Abstract
Haptoglobin (Hp), serum amyloid A (SAA), C-reactive protein (CRP), and white blood cells (WBC) were assessed in 20 dogs divided into 2 groups. The dogs of group A were not subjected to hunting exercise (control group), while the dogs of group B were subjected to hunting exercise for 3 hr (experimental group). Blood samples were collected from each animal before hunting (T0), immediately after 3 hr of hunting (T1), and after 1 hr of recovery (T2). The general linear model (GLM) repeated measures procedure showed a significant difference between the 2 groups (P < 0.0001) and a significant rise (P < 0.0001) in concentration of Hp, SAA, and CRP after hunting exercise, with a consequent decline during recovery period in group B. These parameters could be considered valid and easily obtainable biomarkers in relation to hunting stress in dogs. Additional studies will continue to elucidate the magnitude and the time of response of other acute phase proteins.
Introduction
Hunting with dogs, for sport or in competitive events, is a popular pastime in Italy. There are several types of hunting dogs developed for various tasks: the breed or type of dog varies with the type of game that is hunted as well as the terrain and distances that are covered. Regardless of these differences, most hunting work involves several hours of endurance activity interrupted with short periods of intense running or sprinting. 9 So, in the past few years, an increased number of scientific reports addressing the systemic changes that happen during exercise as well as the time needed to achieve resting values after exercise have been considered.3,23 It has been demonstrated that in dogs, exercise induces a variety of physiological and blood changes depending on the duration and intensity of exercise and on the fitness and training level of the athlete. 23 It has been shown that moderate treadmill exercise involves changes on some physiological parameters in trained and untrained dogs,22,24 and endurance exercise influences the serum concentration of biochemical parameters and acute phase proteins (APPs) in racing sled dogs. 15 But although it is important to know these modifications because they reflect changes in the functions of different systems and in the type of energy utilized, 20 few authors have studied the APPs during a specific exercise such as hunting.
Acute phase proteins are a group of blood proteins whose concentrations decrease or increase in animals subjected to external or internal challenges. 19 The origin of acute phase response (APR) can be attributable to infection, inflammation, surgical trauma, or other causes,5,7,13,19,21 and the purpose of the response is to restore homeostasis and to remove the cause of its disturbance.5,11 Particularly, APPs in dogs can be classified by the magnitude of their response to stimuli as positive (major and moderate) or negative reactants.5,17 Haptoglobin (Hp), serum amyloid A (SAA), and C-reactive protein (CRP), positive APPs that increase its rate of synthesis and release when an inflammation occurs,5,8,12 can be used in dogs as a screening test for systemic response to an inflammatory stimulus and are considered to be the most accurate markers of inflammation. 6 In fact, APPs have received attention as biomarkers for APR due to the facilitating properties of low physiological levels, the fast incline, the marked rise in concentration during APR that eases detection, and the fast decline after a ceased stimulus. 5 Exercise, often considered as a physical stress, represents a variety of physical and psychological stimuli that alter homeostasis and metabolism of animal organisms.1,10,14,18 Considering the important role of APPs as biomarkers and that there are some aspects of the APR in dogs that have not been widely evaluated in relation to exercise, the aim of the current study was to investigate the modifications of serum concentrations of Hp, CRP, SAA, and white blood cells (WBC) in order to identify the influence of exercise during hunting on these biomarkers, which could be increasingly useful in the future for the health and animal welfare of hunting dogs.
Materials and methods
The current study was carried out in November 2012 in Sicily (Italy) and involved a laboratory component and a veterinary clinic component of the University of Messina. Protocols of animal husbandry and experimentation were reviewed and approved in accordance with the standards recommended by the U.S. National Research Council’s Guide for the Care and Use of Laboratory Animals and European Union’s Directive 86/609 CEE.
