Abstract
Waldrapp ibis (Geronticus eremita) are a critically endangered species, and there are currently more birds in captivity than in the wild. A juvenile, male Waldrapp ibis housed in a mixed-species exhibit was found dead with no premonitory signs. Necropsy revealed extensive necrotizing hepatitis associated with numerous pleomorphic protozoa that were immunohistochemically reactive with antibodies raised against Tritrichomonas foetus, a parasite of cattle. Electron microscopy confirmed the organisms as members of family Trichomonadidae, and sequence analysis of the first ribosomal internal transcribed spacer region (ITS1), 5.8S ribosomal RNA, and ITS2 regions indicated high genetic similarity (96–97%) to members of the Tetratrichomonas gallinarum complex. The animal was born in captivity, and no introductions in this exhibit had occurred since 2009. Other Waldrapp ibis that had contact with the infected male were negative for flagellate infections by fecal examination, thus cross-species transmission is proposed as the source of infection. The host range of the T. gallinarum complex is very large and although the pathogenicity of its members, especially for wild birds, is controversial, these parasites should be considered as a possible cause of acute mortality in Waldrapp ibis. In addition, immunohistochemistry with T. foetus antibodies and molecular diagnostics may be useful tools for preventative veterinary care of endangered bird populations. A greater understanding of the ecology and pathogenesis of this pathogen may also be vital for screening subclinical captive populations and existing wild populations prior to reintroduction efforts.
The Waldrapp ibis (Geronticus eremita), also known as the northern bald ibis and the hermit ibis, is listed as highly endangered by the International Union for Conservation of Nature. To date, only approximately 100 breeding pairs are thought to remain in the wild, and captive breeding programs are critical for the survival of this species (www.iucnredlist.org). However, descriptions of diseases arising in Waldrapp ibis and of their relative sensitivity to common pathogens are sparse. Previous reports have cited mycobacteriosis, streptococcal endocarditis, gastric foreign bodies, ulcerative dermatitis, and trauma as causes of disease in captivity (Quevado MA, von Houwald F: 2003, Skin problems in northern bald ibis. In: Proceedings of the Northern Bald Ibis Conservation and Reintroduction Workshop, Innsbruck, Austria).4,14,16
Flagellated protozoa of the genus Tetratrichomonas have a broad host range including birds, mammals, reptiles, mollusks, and bony fish. 5 Tetratrichomonas gallinarum, originally detected in chickens, was subsequently recognized to be a species complex that has a wide host range including several different bird species and human beings.5-7,17,19,20,22 The pathogenicity of this flagellate is controversial as experimental infections have frequently failed to produce disease, and it has been suggested that clinical disease in infected poultry only occurs in birds coinfected with other pathogens.3,9,13,15,18 However, sporadic case reports implicating T. gallinarum as a primary pathogen in other species have been published; one of which was a case of encephalitis in a wild bird.19,20 The current study describes a fatal case of hepatic tetratrichomoniasis in a captive juvenile Waldrapp ibis. The protozoan organism was identified as a member of the T. gallinarum complex by electron microscopy, polymerase chain reaction (PCR), and sequence analysis.
The male Waldrapp ibis was born in captivity 16 months prior to death and was housed in an outdoor, netted multispecies exhibit along with 6 other Waldrapp ibis. During this time, no animals were added to this exhibit. Other species inhabiting the aviary included spotted pigeon (Columba guinea), European roller (Coracias garrulus), smew (Mergellus albellus), spotted dikkop (Burhinus capensis), buff-banded rail (Gallirallus philippensis), and marbled teal (Marmaronetta angustirostris). The collection continues to be relatively healthy with the most recent mortalities being associated with geriatric animals or with aggression. The bird had no previous health problems, received annual physical exams including hematology and clinical chemistry, and was regularly vaccinated for West Nile virus. a The Waldrapp ibis was found dead in the exhibit with no premonitory signs and was promptly submitted for necropsy.
