Abstract
During the 10 days commencing April 29, 2013, the Iowa State University Veterinary Diagnostic Laboratory received the first 4 of many submissions from swine farms experiencing explosive epidemics of diarrhea and vomiting affecting all ages, with 90–95% mortality in suckling pigs. Histology revealed severe atrophy of villi in all segments of the small intestines with occasional villus-epithelial syncytial cells, but testing for rotaviruses and Transmissible gastroenteritis virus (
Keywords
During the 10 days beginning on April 29, 2013, the Iowa State University Veterinary Diagnostic Laboratory (ISUVDL; Ames, Iowa) received the first 4 of many similar submissions from swine farms experiencing explosive epidemics of diarrheal disease and vomiting affecting all ages, with high mortality in suckling pigs. Farm 1 was a 2,400-sow breed-to-wean farm in central Iowa on which the referring veterinarian reported, beginning on April 28, watery fetid diarrhea in 90% of piglets in 4 farrowing rooms that, by the following day (April 29), had spread to all farrowing, breeding, and gestation rooms. All piglets within 24 hr of birth developed watery yellow fetid diarrhea that contained flocculent undigested milk and many were observed vomiting. Piglets dehydrated rapidly, were covered with feces, and mortality was 95% within 2–3 days. Ninety percent of sows in farrowing rooms, 90% of gilts in breeding and gestation rooms, and approximately 15% of parity 2 and older sows in breeding and gestation rooms developed diarrhea and were anorectic. Some vomited. Based on clinical disease consistent with an epidemic of transmissible gastroenteritis (TGE), feedback of pooled diarrheic feces and homogenized intestines from suckling pigs (FB) was initiated to all adult animals on the site. Fecal samples and acutely affected live suckling pigs of various ages were received at the ISUVDL in 3 separate submissions on April 29, May 1, and May 7. Farm 1 was a shower-in–shower-out facility without air-filtering or perimeter fencing, and management was considered excellent by the referring veterinarian. The most recent introduction of breeding gilts was more than a month earlier.
Submissions were received at ISUVDL from farms 2 and 3 on May 6. Farm 2 was a 1,200-sow breed-to-wean farm in northeastern Iowa that, on April 29, had 1 sow observed with diarrhea and vomiting. Three more sows were similarly affected by May 2, and by May 6, 1 week after it began, 60–70 gestating and lactating sows were sick along with large numbers of suckling pigs. Disease in individuals was as described in farm 1. Mortality in suckling pigs exceeded 95%. Tissue samples and feces were collected from 3-, 7-, and 14-day-old euthanized diarrheic pigs and shipped to ISUVDL on May 5. A preliminary clinical diagnosis of TGE led the attending veterinarian to recommend FB to all adult animals on the site, which was accomplished on May 6. Farm 2 was also a shower-in–shower-out site with management and biosecurity described by the submitting veterinarian as excellent.
Farm 3 was a 400-sow breed-to-wean farm in northwestern Iowa that, on May 2, broke with severe watery diarrhea and vomiting in suckling pigs in 1 farrowing room. The next day, May 3, suckling pigs in 3 more farrowing rooms were similarly affected, and 10% of lactating sows in those rooms were vomiting and anorexic. Tissue samples were collected from euthanized 3- and 14-day-old diarrheic pigs and shipped to ISUVDL on May 3. Based on a presumptive diagnosis of TGE by the attending veterinarian, FB to all adult animals was done on May 3. Mortality in suckling pigs less than 2 weeks of age was 90% and of 2–4-week-old pigs was 40%. Farm 3 was also a shower-in–shower-out site with reportedly good biosecurity; air was unfiltered, and the last introduction of gilts was 3–4 weeks prior to the outbreak; the newly introduced gilts reportedly also got sick during the epizootic that started on April 3.
