Abstract
Pasteurella multocida is a common pathogen of swine that causes specific diseases with great economic impact. However, the importance of this pathogen in wild boar is still unknown. In the current work, an outbreak of systemic pasteurellosis in wild boar with a high mortality rate is described. A total of 23 wild boar of all ages were found dead over a 5-day period on a game estate in southwest Spain (11.11% mortality). Three animals were necropsied and showed subcutaneous edema, a generalized congestion, and fibrin deposits in the peritoneal cavity. Hemorrhages, general congestion, and intravascular thrombosis were microscopically observed. Pasteurella multocida type B was isolated from all of the studied organs. Outbreaks of systemic pasteurellosis have been described in domestic pigs from Asia and Australia, but not to date in Europe. This outbreak suggests that systemic pasteurellosis affecting wild boar populations may be an important cause of mortality.
Pasteurella multocida is a nonmotile Gram-negative rod or coccobacillus with 5 recognized capsular serovars: A, B, D, E, and F. 11 The organism infects a wide range of animal hosts causing important diseases such as hemorrhagic septicemia in cattle, fowl cholera in poultry, and snuffles in rabbit. 10
In pigs, the main diseases produced by P. multocida are progressive atrophic rhinitis (PAR) and pneumonic pasteurellosis, although sporadic outbreaks of fatal, acute septicemia attributed to P. multocida have also been registered in pigs from several countries in Asia and Australia.7,8,15 Progressive atrophic rhinitis is produced by toxigenic P. multocida and mainly occurs in intensive farms, affecting animals younger than 3 months with sneezing, nasal and ocular discharges, and low growing rates. 5
Pneumonic pasteurellosis is one of the most widespread diseases of pigs causing great economic losses. 11 Pasteurella multocida is rarely a primary agent of pneumonia in pigs, and coinfections with other respiratory disease agents such as Mycoplasma hyopneumoniae or Pseudorabies virus (Suid herpesvirus 1) are common in swine pneumonic pasteurellosis outbreaks. 11 This syndrome mainly affects finishing pigs resulting in coughing, intermittent fever, anorexia, and depression. In acute cases, P. multocida may produce abscesses and pleuritis with more severe symptoms such as cyanosis or sudden death. 9
The domestic pig and the wild boar belong to the same species (Sus scrofa) and are thus likely to be susceptible to the same pathogens. 12 The presence of P. multocida in wild boar has been related with cases of PAR in young animals from Germany. 13 However, to the authors’ knowledge, outbreaks of pneumonic pasteurellosis or systemic pasteurellosis have not been described in wild boar. The current study describes an outbreak of systemic pasteurellosis in a wild boar population from southwest Spain, including epidemiological, pathological, and microbiological features of the outbreak.
The outbreak occurred in a wild boar population located in Villar del Rey (northeast Badajoz Province, Extremadura, southwest Spain). This area has a continental thermo-Mediterranean climate, with hot dry summers and mild and moderately wet winters. The vegetation consists mainly of scrubland (genus Cistus; e.g., C. ladanifer) and evergreen cork oak forests (Quercus suber).
The home range of this population is surrounded by a fence that encloses approximately 1,000 ha where wild boar share the habitat with red deer (Cervus elaphus), fallow deer (Dama dama), and mouflon (Ovis musimon). Supplementary food (mainly corn) is provided to the wild boar every 3 days across the year in 4 feeders strategically distributed throughout the estate. When the outbreak occurred, approximately 207 wild boar were living on the estate. The total number of animals (July 2012 census) was estimated by counting simultaneously the number of wild boar eating at each of the 4 feeders. The sex and age of counted animals are detailed in Table 1.
Number of animals found daily during the outbreak, and final mortality rates.*
Animals counted in the last census (July 2012) are shown in the “Live animals” column. Mortality rates are calculated for adult males, adult females, and nonadult animals. Day 1 was September 23, 2012.
In the week beginning September 23, 2012, a total of 23 wild boar presenting a profuse epistaxis were found dead, close to the feeders, by the farm workers. The number of dead animals found each day is shown in Table 1. No clinical signs were observed, and no important health problems had been detected on the farm prior to the deaths.