During gross examination, the animal was found to be in fair nutritional condition, with adequate pectoral musculature and coelomic fat, and weighed 1.3 kg. There was a single, chronic 0.3 cm in diameter puncture wound in the right caudolateral coelomic quadrant from which a firm, dark black crust protruded. The wound extended to, but did not penetrate, the coelomic cavity. There were multiple tenacious, fibrous adhesions between the peritoneum, liver, and gastrointestinal tract. The liver weighed 5.1% of body weight and was dark red with multifocal pale tan to yellow, granular, pin-point to 0.4 cm in diameter depressed regions of necrosis that extended throughout the parenchyma (Fig. 1). A liver sample was submitted for aerobic culture, and insignificant numbers of Escherichia coli and rare mixed growth were isolated (interpreted as contaminants). Representative samples of feathered skin (wound), liver, gastrointestinal tract, cloacal bursa, pancreas, spleen, kidneys, testes, heart, trachea, lungs, air sac, thyroid glands, adrenal glands, and brain were fixed in 10% phosphate buffered formalin, embedded in paraffin, cut at 5 µm, and stained with hematoxylin and eosin.
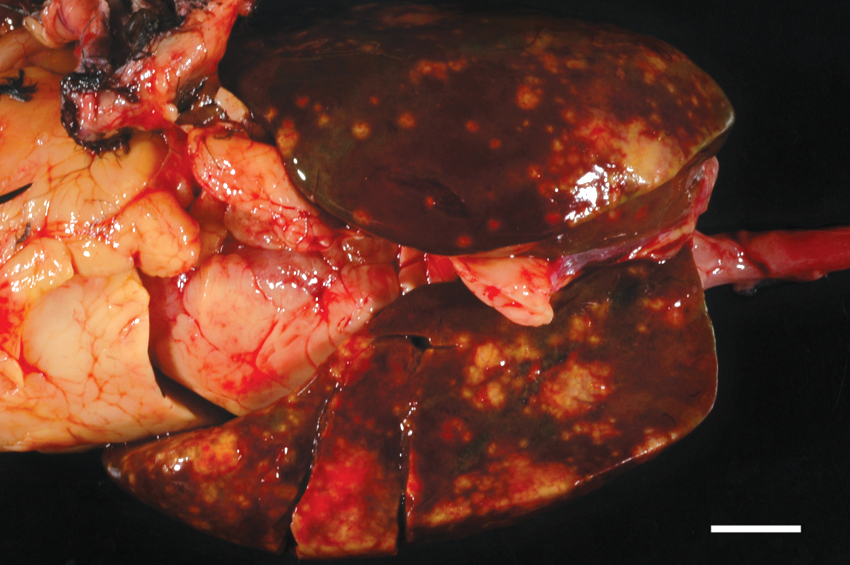
Waldrapp ibis (Geronticus eremita). The liver is enlarged and exhibits multiple individual to coalescing regions of necrosis. Bar = 1 cm.
Histology of the liver revealed extensive, multifocal to coalescing regions of coagulative and caseous necrosis, frequently characterized by central accumulations of degenerate heterophils. Within and at the periphery of the regions of necrosis were numerous pleomorphic round, pyriform, or irregularly shaped, 5–20 μm in diameter protozoan organisms. The organisms typically had abundant pale amphophilic to gray cytoplasm that stained positively with periodic acid–Schiff, and 1–4 small, round, eccentric, hyperchromatic nuclei (Fig. 2). Portal regions were expanded by numerous lymphocytes and plasma cells, fewer macrophages, and rare Mott cells that extended into the adjacent parenchyma. Sinusoids were congested, and hepatocytes were frequently vacuolated, interpreted as degeneration. Tissue Gram stains and Gomori methenamine silver (GMS) stain did not reveal any bacterial or fungal organisms in the sections of liver. Extensive sectioning of the gastrointestinal tract did not reveal similar protozoa. There was mild lymphoplasmacytic enteritis of the small intestine that was likely clinically insignificant.
