets and performing the high throughput testing in a targeted BBMA. For example, a gene expression microarray assay was performed to determine genes expressed that differentiated naïve from memory T cells. Since memory T cells are important in immunity to pathogens, screening for gene expression would help in determining vaccine efficacy and treatment responses. A consistent gene expression profile was determined from the microarray and narrowed to 55 analytes to be detected simultaneously in subsequent screening of a large number of samples through the BBMA. 63
Human disease diagnostic assays as a preview of potential veterinary diagnostic assays
Before discussing veterinary applications, the impact of a few of the current human multiplex assays will be reviewed as examples and possible templates for the development of veterinary-related assays. In January 2013, the U.S. Food and Drug Administration allowed marketing of an assay for a multianalyte profile GPP, d which is a multiplexed nucleic acid–based test that can simultaneously detect 14 bacteria or bacterial toxins, 3 viruses, and 3 parasites. This assay detects genera Salmonella, Shigella, Campylobacter, Giardia, and Cryptosporidium, as well as Clostridium difficile toxin types A and B, the genes for the heat labile toxin and heat stabile toxin of enterotoxigenic Escherichia coli (ETEC), E. coli O157, the gene for the Shiga-like toxin (stx) produced by Shiga toxin–producing forms of E. coli (STEC; stx1 and stx2), Rotavirus A, and Norovirus groups I and II from a single fecal sample. Another human diagnostic test, the RVP, c simultaneously detects 8 viruses and subtypes including Influenza A virus subtypes H1 and H3, Influenza B virus, RSV, hMVP, Rhinovirus A, B, and C, and some adenoviruses. Bead-based multiplex assay strategies for detecting human respiratory viruses has been reviewed, 81 and these assays are performed routinely in some hospitals. 115 Another commercially available BBMA e has been used for the detection of 6 respiratory bacterial pathogens including Streptococcus pneumoniae, Neisseria meningitidis, encapsulated or nonencapsulated Haemophilus influenzae, Legionella pneumophila, Mycoplasma pneumoniae, and Chlamydia pneumoniae. Although this assay had less sensitivity when compared to real-time PCR, it was useful for surveillance purposes 15 and, when combined with a commercial assay including viral respiratory pathogens f for a 21-plex assay, significant coinfection rates could be established.22,25
In-house developed assays have also been used97,98 to detect Norovirus, Rotavirus, and Sapovirus species, as well as astroviruses and adenoviruses from feces. In addition, another in-house developed nucleic acid–based multiplex assay described as a variation-tolerant capture multiplex assay was developed. 112 This assay used PCR multiplexing (i.e., up to 22 PCRs) with a strategy of using long primers and probes that allow for variation at known sites and degenerate and universal bases that give good sensitivity for the detection of gastrointestinal bacteria, fungi, viruses, and antibiotic-resistant genes. 112
Numerous commercial assays are available for inflammatory and acute phase proteins, immune cytokines, and growth factors and their receptors for use in human, rodent, and primate diagnostics and research. Some of the early applications included multiplexed assays to detect cytokines.26,113,145 A comparative evaluation of several commercial BBMAs for human cytokines has been published. 42 Cytometric bead array b is also commercially available to measure 7 cytokines. o The assay was important in determining anti–Human immunodeficiency virus cytokine responses for those individuals advancing to acquired immunodeficiency syndrome. 159 A previous study 45 compared 2 BBMAa,b methods and a microarray method p and concluded there was lack of comparability among the 3 methods. However, analytical performances were adequate for longitudinal studies using any 1 method, but standardization was required before results obtained with different methods could be compared. 45 Because comparisons between various commercially available cytokine BBMAs are not completely similar, it will be important to include positive controls (high, medium, low), dilute standards within the given sample matrix, and validate the standards prior to performing the assay on large-scale experimental studies, checking for linearity, recovery, repeatability, and obtaining a dynamic range that is acceptable for the samples to be tested.
Many other “noncommercial” human assays have been developed, and it is estimated that there are >15,000 publications describing uses of BBMAs (http://www.luminexcorp.com/publications). Such assays are used for detecting nucleic acids, viruses, bacteria, and antibodies to other biomolecules (such as toxins).16,114,125,127,133,135,140 Genetic typing of HLA has also been performed using BBMA technology and had good correlation with single-antigen assay flow cytometry cross-match outcomes. 110 In cancer studies, the humoral responses to peptide cancer vaccines have correlated with cancer survival rates using BBMAs developed to measure the responses to specific peptides. 80 Other applications are for the detection of multiple antibodies14,16,53,60 and hormones. 13 The technology was also expanded for measurement of cancer biomarkers in human saliva samples. 6 A review of markers in cerebrospinal fluid for the discovery of inflammatory profiles in neurodegenerative diseases using BBMA has been published 104 and explored potential uses for BBMA in identifying biomarkers of immunotoxicity for environmental and public health research. 46
Veterinary disease diagnostic assays
Commercial assays for serologic responses
Typical ELISA technology is used extensively in veterinary diagnostic laboratories for antibody detection. Differences between the ELISA and BBMAs are in the number of targets per sample, and therefore, the volume of sample needed for the assays, with the BBMA requiring less sample volume and being able to evaluate more targets per sample. In addition, the BBMAs involve a fluid incubation step with beads suspended in solution resulting in more surface area exposed to the sample in 3 dimensions. This results in a short diffusion path to antibody binding sites on the beads for a rapid reaction time. In addition, beads in the BBMA have a large surface area of approximately 106 capture molecules per bead so more capture biomolecules can be immobilized in the assay. 70 In ELISA technology, there is an enzymatic reaction for detection, but for the BBMAs, direct fluorescence is used for detection without the requirement of enzyme activity, which may also reduce the time for detection and increase the test sensitivity. Some commercial and “in-house” developed BBMAs for detection of antibodies and antigens are available for veterinary use and are discussed below. However, this is not an all-inclusive list because of time and space limitations, with many tests being currently developed.