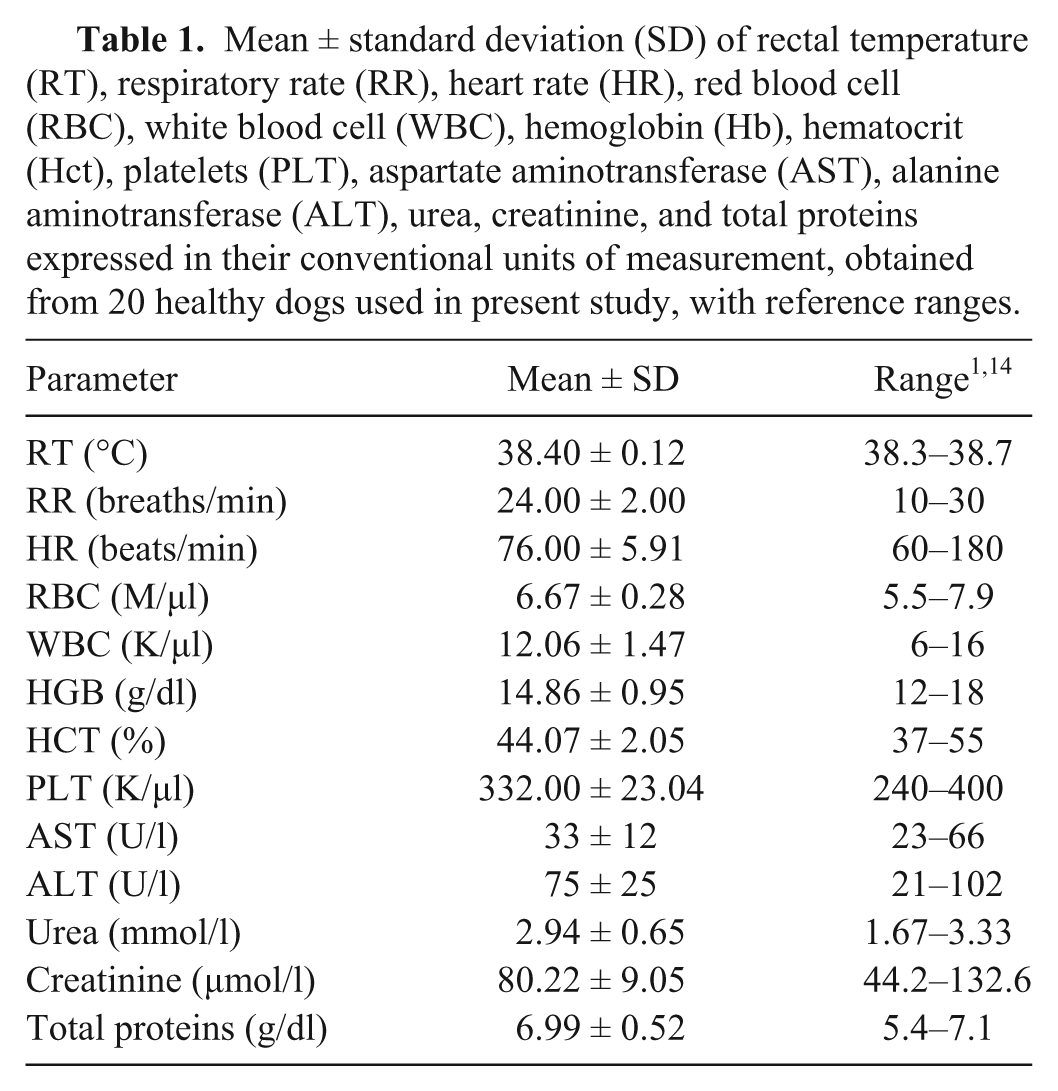
Twenty clinically healthy dogs (6–8 years old) of various breeds, including English Setters (5), Pointers (5), Spinone Italiano (5), and Cirneco dell’Etna (5), were used. Dogs were fed a high-quality commercial diet a in accordance with their body condition score, as assessed by their owners. Before recruitment, a full clinical history, including dietary regimens and average daily exercise, was obtained from every dog. All dogs underwent a full laboratorial examination in order to exclude animals with injuries, swelling, or any form of apparent diseases (Table 1).
Mean ± standard deviation (SD) of rectal temperature (RT), respiratory rate (RR), heart rate (HR), red blood cell (RBC), white blood cell (WBC), hemoglobin (Hb), hematocrit (Hct), platelets (PLT), aspartate aminotransferase (AST), alanine aminotransferase (ALT), urea, creatinine, and total proteins expressed in their conventional units of measurement, obtained from 20 healthy dogs used in present study, with reference ranges.
The following experimental protocol was used: the dogs were divided into 2 groups (A and B), each of which contained 10 animals. The dogs of group A were not subjected to hunting exercise (control group), while the dogs of group B were subjected to hunting exercise for 3 hr (experimental group).
During the experiment, the dogs were not fed 12 hr prior to exercise to ensure that the dogs were in a postabsorptive state. Blood was collected from every dog via cephalic venipuncture into evacuated glass tubes and stored in 2 different types of venous blood collection tubes: with no additive b and containing ethylenediamine tetra-acetic acid (EDTA). c The serum samples obtained after centrifugation d were stored for 20 days at −80°C before all measurements. Dogs were restrained gently with a halter during blood collection. On all samples, collected before hunting (T0), immediately after 3 hr of hunting (T1), and after 1 hr of recovery (T2), Hp, SAA, CRP, and WBC were assessed.
