Abstract
The world’s poultry industry has grown into a multibillion-dollar business, the success of which hinges on healthy intestinal tracts, which result in effective feed conversion. Enteric disease in poultry can have devastating economic effects on producers, due to high mortality rates and poor feed efficiency. Clostridia are considered to be among the most important agents of enteric disease in poultry. Diagnosis of enteric diseases produced by clostridia is usually challenging, mainly because many clostridial species can be normal inhabitants of the gut, making it difficult to determine their role in virulence. The most common clostridial enteric disease in poultry is necrotic enteritis, caused by
Introduction
Since the 1950s, the world’s poultry industry has grown dramatically, and with this growth has come many new demands that have changed the very infrastructure of the industry. In 1945, the U.S. broiler industry was valued at $327 million, producing a total weight of 1.11 billion pounds of meat, while in 2010 the industry was valued at $23.7 billion, producing a total weight of 49.2 billion pounds of meat.125,126 In 2010, the U.S. poultry industry produced 8.63 billion broilers, 244 million turkeys, and 91.4 billion eggs, with a total value of $34.7 billion. 126 In Europe, poultry production has also increased dramatically over the past few decades; Swedish broiler production has increased from 23.8 million to over 65 million broilers annually. In Denmark, broiler production over the past 10 years has also grown significantly, from 113 million to over 137 million broilers annually.44,46 On a global scale, 34 billion broilers, 4 billion layers, 800 million breeders, 600 million turkeys, and 800 million ducks were produced in 2000. Growth in the world’s poultry industry has resulted from increased poultry production in many countries, but particularly in India, Pakistan, and Bangladesh. 123
With ever-increasing demands for massive volumes of high-quality food, the global poultry industry has been forced to undergo numerous changes. 3 Genetic selection has resulted in faster growth, improved feed conversion, higher meat yields, and lower mortality rates.29,37 For example, in the United States in 1945, the average final live weight per broiler chicken was 1.37 kg, but by 2001 the average final live weight was 2.3 kg. 125 In 1976, poultry producers required 64 days to grow birds to 2.41 kg, but as of 2003 this is accomplished in only 38 days. 112 Because it is estimated that 65–70% of producers’ total costs are in feed, faster growing birds with high feed efficiency are required for profitability.1,110 Improvements in housing and equipment have led producers to dramatically increase the number of birds per chicken house.
In the 1990s in Denmark, the average broiler house contained approximately 27,000 birds, but as of 2004, the stocking density has been increased to 40,000–50,000 birds per house.37,45 Even in smaller countries such as Norway, the average flock is 10,000–20,000 birds per house.62,92 Due to higher demands for poultry and improvements in housing, equipment, genetic selection, and other factors, poultry production has become highly specialized and requires a significant economic investment to turn a profit. Poultry producers must do everything possible to grow healthy birds as fast as possible if they are to survive in the market. 10
One major concern in poultry production is disease prevention, as outbreaks of various poultry diseases can dramatically slow growth and increase mortality rates. One of the most critical concerns of producers is that the birds have healthy and functional intestinal tracts to grow to proper weights 110 ; outbreaks of enteric disease can cause poor feed efficiency, decreased weight gain, and an overall increase in mortality, all of which can dramatically increase production costs. 110
Enteric disease in poultry can be caused by a wide variety of infectious agents, and clostridia are among the most important of these. Diagnosis of enteric diseases produced by clostridia is unusually challenging, in that many clostridial species can be normal inhabitants of the gut, making difficult the determination of their pathogenic roles. The most common clostridial enteric diseases of poultry are reviewed herein, and the diagnostic criteria for these diseases are discussed.
Genus Clostridium
The genus
Several clostridial enteric diseases occur in poultry, but probably the most common and severe is necrotic enteritis (NE), caused by
Clostridium perfringens (necrotic enteritis)
Host range
Outbreaks of NE in poultry are typically sporadic,60,94,105,113 and the disease has been reported by poultry producers worldwide.23,39,67,99,132 Outbreaks are most common in broiler chickens 2–6 weeks of age,12,17,47,56,91 which is believed to be due to a window in the chick’s anti-clostridial immunity. 85 This window develops when titers of maternal antibodies wane around 2 weeks of age and before the chick’s immune system reaches maturity at around 3–4 weeks of age.85,91 However, outbreaks also occur in broilers up to 11 weeks of age.64,87,101 The disease has also been reported in 3–6-month-old commercial layers50,110 and also in 12–16-week-old replacement pullets.24,50,51,101,110 An outbreak of NE in 9-month-old chickens was reported in India in 1974, 83 but this is the only reported case of the disease in older birds. Necrotic enteritis has also been reported in a wide variety of avian species other than chickens, including turkeys,40–42,121 ostriches, 84 quail, 9 capercaillies,121,122 geese, 134 bluebirds, 18 lorikeets,98,104 and crows. 8
Pathogenesis and host response
Necrotic enteritis develops when
During the past 10 years, the role of CPA in the pathogenesis of NE has been called into question. A previous study
58
found that isolates from NE cases did not produce more CPA in vitro than isolates from healthy birds. A 2006 study
76
found that a CPA null mutant produced lesions typical of NE in an experimental model and concluded that CPA was not critical for pathogenesis of NE. However, a 2010 study
33
examined the same CPA null mutant, not only finding lesion production but also detecting CPA in the intestinal tract of conventional birds challenged with the mutant, suggesting that wild-type strains of
Others have examined serum from birds during NE outbreaks for anti-CPA antibodies and found high levels of anti-CPA antibodies in birds with a history of clinical or subclinical NE. 92 In a study 92 using ELISA to examine broiler flocks with a history of NE and from healthy flocks for anti-CPA antibodies, the authors found 2.9% of birds from healthy flocks to be seropositive, compared to 15.7% of birds from flocks with a history of NE. Eighteen percent of birds from flocks with clinical NE were seropositive at 35 days of age, but the seropositivity level rose to 78% in those birds at 53 days of age. 92 In experimental NE, the authors found that 59–79% of birds were seropositive for anti-CPA antibodies. 92 However, the exact role these antibodies play in immunity to NE is still not known.91,95
Transcriptional profiling of chicken genes during experimental production of NE has showed that both cell-mediated and antibody-mediated immune responses are raised during infection.114,136 Overall, very little is known about the immune response of poultry to NE.
Recently, the so-called NE toxin B (NetB) was identified and found to have a role in the pathogenesis of NE.
75
NetB has amino acid sequence similarity to
Beta2 toxin (CPB2) was originally purified from a type C isolate from a piglet with hemorrhagic NE,26,127,130 but the toxin also has been suggested to have a role in poultry NE.