Avian disease diagnostic assays
One commercially available serologic test q detects antibodies to 4 avian viruses, including Infectious bursal disease virus, Newcastle disease virus, Infectious bronchitis virus (Avian coronavirus), and avian reoviruses. This assay uses 50 µl of a 1:500 dilution of sera, so it has an advantage over other serologic tests because only small volumes of sera can be obtained from some birds. The assay takes approximately 3.5 hr to perform, and multiple control beads are included to make sure the secondary antibody and serum are added to the sample. Investigators showed that results were equivalent between the BBMA and ELISA (R 2 > 0.99) when tested with identical samples under the same conditions (Comparison of Luminex® 200™ to MAGPIX® using the poultry serology assay: technical notes, http://www.luminexcorp.com/prod/groups/public/documents/lmnxcorp/309-poultry-serology-compar.pdf).
In another avian assay, antibody responses to avian Influenza A virus have been monitored over time using a BBMA. For this assay, baculovirus-expressed proteins, including the nucleoprotein, matrix protein, and nonstructural protein (NSP) 1, were coupled to fluorescent beads for the basis of an assay that differentiated between serologic responses to various influenza subtypes. 157
A BBMA 11 was developed to detect antibodies to West Nile virus (WNV), St. Louis encephalitis virus, and Eastern equine encephalitis virus from avian and other species through biotinylation of the serum sample. This allowed for a multispecies BBMA without the need for species-specific reagents (i.e., anti-species capture antibodies and detection conjugates) or the use of blocking ELISAs. When this assay was used on multiple avian and other species samples, initial results showed high background levels that were alleviated by changing the phosphate buffered saline–1% bovine serum albumin (BSA) buffer to a commercial buffer. r The use of biotinylated protein A/G (i.e., an Ig binding protein) with subsequent detection by SAPE has also been used in place of species-specific detector antibodies (Achterberg R, van der Wal F, Maasen K: 2010, Trichinella serology in swine on the Luminex platform. Poster, EAVLD Congress, September 2010, Lelystad, The Netherlands).
Rodent disease diagnostic assays
An advantage of using BBMA in small animals such as rodents is that only small amounts of sample are needed to obtain many results. For example, one particular assay only used 1 µl of a 1:500 dilution of sera for the serodetection of 10 pathogens in mice including Vaccinia virus, Ectromelia virus, Epizootic diarrhea virus of infant mice, Enteric orphan virus, Mouse hepatitis virus, Sendai virus, Theiler’s mouse encephalitis virus (GDVII strain), Mouse parvovirus virus rVP2, Mouse cytomegalovirus, Minute virus of mice, and Mycoplasma pulmonis. 75 Other BBMAs have been developed to detect serologic responses to pathogens in rodents.65,66 Additionally, some commercial laboratories provide BBMAs for use in rodents, or use the technology within their laboratory. s
Porcine disease diagnostic assays
To date, most porcine disease diagnostic BBMAs have focused on the detection of antibodies to specific pathogens. For example, a fluorescent microsphere immunoassay was developed for detection of antibodies against Porcine reproductive and respiratory syndrome virus (PRRSV) and affirmed that pen-based oral fluid sampling could be an efficient, cost-effective approach to PRRS surveillance in swine populations. 83 Another swine-specific BBMA used baculovirus-expressed proteins of the Porcine circovirus-2 (PCV-2) capsid and bacterial-expressed PRRSV nucleocapsid for bead conjugation and subsequent detection of serum antibodies to PCV-2 and PRRSV. 96
Additional BBMAs detecting porcine serologic responses included an assay to investigate the dynamics of the anti-staphylococcal humoral immune response to methicillin-susceptible Staphylococcus aureus. That study reported both age- and litter-associated responses. 34 Bead-based multiplex assays were also developed to detect antibodies to the nematode Trichinella 142 or simultaneous antibody detection of both Trichinella spiralis and Toxoplasma gondii antigens. 19
It was found that a BBMA for detection of swine antibodies against the Gram-positive bacteria Erysipelothrix spp. was more sensitive than 2 commercial ELISAs. 56 The BBMA used a recombinant polypeptide from the major surface protective antigen coupled to beads. The authors concluded that the assay would be useful for early detection of Erysipelothrix spp. and monitoring vaccine compliance. 56 Clearly, a combination of the aforementioned BBMAs could be developed for broad serologic monitoring of swine herds.
