Abstract
A 22-month-old, female red deer (Cervus elaphus) was submitted to the University of Minnesota Veterinary Diagnostic Laboratory for necropsy and chronic wasting disease (CWD) testing. The deer was found positive for the abnormal prion protein in the obex and the retropharyngeal lymph node by immunohistochemical staining. Microscopic lesions of spongiform encephalopathy and immunohistochemical staining patterns and intensity were similar to those in CWD-positive elk and experimentally infected red deer.
Chronic wasting disease (CWD) is a fatal, neurodegenerative disease of cervids caused by the accumulation of a misfolded prion protein in brain, and is classified as a transmissible spongiform encephalopathy (TSE) along with other diseases such as bovine spongiform encephalopathy (BSE, mad cow disease), scrapie, and transmissible mink encephalopathy. The disease was first recognized in captive mule deer (Odocoileus hemionus) in 1967, in captive Rocky Mountain elk (or American elk; Cervus elaphus nelsoni) in 1979, and in wild moose (Alces alces shirasi) in 2005.1,10 In 2010, 5 red deer–sika deer cross-breeds in the Republic of Korea were found to be CWD positive; the disease was traced to accidental introduction of infected elk imported from Canada in 1994 and 1997 (Sohn HJ, Lee YH, Kim MJ, et al.: 2011, Chronic wasting disease (CWD) outbreaks and surveillance program in the Republic of Korea. Prion Pre-Conference Workshop, Montreal, Ontario, Canada). Chronic wasting disease has been reproduced experimentally by intracerebral and oral inoculation of red deer.2,6
A deceased, 22-month-old, female red deer (Cervus elaphus) was submitted to the University of Minnesota Veterinary Diagnostic Laboratory (St. Paul, Minnesota) for necropsy and CWD testing on May 10, 2012. The deer was part of a 500-head, captive herd located in the city of North Oaks, Ramsey County, Minnesota, a northern suburb of St. Paul. According to farm staff, the animal was found recumbent on the morning of May 10, 2012 within the outdoor enclosure. The deer was able to rise when approached but stayed removed from the herd and was found dead later that afternoon. None of the deer’s herd mates appeared to be ill.
Upon gross examination, the red deer was found to be in good nutritional and postmortem condition. There were multifocal, mild, pulmonary hemorrhages, and congestion. A single, complete tape worm was found in the small intestine. The hind (female red deer) was pregnant with a near-term male fetus. The uterus contained copious, cloudy yellow fluid. The cotyledons and caruncles were prominent and contained fibrin, catarrhal exudate, and/or mineralization. The fetal stomach contents were also cloudy and yellow and had flecks of white debris (fibrin, purulent exudate, and/or mineralization). No bacteria were isolated from tissues, uterine swab, or fetal stomach contents. The reverse transcription polymerase chain reaction for Bovine viral diarrhea virusa,b was also negative on a tissue pool. Fecal floatation for endoparasites revealed a subclinical level of Coccidia sp., Moniezia sp., and Strongyloides sp. oocysts.
Formalin-fixed tissue samples were paraffin embedded, sectioned at 5 µm, and stained with hematoxylin and eosin (HE). The placenta had multifocal endometrial gland dilation with extensive mucus and fibrin. The spleen contained mild, multifocal hemosiderosis. The lung contained multifocal hemorrhage and congestion. There was diffusely widespread spongiform degeneration of the brainstem neuropil with occasional neuronal degeneration and intracytoplasmic vacuolation. Spongiform degeneration of gray matter neuropil was diffusely spread throughout the cerebrum and cerebellum (Fig. 1A). In the cerebral cortex, vacuoles were present in the neuropil but not distinctly associated with neurons. In the cerebellum, intracytoplasmic vacuolations were detected but not exclusively affecting Purkinje cells. Vacuoles were seen in Purkinje cell processes, and numerous, necrotic Purkinje cells were present (Fig. 1B). Vacuoles were particularly common in the molecular and granular layers.
