Abstract
Procalcitonin is considered an acute phase protein used as both a marker of infection and prognosis in human medicine. Canine procalcitonin has been previously sequenced; however, its use as a diagnostic or prognostic tool in dogs has never been assessed. A quantitative reverse transcription polymerase chain reaction (qRT-PCR) assay for canine procalcitonin messenger RNA (mRNA) was developed. Whole blood samples were collected from ill and healthy dogs. RNA was extracted and the realtime PCR was assessed. The patients' diagnoses, complete blood cell count, and differential leukocyte count results were recorded. Based on the diagnosis, dogs were divided into 5 groups: inflammatory, infectious, neoplastic, other diseases, and healthy controls. Procalcitonin mRNA expression and the hematological measures were compared between groups, and their correlations were assessed. Procalcitonin mRNA expression was assessed in 70 dogs, including infectious (17), noninfectious inflammatory (17), neoplastic (18), other diseases (7), and healthy controls (11), and was significantly (P < 0.001) higher in all ill dogs versus controls. Procalcitonin may therefore be considered an acutephase protein in dogs. However, there were no significant differences in procalcitonin mRNA expression between ill dog groups and no correlations between its expression levels and hematological measures. In 5 dogs of all disease categories, procalcitonin mRNA expression was measured twice during the course of disease. The changes in its levels were in agreement with the clinical evaluation of improvement or deterioration, suggesting a possible prognostic value.
The acute phase response (APR) is a nonspecific inflammatory reaction that occurs after any tissue injury that may be triggered by infectious, inflammatory, neoplastic, or other mechanisms early in the disease course. 3,20,21 During an APR, serum acute phase protein (APP) concentration alterations are observed. Often, APP concentrations are increased prior to the onset of clinical signs. 3,9,20,21 Several APPs have been studied in different animal species. Each APP has a characteristic response pattern and distinctive significance in different animal species, disease category, and individual patients.
Calcitonin (CT), secreted from thyroid C-cells, is synthesized as part of a large precursor molecule, preprocalcitonin, which dissociates to procalcitonin (PCT) and is later processed to mature CT in a regulated secretory pathway. 1 In addition, PCT is secreted by nonthyroidal tissues as the major product of a secondary, constitutive (unregulated) pathway activated by proinflammatory cytokines during disease and thus is considered an APP in humans. Its serum concentration increases during inflammation, while CT concentrations remain within the reference interval. 1,5,17 Studies in humans have shown that PCT levels were significantly higher in septic shock and severe infectious diseases compared with noninfectious inflammatory and neoplastic diseases and were diagnostically superior to serum amyloid A and C-reactive protein. 2,7,18 In addition, PCT expression was found to be only slightly induced by viral infections, autoimmune disorders, neoplasia, and surgical trauma. 4 Thus, in humans, PCT is considered a specific APP marker of infectious bacterial diseases, differentiating between infectious and other inflammatory and neoplastic diseases. 4 Procalcitonin may also be used to monitor response to treatment. A decrease in its serum concentration often precedes decreases in levels of other commonly used markers, including the leukocyte count. 2,7,18 Procalcitonin has also been used as a prognostic marker for assessing disease severity in septic, critically ill human patients. 2,12
Due to the usefulness of PCT in human medicine, PCT RNA expression in whole blood was evaluated in septic foals. No differences were found between septic foals versus healthy controls. Thus, the authors concluded that whole blood PCT RNA expression should not be considered a useful marker of infection in foals. 16
Canine PCT has been sequenced; however, its diagnostic usefulness has yet to be evaluated. 15 In humans, serum PCT is measured using an enzyme-linked immunosorbent assay-based 2-site monoclonal antibody targeted against the CT carboxyl terminal peptide (CCP) and the n-procalcitonin part, which allows differentiation of PCT from CT. 1 Because the carboxyl terminal parts of canine and human PCT are completely different, the enzyme-linked immunosorbent assay-based 2-site monoclonal antibody cannot be utilized in dogs. 15
The aim of the current study was to develop a real-time polymecase chain reaction (real-time PCR) assay for canine PCT messenger RNA (mRNA) and to evaluate its whole blood mRNA expression in diseased versus healthy control dogs. Presumably, because leukocytes have never been documented to contribute to mature CT serum concentrations and its levels have been reported to be unaffected by inflammation in humans, an increase of the leukocyte preprocalcitonin mRNA expression will only result in increased PCT production, whereas mature CT serum concentration will be unaffected. 1,11 The second aim of this preliminary study was to provide evidence as to whether future development of a canine PCT peptide assay is warranted for diagnostic and prognostic purposes.