Bovine disease diagnostic assays
Several multiplex assays have been developed for detection and differentiation of bovine viruses. Species-specific and pestivirus-common oligonucleotide probes were used to detect Bovine viral diarrhea virus 1 and 2 (BVDV-1 and BVDV-2, respectively), Classical swine fever virus (CSFV), and Border disease virus (BDV). 37 A 4-plex assay was developed for detection of antibodies to several bovine viruses, such as BVDV, Bovine respiratory syncytial virus, Bovine parainfluenza virus 3, and Bovine herpesvirus 1 using only crude viral lysates coupled to fluorescent beads. 4 However, when a purified BVDV rE2 protein was used, better sensitivity was achieved demonstrating the importance of using purified proteins for capture antigens. 4 When BVDV-specific mAbs were used in a “blocking” BBMA format, the assay detected both antibodies to BVDV-1 and BVDV-2 and was comparable to a commercially available ELISA (i.e., sensitivity of 99.4% and specificity 98.3%), demonstrating that the assay would be useful for bovine viral diarrhea eradication programs. 163 When the performance of different immunoassays were compared, a BBMA was the preferred assay to detect specific antibodies against emerging atypical bovine pestiviruses. 84 In addition, this BBMA used a baculovirus-expressed glycoprotein that retained the posttranslational modifications that are recognized in an adequate immune response. 146
As an alternative to purchasing “anti-tagged” primers and “tagged” beads, an internal probe can be coupled to fluorescent beads. For example, a 7-plex assay was developed by directly labeling each of the microspheres with a user-defined probe and biotinylating one of the primers to identify Mycoplasma sp. causing contagious bovine and caprine pleuropneumonia. 129 This format was also used for genotyping Mycobacterium subsp. paratuberculosis into types I (sheep), II (cattle), and III (intermediate) based on the gyrB locus. 54 The BBMA consisted of a duplex PCR with 4 specific probes linked to 4 beads in the assay, 54 but could be expanded to include further genotyping for determining associations of clinical isolates with possible food or environmental origins. 27
Equine disease diagnostic assays
A BBMA was developed to detect antibodies specific to Equine arteritis virus and compared it with the virus neutralization test (VNT). 57 Of 8 recombinant proteins coupled to fluorescent beads, the antibody response to a partial gp5 conjugated bead had the highest correlation with the VNT. Although the sensitivity of the BBMA was less than the VNT, it was suggested that high-throughput screening could be performed using the BBMA, with follow up VNT confirmatory testing. 57
Lyme disease diagnostics have been developed for horses, dogs, and deer based on detection of antibodies to Borrelia burgdorferi OspA, OspC, and OspF antigens as indicators for infection.78,149–152 The assay distinguishes vaccination from infection (i.e., DIVA responses) and determines early- versus late-infection stages to support treatment decisions. Such tests have also enabled researchers to use these host species as sentinels of Lyme disease ticks and diagnose Borrelia-associated uveitis in horses.126,147 The same group has developed new tools for equine Ig isotyping using multiplex analyses. 72
Exotic animal disease diagnostics
A BBMA has been developed for simultaneous and simple detection of 4 pestiviruses, CSFV, BDV, BVDV-1, and BVDV-2. 89 African swine fever virus is a large DNA virus with at least 22 genotypes. 89 Therefore, in genotyping and differentiating the various types, a new BBMA was developed targeting a 450-bp region of the p72 gene. Fifty-two probes that identified 24 SNP pairs and 4 probes for general detection were coupled to beads and hybridized to their appropriate target (if present) in a BBMA after PCR amplification. 88
Although not currently found in the United States, Hendra virus and Nipah virus (both paramyxoviruses) have been identified in Australia and Southeast Asia. These viruses use amplifying hosts of pigs (Nipah virus) or horses (Hendra virus) and can be transmitted to human beings. 144 A TSPE BBMA was developed to detect and differentiate between these 2 viruses and used the nucleoprotein and phosphoprotein encoding genes as PCR targets. 51 The 5′-ends of the TSPE primers contained a 24-base TAG sequence complementary to microsphere sets and detected both viruses and differentiated between them.
A BBMA using multiple probes to detect Babesia divergens, Babesia bovis, Babesia bigemina, Babesia major, Babesia occultans, Theileria annulata, and Theileria buffeli, has been developed. 130 Because there are many species of Babesia and Theileria, 132 some of which are pathogenic and some that are not, this BBMA 131 included a catch-all Theileria and Babesia control probe for the possibility of identification of other species, along with an internal amplification control to monitor for inhibition from bovine blood samples. 131
Other diagnostic assays
Additional pathogen detection assays could be added to current multiplex PCRs, in the form of a BBMA. This has been suggested for aquaculture pathogen testing.1,165
Influenza A virus and resistance to antivirals are ongoing issues in multiple species. Therefore, it is of interest that a TSPE BBMA was developed that used 8 “tagged” beads detecting the seasonal and pandemic H1, seasonal H3, seasonal N1 antiviral (oseltamivir) sensitive and resistant, pandemic N1 antiviral sensitive and resistant, and N2 seasonal antiviral sensitive genotypes. 103 This BBMA, which could be completed within a day, was useful for influenza subtyping and genotyping; it could provide a format for further influenza subtyping and detection of antiviral and antibiotic resistance genes found in other pathogens.
