Abstract
Two putative zinc metalloproteases encoded by
Introduction
Necrotic enteritis imposes a heavy economic burden on poultry producers and has been estimated to cost the world poultry industry U.S.$6 billion a year.
39
The impact of necrotic enteritis has led to a surge in research on this disease
4
and on the causative bacterium,
The pore-forming toxin, NetB, is recognized as a major virulence factor, but there is still much to be learned about the pathogenesis of this complex, multifactorial disease.12,30 Few bacterial factors, other than NetB, have been shown to contribute to disease. A putative fimbrial operon has been shown to have a role in the adherence, colonization, and necrotic enteritis pathogenesis of
These 2 putative proteases belong to a family of M60-like/PF13402 domain–containing proteins 25 that carry a classical HEXXH gluzincin–like motif that is responsible for zinc-binding and acts as the catalytic core. 10 The proteins are usually secreted and are generally produced by bacteria that reside on or in the mucosal surfaces of their animal hosts. The proteins often also have accessory domains that have binding specificity for eukaryotic extracellular matrix molecules, specifically mucin, the primary constituent glycoprotein of mucosa.23–26,28 These M60-like proteases have been shown to degrade mucin and have been implicated in the disease processes of pathogens, especially enteric pathogens.23,24,28 Of the 2 putative zinc metalloproteases of this family implicated in necrotic enteritis, one, designated ZmpB, has been shown to be mucolytic. 26
The ~190 kDa zinc metalloproteases from
Little relevant
We report herein the prevalence of both
Materials and methods
Bacterial strains and growth conditions
The growth conditions of strains used in our study (Table 1) were identical to those described previously, 41 except that the brain–heart infusion (BHI) medium did not contain soluble starch. Growth curves were generated by inoculating 20 mL of BHI broth with 200 µL of an overnight culture and growth was monitored over 8 h, with 1-mL aliquots being taken every 2 h and the optical density at 600 nm determined. Growth curves were performed in triplicate.