Primers sequences used for real-time polymerase chain reaction procalcitonin expression.
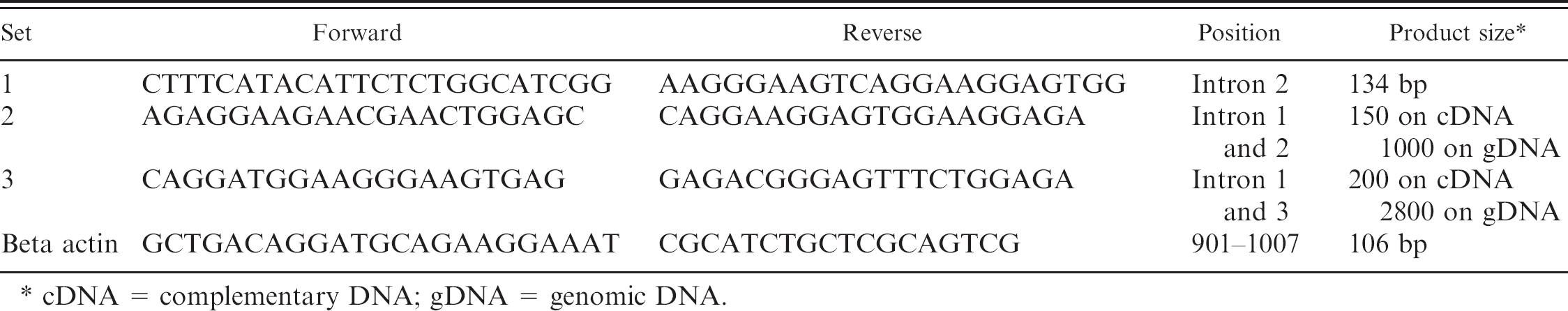
cDNA = complementary DNA; gDNA = genomic DNA.
A quantitative reverse transcription PCR (qRT-PCR) assay was developed to assess canine PCT mRNA expression. Three primer sets were designed based on GenBank accession number NM_001003266 of the Canis familiaris CT/CT-related polypeptide alpha (CALCA) mRNA. Two sets, which gave a product on both genomic DNA (gDNA) and complementary DNA (cDNA), were assayed on gDNA isolated from 10 μl of blood samples using FreezeFree PCR mix a and visualized by ethidium bromide on a 1.5% agarose electrophoresis gel. The products were sequenced, and the results were compared with GenBank sequences to verify the identity of the product as PCT.
The total RNA from 2 thyroid samples was extracted using TRIzol b and cleaned from gDNA contamination using DNasel c and the vendor's protocol. The RNA was amplified with PCR to validate that there was no gDNA contamination. Complementary DNA was prepared from 5 μg of RNA using the Superscript III d kit, and PCR of the sample was run with all 3 primer sets on regular PCR and qRT-PCR with SYBR Green FreezeFree QPCR mix. All 3 sets gave a positive result, and a standard curve (based on plasmid clone prepared from gDNA PCR product) of 6 2-fold dilutions was assayed (Table 1). Set number 2 produced a superior result, with a wider dynamic range and higher sensitivity at lower dilutions, and was therefore selected for the assay.