Fungal infections
Cryptococcosis is a systemic fungal infection that affects multiple animal species. 139 One assay designed for detecting Cryptococcus neoformans in immunocompromised people may be of use in veterinary medicine. 41 The assay uses 8 oligonucleotide probes from a rRNA gene region coupled to fluorescent beads to detect and genotype Cryptococcus species. The lowest limit of detection was 101–103 genome copies and could discriminate a 1-bp mismatch. Other fungal infections may affect entire animal populations and immune-compromised individuals, 24 so multiplexed detection of these pathogens may be needed.
Food safety assays
Multiplex assays have been developed to provide early diagnosis of food-borne illness. For example, quick identification of STEC in food would be useful for treatment considerations, and if differentiated by their O groups, the matches might be quickly identified as to their source. 95 One particular BBMA identified 10 different O groups of E. coli using a PCR-based BBMA method, 95 whereas another 7-plex BBMA, which serotyped STEC, used polyclonal antibody–coated beads that bound each bacterial serogroup. 32 In addition, commercially available mAbs to stx1 and 2 were conjugated to beads to simultaneously identify E. coli O157, stx1, and stx2 in “spiked” ground beef, lettuce, and milk. 33
To identify bacteria causing food-borne illness, including E. coli, Salmonella spp., Listeria monocytogenes, and Campylobacter jejuni, the 23S rRNA gene was used for PCR, with subsequent denaturation and binding of the PCR product to specific probe-labeled beads. 43 Antibodies to Salmonella spp., Campylobacter spp., E. coli, Listeria spp., and staphylococcal enterotoxin B were also conjugated to fluorescent beads in a BBMA. 77 However, when the assay was tested on “spiked” apple juice, green pepper, tomato, ground beef, alfalfa sprouts, milk, lettuce, spinach, and chicken washes, some of these complex matrices compromised the sensitivity of the assay. It was suggested that washing samples may be preferable to homogenization of the foodstuffs for optimal sensitivity of that assay. 77 Because salmonella is a frequently reported cause of food-borne illness, a commercial BBMA t was developed using PCR for the O and H antigens, along with some additional targets (i.e., designated as “AT,” which includes Salmonella Enteritidis and Salmonella Typhi and a positive control for the second motility phase of Salmonella) and then hybridizing the PCR products to 100 fluorescent beads for salmonella serotyping (Fig. 4). This BBMA only takes a few hours to complete and is used for direct serotyping of Salmonella isolates from culture. The genes involved in antigen expression are detected in the assay without the use of plate or tube agglutination, which removes some of the subjectivity of agglutination analysis. Another Salmonella serotyping BBMA compared 189 Salmonella isolates to traditional methods for classification as serotype Paratyphi A or serogroup B, C1, C2, D, E, or O13. 50
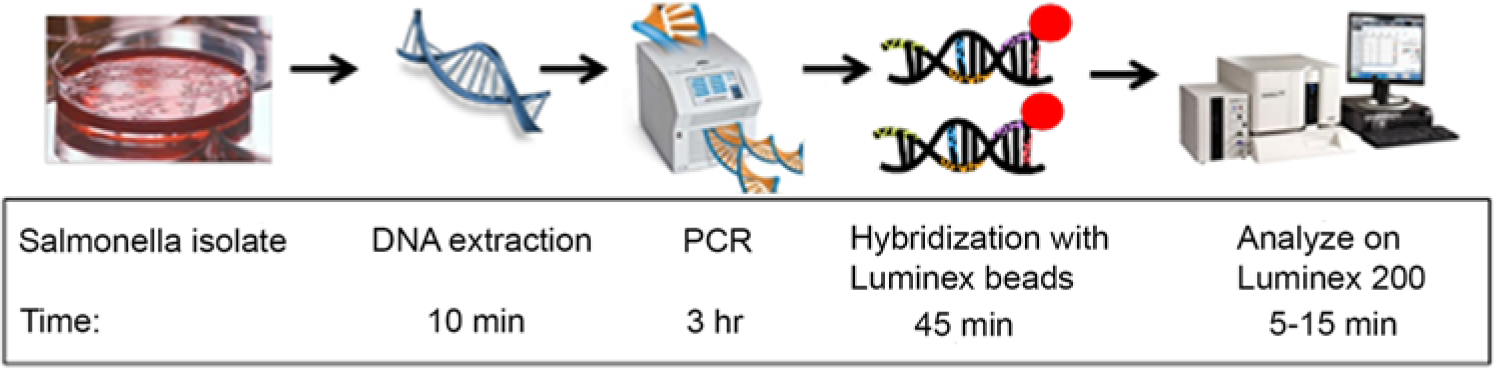
Timeline of Salmonella serotyping bead-based multiplex assay (BBMA). Process includes culture and isolation of Salmonella, DNA extraction, polymerase chain reaction (PCR) assay, hybridization of the PCR product with probe-coated BBMA beads, and analysis on the Luminex BBMA instrument.