127
However, surveys of isolates from normal chickens and chickens suffering from NE revealed that prevalence of
Enterotoxin (CPE), produced by some isolates of
Clinical signs
Typical clinical signs of NE include depression,4,50,87,110 reluctance to move,50,64 diarrhea,64,110,121,127 ruffled feathers,50,64,87,110 somnolence,4,127 decreased appetite or anorexia,23,50,121,127 huddling,87,110 and in some cases, dribbling from the beak 101 and dehydration. 88 Clinical illness is often very short, with birds dying within 1–2 hr after onset of clinical signs 64 ; frequently, birds are found dead without premonitory clinical signs.22,23,50,101 Similar clinical signs are seen in birds of other avian species suffering from NE.41,42,84
Gross lesions
Birds with acute NE typically have gross lesions that are restricted to the small intestine,17,87,110 most often in jejunum and ileum.84,88,105,127 However, lesions can also occur in duodenum and ceca.4,88,105,117 A previous study
88
suggested that cecal lesions only occur when necrotic debris filled with
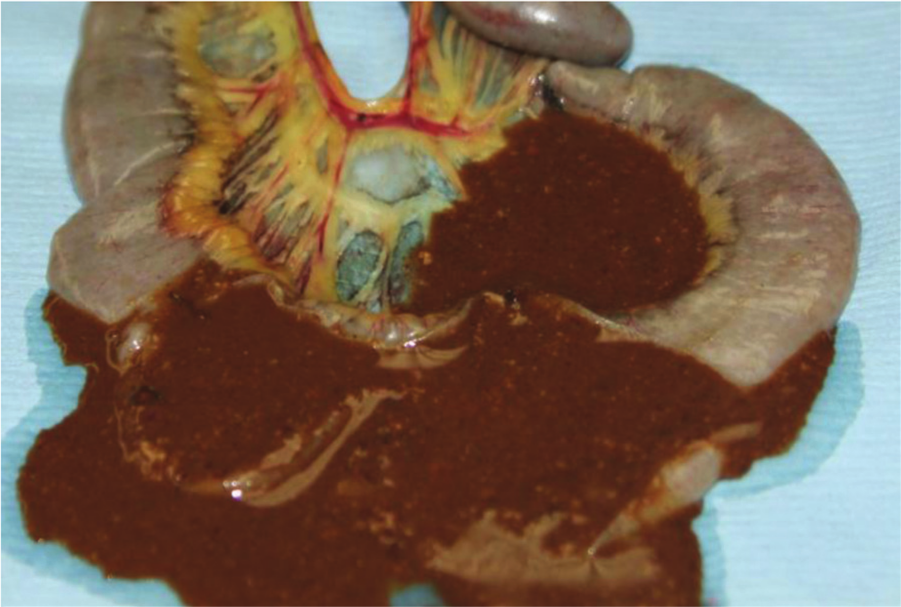
Jejunum, chicken. Clinical, acute necrotic enteritis. The lumen of the intestine is filled with thick, dark brown exudate.
Jejunum, chicken. Clinical, subacute necrotic enteritis. There is diffuse necrosis of the intestinal mucosa, which is completely covered by a pseudomembrane, which is forming a thick cast in the lumen of the intestine. The wall of the intestine is thickened.
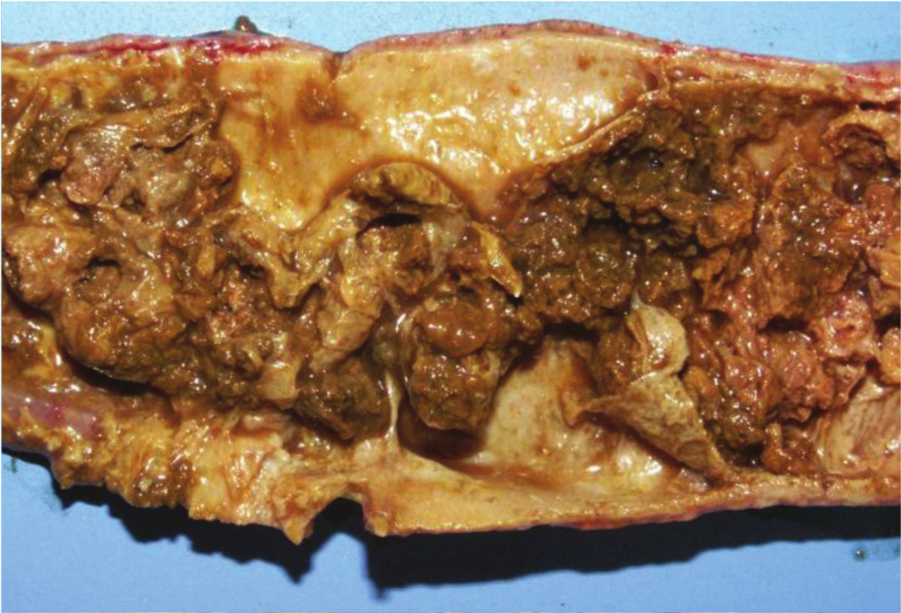
Jejunum, chicken. Clinical, chronic necrotic enteritis. The intestinal wall is severely thickened by inflammatory exudate and fibrous tissue, reducing significantly the intestinal lumen.
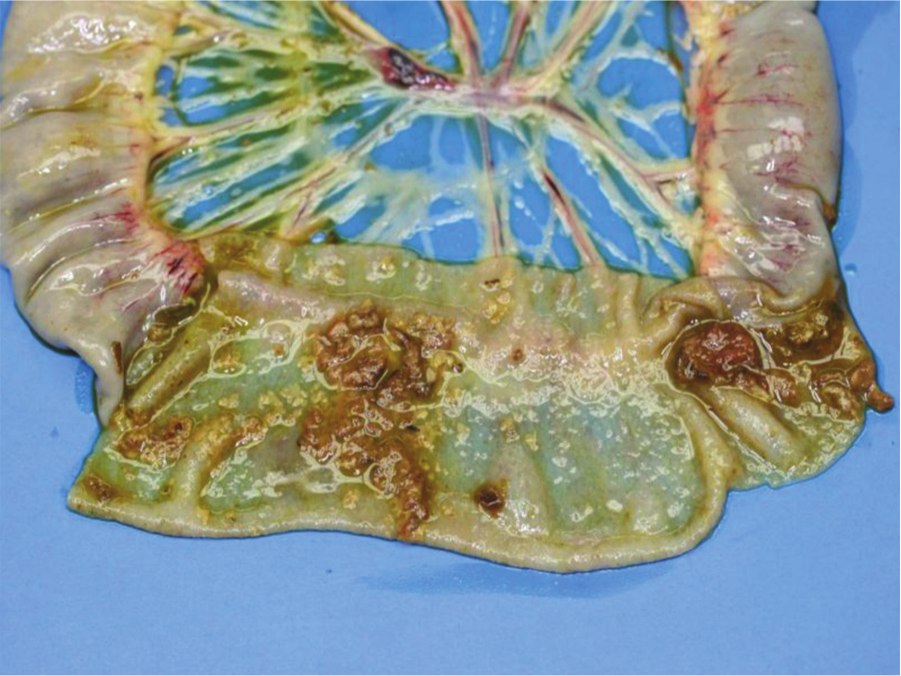
Jejunum, chicken. Subclinical necrotic enteritis. Multifocal to coalescing areas of mucosal necrosis covered by pseudomembranes can be seen on the intestinal mucosa.
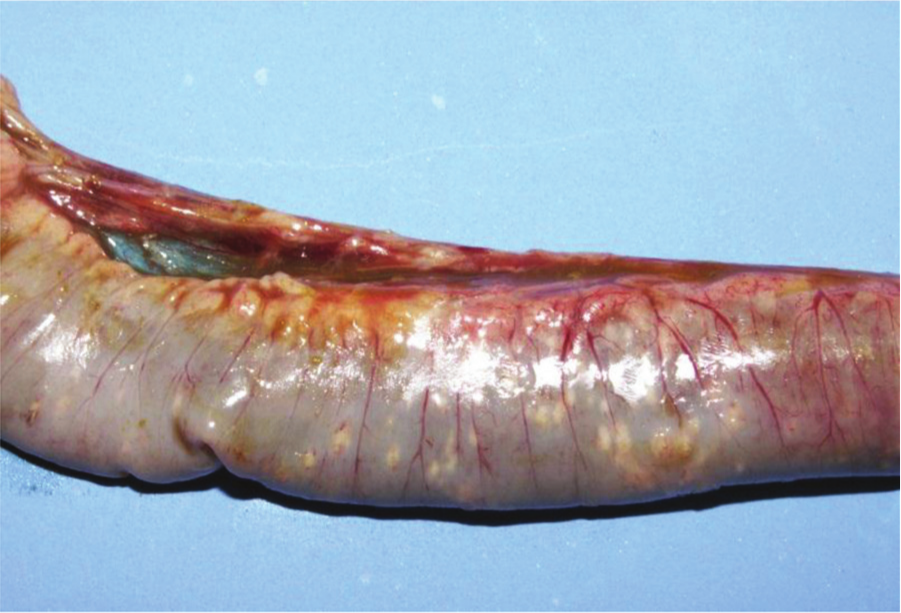
Jejunum, chicken. Subclinical necrotic enteritis. Multifocal pale foci of mucosal necrosis can be seen from the serosa.