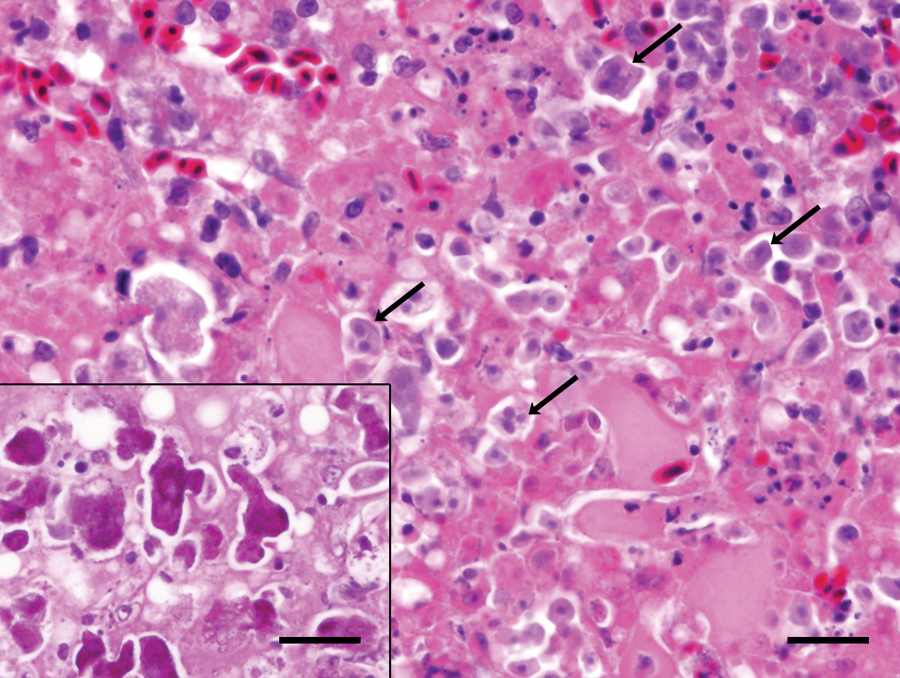
Waldrapp ibis (Geronticus eremita); liver. Admixed with regions of hepatic necrosis are numerous amphophilic protozoa with 1–4 hyperchromatic, round nuclei (arrows). Hematoxylin and eosin. Bar = 20 μm. Inset: the parasites stain positively with periodic acid–Schiff. Bar = 20 μm.
The lesion of the coelomic wall was a chronic heterophilic granuloma, with a thick fibrous capsule that separated it from the peritoneal cavity. No protozoan organisms were present in this lesion and a tissue Gram stain revealed low numbers of mixed Gram-positive and -negative bacteria. The GMS stain did not reveal any fungal organisms in the coelomic wall. Other lesions, including lymphoid hyperplasia and plasmacytosis of the spleen, mild myeloid hyperplasia of the bone marrow, and involution of the thymus and cloacal bursa, were considered to be secondary to antigenic stimulation and chronic disease.
Polyclonal antibodies reactive with Tritrichomonas foetus, b Naegleria fowleri, c Balamuthia spp., c and Acanthamoeba spp. c were applied to the sections of liver as previous described.1,8 The nuclei of the organisms reacted strongly and uniformly with polyclonal T. foetus antibodies (Fig. 3). As T. foetus is a parasite of cattle and not birds, this result was interpreted as a cross-reaction of the antibody with a related trichomonad species. Tritrichomonas foetus antisera have previously been noted to also react positively with Trichomonas gallinae in avian tissues (B Barr, personal communication, 2012). No other antibodies were reactive with the liver sections.