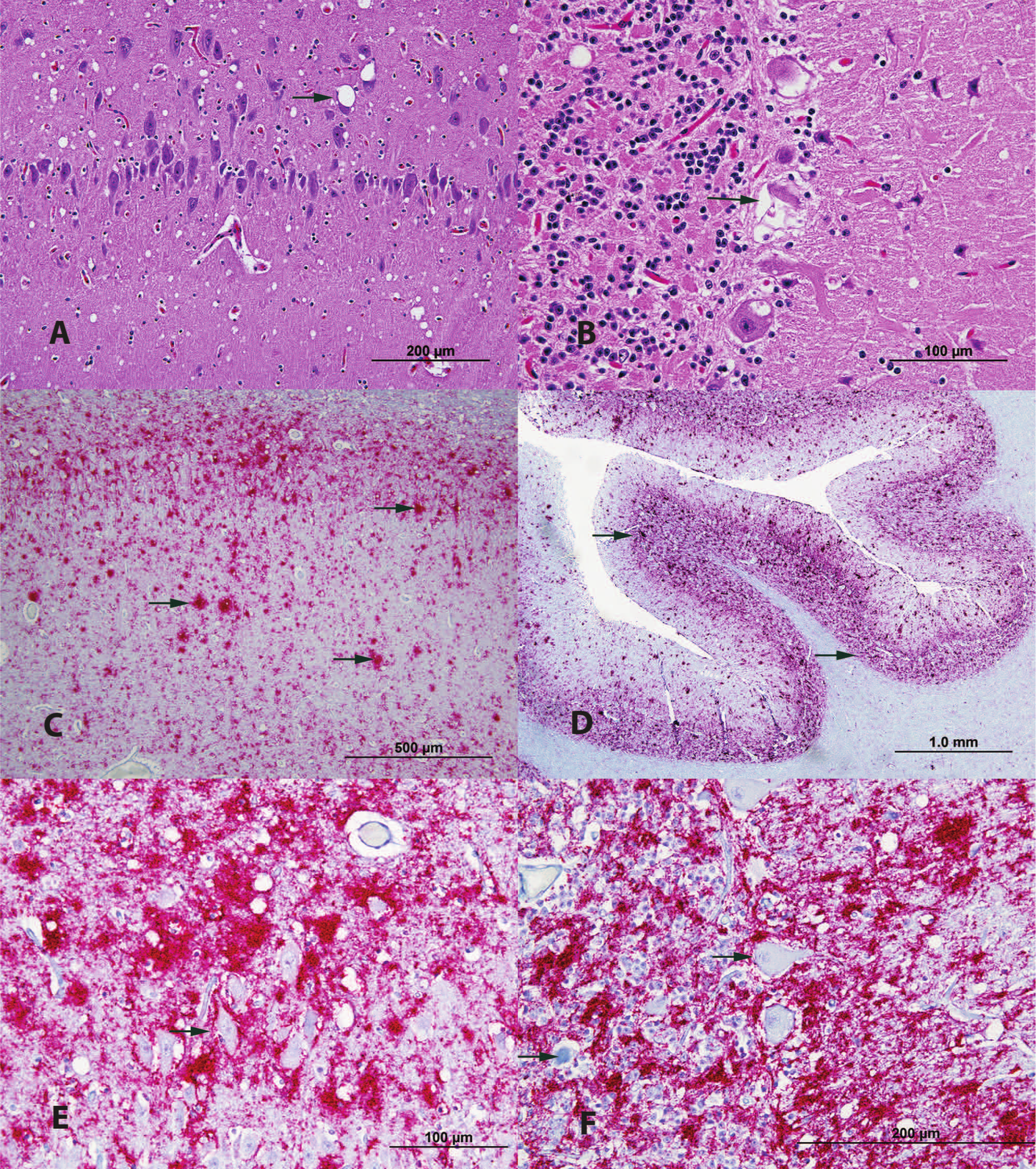
Red deer (Cervus elaphus) with chronic wasting disease.