Some chickens with NE also develop cholangiohepatitis, which is hypothetically caused by the increased numbers of
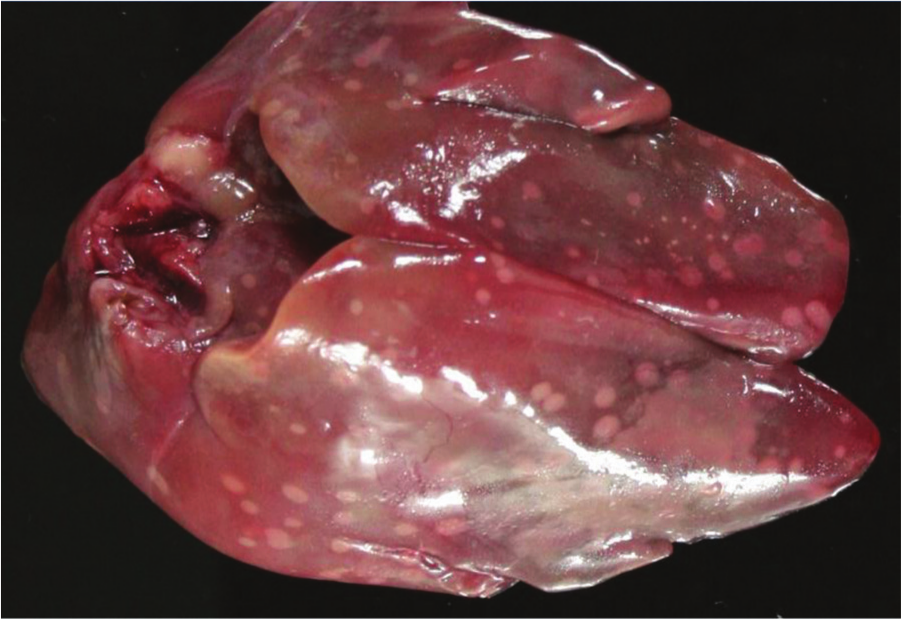
Liver, chicken. Clinical, acute necrotic enteritis. The liver contains multiple, scattered yellow necrotic foci. Photo courtesy of Dr. G. Sentíes-Cué.
Gross lesions of NE in turkeys are very similar to those in chickens,8,41,56,115 but can extend into the colon, often contain hemorrhagic debris, 42 and the duodenum is frequently more affected than in chickens. 56 In outbreaks of NE in ostriches, gross lesions comprise diffuse, small intestinal fibrino necrotic enteritis. The small intestine is dilated with gas and yellowish fibrinohemorrhagic fluid. 84
Histology
Microscopic lesions in field cases of NE are typically characterized by extensive mucosal necrosis (Fig. 7),24,51,83 although in some cases necrotic changes reach the submucosa,50,101 and in the most severe cases the necrosis extends into the muscularis mucosa.83,101 Lesions typically develop at the villus tips (Fig. 8), with coagulation necrosis and sloughing of the epithelial cells (Fig. 9).50,105,117 As lesions progress, there is a sharp line of demarcation between necrotic and viable tissue87,88,101 due to accumulation of inflammatory cells in outer areas of the viable tissue (Fig. 8).24,87,88 Heterophils are the dominant inflammatory cells infiltrating the mucosa in the initial stages of the disease,6,50,117 but mononuclear cells are also present in more chronic lesions.
88
In the lumen, there is fibrinonecrotic material, which frequently forms a pseudomembrane composed of cell debris, bacilli, and inflammatory cells trapped in fibrin (Fig. 10).4,64,88 Gram-positive rods with square ends are commonly associated with areas of necrosis.50,64,87 Immunohistochemistry has revealed
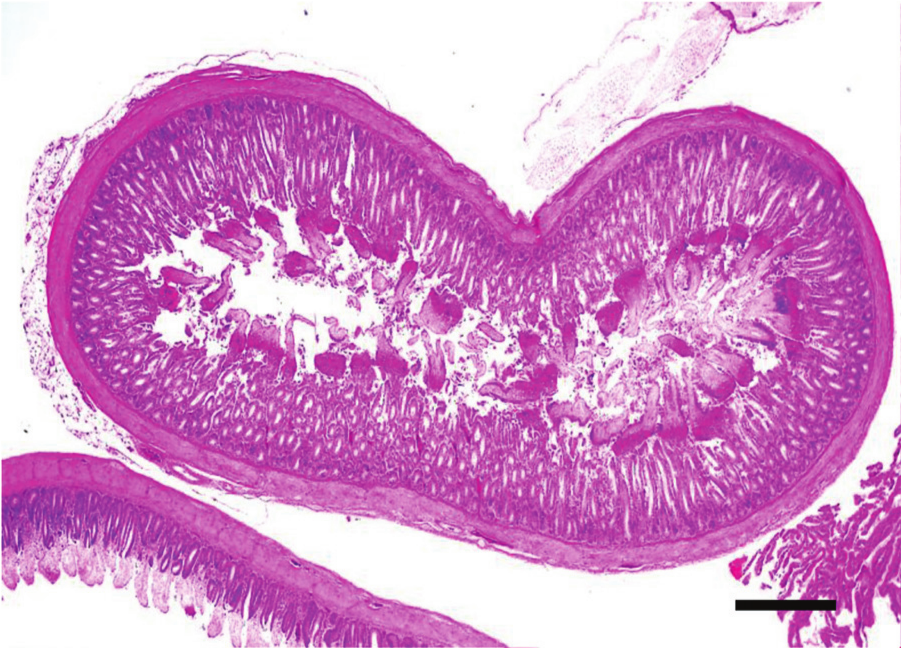
Jejunum, chicken. Clinical, acute necrotic enteritis. There is diffuse severe necrosis of the superficial intestinal mucosa with a clear line of demarcation from the more preserved deeper mucosa. Hematoxylin and eosin. Bar = 100 µm.
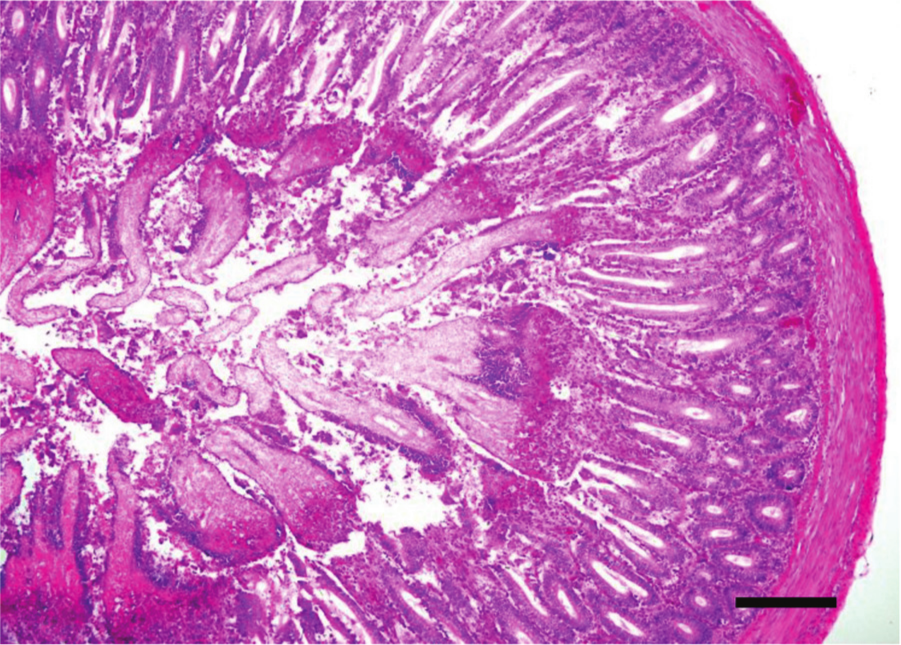
Jejunum, chicken. Higher magnification of Figure 7. Clinical, acute necrotic enteritis. There is diffuse severe necrosis of the superficial intestinal mucosa with a clear line of demarcation from the more preserved deeper mucosa. Large amounts of bacteria can also be seen. Hematoxylin and eosin. Bar = 50 µm.