Farm 4 submitted tissues from diarrheic suckling pigs to ISUVDL on May 8. Two breed-to-wean farms in the same production system in southeastern Indiana were involved. The first, a 6,000-sow farm, observed mild-to-moderate diarrhea in some suckling pigs on May 4 and the following day, May 5, sows in 1 gestation room exhibited watery green diarrhea and vomiting. By the next day, May 6, 10% of 1–3-day-old pigs and 50% of 7–14-day-old pigs likewise had watery diarrhea and were vomiting. That same day the attending veterinarian initiated FB to all adult animals based on a presumptive diagnosis of TGE. Samples of tissues from diarrheic pigs were shipped to ISUVDL on May 6 and received on May 7 to confirm TGE. On May 7, identical clinical signs were observed in suckling pigs in 2 farrowing rooms of a second 3,600-sow farm. Tissue samples from diarrheic pigs were collected and shipped to ISUVDL on May 7 and received on May 8 to confirm TGE, and FB was accomplished on April 8. Mortality in suckling pigs in both farms was 90–95%. Both farms were part of a system that has been closed to introduction of live gilts from outside for several years. The 2 Indiana farms were shower-in–shower-out, well managed, had excellent biosecurity, and used the same source of feed.
Laboratory findings were similar for all 4 farms. Complete necropsy examinations were performed on pigs received from farm 1, and gross lesions were reported by referring veterinarians for farms 2–4. Acutely affected euthanized suckling pigs had yellow feces coating the skin and hair, the stomachs contained little milk curd, the small and large intestines were distended by watery contents that contained floccules of white-to-yellow undigested milk, and small intestinal walls were thin (Fig. 1). Intestinal contents, when collected and allowed to settle, partitioned into a top layer of clear yellow watery fluid and a bottom layer of loosely aggregated white-to-light yellow flocculent ingesta. Suckling pigs that died naturally were also coated with yellow feces and were gaunt and markedly dehydrated as evidenced by sunken eyes, inelasticity of the skin, and tacky subcutaneous tissues. The gastrointestinal tract was usually empty, and the small intestinal walls were thin. Tissues from suckling pigs from all 4 farms were fixed in 10% neutral buffered formalin, and tissue sections stained with hematoxylin and eosin were prepared for microscopic examination by standard methods. Microscopic lesions were limited to the small intestines. The most acute lesions were of degenerate epithelial cells on the lateral surfaces and tips of villi (Figs. 2, 3). Cells were variably swollen with granular eosinophilic cytoplasm that sometimes contained variably sized round-to-oval light pink proteinic vacuoles. Swollen cells resulted in a brush border with a scalloped rather than a more normal linear luminal surface. Some cells were losing attachment with adjacent cells on their lateral surfaces and with the basement membrane on their basal surfaces. Syncytial epithelial cells were observed containing numerous nuclei. Villi were variably shortened with condensation of the lamina propria near the tips of villi. Lesions in other pigs, presumably slightly later in the viral infection, were observed throughout the entire length of the small intestine (Fig. 4). The villi were severely atrophic with a villus-to-crypt ratio of 1:1 or less. Tips of villi were eroded or covered by markedly attenuated epithelial cells with low cuboidal to squamous morphology. The lamina propria was condensed throughout the length of villi and often contained nuclear debris and few neutrophils.