The current study was designed as a prospective study. Potassium ethylenediamine tetra-acetic acid blood samples were consecutively collected from sick dogs presented to the Hebrew University Veterinary Teaching Hospital and from healthy, staff-owned controls and underwent RNA extraction and qRT-PCR. Signalment, clinical signs, diagnoses, complete blood cell count, and differential leukocyte counts were recorded. RNA from 500 μl of 112 fresh whole blood samples was extracted using PureLink whole blood extraction kit. d Only samples that yielded total RNA >500 ng were used (70 of 112 samples). Samples were assayed for gDNA contamination by running a PCR reaction on the RNA. Samples showing contamination were retreated using DNasel c and retested. Once clean of gDNA, the RNA was pelleted down using ethanol precipitation and resuspended in 10 to 15 μl of nuclease-free water. Complementary DNA was prepared from all 500 ng of RNA. Each set was tested first for cDNA amount and quality by qRT-PCR with canine beta-actin primer. Quantitative RT-PCR was done in triplicate for each sample with PCT set number 2 and beta-actin primer using 50 ng of total RNA per reaction (Table 1). First, threshold cycle (Ct) values were assigned according to the cycle number at which a fixed fluorescent intensity was achieved. Then, a ΔCt value was calculated by Ct(PCT) – Ct(beta actin). Next, fold induction of genes in the ill samples relative to the healthy samples was calculated according to the following expression, in which ΔΔCt is the difference between ΔCt PCT of ill and ΔCt of healthy controls. 14