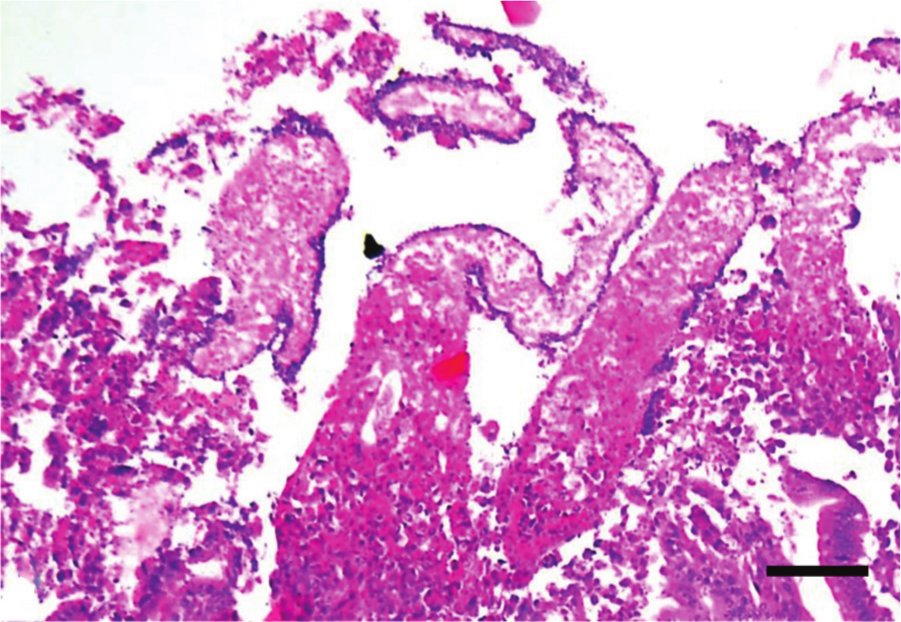
Jejunum, chicken. Clinical, acute necrotic enteritis. Observe diffuse necrosis and sloughing of the enterocytes at the tip of the villi. The denudated basement membrane is lined by rods. A belt of inflammatory cells can be seen at the bottom of the image. Hematoxylin and eosin. Bar = 20 µm.
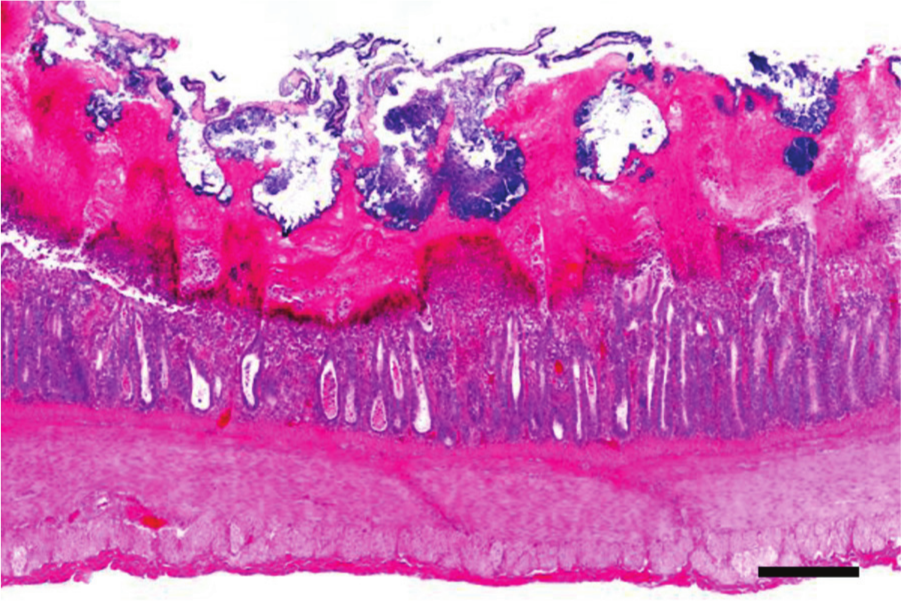
Jejunum, chicken. Clinical, subacute necrotic enteritis. The mucosa is necrotic and covered by a thick fibrinous pseudomembrane. The crypts are dilated and filled with necrotic debris. Hematoxylin and eosin. Bar = 50 µm.
In subclinical cases of NE, the intestinal necrosis is usually observed at the villous tips, and clusters of large Gram-positive rods are associated with the necrotic areas. 73 Heterophilic leukocytes create a sharp line of demarcation between necrotic and healthy tissue. 73
Regeneration of the intestinal tract in field cases of NE is characterized by proliferation of epithelial cells, beginning in the crypts, production of granulation tissue (Fig. 11), decreased numbers of goblet and columnar epithelial cells, and an increase in numbers of cuboidal cells. Overall, the regeneration process leaves the intestinal tract with short villi that have a reduced absorptive surface50,88; however, to the authors’ knowledge, the duration of this condition has not been investigated.
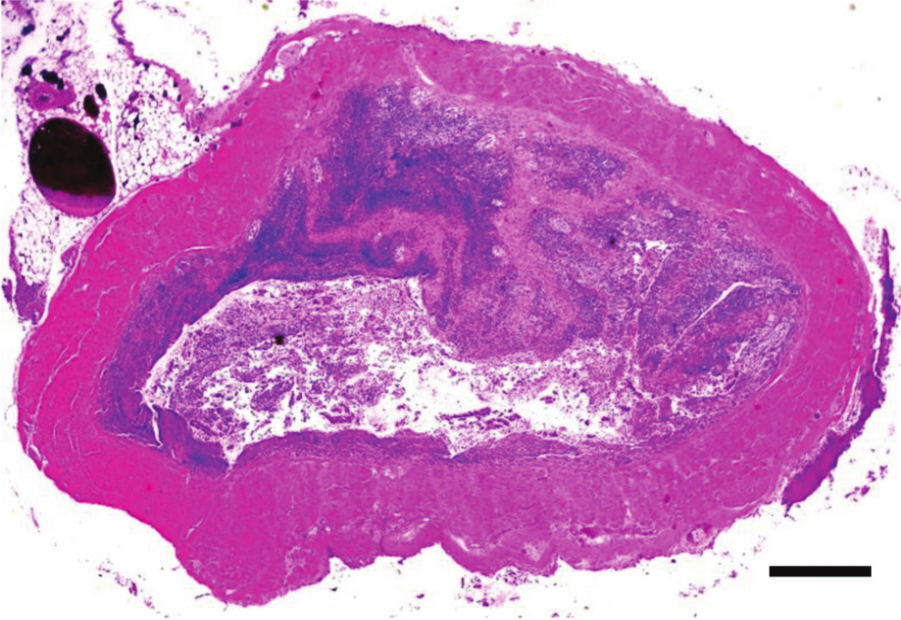
Jejunum, chicken. Clinical, chronic necrotic enteritis. The mucosa is necrotic and replaced by a thick layer of granulation tissue. Hematoxylin and eosin. Bar = 100 µm.
