Abstract
A rare barbastelle bat (Barbastella barbastellus) died shortly after being found in emaciated condition in Devon, England. The skin over the muzzle and face was grossly thickened, crusty, and in places was sloughing and ulcerated. There were numerous nodules up to 3 mm in diameter on both wings and ear pinnae. Histologically, multiple foci of epidermal hyperplasia, hyperkeratosis, and crateriform erosions containing masses of fungal spores and septate hyphae were found in the wing. Epidermal hyperplasia and follicular hyperkeratosis, with fungal masses within keratinized follicles and also in fissured stratum corneum, were found in the pinna. Hyphae did not invade the dermis, and there was no inflammation, but there was multifocal serous exudation and crusting. No parasites or other significant organisms were identified. Microscopic and multiple cultural analyses of face and wing lesions demonstrated (10/10) a fine, septate fungus bearing laterally oval to clavate conidia; morphologically and culturally this was entirely consistent with Hyphopichia burtonii, and polymerase chain reaction analysis and sequencing gave 100% identity with the type strain. The organism isolated was morphologically consistent with that repeatedly seen in histology sections and demonstrates that although H. burtonii has not previously been recognized as a dermatophyte, it clearly has the ability to invade the skin of live bats. Although not identical, the lesions in this case show similarity with those of white nose syndrome and therefore H. burtonii should be considered as a potential pathogen of bats.
The barbastelle bat (Barbastella barbastellus) is one of the rarest mammals in the United Kingdom (http://jncc.defra.gov.uk/publications/jncc312/species.asp?FeatureIntCode=S1308),is defined as a Near Threatened species on the International Union for Conservation of Nature Red List (http://www.iucnredlist.org/search), and is also 1 of 7 species of British bats that are covered by the UK Biodiversity Action Plan (http://jncc.defra.gov.uk/page-5170). In common with many other bats species in the United Kingdom, its numbers are believed to have declined since the 1950s, but the reasons for this are poorly understood (http://www.bats.org.uk/pages/barbastelle-1.html).