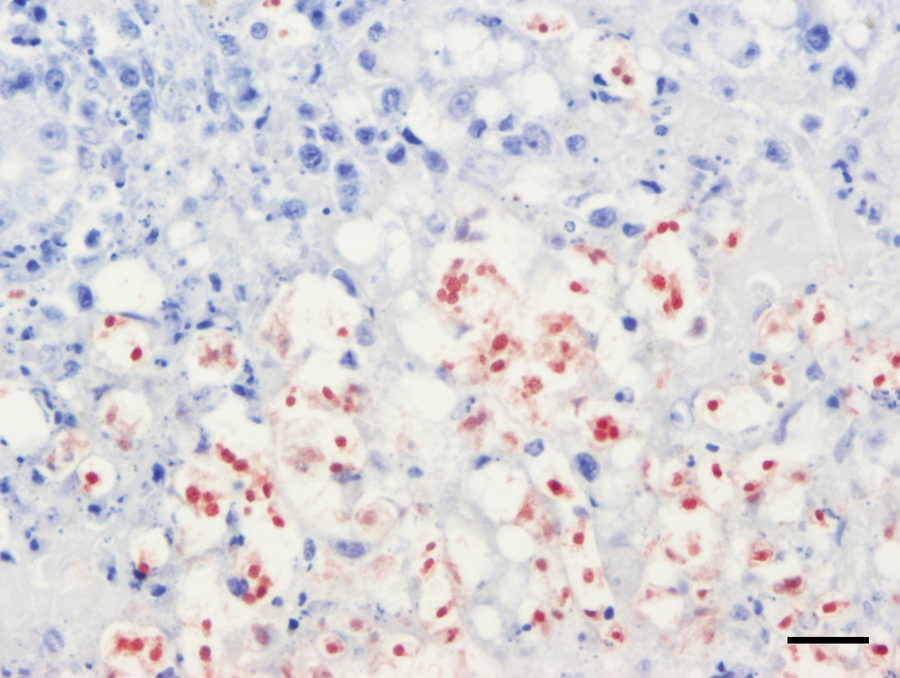
Waldrapp ibis (Geronticus eremita); liver. The protozoa exhibit strong nuclear immunoreactivity with anti–Tritrichomonas foetus antibodies. Rabbit polyclonal anti–T. foetus immunohistochemistry. Bar = 20 μm.
Sections of affected liver were processed for transmission electron microscopy. Liver fixed in 10% neutral buffered formalin was transferred into glutaraldehyde before washing in cacodylate buffer and post-fixation in 2% osmium tetroxide with 2.5% potassium ferrocyanide. Dehydration was by a graded ethanol series before transition into propylene oxide and epoxy resin. d Thin sections were examined in a transmission electron microscope at 100 kV accelerating voltage, and images were captured with a digital camera. e The parasites varied from 5–20 μm in diameter, and their cytoplasm contained abundant organelles and glycogen granules (Fig. 4). The organisms exhibited typical features of trichomonad species including hydrogenosomes, a striated costa, an axostyle formed by concentrically arranged microtubules, a basal body, and flagella.