Occasionally, multifocal coagulative necrosis of the liver101,115 and bile ducts 74 is observed. A fibrinous exudate and Gram-positive bacilli are present in the necrotic areas (Fig. 12).74,106,115 In such cases, the gall bladder and extrahepatic bile ducts are thickened and distended with yellow inspissated material, 116 and there is hyperplasia in the bile ducts and occasional granulomatous inflammation.89,106,127
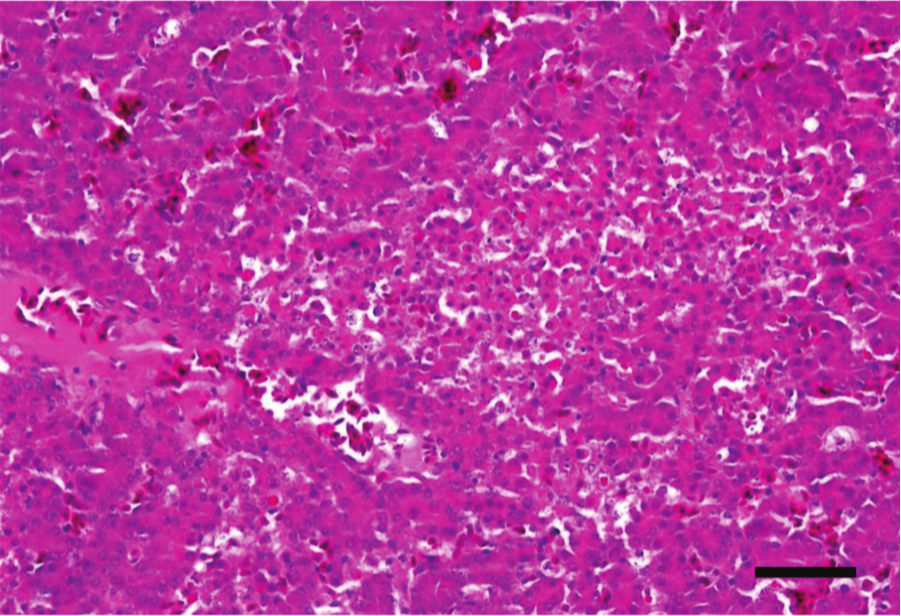
Liver, chicken. Clinical, subacute necrotic enteritis. There is a large, irregular, and more or less well-delimited paler area of necrosis in the center of the image. Hematoxylin and eosin. Bar = 20 µm.
Diagnosis and diagnostic criteria
A diagnosis of NE should be based on several criteria, and there is not a single test or finding that can confirm this diagnosis. On postmortem examination, focal, multifocal, or diffuse necrotizing lesions affecting the small intestine and occasionally the proximal colon and/or cecum are highly suggestive of NE. However, these lesions are not pathognomonic for NE, and a final diagnosis cannot be based on gross lesions alone. Histology is also helpful to establish a presumptive diagnosis of NE, but as is the case with gross lesions, it cannot be used to establish a final diagnosis. However, histology is particularly useful to rule out other conditions such as coccidiosis. Isolation of
Differential diagnosis
Coccidiosis is a known predisposing factor of NE and can occur prior to or simultaneously with the disease, and thus it must be differentiated from NE. Typically, coccidiosis in chickens will result in blood in the intestinal tract, which is rarely observed in cases of NE. 70 Histologically, in all avian species, the presence of oocysts at the tips of the intestinal villi or large schizonts in the lamina propria indicates coccidiosis, 4 whereas large Gram-positive rods in the lesions would indicate a clostridial infection such as NE or UE. 131
Ulcerative enteritis is another clostridial enteric disease that must be differentially diagnosed from NE and is described in detail in the following. In short, UE will present with ulcers in the small intestine that often perforate the intestinal wall and produce peritonitis and intestinal adhesions. Isolation of
Histomoniasis must also be differentiated from NE. Gross pathology of histomoniasis includes necrotic liver lesions with depressed centers and well-defined edges, as well as cecal lesions composed of necrotic mucosa, with blood and other debris. The observation of living organisms by phase contrast microscopy can assist in diagnosing histomoniasis. 97
Clostridium colinum (ulcerative enteritis)
Ulcerative enteritis is an infectious disease characterized by sudden onset and rapidly increasing flock mortality. Because the disease was first described in quail, it is also called quail disease. The disease has been described in quail,11,54 grouse,86,108 turkeys,27,118 pigeons, 59 young chickens, 118 pheasants, blue grouse, California quail, 28 other game birds, and occasionally other avian species. A single case in a lory has been described. 109 The disease has a worldwide distribution.