Prevalence of
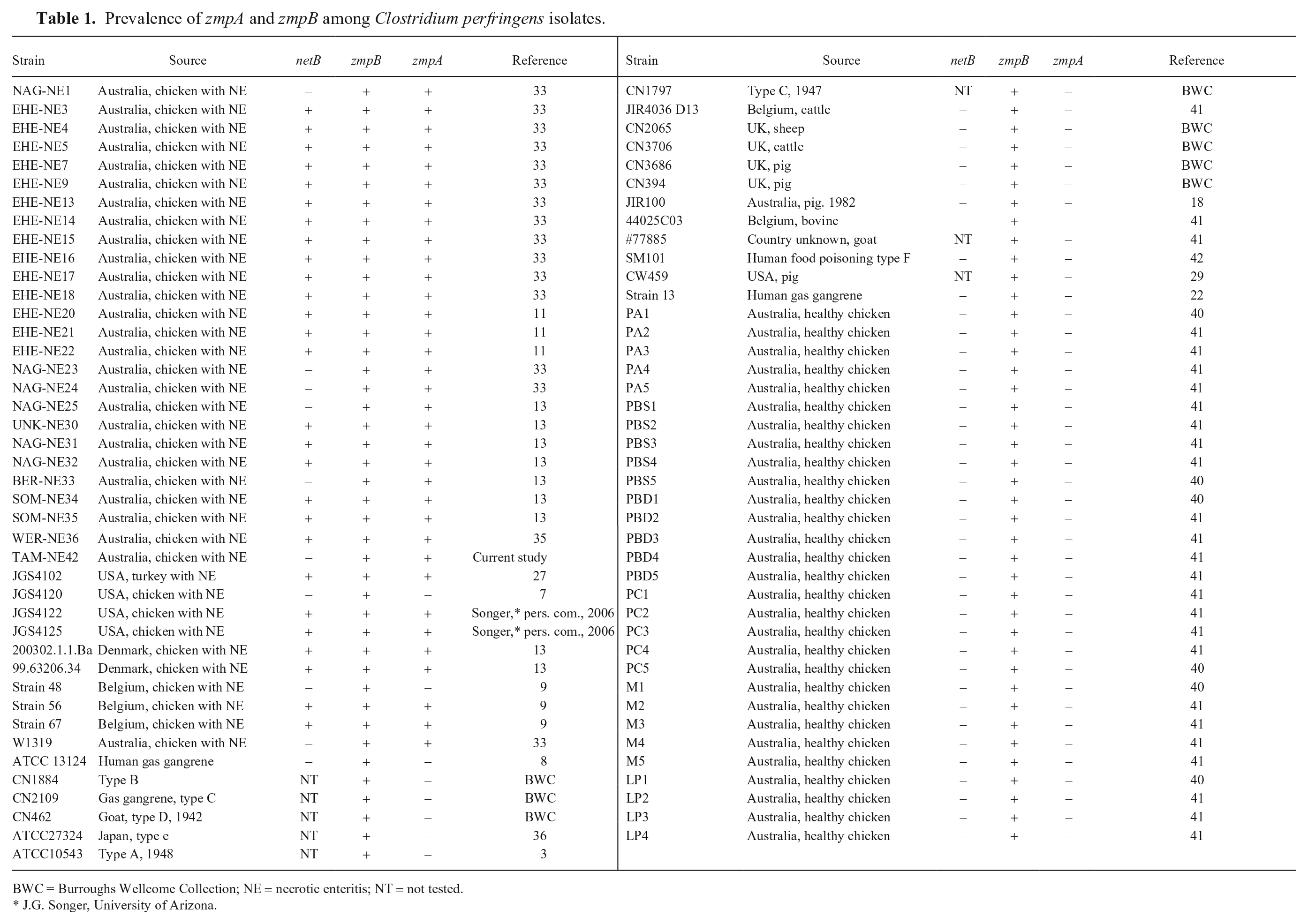
BWC = Burroughs Wellcome Collection; NE = necrotic enteritis; NT = not tested.
J.G. Songer, University of Arizona.
PCR for detection of zmpA and zmpB and for Sanger sequencing
Screening for
Targeted mutagenesis of zmpA and zmpB
The pALK1 vector was used as before11,41 for the generation of the suicide vector pBWP4. A 2,156-bp
The insertionally inactivated
High-throughput pyrosequencing
Strains EHE-NE18
The resultant sequences from the 2 mutants were compared to the wild-type EHE-NE18 genome (gsMapper of the Newbler package v.2.6; Roche). Predicted differences were collated and then filtered based upon the parameters that any variation must be present in ≥ 10 reads, and that the variation must be supported by > 75% of reads covering that sequence. The sequence differences that were found to be either non-synonymous, or had the potential to disrupt promoter sequences, were confirmed via Sanger sequencing.
Necrotic enteritis disease induction model
A necrotic enteritis induction protocol was used as described previously.
14
Briefly, unvaccinated 1-d-old Ross 308 birds were fed an antibiotic-free commercial starter feed. Animals were housed in 1.5 × 1.5-m pens with 3-cm deep wood shavings. On day 15, a wheat-based feed containing 50% (w/w) fishmeal was introduced. Feed mixed with a
Ethics statement
All animal experiments were undertaken with the approval and oversight of the CSIRO Australian Animal Health Laboratory Animal Ethics Committee (Approval AEC 1517) and in accordance with national (Australian Code for the Care and Use of Animals for Scientific Purposes, 2013) and state (Victorian Prevention of Cruelty to Animals Act 1986 and Part 4 of the Prevention of Cruelty to Animals Regulations 2008) legislation.
Statistical analysis
All statistical analysis was undertaken using Prism v.5.02 (GraphPad Software, San Diego, CA). The nonparametric Kruskal–Wallis test with Dunn post-test was used to statistically compare disease outcome results across all groups.