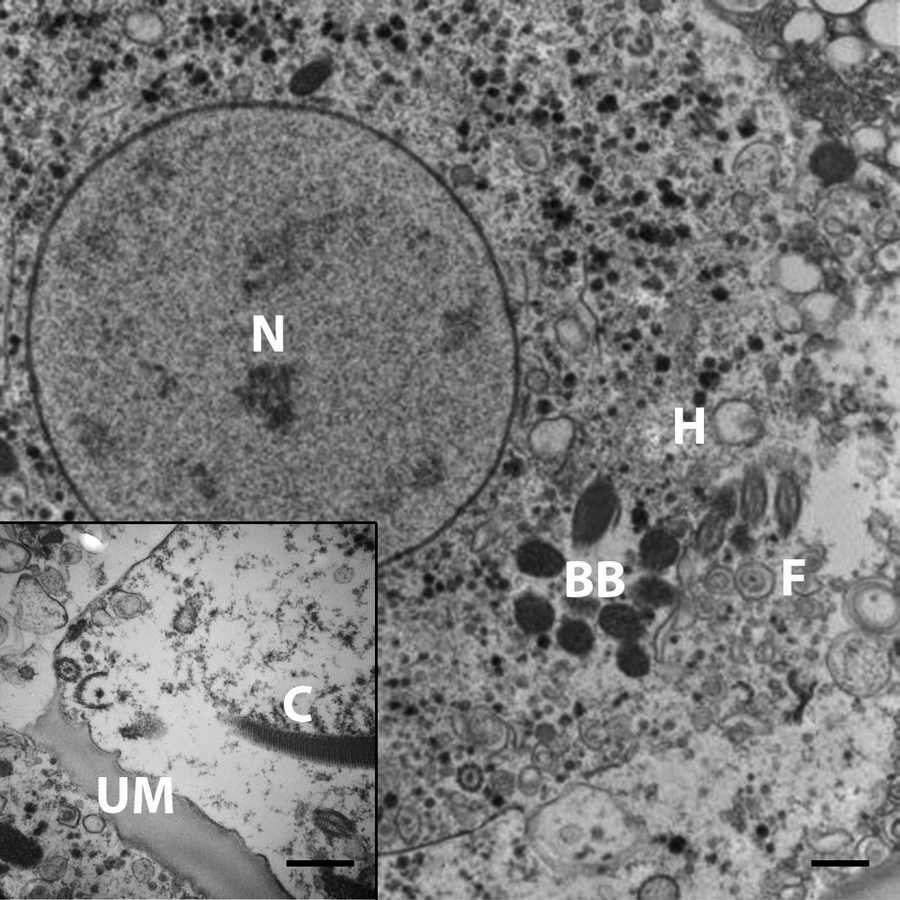
Waldrapp ibis (Geronticus eremita); liver. The protozoa exhibit several organelles consistent with trichomonads. N = nucleus; BB = basal body; F = flagella; H = hydrogenosome; C = costa; UM = undulating membrane. Electron microscopy. Bars = 500 nm.
DNA was extracted from the liver (2 independent 5 × 5 mm biopsies) using extraction kits per the manufacturer’s instructions for tissue samples. f DNA amplification of the first ribosomal internal transcribed spacer region (ITS1), 5.8S ribosomal RNA, and ITS2 regions was performed using trichomonad-specific primers TFR1 (5′-TGCTTCAGTT CAGCGGGTCTTCC-3′) and TFR2 (5′-CGGTAGGTGA ACCTGCCGTTGG-3′). 12 The PCR reactions included 5 µl of DNA in a 25-µl reaction containing 10 mM Tris-Cl (pH 9.0), 50 mM KCl, 0.01% Triton X-100, g 1.5 mM MgCl2, 0.2 mM of each deoxyribonucleotide triphosphate, 0.75 U GoTaq Flexi DNA polymerase, h and 0.5 µM of primers TFR1 and TFR2. h Cycling parameters for the amplification were 94°C for 1 min followed by 35 cycles of 94°C for 30 sec, 66°C for 30 sec, and 72°C for 2 min, and a final extension at 72°C for 5 min. A negative water control was extracted at the same time as tissues, and an individual water control was included with each PCR reaction. Amplicons were purified from a 1.8% agarose gel using a gel extraction kit per the manufacturer’s instructions f and bidirectionally sequenced at the Integrated Biotechnology Laboratories, The University of Georgia, Athens, Georgia. Sequences were analyzed with Sequencher 5.0, i and along with sequences obtained from GenBank, were aligned; phylogenetic analyses were conducted using the MEGA (Molecular Evolutionary Genetics Analysis) version 5.05 program. 21 Both neighbor-joining analysis using the Kimura 2-parameter model and maximum parsimony using a heuristic search were conducted.