Etiology
Ulcerative enteritis is caused by
The best medium on which to isolate
Pathogenesis and host response
Ulcerative enteritis is transmitted by ingestion of feed, water, or litter contaminated with feces. In an outbreak of UE, the spores of
Bobwhite quail is the most susceptible species. Most cases are reported in captive populations (as opposed to wild quail), suggesting that management plays a role in the incidence of UE. 129
Chickens can be naturally infected, but experimental infections with
Clinical signs
In bobwhite quail, sudden death may occur without signs or with only weight loss observed. Mortality may reach 100% in just a few days. However, watery, whitish diarrhea is frequently observed, and as the disease progresses, depression and listlessness, with humped backs, ruffled feathers, and bloody diarrhea, are observed. In chronic cases, extreme emaciation is common. Mortality in chickens is usually lower and typically ranges from 2% to 10%. 13 The clinical signs of UE in chickens and other game birds are similar to those described for quail.
Gross lesions
Quail with acute UE have hemorrhagic enteritis affecting mainly the duodenum. At this stage, punctate hemorrhages may be visible from the serosa (Fig. 13). As disease progresses, larger, round, yellow ulcers surrounded by hemorrhages develop in any part of the small or large intestine and ceca (Fig. 14). These ulcers later coalesce to form larger, sometimes perforating, ulcers, with no discrete borders and surrounded by a pale halo (Fig. 15), causing local or diffuse peritonitis and intestinal adhesions. The ulcers may be lenticular or roughly circular and may be covered by pseudomembranes (Fig. 16). The lenticular shape is more common in the upper portion of the intestine. Blood is commonly found in the gut. Liver lesions vary from light yellow mottling to large, multifocal, irregular yellow areas. Multifocal gray or yellow circumscribed foci, sometimes surrounded by a pale yellow halo, can also be observed (Fig. 17). Spleen enlargement and hemorrhage with multifocal necrotic areas may be present. Gross lesions are usually absent from other organs. 129 The gross lesions in chickens and other avian species are similar to those described in quail. 129
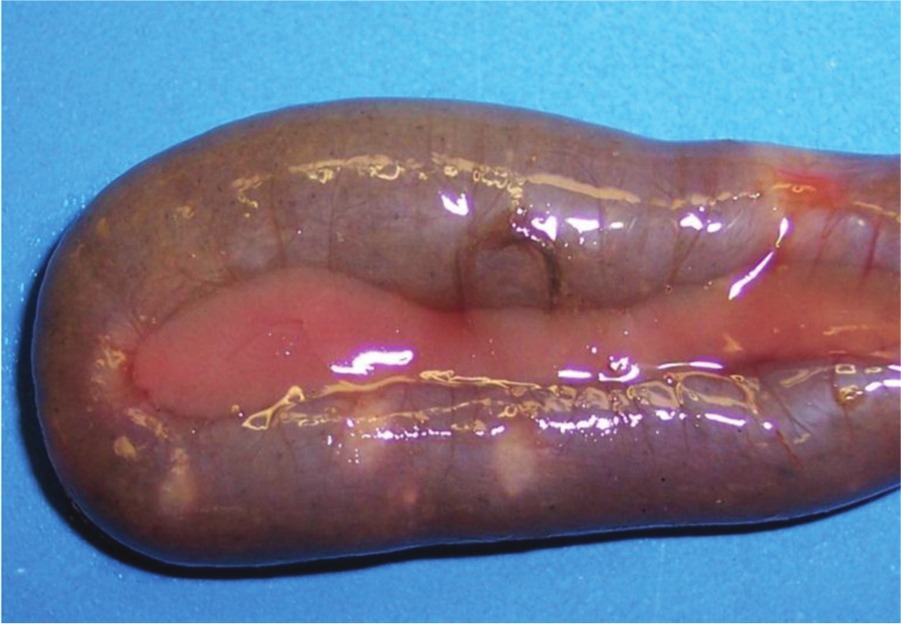
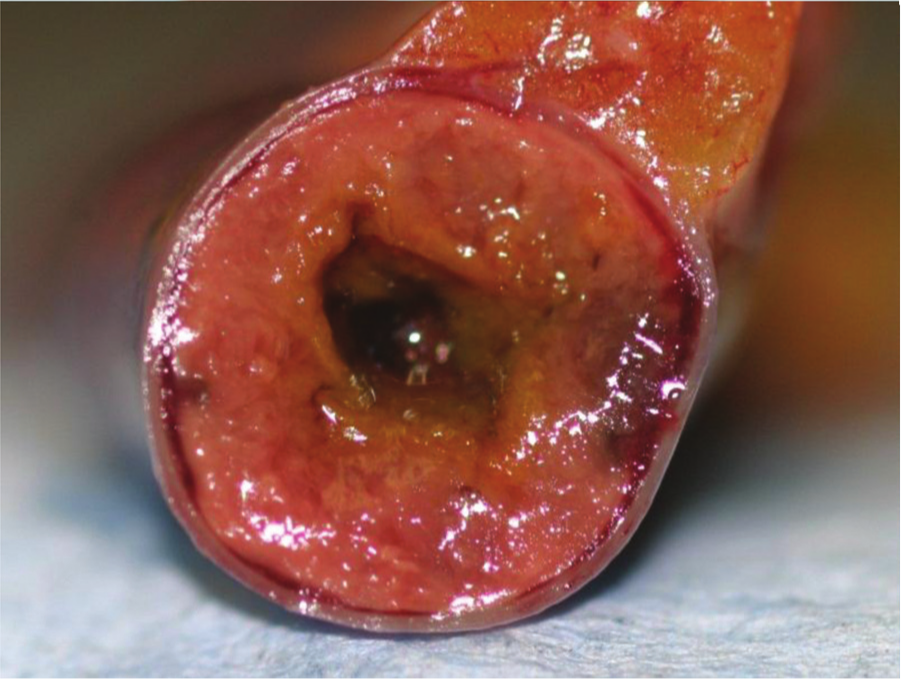
Duodenum, quail. Ulcerative enteritis. Multifocal pale foci of mucosal necrosis can be seen from the serosa.
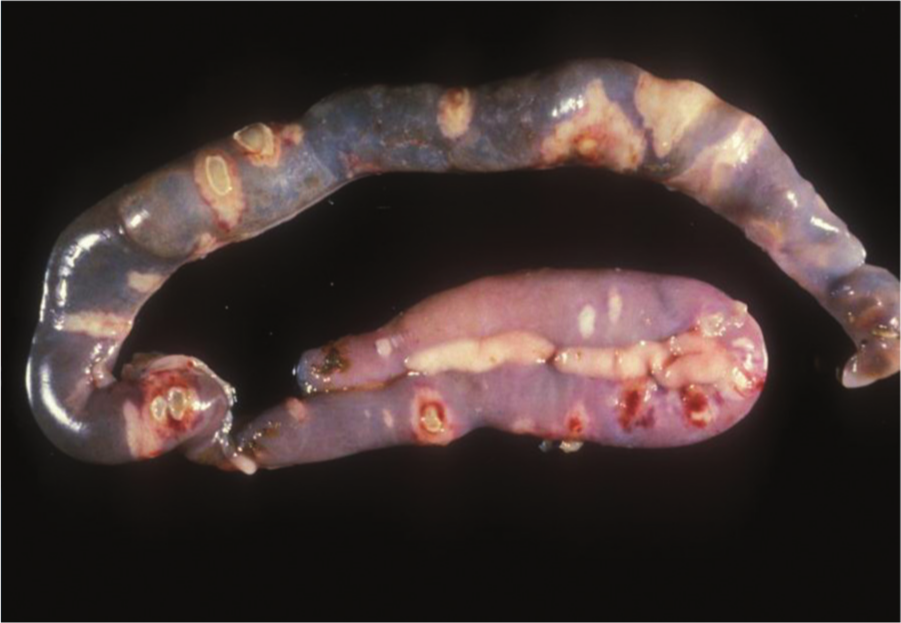
Duodenum and jejunum, quail. Ulcerative enteritis. Many multifocal transmural (perforating) foci of necrosis can be seen from the serosa. Photo courtesy of Dr. H. L. Shivaprasad.
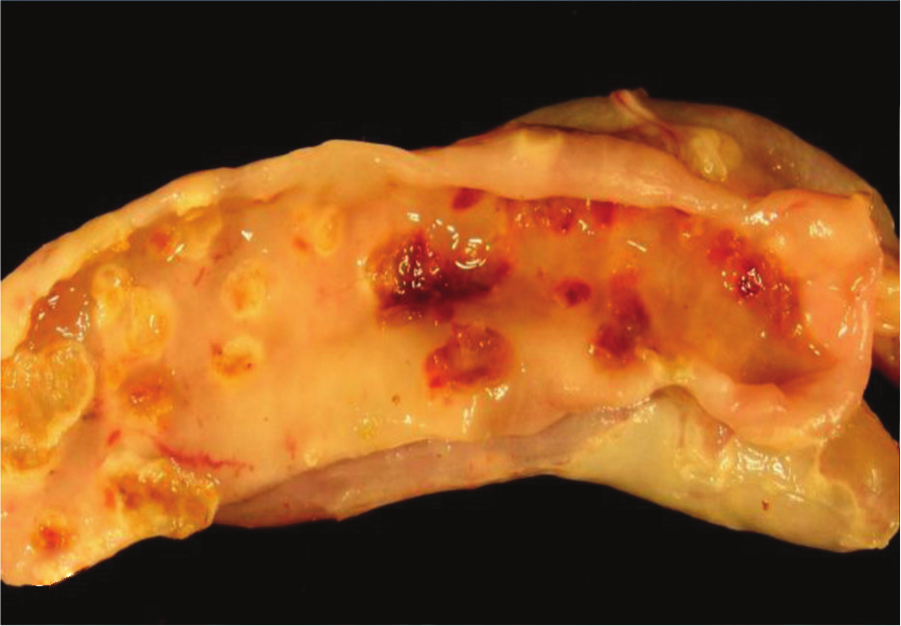
Jejunum, quail. Ulcerative enteritis. Many multifocal hemorrhagic ulcers surrounded by a pale halo can be seen on the mucosa. Photo courtesy of Dr. G. Sentíes-Cué.
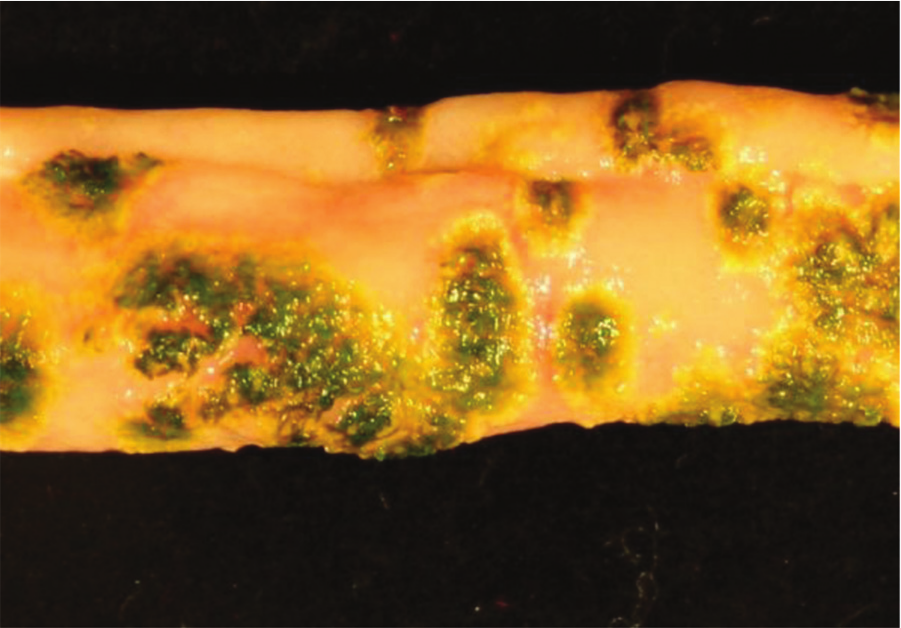
Jejunum, quail. Ulcerative enteritis. Many multifocal hemorrhagic ulcers covered by a green stained pseudomembrane and surrounded by a pale halo can be seen on the mucosa. Photo courtesy of Dr. G. Sentíes-Cué.