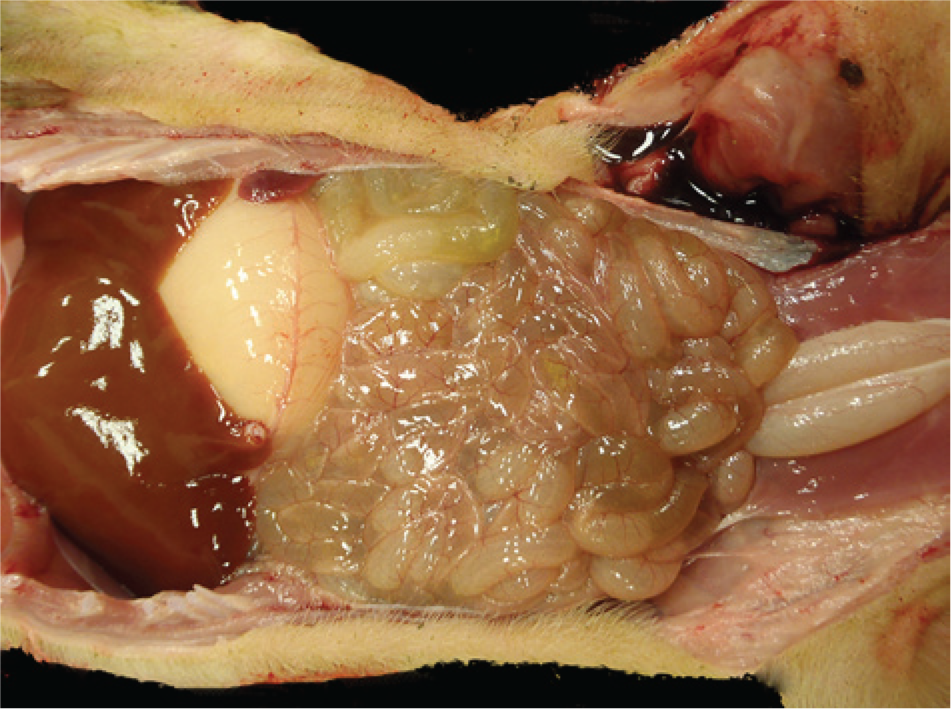
Four-day-old pig from farm 1 with acute porcine epidemic diarrhea. Small intestines are thin walled and dilated with fluid contents.
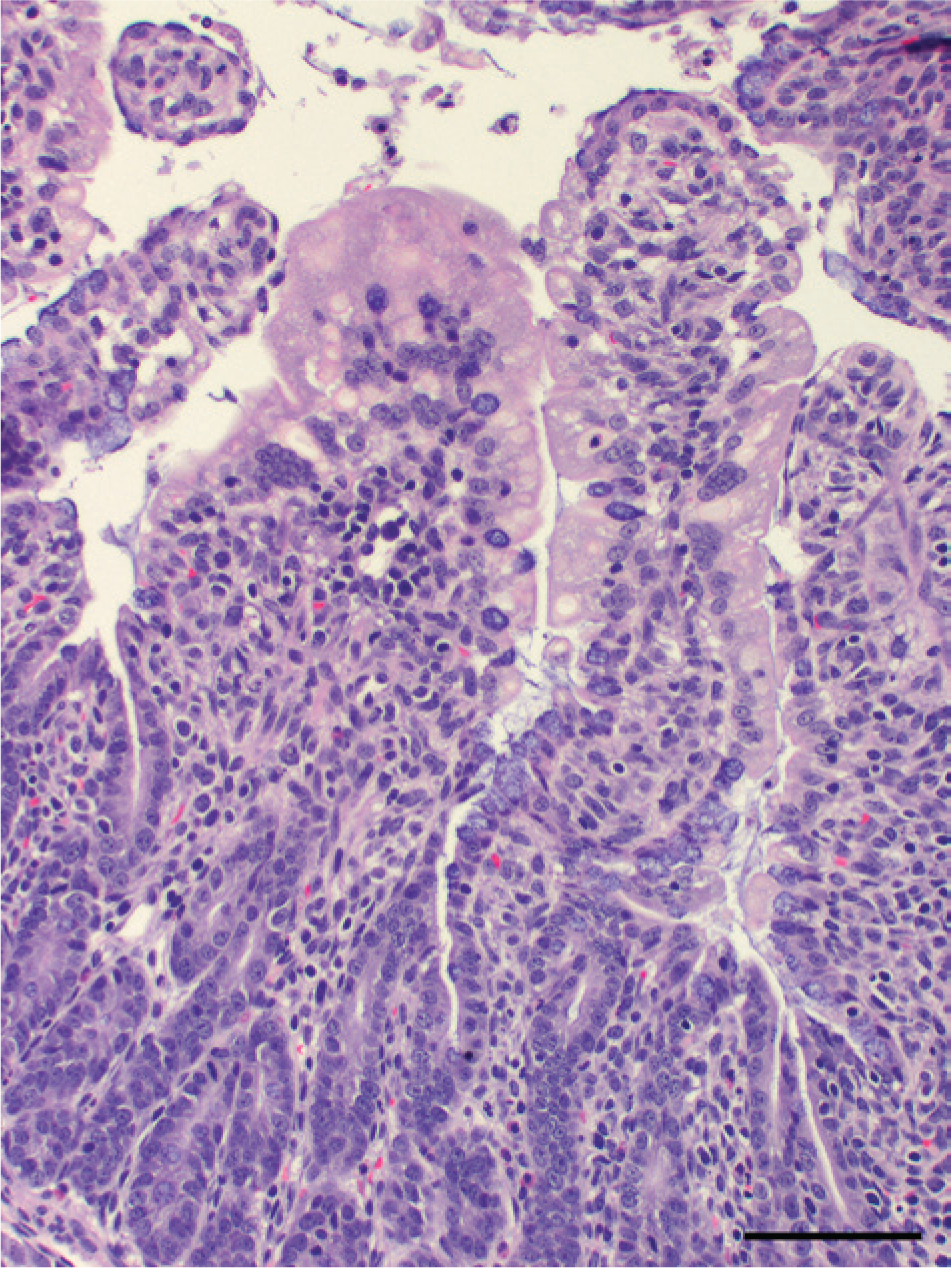
Hematoxylin and eosin–stained tissue sections of small intestines from a diarrheic 19-day-old pig from farm 4 with acute porcine epidemic diarrhea. Villus-epithelial cells are swollen with intracytoplasmic vacuoles, some are partially detached, and few form multinucleate syncytia. Bar = 200 µm.