Both sequences from the ibis were identical, and were most similar (97%) to oral and respiratory Tetratrichomonas spp. from human beings, followed by a Tetratrichomonas spp. associated with salpingitis in Pekin ducks (Anas platyrhynchos) from California (96.4%), and several T. gallinarum strains (96.4–97%). 7 Phylogenetically, the ibis Tetratrichomonas sp. from the current study was included in a group of T. gallinarum species and a Tetratrichomonas sp. reported from human beings (Fig. 5).
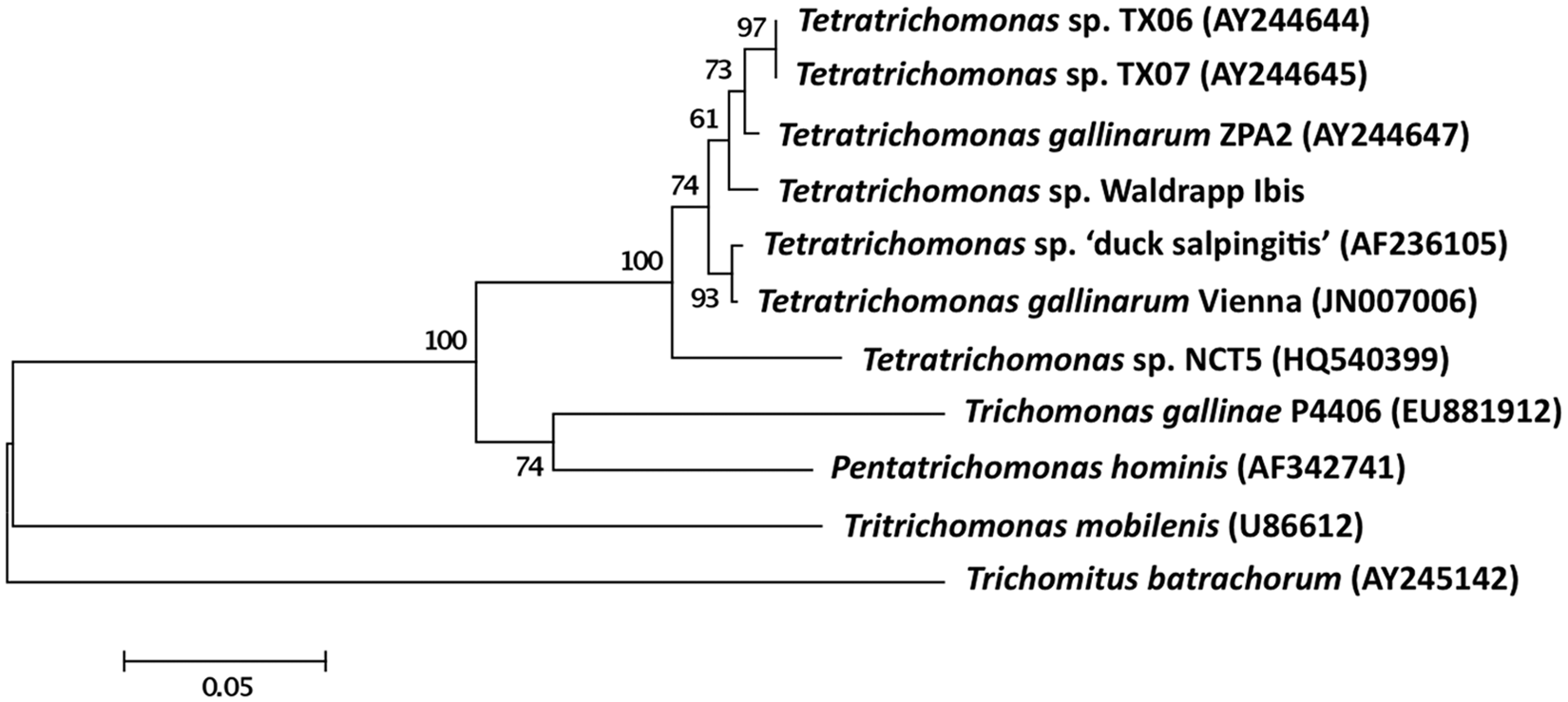
Phylogenetic analysis of trichomonad first and second ribosomal internal transcribed spacer region (ITS1, ITS2). The isolate from the affected Waldrapp ibis (Geronticus eremita) was most closely related to Tetratrichomonas spp. from human beings followed by one isolated from Pekin ducks (Anas platyrhynchos) with salpingitis. Bar = 0.05 nucleotide changes. The values at the nodes are bootstrap values and were derived from 1,000 bootstrap replicates and expressed as percentages.