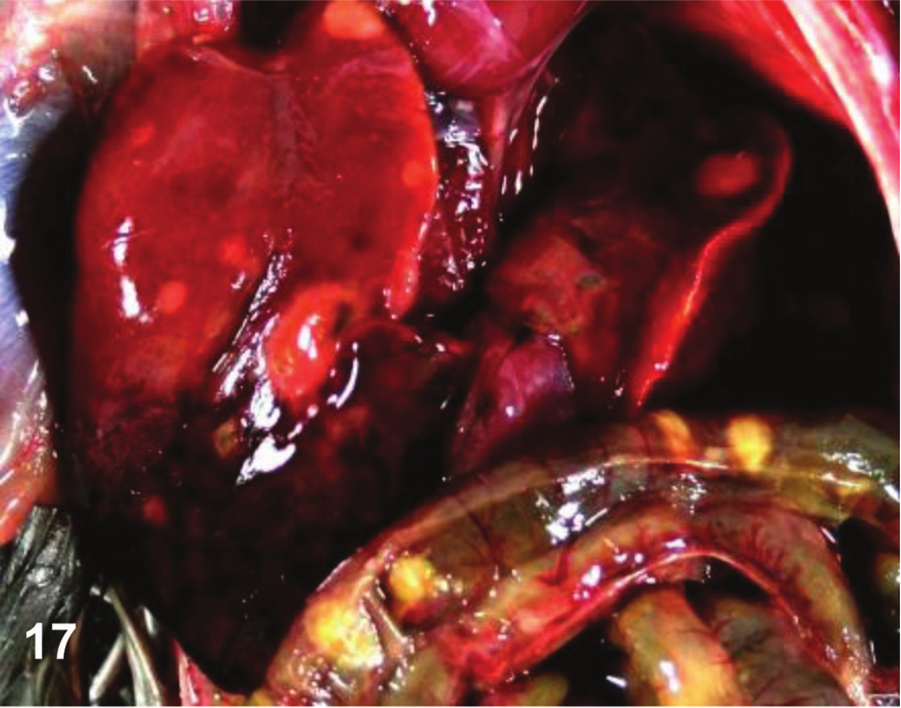
Abdominal cavity, quail. Ulcerative enteritis. Many multifocal hemorrhagic white foci of necrosis can be seen on the surface of the liver. Multifocal pale foci of mucosal necrosis can be seen from the serosa of the duodenum and jejunum. Photo courtesy of Dr. G. Sentíes-Cué.
Histology
Microscopic lesions in acute cases include diffuse necrosis and desquamation of the intestinal epithelium, mucosal edema, vascular congestion, and heterophil infiltration. In later stages, mucosal ulceration is observed (Fig. 18). Early ulcers consist of small hemorrhagic and necrotic areas involving villi and extending into the submucosa (Fig. 19). Areas of coagulation necrosis and a rim of heterophils, lymphocytes, plasma cells, and macrophages surround the ulcers. Clumps of Gram-positive rods are usually present in the areas of necrosis (Fig. 20). As the lesions progress, ulcers are covered by thick pseudomembranes composed of fibrin, cell debris, mixed inflammatory infiltrate, and bacilli. At later stages, transmural necrosis and inflammation can be observed. Vascular thrombosis in the mucosa is an almost constant finding in both the acute and chronic lesions. 129 Hepatic lesions consist of multifocal, random foci of coagulation necrosis, with mild mixed inflammatory infiltrate and, occasionally, small colonies of Gram-positive bacilli (Fig. 21). 129
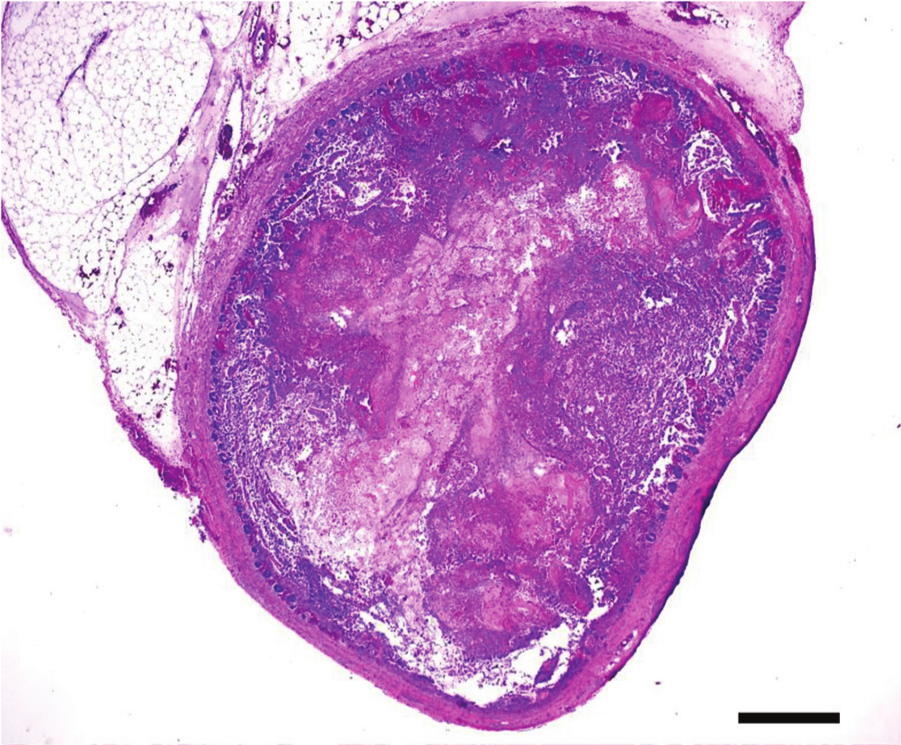
Jejunum, quail. Ulcerative enteritis. Multifocal to coalescing ulceration of the mucosa. Hematoxylin and eosin. Bar = 100 µm.