Vaccine evaluations—DIVA applications
To determine serologic responses to multivalent (such as trivalent and pentavalent) vaccines, several serotypes or strains can be evaluated using BBMAs when vaccine proteins are conjugated to fluorescent beads. To differentiate vaccinated animals from those exposed to a field strain of virus (DIVA assay), fluorescent beads are coated with field virus proteins that are not found in the vaccine strain. For example, animals exposed to Rift Valley fever virus reacted serologically to field virus proteins coupled onto beads that included the surface glycoproteins (Gn, Gc) and nucleocapsid protein (N), whereas vaccinated animals were only serologically positive to Gn and Gc proteins but not to the N proteins because the vaccine is devoid of the N protein. 141
Circulation of FMD would bring significant economic consequences in regard to international trade and animal health. 67 Therefore, if detected, FMD is controlled by culling or vaccination. Because the vaccine does not result in sterilizing immunity, in order to determine an FMD-free status, postvaccination surveillance would require the absence of persistent infection in vaccinates. 161 Currently, ELISAs, which detect antibodies to NSPs from infected animals, are able to differentiate between infected and vaccinated animals; vaccinated animals would not have antibodies to NSPs, whereas infected animals would have antibodies to NSPs. 31 It is useful to have “multiple signatures” of these NSPs to verify that the antibody responses are indicative of vaccine-only responses, which is the reason an 8-plex BBMA was developed that used 4 NSP-coated beads (i.e., a 3ABC recombinant protein; 3A, 3B, 3D peptides) and 4 controls. 121 In this particular assay, 3 peptides rather than recombinant proteins were used because they could be easily produced in large quantities without the need for a high level containment facility. A BBMA incorporating 4 recombinant proteins (1 envelope protein and 3 NSPs) onto beads enabled detection of antibodies to WNV; thus, it distinguished between horses vaccinated with an inactivated WNV vaccine from horses that were naturally infected. 8
Measurement and differentiation of immune responses to various pathogen proteins such as those associated with Borrelia burgdorferi, the etiologic agent of Lyme disease, 152 may determine if an animal has been exposed to field virus or vaccination. In addition, antibody responses to various Borrelia proteins may represent an acute or chronic infection. 91 Typically, for disease diagnosis in dogs, both ELISA screening and Western blots have been used for these determinations. 91 However, using a BBMA consisting of multiple Borrelia protein–coated beads, which is currently commercially available for human use, u may provide a quicker, broad-spectrum approach to diagnosis in veterinary species compared to screening ELISAs and Western blots.
Assays to evaluate immune responses in animals
Cytokines are small secreted proteins that mediate and regulate immunity. 28 Therefore, it is useful to have multiplexed cytokine assays that can objectively assess the biological immune responses to pathogens, vaccines, and therapeutics or other interventions for controlling animal diseases. For a swine-specific cytokine assay, 87 commercially available porcine-specific mAbs were used that bound to different epitopes for capture and detection functions in the BBMA. Species-specific mAbs are useful because biological cross-reactivity between cytokines of different species has been described, and a minimum of 60% identity is needed for cross-reactivity. 134 It has been shown that of 11 mAbs specific for ovine, bovine, and human cytokines, only 4 antibodies cross-reacted with cytokines from a majority of species. 119 In another study, 5 commercially available human antibodies to tumor necrosis factor α (TNFα), were not able to detect bovine TNFα. 38 Some studies have also shown a reduced sensitivity in the multiplexed assay compared to single-plex assays if polyclonal antibodies were used. 39 Many species-specific mAbs can be found at the U.S. Veterinary Immune Reagent Network website (http://www.umass.edu/vetimm/). To date, there are many human, rodent, and monkey cytokine BBMAs that are commercially available, but few are available for the diversity of veterinary species.