Brain sections were histochemically stained by hand with Bielschowsky silver, Congo red, and periodic acid–Schiff (PAS) and were immunohistochemical stained by hand for calbindin Dk28 and glial fibrillar acidic protein. Immunohistochemical staining was performed on sections of obex, retropharyngeal lymph nodes (RPLN), and all formalin-fixed tissue sections using protease-resistant prion protein (PrPres) antibodies (clone 99/97.6.1) at 8 µg/ml in an automated immunostainer. c Bielschowsky silver, Congo red, and PAS failed to reveal plaques and amyloid deposition. Calbindin-positive Purkinje cell bodies appeared to have some association with neuropil vacuolization seen with HE staining, but not all vacuoles were associated with Purkinje cells processes. The loss of normal architecture was apparent in necrotic and/or vacuolated Purkinje cells. Glial fibrillar acidic protein staining revealed a mild increase of reactive astrocytes within the capsula interna, the parahippocampal gyrus, and the white matter of the cerebellar cortex. This increase of reactive astrocytes was not appreciated by HE staining. The RPLN was positive for PrPres by immunohistochemical staining. Positive PrPres immunohistochemical staining of additional lymphoid tissues was noted in the spleen, a peripheral lymph node of unknown location, Peyer patches of the small intestine, and the colon wall. Neural tissue within the wall of the small intestine also stained PrPres positive by immunohistochemical staining. Positive PrPres immunohistochemical staining within the brain was characterized by a diffuse pattern mixed with multicentric, granular plaques, mainly associated with the gray matter of the cerebrum and cerebellum and the mesencephalon, including the brainstem, but was also present in the white matter of the cerebrum and cerebellum (Fig. 1C, 1D). Dense PrPres staining was observed in the dorsal motor vagus nuclei and surrounding nuclei. The presence of PrPres plaques seemed not to necessarily correlate with the presence of vacuoles (Fig. 1E, 1F). Cerebral staining was very strong in the cortex of the temporal region and the hippocampus. The cerebellar cortex granular layer and the pedunculus presented very strong PrPres immunohistochemical staining. The immunohistochemical results were confirmed on obex and RPLN at the National Veterinary Services Laboratories (NVSL; U.S. Department of Agriculture, Ames, Iowa).
Histological examination of nervous lesions in multiple TSEs convey some variability but are morphologically similar in many ways. 5 The microscopic characteristics of encephalopathy found in red deer described herein closely relate to those found in CWD-positive elk in natural and experimental settings and CWD-positive red deer in experimental settings, although inter- and intraspecies variation in the neuroanatomic distribution and severity of histological lesions associated with CWD prion infection have been reported.2,5,6,7,10,11 Cerebellar astrocytosis and vacuolation in the molecular layer and intracytoplasmic vacuolation of Purkinje cells were demonstrated in this red deer; similar lesions are common in elk and experimentally infected red deer but not in other cervid species.1,2,6,9,11 Neuronal degeneration and widespread vacuolation within the brainstem, cerebellum, and cerebrum is typical in cervids with CWD and was observed in this red deer.1,2,5,6,11 Distribution of PrPres within the brain does not necessarily correspond to areas of vacuolation as has been reported in CWD-infected cervids.2,6,8 Chronic wasting disease–affected white-tailed (Odocoileus virginianus) and mule deer have been reported to have amyloid plaque accumulation within the gray matter, which can be appreciated with HE staining and strongly expressed by Congo red and PAS staining. 11 These plaques were not seen in this red deer nor are they normally seen in naturally and experimentally infected elk or experimentally infected red deer with CWD.2,5,6
The positive immunohistochemical staining in multiple lymphoid tissues and peripheral nervous tissue is consistent with that of CWD-positive, orally inoculated red deer in experimental studies.2,6 This also suggests that neither BSE prion nor scrapie prion is the source of infection in the red deer described herein because experimentally BSE-infected red deer and scrapie-infected elk lacked positive PrPres immunohistochemical staining in those tissues.3,4,6 The staining patterns observed within the obex were analogous to those described in severe cases of CWD in other cervid species.7,9 The intensity and location of staining within the brain is consistent with orally exposed CWD-positive red deer in experimental settings; however, differences were observed within and adjacent to neurons.2,6 Neurons and Purkinje cells did not directly take up stain (Fig. 1E, 1F) in the red deer of the present case report unlike the observed fine punctate to granular deposits within the cytoplasm of neurons in experimentally infected red deer.2,6 The perineuronal staining patterns observed in experimentally infected red deer were not as remarkable in the red deer of the current case report because of the overall density of positive staining.2,6
Histological and immunohistochemical observations strongly indicate a CWD prion infection in the red deer of the current case report. Further testing at NVSL revealed brain tissue to be positive for a TSE consistent with CWD by Western blot d and enzyme-linked immunosorbent assay. e Genetic characterization of this prion will be presented in a separate publication. The impact of the CWD-induced brain lesions on mortality was not clear. The red deer was in good body condition and other than being found recumbent the day of death did not appear to demonstrate clinical signs of CWD infection. The hind may have had late-term pregnancy metabolic complications, which lead to death.