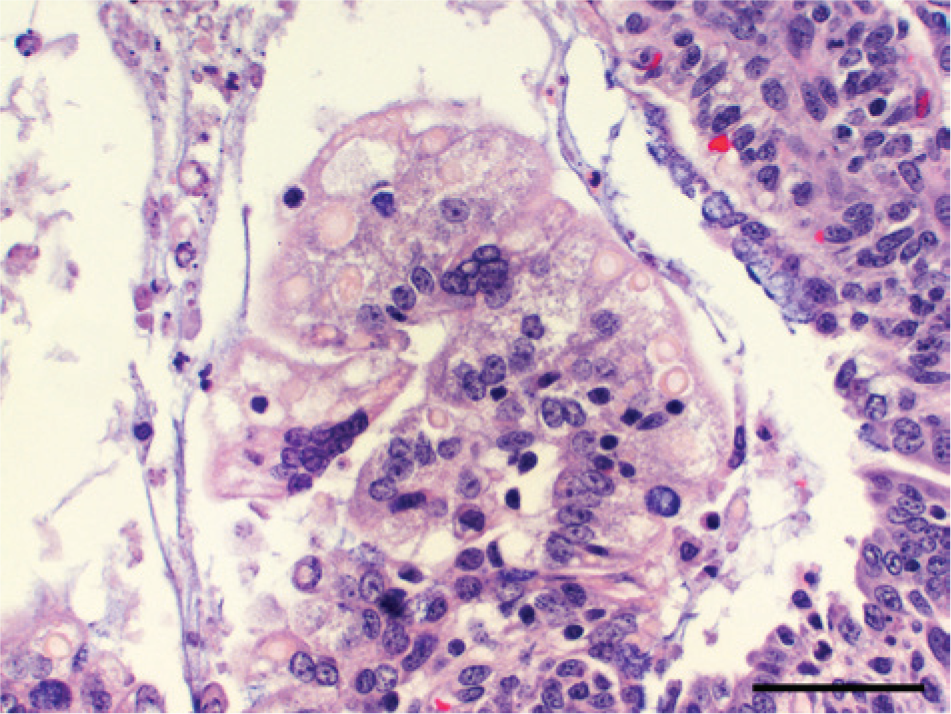
Hematoxylin and eosin–stained tissue sections of small intestines from a diarrheic 19-day-old pig from farm 4 with acute porcine epidemic diarrhea. Swollen villus-epithelial cells have granular cytoplasm that sometimes contains variably sized, round-to-oval light pink vacuoles. Few multinucleate syncytial epithelial cells are observed. Bar = 100 µm.