Porcine immune assays
One of the first porcine cytokine BBMA used anti-mouse IgG-coated microspheres that attached to the anti-cytokine mAbs (instead of covalently linking the mAb directly to the microsphere). The coated bead then captured the target cytokine. The assay detected nanogram (ng) quantities of 3 porcine proinflammatory cytokines, including TNFα, interleukin-8 (IL-8), and IL-1β. 69 In 2009, another porcine BBMA cytokine assay was developed for detection of IL-1β, Il-6, IL-8, IL-10, TNFα, and heat shock protein 32 (Hsp32); however, several polyclonal antibodies were used for capture and detection; the sensitivity was enhanced if the BBMA was performed as 2 separate assays (i.e., a 4-plex of IL-6, IL-8, IL-10, and TNFα and a 2-plex of IL-1β and Hsp32). 18 An 8-plex “sandwich” BBMA, involving capture and detection with swine-specific mAbs, simultaneously detected picogram quantities of innate (IL-1β, IL-8, interferon α [IFNα], TNFα, IL-12), regulatory (IL-10), T helper 1 (Th1; IFNγ) and Th2 (IL-4) cytokines. 87 This BBMA measured serum cytokine levels associated with vaccination strategies for PRRSV, although it was determined that a significant increase in serum IL-12 in vaccinated pigs did not correlate with protection against PRRSV viremia. 87 In 2013, a swine cytokine and inflammatory marker BBMA was developed for swine when used as biomedical models in studying cardiac, limb, or ischemic injury and shock. 20 The assay detected and quantified basic fibroblastic growth factor, vascular endothelial growth factor, platelet-derived growth factor-bb, injury markers (cardiac troponin-I), and complement activation markers (C5a and sC5b-9) along with the typical inflammatory cytokine markers (IL-1β, IL-6, IL-10, and TNFα). 20 Commercial swine cytokine assays are available from various companies including a 7-plex quantitative assay v (IL-1β, IL-4, IL-8, IL-10, IFNγ, IFNα, TNFα), 87 another BBMA w that offers a choice of 10 analytes (IFNα, IFNγ, IL-1β, IL-4, IL-6, IL-8, IL-10, IL-12/IL-23 p40, TGF-β1, TNFα), and a third BBMA with 13 analytes (GM-CSF, IFNγ, IL-1α, IL-1β, IL-1ra, IL-2, IL-4, IL-6, IL-8, IL-10, IL-12, IL-18, TNFα). x In 2013, a CBA 3-plex assay was developed for the major pro-inflammatory cytokines TNFα, IL-1β, and IL-6 with an additional CBA assay for the major acute phase proteins, including C-reactive protein, haptoglobin, and pig major acute phase protein in porcine plasma. 162
Equine immune assays
The first BBMA for equine immune studies was a 3-plex assay that detected IL-4, IL-10, and IFNα. 148 Cornell University Animal Health Diagnostic Center (https://ahdc.vet.cornell.edu/test/list.aspx?Species=Equine&Test_Name=&TstTyp=&WebDisc=) offers equine BBMA analysis, which includes detection and quantitation for IFNα, IFNγ, IL-4, IL-10, and IL-17. The assay helped affirm that IFNα is induced with neuropathogenic Equid herpesvirus 1 (EHV-1) strain Ab4 and that IL-10 is suppressed. 153 The assay identified immune correlates of vaccination and infection for EHV-1. 58 Markers of systemic inflammation in horses with heaves have been reported. 86 An additional BBMA was developed for measurement of equine IFNγ and IL-12 using bovine antibodies that had an 89% and 78% species-specific amino acid similarity, respectively. This BBMA was described as useful in the measurement of immune-mediated equine melanoma therapy. 47
Other immune assays
For canine immune measurements, a 12-analyte BBMA y is commercially available that detects β-NGF, IFNγ, IL-2, IL-6, IL-8, IL-10, IL-12p40, TGF-β1, TNFα, CCL2, SCF/c-kit Ligand, and VEGF-A. Another canine-specific cytokine BBMA z was used to determine prognostic outcomes of immune-medicated hemolytic anemia in dogs. 79 There is a cattle 3-plex BBMA for the detection of IL-1 β, IL-6, and TNFα. 39
An optimized feline cytokine BBMA was also developed using ELISA-characterized antibodies for capture and detection. aa The 3-plex assay detected IL-10, IFNγ, and IL-12/IL-23 p40 with equal to better lower limits of detection and dynamic range compared to ELISAs and used 50 µl of sample compared to 300 µl needed for the 3 ELISAs. 160
Many cytokine BBMAs are commercially available for use in rodents.v,bb,cc For example, a commercially available 23-plex rodent BBMA cytokine assay bb was used for evaluations of the immune responses to naked DNAs encoding 6 open reading frame antigens of PCV-2. 2 The BBMAs are also adaptable for use on several sample matrices including brain tissue, 35 seminal plasma, 59 and lung. 71
Polymorphic genotyping
As mentioned for human studies, BBMAs have been used for SNP genotyping and genetic typing of HLA alleles. In 2011, a BBMA for detection of class II swine leukocyte antigen (SLA) alleles was developed. 5 These host antigens are known to be important in the genetic control of immune responses to foreign antigens, and, as such, potentially could be used in predicting the efficacy of vaccine responses. The assay uses 7 primer pairs (with 1 biotinylated primer per pair) and 20 probes conjugated to fluorescent beads. This assay was useful for high-throughput screening of multiple animals and had a 95% concordance with DNA sequencing. 5
Assays for other diagnostics
Agricultural and environmental assays