Dogs were divided into groups based on the primary disease diagnosis. Categories were 1) infectious, 2) noninfectious inflammatory, 3) neoplastic, and 4) other (i.e., degenerative, idiopathic, and traumatic) diseases. In 5 animals, PCT mRNA expression was measured twice during their disease course, and their clinical status (i.e., improved or deteriorated) was assessed blindly (to PCT mRNA expression results) by their attending clinicians. Procalcitonin mRNA expression was compared between disease category groups using Student's t-test when data had normal distribution. Spearman rank correlation was used to assess the association between PCT mRNA expression and hematological measures within groups and in the whole study population. P < 0.05 was considered statistically significant for all tests. A receiver operating characteristic analysis was used to assess the potential sensitivity and specificity of PCT mRNA expression for prediction of disease.
Blood samples were collected from 101 ill dogs at presentation to the hospital and from 11 healthy, staff-owned dogs (controls). The control group included dogs with an age range of <1 to 11 years (median: 3 years), 5 males (4 castrated), and 6 females (4 spayed). The ill dog group included 28 males (11 castrated) and 31 females (21 spayed) with an age range of 2 months to 15 years (median: 7 years). Fifty-nine ill dogs and 11 healthy controls yielded sufficient amounts of extracted RNA and were thus included in the study. Unsuccessful RNA extraction was the only exclusion criteria. Data of the different disease categories and diagnoses are summarized in Table 2.
Disease categories and diagnoses.