In October 2011, a barbastelle bat was found hanging in a house doorway in a small town in Devon, southwest England. It was taken in to care but died a short while later. The bat was a juvenile female in emaciated condition with a staring coat. The skin over the muzzle and facial area was grossly thickened, crusty, and in places was sloughing and ulcerated. There were numerous nodules, ranging from less than 1 mm to approximately 3 mm in diameter, on the skin over the forearm and chiropatagium of both wings and on the ear pinnae. Most of the nodules were smooth, dark, and circumscribed but larger ones were often irregular with whitish foci (Fig. 1A).
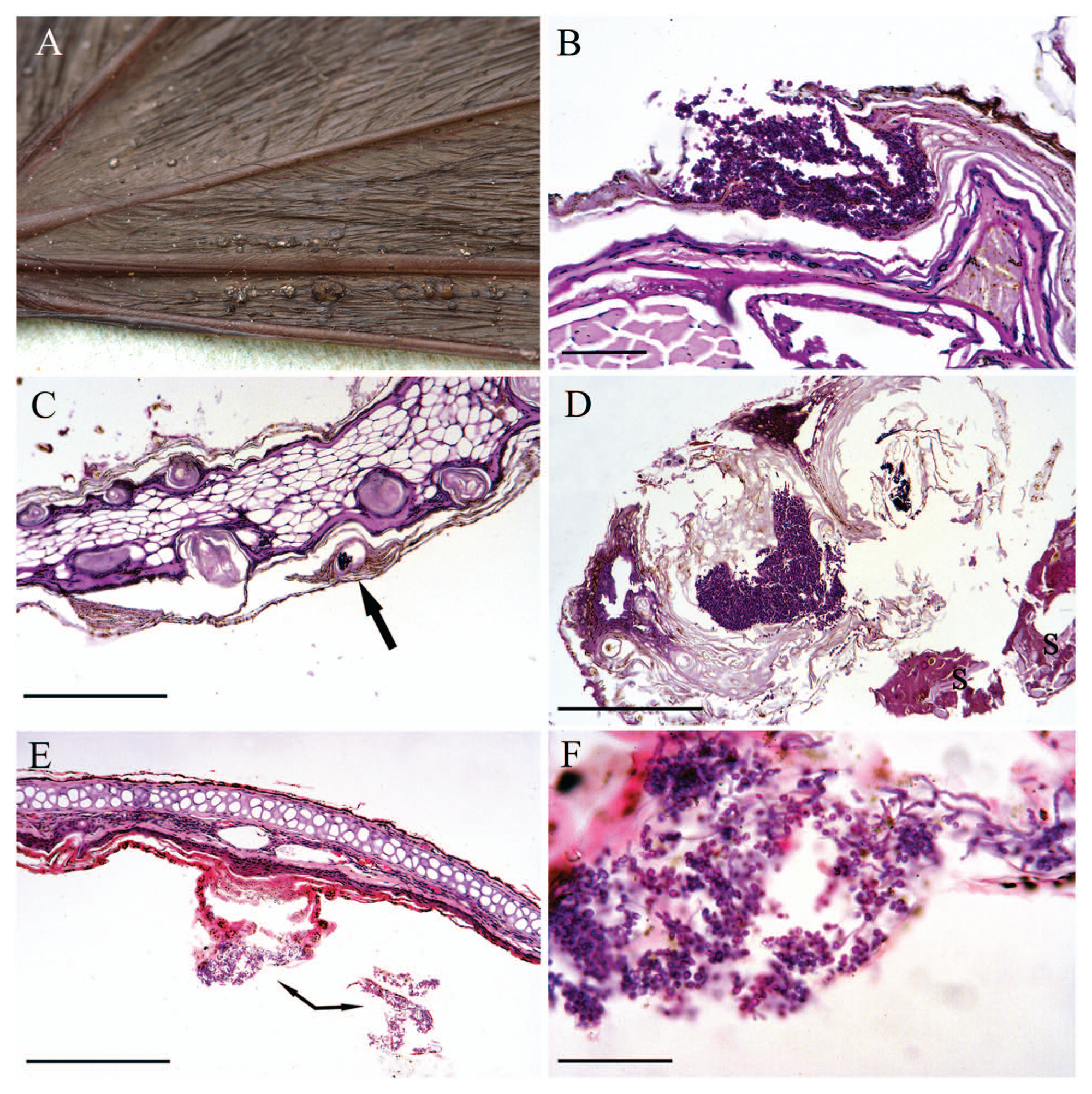
Barbastelle bat (Barbastella barbastellus); wing and pinna lesions.
A postmortem examination was not performed but samples were retained for laboratory examination (Natural England license no. 20103751). An ear pinna fixed in 10% buffered formalin was submitted for histological examination, and an unfixed wing and a sample of the facial lesion were submitted for microscopic and cultural examination. The wing sample was retrospectively fixed in 10% buffered formalin for histological examination. The fixed ear pinna and wing samples were embedded in paraffin wax, sectioned at 4 µm, and stained by hematoxylin and eosin, Gram, Giemsa, and periodic acid–Schiff.
Histological sections of wing showed focal areas of epidermal hyperplasia and hyperkeratosis associated with crateriform erosions, within which were masses of fungal spores and septate hyphae. No inflammation was noted. Hyphae infiltrated the stratum basale but were not observed in the underlying dermis (Fig. 1B). It is likely that these crateriform lesions represented the foci seen at gross examination and that the superficial layers were lost during processing.
Transverse sections of the pinna showed marked follicular hyperkeratosis and epidermal hyperplasia along both the inner and outer margins. Centrally within some of the keratinized follicles there were masses of fungal spores and septate hyphae. There was no inflammation around thefollicles but there was multifocal serous exudation and crusting (Fig. 1C, 1D). In the central area of the pinna there were occasional small focal areas of epidermal hyperplasia. In some of these foci the superficial stratum corneum contained small groups of septate periodic acid–Schiff-positive fungal hyphae, but in others, the layers of the stratum corneum were fissured and contained fungal hyphae and numerous spores (Fig. 1E, 1F). No parasites or other significant organisms were identified in any of the sections.