Administration of recombinant bovine somatotropin (rbST) to enhance milk production in dairy cows is banned within the European Union. 136 Therefore, a 3-plex BBMA was developed for detection of rbST-dependent markers, including insulin growth factor-1 (IGF-1), IGF binding protein (receptor for IGF-1), and antibodies to rbST. 136 Because no one marker alone provides enough sensitivity or specificity, it was thought that the combination of targets would be advantageous for detecting rbST abuse. However, it was found that an additional marker was needed to detect the 95% true prediction (5% false compliant rate). The fourth marker, consisting of osteocalcin (a marker of bone turnover), was added; this addition combined with detection of antibodies to rbST was sufficient to detect the noncompliant population. 101
A BBMA was used to detect 3 vegetable sources of proteins in milk powder to check for adulteration of the powder. 62 A BBMA was developed to verify antibiotic-free milk. The assay used specific mAbs to detect tylosin, tetracycline, gentamicin, streptomycin, and chloramphenicol. 137 Inhibitory substances in milk were removed by using a diethyl ether pretreatment with ultrafiltration. A BBMA was also developed to detect 6 selected priority coccidiostats in feed and eggs, as these are the only veterinary drugs still permitted as feed additives to treat poultry for coccidiosis. 17 A BBMA for clenbuterol detection in livestock urine has been developed 120 and could be incorporated into a screen for multiple veterinary drugs. This technology is being tested for simultaneous quantitative determination of 7 kinds of pesticide and veterinary drug residues (i.e., atrazine, chloramphenicol). 99
The potential of the BBMA f system for water quality applications has been investigated and is considered to be important to human and animal health. 12 In that study, multiple fecal indicator targets indicating human fecal contamination of coastal waters were simultaneously detected. However, the study concluded that more efficient and concentrated methods of detecting environmental contaminants are needed. Another BBMA 9 was able to differentiate Cryptosporidium hominis from Cryptosporidium parvum in stool and environmental samples and is particularly useful for epidemiologic investigations as C. hominis primarily infects people, whereas C. parvum infects many animal species as well as people. 9 This BBMA was significantly less expensive than other techniques such as PCR amplification followed by DNA sequencing analysis or direct fluorescent antibody testing. 9 A BBMA was also developed for simultaneous detection of Cryptosporidium spp. and Giardia spp. in fecal and environmental samples and was equivalent to microscopic examination of fecal smears. 93 Both organisms are of importance because they can cause diarrhea in human beings and animals.164,168
Other pathogens, including Bacillus anthracis, Yersinia pestis, Francisella tularensis, and Coxiella burnetii were detected in animal and environmental samples using a BBMA. 68 As previously described, the MOL-PCR assay detected 13 signatures of B. anthracis, Y. pestis, and F. tularensis. 40 This methodology was particularly useful because the gene-specific areas detected were short (i.e., DNA targets of 40–50 nucleotides) and could be amenable to detection of degraded targets that can be found in environmental, forensic, and clinical samples.
Mycotoxin assays
Mycotoxins in feed can compromise animal health. The primary assays for detection of mycotoxins include ELISA and liquid chromatography–tandem mass spectrometry (LC-MS/MS). However LC-MS/MS may not be as efficient for screening a large number of samples and ELISA is not as efficient for determining multiple mycotoxin species. 122 A BBMA was developed to detect 6 different mycotoxins. 122 This BBMA used mycotoxin-BSA conjugates coupled to beads to test whether feed samples containing a specific mycotoxin would bind to the bead and block binding of a secondary mAb specific for the mycotoxin analyte. Quantitative results were difficult to obtain in some feed matrices, and a better “clean up” procedure was recommended. 122 An alternate BBMA was developed for simultaneous detection of 4 different mycotoxins in corn and peanuts. 154 An indirect competitive BBMA has been used to detect fumonisin B1 and ochratoxin A contaminants in grain samples. 3 In that assay, both toxins were conjugated to the fluorescent beads, and biotinylated mAbs specific for each toxin were competitively inhibited by various concentrations of the toxins in solution. The mycotoxin concentration was measured by a decrease in antibody binding and was inversely proportional to the amount of mycotoxin in the sample, as routinely described for a competitive assay. 3
Evaluation of enzyme activity related to cancer biomarkers, treatments, and cell signaling
Tyrosine kinase inhibitors have been used to treat cancer and other abnormalities in both human and veterinary medicine.90,100,138 As enzyme (peptide) substrates can be coupled to fluorescent beads in a BBMA, this allowed for an in vitro, biological activity measurement of tyrosine kinase activity and its inhibitors. In a previous human study, which evaluated treatments for chronic myelogenous leukemia (a leukemia where kinase activity affects the clinical phenotype), a BBMA was used to evaluate small molecular inhibitors of kinase activity for treatment and prognostic evaluations. 138 Receptor tyrosine kinases are also used as “cancer biomarkers” because they are receptors for growth factors, cytokines, and chemokines. Other kinases promoting inflammatory processes have been evaluated using BBMAs. 85 Such assays coupled fluorescent beads with the enzyme substrates, and subsequent phosphorylation was measured with a biotinylated anti-phosphate antibody detectable with SAPE. 85
Considerations for new assay development