Chronic wasting disease transmission within and between susceptible host species is not completely understood, and the source of disease in this red deer is currently undetermined. Full blood red deer have not been reported to naturally acquire CWD, and, to the authors’ knowledge, no “atypical” CWD has ever been described for any cervid species in contrast to the “atypical” TSEs described in ovine and bovine species. 12 The 22-month-old red deer presented severe CWD prion accumulation within the brain and RPLN and widespread distribution throughout multiple other tissues. In accordance with the Spraker et al. scoring system, the CWD prion accumulation and spongiform degeneration of the obex fit the most severely affected category (Br+4). 7 The red deer in the current case report presented comparable CWD prion distribution and intensity to the orally exposed red deer in previous experiments, which developed behavioral changes progressing to neurological deficiencies at 18–25 months postinoculation.2,6 It appears the red deer in this case report may have established a CWD prion infection at an early age although severity of disease may also depend on host genotype and prion strain. 7
An epidemiological investigation was completed by the U.S. Department of Agriculture, Animal and Plant Health Inspection Service on the captive herd from which this red deer originated, in which no obvious biosecurity deficiencies were revealed. The herd has been closed to the introduction of outside animals since 2002, and the animals that populated this farm originated from a herd that had been closed to the introduction of outside animals for the previous 15 years. The farm has sustained a level 6 CWD status, the highest attainable level, with the Minnesota Board of Animal Health (MBAH) since its induction through proper recordkeeping, deliberate biosecurity measures, and compliance with the state and federal CWD monitoring programs. The herd is currently under MBAH mandated quarantine, restricting the movement of animals. Approximately half of the herd, including this red deer’s life-long enclosure cohorts, has tested negative for CWD prion infection by immunohistochemical staining of obex and RPLN at the time of this publication. The farm’s epidemiological status does not indicate a CWD infection risk.
Chronic wasting disease surveillance program changes, made in 2012, at the national level apportion implementation of captive cervid rules to state agencies. The rules currently include red deer as a susceptible species to be included in surveillance; the natural infection of the red deer described herein solidifies that policy. A diagnosis of CWD is devastating to a farm but as new scientific information is uncovered and herd situations such as this come to light, progression in policy is to be expected as governing agencies and stakeholders collaborate.
Footnotes
Acknowledgements
The authors thank James E. Collins for helpful suggestions and guidance.
a.
One Step PCR Reagent Kit, Qiagen Inc., Valencia, CA.
b.
Applied Biosystems RNase Inhibitor, Life Technologies, Foster City, CA.
c.
Ventana NexEs, Ventana Medical Systems Inc., Tucson, AZ.
d.
TeSeE Western Blot, Bio-Rad Laboratories, Hercules, CA.
e.
CWD TeSeE ELISA, Bio-Rad Laboratories, Hercules, CA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) declared that they received no financial support for their research and/or authorship of this article.