Microscopic analysis of unfixed wing and facial samples by potassium hydroxide digestion with a fluorescence enhancer a revealed the presence of large amounts of filamentous fungal elements. These were fine, regularly septate, and acutely branching hyphae and were mixed with large quantities of small, oval to round spores (“yeast blastospores”) measuring approximately 4 μm in diameter. Several of these “yeast blastospores” appeared attached to the filamentous fungal hyphae laterally on fine, shortened denticles.
Culture of multiple wing and facial samples on Sabouraud agar with chloramphenicol yielded identical fungal colonies (10/10 inoculum sites) that were pale cream-gray, moist, and spreading, with fasciculated hyphae. Three colonies of yeast (out of 10 inoculum sites) were also recovered. Microscopic examination of the filamentous fungal colonies revealed morphological characteristics that were very similar to the fungal elements identified via direct examination of affected bat tissues—with numerous oval to clavate yeast-like conidia borne laterally and terminally on short denticles along the parental, septate true hyphae (Fig. 2).
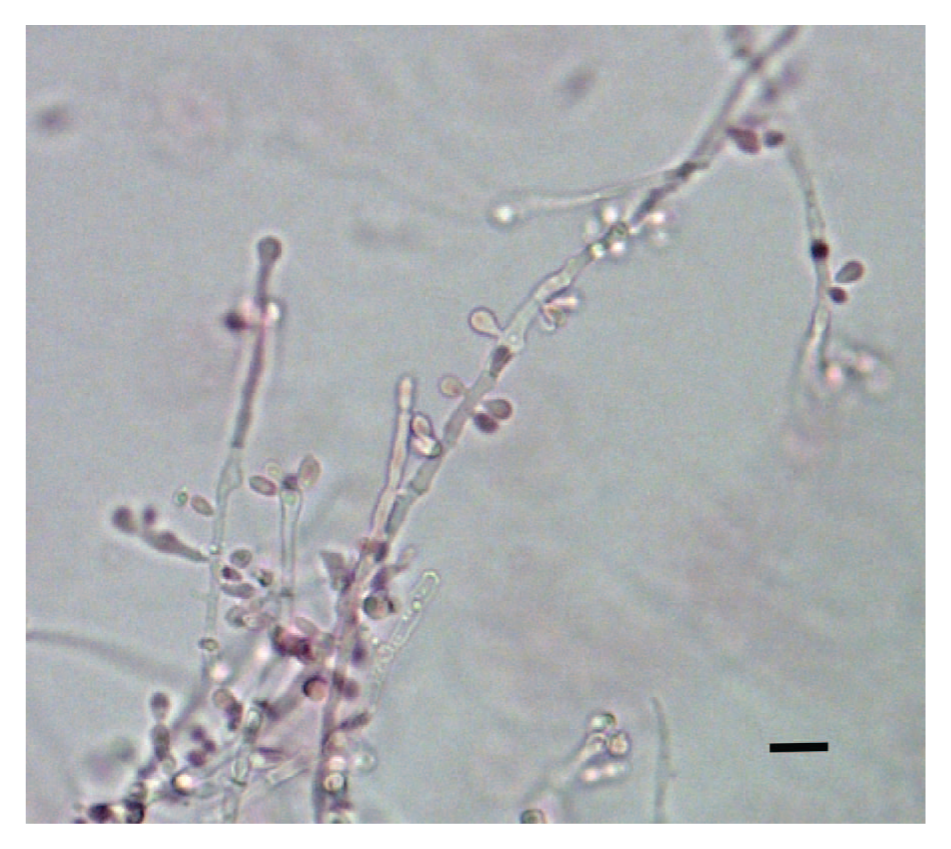
Light microscopy of the isolate of Hyphopichia burtonii. Light microscopy was performed on needle mounts of 7-day-old cultures that had been placed in lacto-fuchsin mounting fluid. Bar = 10 μm.