Three of the dead animals were studied. A postmortem examination was performed, and multiple tissue samples were processed for histology, following standard procedures, and stained with hematoxylin and eosin. a Tissue samples from liver, spleen, kidney, and lung, collected during necropsy, were cultured on blood agar, b chocolate agar, b and MacConkey agar. c The plates were incubated for at least 24 hr at 37°C in aerobic conditions. Isolates obtained were identified using standard methods for phenotypic characterization. 1 Isolates identified as P. multocida were confirmed and serotyped using a specific multiplex polymerase chain reaction (PCR) 14 with a commercial Taq enzyme. d The DNA from the isolates was extracted using a commercial kit. e The PCR products were separated by 1% agarose gel electrophoresis and visualized by ethidium bromide staining under ultraviolet light. The P. multocida isolates were genotyped using enterobacterial repetitive insertion consensus–PCR (ERIC-PCR) as previously described. 16 The antimicrobial susceptibility of the P. multocida isolates (ampicillin, amoxicillin, tetracycline, enrofloxacin, gentamicin, chloramphenicol, and trimethoprim–sulfamethoxazole) was tested using a standardized disk diffusion method. 4
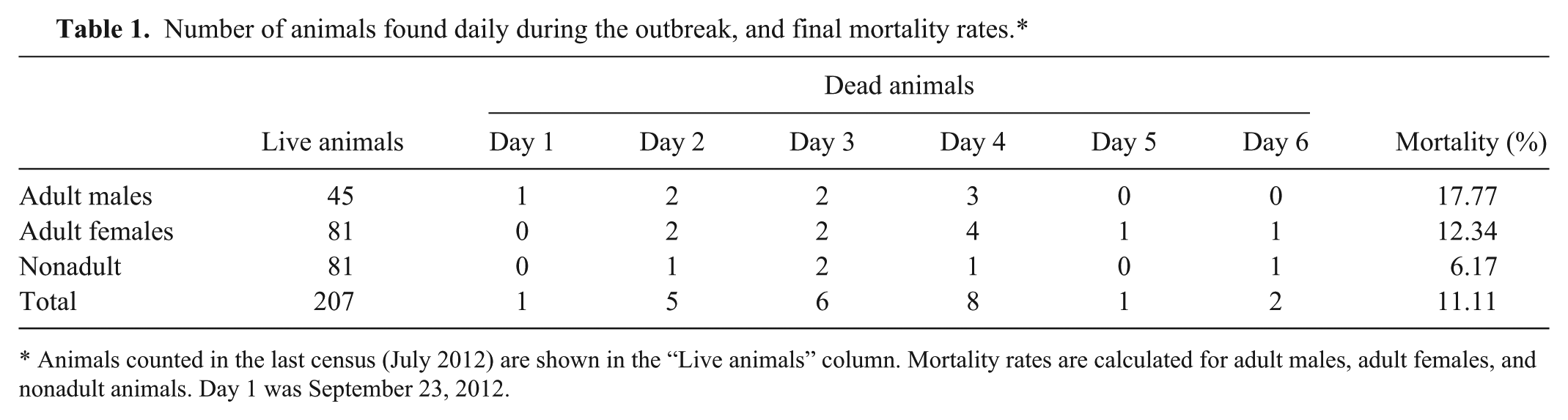
The 3 animals under study (2 females approximately 5 and 3 years old, and 1 male approximately 1.5 years old) showed similar gross lesions. The animals showed good body condition with thick subcutaneous fat layer, subcutaneous edema in low areas of the neck (Fig. 1A), generalized congestion and hemorrhages in all organs, and a great amount of fibrin in the peritoneal cavity (Fig. 1B). Microscopically, vascular damages with presence of hemorrhages, general congestion (Fig. 1C), and intravascular thrombosis (Fig. 1D) were observed in all the studied organs.
Wild boar (Sus scrofa).
A pure growth of grayish, nonhemolytic, oxidase-positive colonies consistent with P. multocida were obtained from all samples (lung, spleen, liver, kidney) of each animal, after culture on blood agar and chocolate agar (12 isolates). The MacConkey agar plates showed no bacterial growth. Microscope observation of the isolates obtained showed Gram-negative coccobacilli typical of P. multocida. The PCR products obtained showed 2 bands (460 bp and 760 bp, respectively) indicating that the 12 P. multocida isolates belonged to serovar B. The 12 isolates of P. multocida belonged to the same genotype showing the same fingerprint pattern in the ERIC-PCR (Fig. 2). Specific interpretative criteria for porcine P. multocida and the antimicrobials examined in the present study are not provided in the standardized methodology used. 4 Hence, the interpretations were done using the generic criteria. 4 As no criteria are available for amoxicillin, the criteria for ampicillin 4 were used. All isolates were found to be sensitive to all antimicrobial agents tested.