Procalcitonin mRNA expression was significantly (P < 0.001) higher in all ill dogs and in each disease category group versus controls (Figs. 1, 2). There were no significant PCT mRNA expression differences between different disease category groups or correlations between PCT mRNA expression and total or differential leukocyte counts. A receiver operating characteristic analysis of PCT mRNA expression optimized for prediction of the presence of disease yielded a 70% sensitivity and 100% specificity at a ΔCT > 1 cutoff point. Five of the 59 dogs were followed. Procalcitonin mRNA expression was remeasured during the disease course and was found in agreement with the clinical assessment of the disease progression or remission (Table 3).
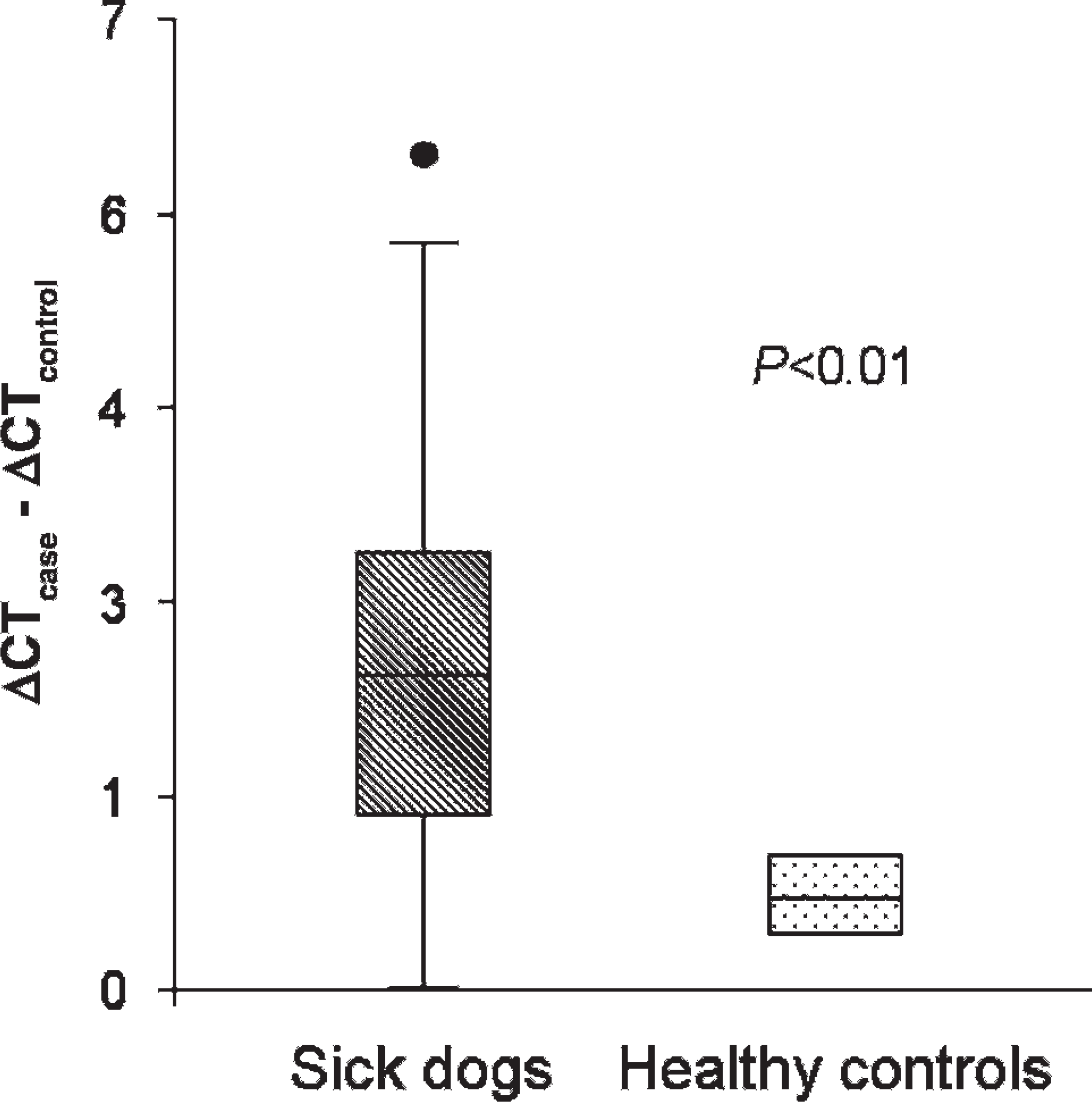
Whole blood procalcitonin RNA expression in all ill dogs combined (59 dogs) versus healthy controls (11 dogs).
To the authors' knowledge, the current study is the first to examine canine PCT mRNA expression. Given the potential usefulness of PCT as a diagnostic and prognostic marker, this study aimed to assess its level of expression in different disease categories. The results suggest that PCT is an APP in dogs, as it is in humans. 1,2,5,12,17,18
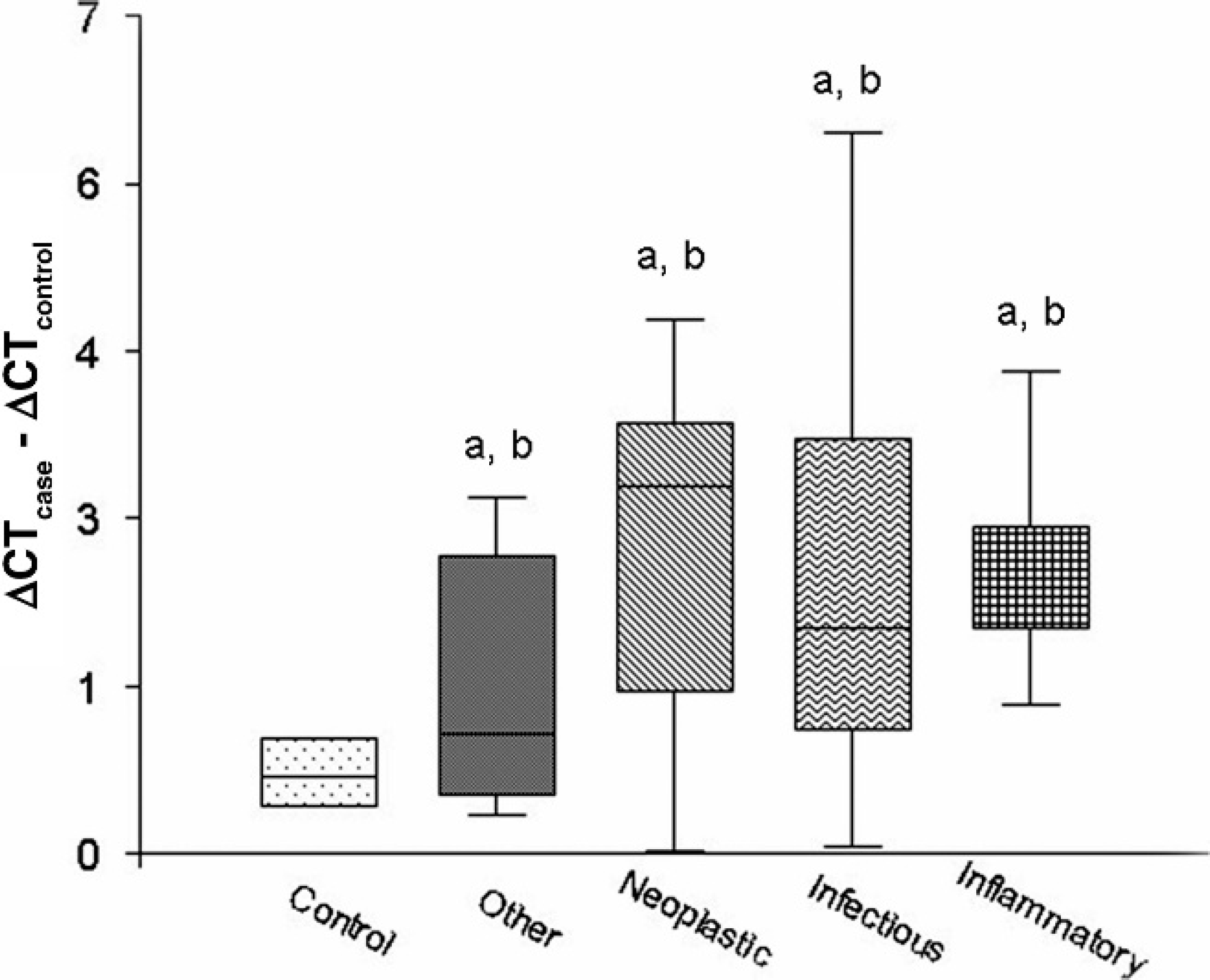
Whole blood procalcitonin (PCT) messenger RNA (mRNA) expression in 4 groups of ill dogs (17 dogs with infectious disease, 17 dogs with inflammatory disease, 18 dogs with neoplastic disease, and 7 dogs with other diseases) and in 11 healthy controls.
Procalcitonin (PCT) messenger RNA (mRNA) expression at presentation and later during the disease course in 5 dogs.
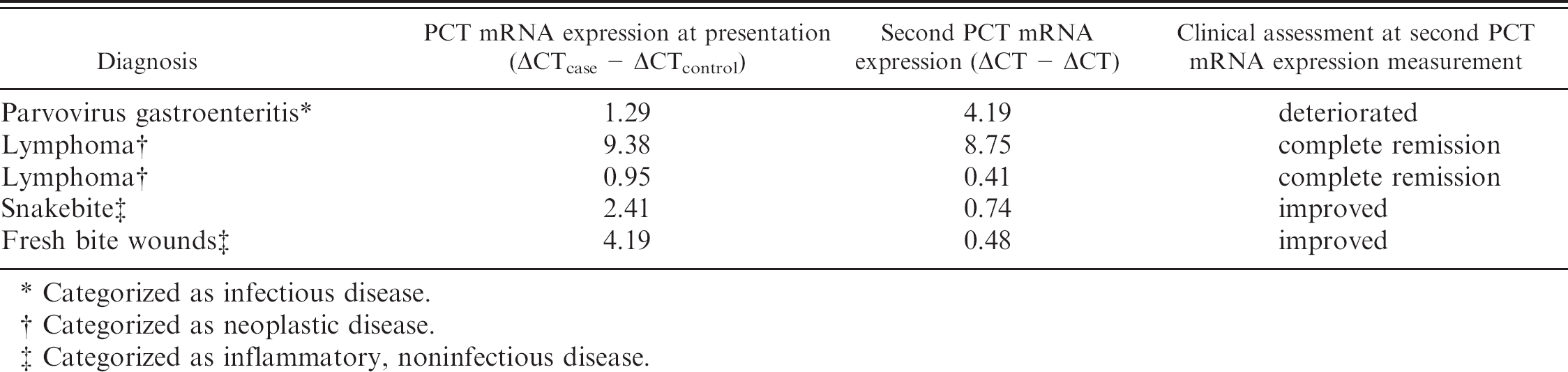
Categorized as infectious disease.