Results
Prevalence of zmpA and zmpB
We screened 83 isolates of
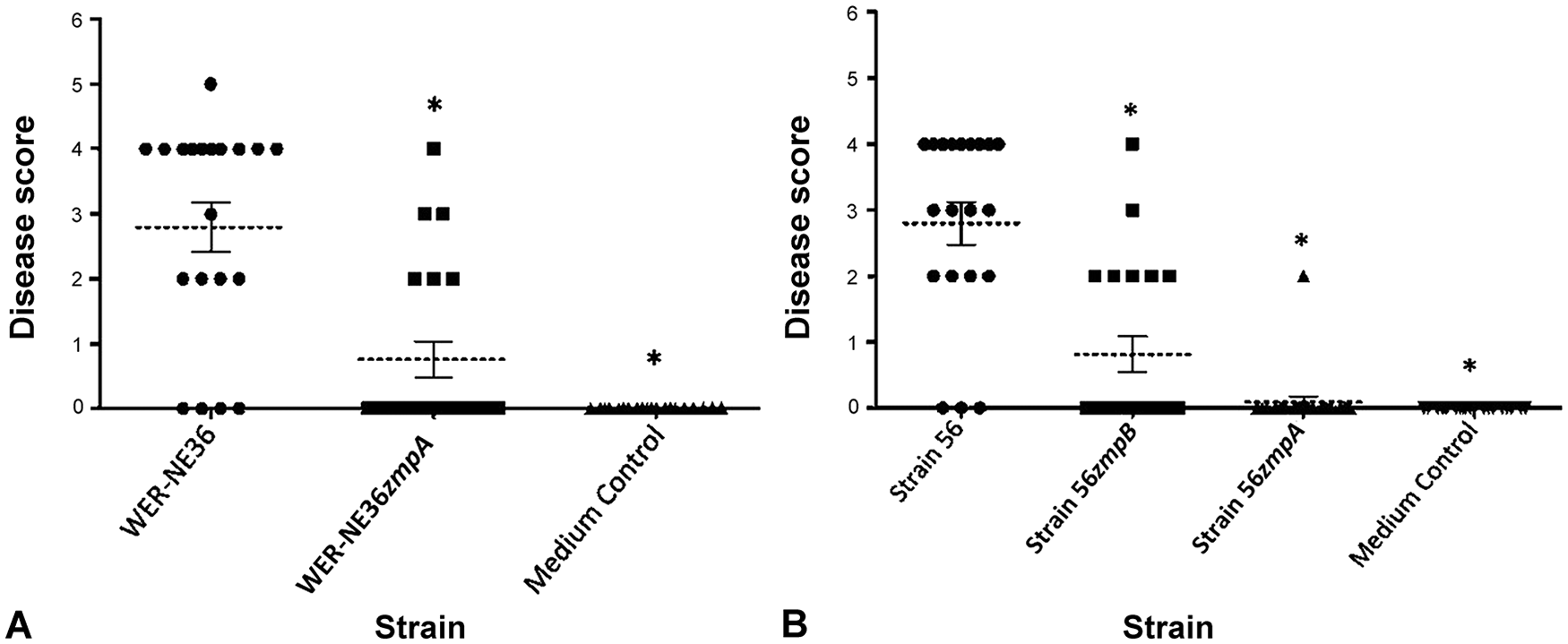
The zmpA and zmpB single and double mutants display reduced virulence
Given that
To investigate if
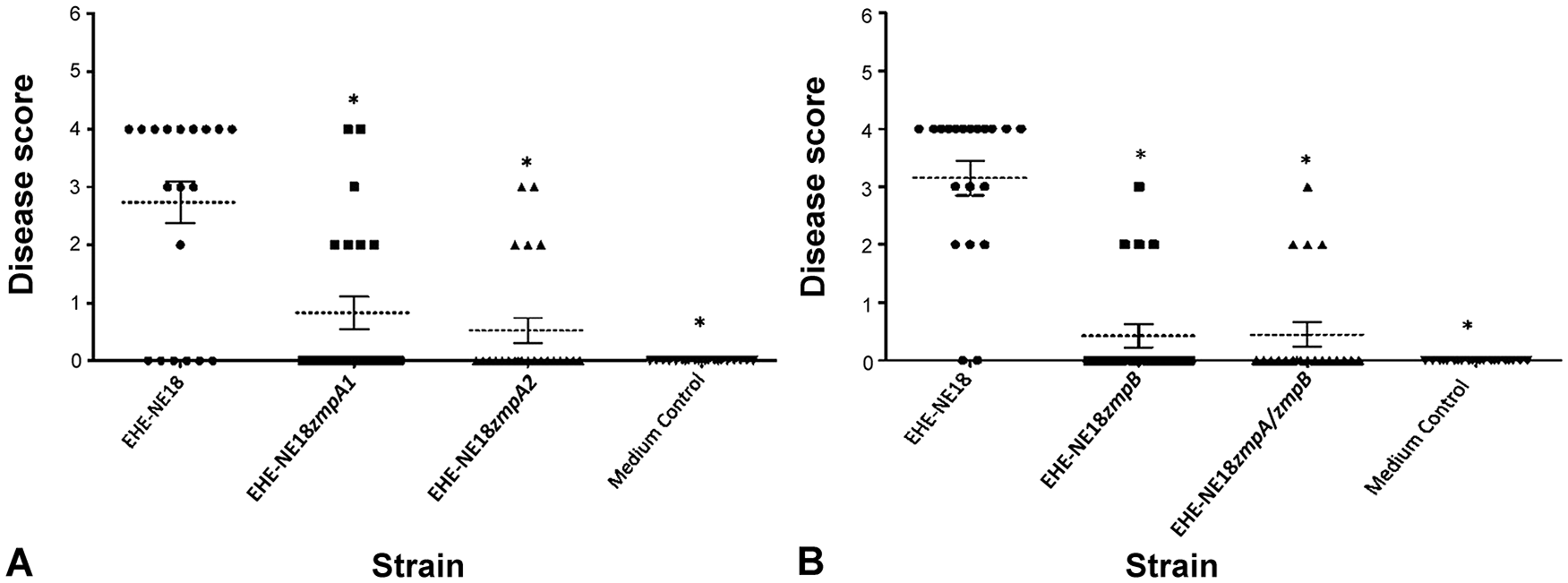
Both
Given that there was a significant reduction in virulence, but not complete abolition, we postulated that the
Both the
Evidence that the phenotypes of the mutants were the result of the targeted mutations
We made numerous attempts to clone the wild-type
Growth assays of the wild-type and the
To confirm that the phenotypic changes resulted from the mutations, independent
In addition, the whole genomes of both the EHE-NE18
In both mutant strains, 2 single-nucleotide polymorphisms (SNPs) were identified (Supplementary Table 2). In the
In the
The sequence data confirmed that there was only a single insertion of the antibiotic resistance marker into the
The occurrence in 2 independently derived protease mutants of 2 off-target mutations that potentially affected the same gene,
Discussion
Our study provides evidence that 2 putative zinc metalloproteases play a role in the ability of avian isolates of
Because it was not possible to clone either
The
Although it has been shown that ZmpB can degrade mucin, its exact affinity, in the context of the different types of mucin within the chicken intestinal mucosa, remains unknown, as does the precise target(s) of ZmpA. If the 2 proteins have different targets, we may have expected to see a cumulative impact upon virulence in the double mutant. Given the 64% amino acid sequence identity of ZmpA and ZmpB, it is possible that they have similar substrates and may be acting in a similar manner during necrotic enteritis. If so, it is difficult to understand why mutation of either gene would attenuate virulence so significantly and why only
We demonstrated that the zinc metalloprotease–encoding genes
Supplemental Material
Supplemental_material – Supplemental material for Two putative zinc metalloproteases contribute to the virulence of Clostridium perfringens strains that cause avian necrotic enteritis
Supplemental material, Supplemental_material for Two putative zinc metalloproteases contribute to the virulence of Clostridium perfringens strains that cause avian necrotic enteritis by Ben Wade, Anthony L. Keyburn, Volker Haring, Mark Ford, Julian I. Rood and Robert J. Moore in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank Honglei Chen for running the 454 pyrosequencing instrument. This work was supported by a PhD scholarship from CSIRO (B. Wade).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with regards to the research, authorship, and/or publication of this article.
Funding
This research was conducted within the Poultry CRC, established and supported under the Australian Government’s Cooperative Research Centres Program, with partial support from project funding 07-15.
Supplementary material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