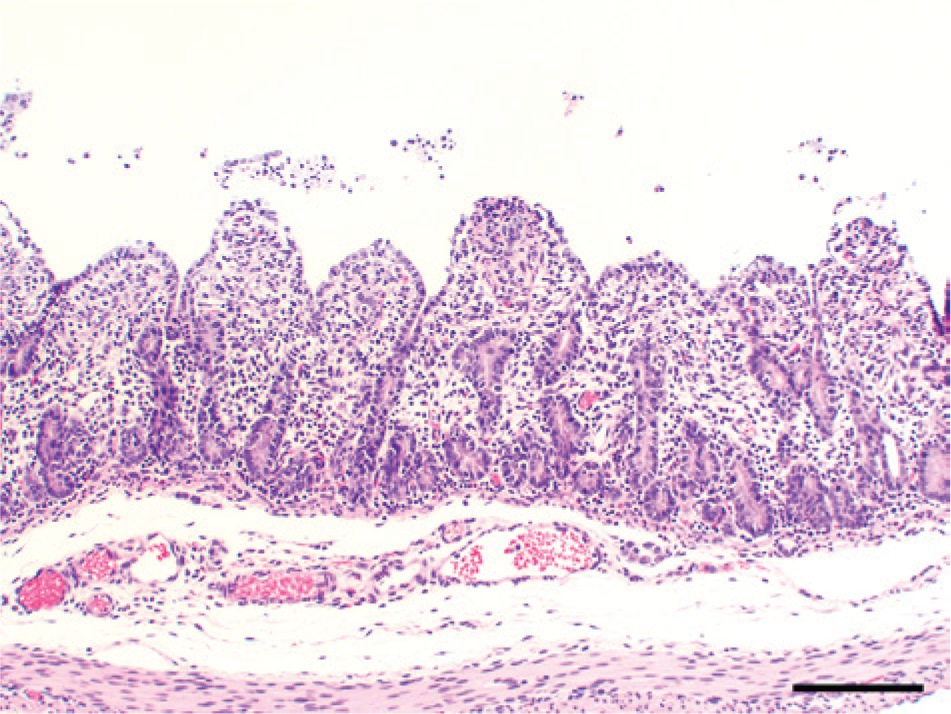
Hematoxylin and eosin–stained tissue sections of small intestines from a diarrheic 19-day-old pig from farm 4 with acute porcine epidemic diarrhea. Severe atrophy of villi (villus-to-crypt ratio of 1:1) with attenuation or erosion of epithelial cells on tips of villi and condensation of the villus lamina propria. Bar = 300 µm.