Categorized as neoplastic disease.
Categorized as inflammatory, noninfectious disease.
Although PCT is a differentiating marker of infectious diseases in humans, canine PCT mRNA expression did not differ significantly between infectious and other diseases. There are several possible explanations for this dissimilarity. First, PCT expression was measured in extracted RNA from peripheral blood leukocytes. Because PCT is produced and secreted by all parenchymatic tissues, leukocytes are not its main source during inflammation and whole blood PCT mRNA expression may not reflect the actual magnitude of its increased expression. Measurement of the serum PCT peptide concentrations may produce a different pattern of expression in different disease categories. Second, whereas the infectious diseases in human studies are composed mostly of septic shock and severe bacterial infections, the dogs included in the present study had a wider range of infectious diseases, yielding a more heterogeneous inflammation and APR. The relatively low number of animals with the wide range of diseases in each group might have rendered the present study a relatively low statistical power for comparison of whole blood PCT mRNA expression in the different disease categories. For example, in the present study, bacterial and viral infections were included in a single infectious diseases group, whereas studies in humans have demonstrated significantly higher serum PCT concentrations in bacterial compared with viral and protozoal infections. 2,5,7,18,19 Therefore, future studies of canine PCT expression should probably focus on specific diseases to overcome this limitation. Third, species differences have been described in other APPs and may exist in PCT expression patterns in dogs. For example, although haptoglobin is a major positive bovine APP, it is much less so in dogs. 8,10
The lack of correlation between PCT mRNA expression and total and differential leukocyte counts is most likely associated with differences in the sensitivity of these latter tests for inflammation and the study population heterogeneity. Although PCT is expected to be a sensitive marker of inflammation, leukocyte and neutrophil counts are less sensitive because their numbers may be lower or higher than or within a reference interval, depending on the disease, its stage and severity, and other factors (e.g., stress, anxiety, age). 13 Serum levels of other APPs in dogs and cats have been reported to change before leukogram changes are detected or clinical signs are observed. 3,21
In human medicine, serum PCT concentration is also used for monitoring response to treatment and as a prognostic marker. 2,6,12 The expression of PCT mRNA was measured twice during the disease course in 5 dogs in the present study and showed a remarkable agreement with the clinical assessment (i.e., improved or deteriorated) of these patients. Although this is a small patient sample, it included dogs of all major inflammation categories. Additionally, both clinically improving and deteriorating patients were followed. These preliminary results, although limited in number, suggest that PCT mRNA expression has a potential use in therapeutic monitoring and planning in dogs.
The receiver operating characteristic analysis results suggest that PCT mRNA expression is a relatively sensitive and highly specific marker for the presence of disease despite several limitations. Measurement of serum PCT peptide levels may improve the diagnostic sensitivity of this APP in selected diseases. Preliminary findings suggest that it can be used both as a prognostic marker and to monitor response to treatment; however, further investigation is needed to fully assess serum PCT concentration and expression for these purposes in dogs. The results of the present study also support the development of an assay to measure serum PCT peptide levels that may change and improve the differentiating qualities demonstrated with whole blood real-time PCR.
Acknowledgement. This work was funded by a grant from the Koret School of Veterinary Medicine Clinical Research Fund.
Footnotes
a.
Karnieli Ltd., Kiryat Tivon, Israel.
b.
Sigma-Aldrich, Rehovot, Israel.
c.
DNase, Applied Biosystems/Ambion, Austin, TX.
d.
Invitrogen Corp., Carlsbad, CA.