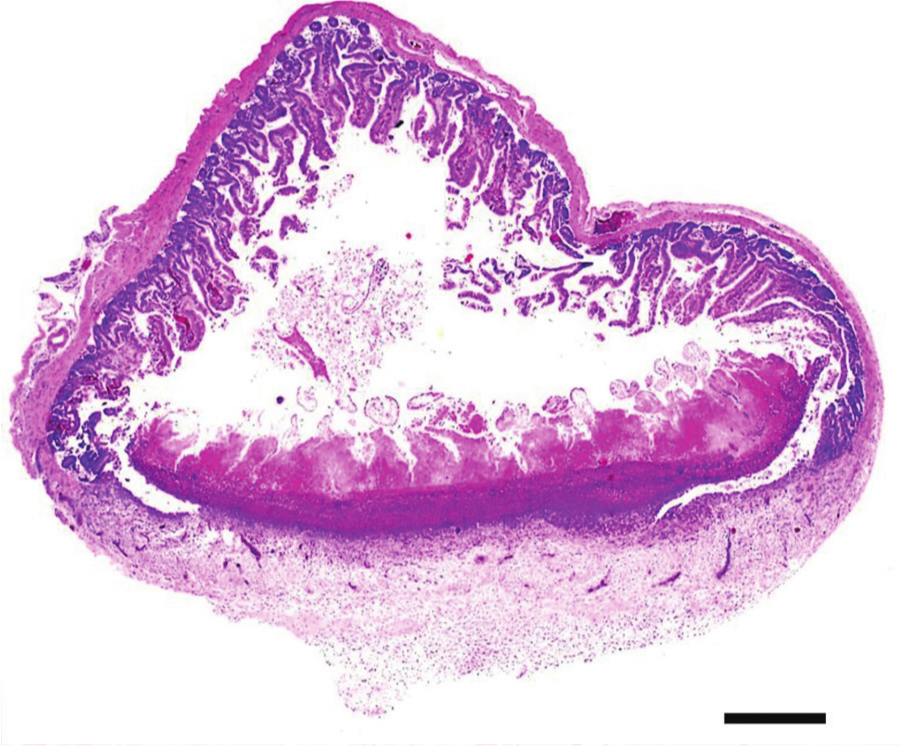
Jejunum, quail. Ulcerative enteritis. Focally extensive focal to coalescing ulceration of the mucosa, submucosa, and muscularis. Hematoxylin and eosin. Bar = 100 µm.
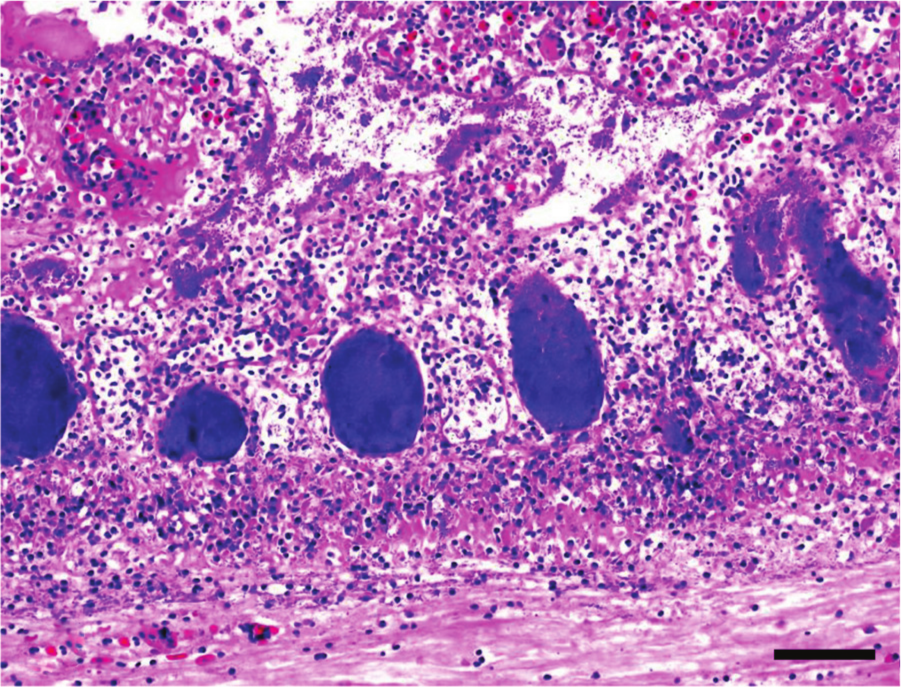
Jejunum, quail. Ulcerative enteritis. Focally extensive mucosal necrosis. Observe inflammatory exudate and the crypts filled with large blue bacterial colonies. Hematoxylin and eosin. Bar = 20 µm.
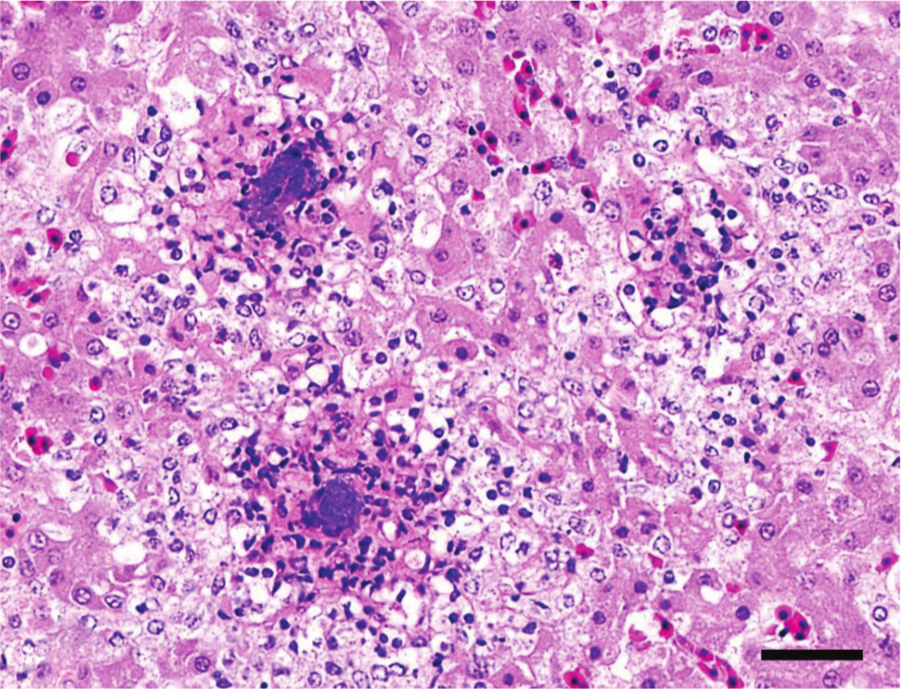
Liver, quail. Ulcerative enteritis. Multifocal hepatic necrosis with intralesional bacterial colonies. Hematoxylin and eosin. Bar = 20 µm.
Diagnosis and diagnostic criteria
A presumptive diagnosis can be based on clinical signs (increased mortality, usually with hemorrhagic diarrhea) and gross and microscopic lesions, including intestinal ulcerations and necrosis, and multifocal hepatic necrosis. The presence of large Gram-positive rods with subterminal spores in liver, spleen, and/or intestinal smears or histological sections strengthens a presumptive diagnosis. However, confirmation of the diagnosis must be based on isolation of
Differential diagnosis
Ulcerative enteritis must be differentiated from coccidiosis,
Histomoniasis produces caseous cores in ceca and necrotic areas of varying size in the liver of chickens, turkeys, and other gallinaceous birds. Differentiation between these 2 diseases can be made on the basis of an enlarged, hemorrhagic spleen and intestinal ulcerations, together with isolation of
Miscellaneous clostridial diseases
Clostridium difficile
Outbreaks of necrotizing enteritis in newly hatched ostriches can be caused by
Tentative diagnosis of the disease is based on clinical signs and gross and microscopic lesions. Isolation of large numbers of
Clostridium fallax
A single outbreak of hemorrhagic enteritis with a high mortality rate caused by
Clostridium baratii
A single outbreak of
Conclusion
Although the diagnostic criteria for clostridial enteric diseases of poultry still require refinement, researchers now have the knowledge and tools to study several important types of poultry enteritis. A prime example of this is the use of reverse genetics that has allowed researchers to establish beyond reasonable doubt that NetB is sufficient to cause NE in chickens. 75 This knowledge, however, still needs to be transformed into diagnostic tools that would allow researchers to confirm a diagnosis of NE in affected birds.
Also, further studies are recommended to investigate other possible predisposing factors and/or etiological agents of clostridial enteric diseases of poultry. It might be worthwhile to conduct metagenomics on some birds with enteritis in order to determine which organisms are present in highest numbers.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