Although lesions were typical of acute viral enteritis, and severity of lesions as well as epidemic manifestation within herds suggested TGE, feces and/or intestinal contents (FCIC) were negative for TGE virus (TGEV;
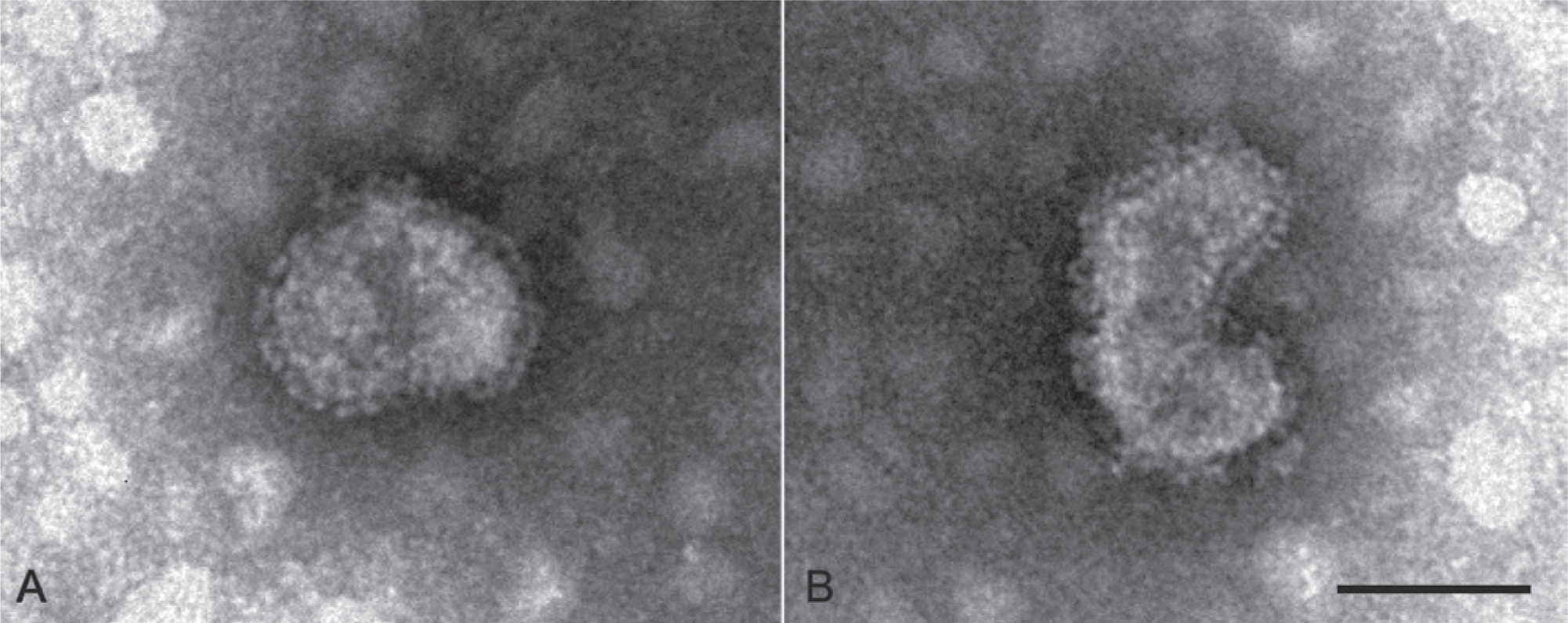
Negative-staining electron microscopy of pooled diarrheic feces of suckling pigs from farm 1. Enveloped pleomorphic virions with club-like spikes protruding from the envelope typical of coronaviruses. Bar = 100 nm.
Identification of a coronavirus that could not be detected by TGEV and/or PRCV PCR prompted further testing. Fecal samples were made to 10% (v/v) homogenates in 0.01 M phosphate buffered saline (pH 7.2). After centrifugation at 3,500 ×
As PEDV had not been reported in the United States, all fecal samples were submitted to National Veterinary Services Laboratories (NVSL; Ames, Iowa) for corroborative testing. Three nested RT-PCR tests designed to detect 2 different regions of the S-gene and 1 of the N-gene of PEDV (Warg JJ: 1998, The development of a reverse transcriptase-polymerase chain reaction (RT-PCR) for diagnosis of porcine epidemic diarrhea virus. Master’s thesis, Iowa State University, Parks Library General Collection, ISU 1998 W395) were performed. All samples were positive for both target genes, and all PCR products were proven to be of PEDV by sequencing, confirming the presence of PEDV in the tested samples. All PCR products from all viruses shared 100% sequence identity with each other.
The PEDV strains in the samples from farm 1 and farm 4 were then sequenced for the whole genome using a next-generation sequencing technique d as per recommended procedure by the manufacturer. The viruses shared 99.9% sequence identity with each other and 96.6–99.5% identity with known PEDV strains whose full-length sequences were available in GenBank (http://www.ncbi.nlm.nih.gov/genbank/). The 2013 U.S. PEDV (GenBank accession nos. KF452322 and KF452323) showed a sequence identity higher than 99.0% to some Chinese PEDV strains reported in 2011–2012 than to older strains, with AH2012 strain of PEDV (GenBank accession no. KC210145) being the closest one at the time of sequence comparison (Fig. 6).