Serum concentrations of Hp were determined by use of the hemoglobin-binding method using a commercial kit e and an automated biochemistry analyzer. f Concentrations of SAA were determined by use of a solid-phase sandwich enzyme-linked immunosorbent assay (ELISA). The ELISA was designed for use in determining concentrations of SAA in different animal species such as dogs. Final absorbance of samples was measured by use of a microtiter plate reader at 450 nm. Both methods were validated in the authors’ laboratory for canine serum. 16 A solid-phase sandwich immunoassay specific for canine CRP g was used in accordance with directions provided by the manufacturer. Final absorbance of samples was measured by use of a microtiter plate reader h at 450 nm. This method was validated in the authors’ laboratory for canine serum. 16 The WBC count was assessed in blood samples containing EDTA using a multiparametric automatic analyser. i
All samples were analyzed in duplicate by the same operator. The samples exhibited parallel 98 displacement to the standard curve. The overall intra-assay coefficient of variation was <5%.
All results were expressed as mean ± standard deviation (SD). Data were normally distributed (P < 0.05, Kolmogorov–Smirnov test). The general linear model (GLM) repeated measures procedure was used to determine the differences between the experimental and control group and the statistically significant effect of sampling time (before hunting, after hunting, and after recovery period). Values of P < 0.05 were considered statistically significant. Duncan multiple post hoc comparison test was applied. The data were analyzed using commercial software. j
Results
Figure 1 shows average values of all studied parameters, expressed in conventional units of measurement with SD, measured during experimental trial in hunting dogs. The application of statistical analysis showed a significant difference between groups A and B (P < 0.0001) and the influence of sampling time (P < 0.0001) on Hp, SAA, and CRP in group B. Particularly, Hp, SAA, and CRP values showed a statistically significant increase at T1 in respect to T0 and T2.
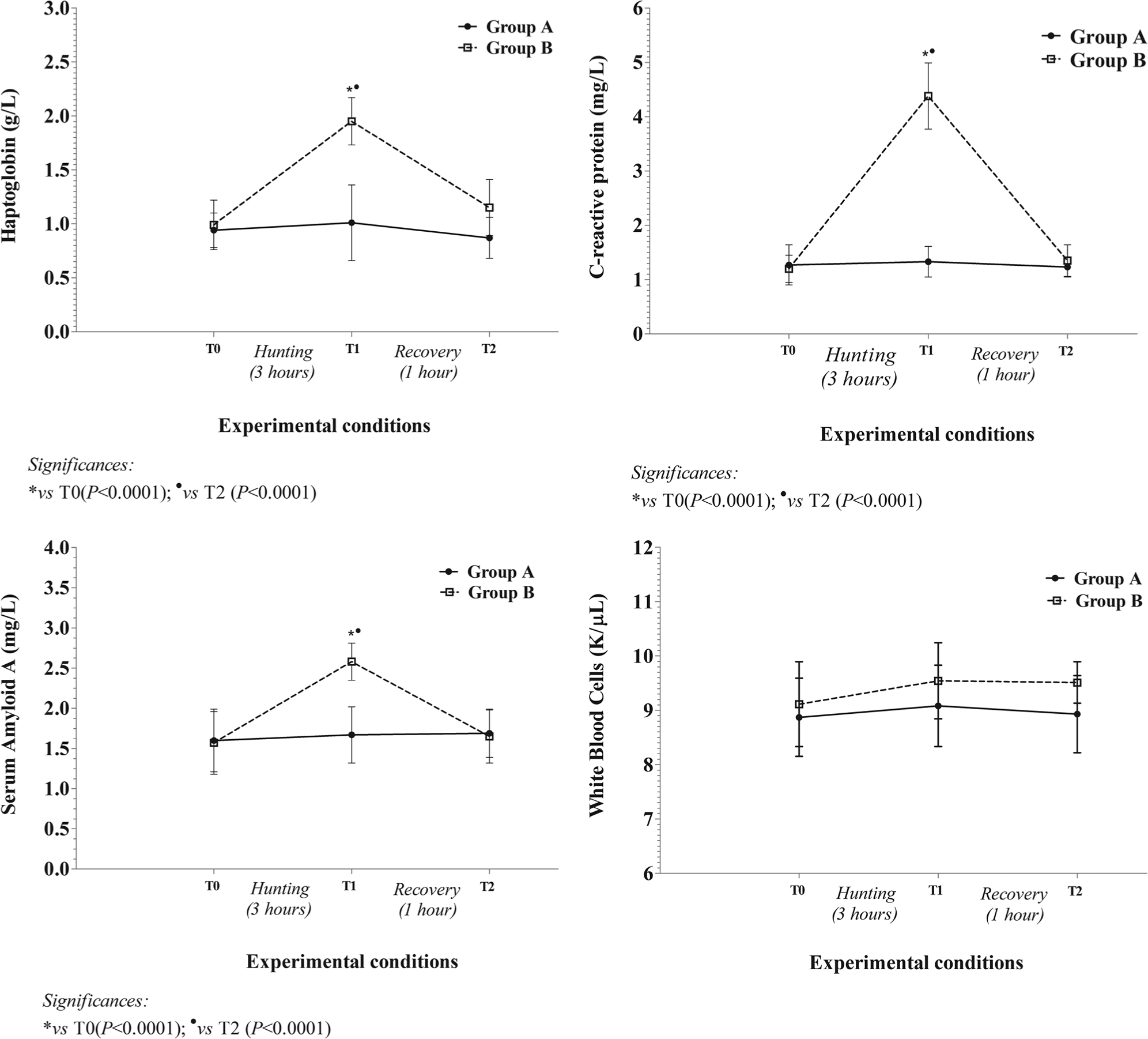
The pattern of mean value (± standard deviation) of haptoglobin, serum amyloid A, C-reactive protein, and white blood cells, together with statistical significances obtained in group A (dogs not subjected to hunting exercise) and in group B (dogs subjected to hunting exercise) during experimental period (T0: before hunting; T1: immediately after 3 hr of hunting; T2: after 1 hr of recovery).
Discussion