For development of new veterinary-related assays, a consortium of laboratories with specific areas of expertise may be needed to devise the optimum assays (Table 2). Expertise is needed in knowing the pathogenesis of the disease (i.e., knowledge of when antigens and antibodies are likely to be present; their expression level and duration during the disease progression or vaccine response). This information would be important for selecting the targets to include in a new BBMA. In addition, standard samples are important for new assay development and are essential for comparison of the new BBMA with the gold standard tests that are already being used in the field. Furthermore, for test validation, sufficient field samples with known status are required, along with knowledge of how the current assays are being used “in the field” and what is needed for improvement. It is essential to contact clients and laboratories to determine the feasibility of using the new assay. For future BBMA development, several resources are available to help the conversion of currently used singleplex ELISAs into BBMA formats, including a new video journal.6,7 However, not all ELISAs translate into optimized BBMAs, so it will be important to develop additional veterinary-specific reagents in the future. 87
Future use of BBMAs in veterinary medicine
Costs are a major consideration for clients and for the laboratory, whether private or public, and testing for more analytes simultaneously would decrease expenses. Therefore, it may be advantageous to develop assays for endemic disease surveillance (either antigen or antibody tests) and then combine this with foreign animal disease surveillance for a possible public and private partnership and shared costs. User-defined tests will also be important for veterinary diagnostic testing. For example, Table 3 illustrates potential swine-specific BBMAs (i.e., an RVP on lung or oral fluid samples; septicemia panel on spleen or liver tissue; or nursery or a finisher enteric panel on feces). Currently, some of these pathogens are being detected in multiplex PCR assays. 52 Other “user-defined (customized) assays” may be developed and performed. Currently, individual antibody ELISAs are routinely used to detect antibodies to specific antigens. In the future, routine testing for antibodies to multiple antigens could be performed simultaneously using BBMAs, resulting in decreased costs, time, and sample volume for testing. Initially, in-house assays will need to be properly validated with diagnostic and analytical sensitivity and specificity accurately measured. For long-term routine testing, validated procedures and reagents (i.e., precoupled beads, standards, and biotinylated secondary primers or antibodies) need to be developed, made available, and standardized among laboratories.
Proposed bead-based multiplex assay swine pathogen panels for routine diagnostics or combined with foreign animal disease surveillance.

Footnotes
Acknowledgements
The authors thank Dr. Charles Martin, Luminex Corp., Austin, TX, for his technical advice and recommendation of numerous articles on veterinary-related multiplex assays.
a.
Multianalyte profiling (xMAP), Luminex Corp., Austin, TX.
b.
BD cytometric bead array (CBA), BD Biosciences, San Jose, CA.
c.
Extended tag (xTAG) respiratory viral panel (RVP) multianalyte profiling assay, Luminex Corp., Austin, TX.
d.
Extended tag (xTAG) gastrointestinal pathogen panel (GPP) multianalyte profiling assay, Luminex Corp., Austin, TX.
e.
Multiplex PCR ResPlex I assay, Qiagen Inc., Valencia, CA.
f.
Multiplex PCR ResPlex II assay, Qiagen Inc., Valencia, CA.
g.
Multicode-PLx, EraGen Biosciences, Madison, WI.
h.
Extended tag (xTAG) beads (MagPlex-TAG), Luminex Corp., Austin, TX.
i.
Antibody coupling (AbC) kit, Luminex Corp., Austin, TX.
j.
Luminex 100/200 system, Luminex Corp., Austin, TX.
k.
FLEXMAP 3D, Luminex Corp., Austin, TX.
l.
MAGPIX, Luminex Corp., Austin, TX.
m.
Biotek Elx405, BioTek Instruments Inc., Winooski, VT.
n.
BSA-free StabliGuard immunoassay stabilizer (SG01), SurModics Inc., Eden Prairie, MN.
o.
CBA human and mouse Th1/Th2/Th17 kits, BD Biosciences Pharmingen, San Diego, CA.
p.
Protein biochip array technology, investigator evidence; Laboratoires Randox, Roissy en France, France.
q.
xMAP Flock Monitor, Biovet USA Inc., Bloomington, MN.
r.
Candor low cross buffer, CANDOR Bioscience GmbH, Wangen, Germany.
s.
RADIL, IDEXX Laboratories Inc., Westbrook, ME.
t.
Luminex xMAP salmonella serotyping assay, Biovet USA Inc., Bloomington, MN.
u.
recomBead Borrelia IgG, recomBead Borrelia IgM; Mikrogen GmbH, Neuried, Germany.
v.
Life Technologies, Grand Island, NY.
w.
Procarta (swine cytokine assay) Plexable porcine cytokine, chemokine and growth factors assay, Affymetrix Inc., Santa Clara, CA.
x.
MILLIPLEX MAP porcine cytokine/chemokine magnetic bead panel, EMD Millipore Corp, Billerica, MA.
y.
Procarta Plexable canine cytokine, chemokine and growth factors, Affymetrix Inc., Santa Clara, CA.
z.
CCYTO-90K, EMD Millipore Corp., Billerica, MA.
aa.
DuoSet ELISA, R&D Systems Inc., Minneapolis, MN.
bb.
Bio-Plex mouse cytokine 23-plex panel, Bio-Rad Laboratories, Hercules, CA.
cc.
Affymetrix Inc., Santa Clara, CA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a Conselho Nacional de Desenvolvimento Científico e Tecnológico-Embrapa Labex postdoctoral fellowship (200602/2010-1) to Karla Araujo, by National Pork Board support through grants 08-189, 09-234, 09-244, and by U.S. Department of Agriculture Agricultural Research Service project funds.