Phylogenetic relationship of
Immunohistochemical staining 2 using a monoclonal antibody specific for PEDV e revealed abundant viral antigen in the cytoplasm of epithelial cells predominantly on villi and also few in crypts in all segments of the small intestines (Fig. 7). Additionally, few epithelial cells were positive in the colon on or near the luminal surface (Fig. 8).
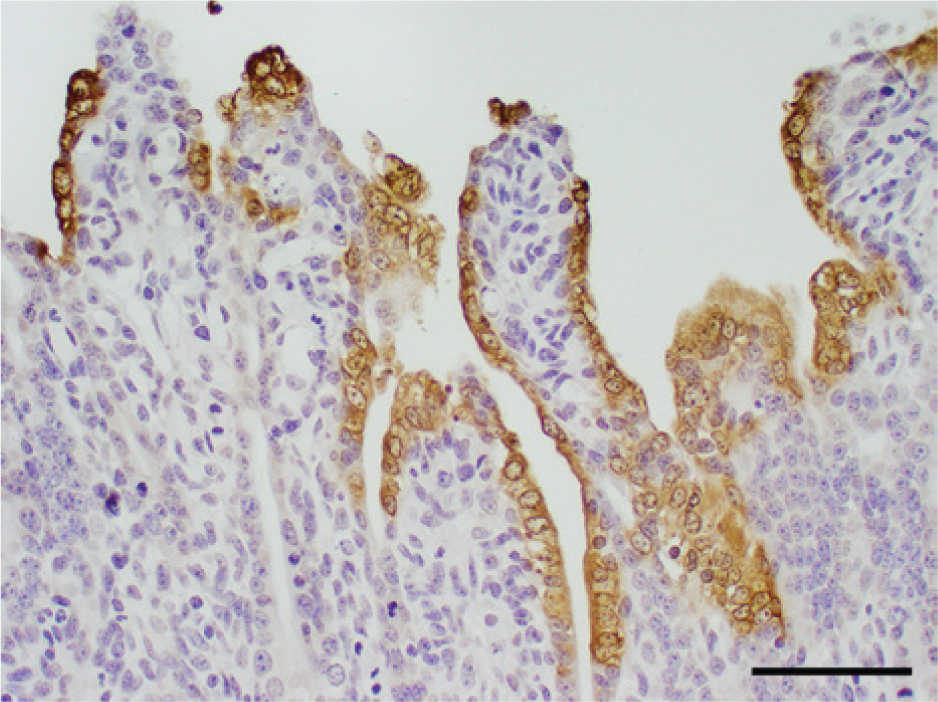
Immunohistochemical staining for
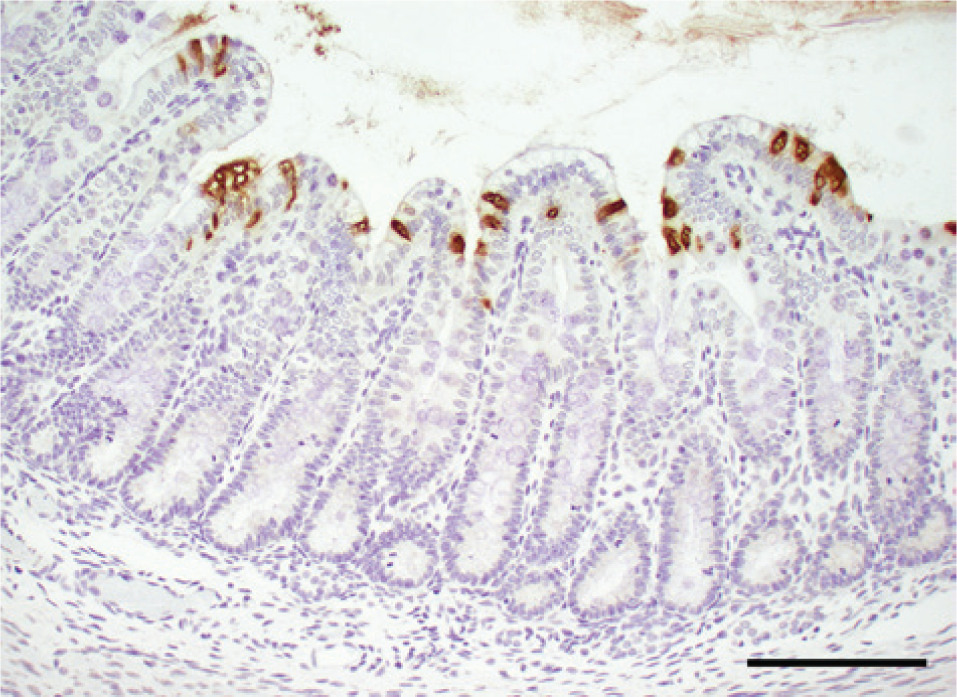
Immunohistochemical staining for
The clinical disease on each farm spread rapidly, affecting all ages with diarrhea and variable vomiting. Mortality was high only in suckling piglets, which was consistent with previous reports of epidemics of PED in naïve populations in Japan, Korea, Thailand, and China. 6 Clinical disease and lesions were indistinguishable from epidemic TGE. Porcine epidemic diarrhea was suspected when PCR and immunohistochemical staining for TGEV were negative and coronavirus-like particles were observed in feces by negative-staining EM. The availability of multiple testing formats and reagents at ISUVDL, the cooperation and expertise of NVSL, as well as the cooperation of submitting veterinarians who provided proper samples, were essential for the initial diagnosis as well as subsequent corroboration and full genome sequencing. The emergence of PEDV in the United States reiterates the importance of preparedness and also stresses the need for the development of better and more comprehensive disease surveillance strategies for transboundary and foreign animal diseases. Rapid recognition of the emergence of a new disease is essential to minimize economic losses to the U.S. livestock industry.