The results of the present study indicated that there was a statistically significant influence of hunting exercise on the studied APPs in hunting dogs. In particular, Hp, SAA, and CRP values had a rise in concentration after hunting exercise and a decline during the recovery period.
These results are in agreement with other researchers who emphasized the role of APPs in dogs not only with inflammation but also with some conditions unrelated to inflammation, which can be highly stressful and compromise welfare.5,25,26 The significant increase of Hp, SAA, and CRP after 3 hr of hunting versus T0 and T2 cannot be associated with pathological conditions but could be due to a physical “stress” resulting in numerous changes that occur during hunting exercise. 2
In disagreement with other researchers who interpreted increases in APPs as a sign of possible subclinical diseases such as arthritis, which could be manifested over a period of time,4,5 results of the present study showed a statistically significant increase of Hp, SAA, and CRP concentrations within the interval range of APR. Moreover, WBC count did not change in association with APPs after hunting exercise, thus confirming no pathological conditions. 2 In fact, Hp, SAA, and CRP concentrations returned to the baseline values after 1 hr of recovery, underlying the physiological response to exercise. Particularly, SAA and CRP values (major APPs) decreased rapidly up to the baseline values; Hp (moderate APP) values showed a more gradual decline because it needs more time to return to baseline values. 5
The results, characterized by a fast increase shortly after physical stimulus (hunting exercise) followed by quick normalization when no further stimulus is present, should facilitate the use of canine APPs as biomarkers for routine monitoring and screening purposes that could be used as an indicator of health and fitness in hunting dogs. In conclusion, results showed that Hp, SAA, and CRP are valid and easily obtainable biomarkers in relation to hunting stress in dogs, but additional studies are needed to elucidate the influence that hunting exercise may have on the magnitude and the time of course of response of other positive and negative APPs.
Footnotes
a.
Purina Pro Plan Adult, Nestle Italiana SpA, Milano, Italy.
b.
Venoject evacuated tubes, Terumo Europe N.V., Leuven, Belgium.
c.
Venoject haematology, Terumo Europe N.V., Leuven, Belgium.
d.
CL10 centrifuge, Thermo Fisher Scientific Inc., Waltham, MA.
e.
Tridelta Ltd., Brey, Ireland.
f.
Cobas Mira Plus multiparametric autoanalyser, ABX Diagnostics, Montpellier, France.
g.
Tridelta Development, Ltd., Wicklow, Ireland.
h.
PowerWave XS, Bio-Tek Instruments Inc., Vermont, CT.
i.
Automatic analyzer of haematology, HecoVet, SEAC, Florence, Italy.
j.
Version 4.3, SAS Institute, Cary, NC.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