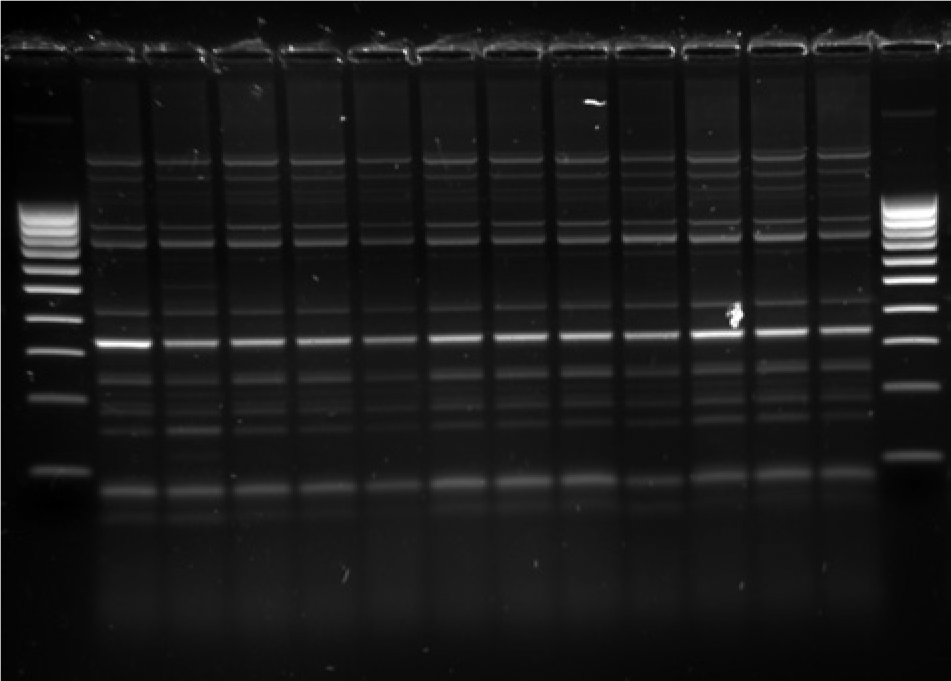
Fingerprint patterns obtained with use of enterobacterial repetitive insertion consensus sequence polymerase chain reaction on 12 Pasteurella multocida serovar B isolates.
In the current work, an outbreak of acute systemic pasteurellosis affecting wild boar has been confirmed. Septicemic disease caused by P. multocida is well recognized in cattle and buffalo. 7 In domestic pigs, sporadic outbreaks have been reported in several countries of the eastern hemisphere,2,8,9 being extremely uncommon in Europe where only 1 outbreak has been reported in Germany. 13
Pasteurella multocida has been previously isolated from wild boar and linked with a case of PAR, 6 but has not been associated with any outbreak of acute septicemia. The P. multocida isolates obtained in the current study were typed as capsular type B, the causal agent of hemorrhagic septicemia. 10 Serovar B is the most prevalent capsular serovar associated with acute systemic pasteurellosis in swine in Vietnam. 15 On the other hand, serovars D and A are the most commonly isolated serovars from pneumonia in pigs. 11
The clinical signs and lesions found in these animals were very similar to those previously described in domestic pigs: sudden deaths affecting all ages, with subcutaneous edema, generalized congestion, hemorrhages, and fibrinous deposits in peritoneal cavity. 8 In the studied outbreak, the estimated mortality rate was 11.11%. The mortality was estimated based on the number of dead wild boar found by estate workers. However, the total number of deaths could be higher because it is very difficult to recover all carcasses in a big estate and many of them could have been removed quickly by scavengers. The mortality rate in the current outbreak was in agreement with those previously reported in acute systemic pasteurellosis outbreaks described in domestic pigs, ranging from 5% to 40%.7,11
Although an outbreak of acute systemic pasteurellosis involving different species has been described in Europe, 13 the current outbreak involved no mortalities in the red deer, fallow deer, or mouflon populations, suggesting that this outbreak was limited to the wild boar population. Some wild species such as roe deer or fallow deer have been suggested as being able to be healthy carriers of P. multocida type B. 13 Thus, the presence of other wild ungulates sharing the habitat with wild boar might be a potential source of P. multocida. However, more research would be necessary to determine the primary source of P. multocida in this outbreak.
Outbreaks of systemic pasteurellosis have been commonly related with environmental factors, such as abundant precipitations. 3 In this sense, the start of this outbreak could have been favored by the extreme wet period that occurred on the estate just when the outbreak was recognized, with rainfall of 45.6 mm between September 23 and 28 (no rain had been registered in the previous 4 weeks).
A therapeutic protocol based on the addition of tetracycline (200 ppm) in food was implemented daily for 10 days, starting 3 days after the first deaths. The antimicrobial was added in the food due to the difficulty of parenteral application in this kind of species. In addition, the addition of tetracycline to food has been widely recommended as a preventive treatment to pasteurellosis. 11 No more dead animals were found 3 days after the beginning of the treatment.
Footnotes
a.
Isokit, Bio-Optica Milano SpA, Milan, Italy.
b.
Oxoid, Thermo Fisher Microbiology Division, Basingstoke, Hampshire, United Kingdom.
c.
Scharlau, Barcelona, Spain.
d.
Kapa Taq, Kapa Biosystems, Woburn, MA.
e.
QIAamp DNA Mini kit, Qiagen Ltd., Crawley, United Kingdom.
Declaration of conflicting interests
The author(s) declared that they had no conflict of interests with respect to their authorship or the publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Ministerio de Ciencia e Innovación (Gobierno de España) PS0900513, Regional Government of Extremadura (PDT09A046 and GRU10142) and FEDER. D. Risco has a Formación de Profesorado Universitario (FPU) grant from the Ministerio de Ciencia e Innovación (AP2009-0704).