The 4 farms were separated by a hundred or more miles and, based on discussions with submitting veterinarians, shared no personnel, service personnel, feed mills, trucks or trucking companies, seed stock suppliers, or veterinarians. Management and biosecurity was rated as excellent by referring veterinarians. Outbreaks of disease within a few days on 4 apparently unrelated and geographically separated sow farms caused by highly related PEDV strains is perplexing, but suggests a common viral source. Furthermore, the PEDV strains are nearly identical to recently reported Chinese strains (2011–2012). Epidemiological studies are needed, including retrospective studies, to identify the source to the farms and any other PED cases prior to the present case report. Because PEDV is new to U.S. swine, animal studies to confirm biological characteristics of host-virus relationships, virological studies to characterize physicochemical properties with this particular PEDV, and methods of surveillance to monitor disease outbreaks and virus spread will be important for implementing control strategies.
Footnotes
Acknowledgements
The authors thank Kerrie Franzen at National Veterinary Services Laboratories (Diagnostic Virology Laboratory) and Judith Stasko at National Animal Disease Center for technical assistance on next-generation sequencing and electron microscopy, respectively. The authors are also grateful to staff of Molecular and Viral Diagnostics Section and Joe Brodie at Histopathology Section of the Iowa State University Veterinary Diagnostic Laboratory for technical assistance during diagnostic investigations. Submitting veterinarians are acknowledged for their extra efforts in supplying clinical and herd histories and for being patient and understanding during the investigation.
a.
TGE monoclonal antibody, Linda Saif, Ohio Agricultural Research and Development Center, Ohio State University, Wooster, OH.
b.
QIAquick Gel Extraction kit, Qiagen Inc., Valencia, CA.
c.
Lasergene, DNASTAR, Madson, WI.
d.
Ion Torrent, Life Technologies, Grand Island, NY.
e.
BioNote, Hwaseong-si, Gyeonggi-do, Korea.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