Severe necrotizing hepatitis associated with tetratrichomonads was diagnosed as the cause of death of this juvenile Waldrapp ibis. Histologically, the protozoa were not initially diagnosed as trichomonads as they lacked apparent flagella, were highly variable in size, and numerous organisms were multinucleated. However, a polyclonal antibody cocktail raised against T. foetus, a related parasite of cattle, reacted strongly with the protozoa, and subsequent ultrastructural and genomic analyses were used to confirm infection with a T. gallinarum–like parasite. The presence of multinucleate, amoeboid forms of trichomonads have previously been reported in human tissues, and it is likely that the organisms in the present case underwent similar morphologic alterations.10,11
Protozoan organisms were not associated with the granuloma of the coelomic wall, and the presence of a thick fibrous capsule is consistent with a more chronic course than the hepatitis. Trauma, such as a penetrating foreign body, is considered the most likely cause of this lesion, and it is possible that it led to debilitation, which allowed the tetratrichomonad infection to progress. However, the route of infection in this case remains elusive. Typically, T. gallinarum is expected to be acquired by ingestion, cause necrotizing typhlitis or colitis, and to gain access to the liver via the portal circulation. 2 Multiple representative sections of the alimentary tract of the bird failed to reveal any protozoa, and there was minimal intestinal inflammation. Despite these findings, enterohepatic spread remains the most likely route of infection as a focal region of mucosal invasion may have been missed in sectioning, or intestinal lesions may have resolved by the time of necropsy.
Organisms that have historically been identified as T. gallinarum are likely to represent a complex of closely related strains or cryptic species. 6 Such strains have been associated with differing host ranges and virulence in a 2005 study, and reports regarding the pathogenicity and life cycle of “T. gallinarum” should be interpreted with caution. 6 However, experimental infections in poultry suggest that T. gallinarum has a rapid reproductive cycle, and a 2011 study documented infection of in-contact animals within 2 days.3,13 Direct fecal examination from Waldrapp ibis and other exhibit animals co-housed with the affected juvenile did not reveal current infection with T. gallinarum or other flagellates, although the large number of birds in the exhibit precluded testing of all of the animals. This may suggest that tetratrichomonad flagellates are not normal fauna of the Waldrapp ibis gastrointestinal tract, and raises the possibility of cross-species transmission. The presence of species subclinically infected with T. gallinarum was recently proposed as a possible source of infection of fatally affected sea ducks in a zoological park. 20
The death of the Waldrapp ibis described herein appears to have been a sporadic case of tetratrichomoniasis, likely associated with prior debilitation of the bird. Transmission from an in-contact species cannot be proven, but further studies are warranted to advise appropriate housing of this critically endangered species. A greater understanding of the ecology and pathogenesis of the T. gallinarum complex may also be vital for future screening of captive populations and existing wild populations prior to reintroduction efforts. Anti–T. foetus antibodies and immunohistochemistry, as well as molecular diagnostics, may be useful screening tools for such an endeavor.
Footnotes
Acknowledgements
The authors would like to acknowledge the assistance of Bob Nordhausen in the preparation of thin sections and interpretation of the ultrastructure of the parasites.
Notes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) declared that they received no financial support for their research and/or authorship of this article.