Polymerase chain reaction amplification and sequencing of the D1D2 and ITS1 regions of the filamentous fungus2,3 returned sequences (EMBL accession nos. HE967323 and HE967324) that were 100% identical to the type strain (CBS 2352, EMBL GU597324, and GU246247) of Hyphopichia burtonii in the European Molecular Biology Laboratory public synchronized database. Similar analyses of the yeast isolated from only a subset of inocula revealed 100% identity with strains of Debaryomyces hansenii (EMBL accession nos. HE967325 and HE967326).
While the wing and ear lesions seen in the bat in the current study might appear to be of limited significance, the lesions could possibly have affected normal physiological function. 4 However, the lesions around the muzzle were severe and would have almost certainly affected feeding and grooming and therefore contributed to the bat’s death. The lesions are also important as they resemble in several respects those caused by Geomyces destructans, the agent of white nose syndrome (WNS), which is responsible for unprecedented bat mortality in North America, currently estimated at approximately 6 million bats (http://www.batcon.org/pdfs/USFWS_WNS_Mortality_2012_NR_FINAL.pdf). The gross lesions of WNS include patches of rough skin on the face, ears, forearms, wing membranes, and feet as well as pinpoint white foci that resemble comedones on the muzzle. 7 Histologically, fungal hyphae are observed forming cup-like epidermal erosions and ulcers in the wing membrane and pinna. The hyphae extend beneath the epidermis, but there is a surprising lack of inflammation. 7 Similar gross and histological lesions associated with G. destructans have been observed in a small number of greater mouse-eared bats (Myotis myotis) in the Czech Republic but without evidence of mass mortality. 9 Although the lesions in the barbastelle bat were somewhat similar to those described in cases ofG. destructans infection, with fungal colonies enclosed within the outer layers of the epidermis and with no underlying inflammatory reaction, H. burtonii was not observed invading beyond the epidermis. The present case also differs significantly from typical WNS cases in that the bat was found alive in the autumn and was not in hibernation. Bats affected by WNS are often observed showing abnormal flying and roosting behavior (http://www.batcon.org/pdfs/USFWS_WNS_Mortality_2012_NR_FINAL.pdf) and it may be relevant, therefore, that the barbastelle bat, a species typically dependent on ancient deciduous woodland, was found roosting in an urban environment.
There have been few studies on the diseases of bats in the United Kingdom although more extensive studies have been carried out elsewhere in Europe. 8 There appear to be no published accounts of disease in B. barbastellus and no reported animal or human mycoses due to Hyphopichia burtonii. The morphology of the fungus isolated from both muzzle and wing of the barbastelle bat in the present study is entirely consistent with the limited morphological descriptions of H. burtonii. 6 It is not possible to suggest the source of the H. burtonii infection, as like most fungi, it is fairly ubiquitous in nature. The fungus has been isolated, for example, from spoiled foodstuffs, caterpillars, silage, pollen, human beings, and occasionally other mammals. 1 Debaryomyces hansenii is a known, but rare, cause of invasive yeast infection in immunocompromised human patients, 5 but from the limited amount of histological material available in the current case there was no evidence to show that D. hansenii was associated with the skin lesions and its presence probably represented secondary or postmortem colonization of damaged bat tissues. By contrast, the current study clearly shows that H. burtonii has the ability to invade the skin of live bats and therefore the role of this organism as a possible cause of bat mortality merits further study.
Footnotes
Acknowledgements
The authors wish to thank Abbey Veterinary Services for histological processing.
a.
Calcofluor: Bactidrop, Remel Microbiology Products, Thermo Fisher Scientific Inc., Waltham, MA, distributed by Oxoid Limited Basingstoke, UK.
Declaration of conflicting interests
The author(s) declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
