Abstract
The objective of the current study was to determine the incidence of contamination by the top 7 Shiga toxin–producing Escherichia coli (STEC) O-groups, responsible for the majority of E. coli infections in human beings, in retail meat from different animal species. Samples from ground beef (n = 51), ground pork (n = 16), ground chicken (n = 16), and game meat (deer, wild boar, bison, and rabbit; n = 55) were collected from retail vendors for the detection of 7 STEC O-groups (O26, O45, O103, O111, O121, O145, and O157). Meat samples were tested by using a multiplex polymerase chain reaction assay targeting the wzx gene of O antigen gene clusters of the 7 STEC O-groups. The positive samples were further tested for Shiga toxin genes (stx1 and stx2). Out of a total of 83 ground beef, pork, and chicken samples, 17 (20%) carried O121, 9 (10%) carried O45, 8 (9%) carried O157, 3 (3%) carried O103, and 1 (1%) carried O145. None of the samples were positive for O26, O111, or the stx gene. All 3 white-tailed deer samples (100%) were positive for O45, O103, or both, 2 (10%) out of 20 red deer samples exhibited the presence of O103, and all 3 bison samples were contaminated with either O121, O145, or O157. One sample from ground deer, contaminated with E. coli O45, carried the stx1 gene. This preliminary investigation illustrates the importance of microbiological testing of pathogens in meat products, as well as the recognized need for increased surveillance and research on foodborne pathogens.
Keywords
Shiga toxin–producing Escherichia coli (STEC) have emerged as important enteric foodborne zoonotic pathogens of considerable public health significance, worldwide. 8 STEC comprise a diverse group that elaborate one or both Shiga toxins (stx1 and stx2) and can cause diarrhea, hemorrhagic colitis, and hemolytic uremic syndrome in human beings.14,15 Meat obtained from farmed and wild game animals contaminated with STEC has the potential to cause infections in human beings, and while most of these E. coli infections are caused by E. coli O157:H7, 20–70% of STEC infections throughout the world are attributed to non-O157 STEC. 8 Of the 81 serotypes identified worldwide, 71% of the isolates recovered from human beings belonged to the “top 6” O-groups (O26, O45, O111, O103, O121, and O145). 8 STEC O157 has been considered an adulterant in beef produced in the United States for some time. In 2012, 6 other non-O157 STEC O-groups have been included as adulterants by the U.S. Department of Agriculture–Food Safety and Inspection Services (USDA-FSIS; http://www.fsis.usda.gov/PDF/Non_O157_STEC_Risk_Profile_May2012.pdf).
The Centers for Disease Control and Prevention estimates that STEC are responsible for 112,000 illnesses every year in the United States. 31 In Germany, more than 60% of the STEC O-groups isolated from food were also isolated from human patients, indicating the importance of food as a potential source of human infection, relative to other forms of transmission.11,23 Escherichia coli O157 can be easily distinguished from other E. coli strains using selective and differential media. 12 However, the non-O157 STEC do not have any such distinguishing physical or biochemical characteristics that readily differentiate them from the large number of harmless serotypes of E. coli that are part of the normal gastrointestinal flora. Therefore, the infectious capacity of non-O157 STEC strains, in human beings, has been difficult to assess and is probably underestimated. 31 Although contamination of ground meat with E. coli O157:H7 has been studied extensively, not much is known about non-O157 “top 6” STEC contamination in retail or game meat, produced under different processing conditions. Several surveys have indicated that game meats can be sources of these pathogens.2,3,16 Beef and a wide variety of wild and farmed game, consumed worldwide, are important sources of protein. In many countries, game meat may be processed under less-than-ideal sanitary conditions. Given the unknown incidence of “top 6” non-O157 STEC in the food supply, the aim of the current study was to assess the frequency of the top 7 STEC, including O157, contamination in retail and game meats.
Retail ground meat samples from beef (n = 51), pork (n = 16), and chicken (n = 16) were purchased at grocery stores, local farmers’ markets, and online vendors. The grocery stores were located in the states of Pennsylvania and Virginia. The online vendors located in Colorado (bison a ), South Carolina (rabbit b ), and New Jersey (deer and boar c ) did not indicate the geographical source of the meats. The samples obtained consisted of game meat (n = 55) from deer (n = 25), bison (n = 24), wild boar (n = 2), and whole rabbit carcasses (n = 4). The samples collected were held at 4°C until testing. Meat samples were enriched using previously described procedures with slight modification.12,26,33 Briefly, 5 subsamples weighing 5 g each were added to 45 ml of tryptic soy broth (TSB) containing 8 mg/l of novobiocin and 16 mg/l of vancomycin d followed by homogenization for 2 min using a stomacher. e Whole rabbit carcasses were aseptically transferred to a sterile bag and rinsed with 400 ml of Butterfield’s phosphate diluent according to USDA-FSIS protocol (http://www.fsis.usda.gov/OPHS/microlab/mlgchp3.pdf). Upon removal of the rabbit carcass in the stomacher bag, 50 ml of the liquid rinse was transferred into plastic test tubes and centrifuged at 12,000 × g for 10 min at 4°C. The pellet was resuspended in 20 ml of pre-enriched TSB and transferred into a stomacher bag containing 25 ml of TSB. All ground meat and rabbit rinse samples were incubated statically for 6 hr as a pre-enrichment step at 37°C, followed by incubation at 42°C for 18 hr with shaking after addition of bile salts (1.5 g/l), rifampicin (2 mg/l), and potassium tellurite d (1 mg/l). Each time a sample was prepared for STEC detection, an additional sample was inoculated with a known reference strain belonging to 1 of the top 6 STEC O-groups to serve as a positive control and subsequently subjected to the same enrichment procedures described above.
Bacterial DNA from 1 ml of the enriched culture was extracted with a commercial kit f according to the manufacturer’s instructions. The DNA pellet was rinsed twice with 200 µl of 70% ethanol, dried at room temperature for 5 min, and resuspended in 35 µl of Tris–ethylenediamine tetra-acetic acid (EDTA; 10 mM, pH 7.0) buffer solution. All DNA preparations were stored at −20°C until use. A multiplex polymerase chain reaction (mPCR) assay targeting the wzx gene of the O antigen gene clusters of O-groups O26, O45, O103, O111, O121, O145, and O157 was performed using the primers and procedure described previously. 9 The negative control was E. coli K12. The primers (2 µM) were mixed with commercial mPCR reaction solutions (47 µl) as described in the mPCR kit protocol g and were subjected to 95°C (15 min, 1 cycle) followed by 30 cycles at 94°C (30 sec), 57°C (90 sec), 72°C (90 sec); and extension at 72°C for 10 min using a thermocycler. h Amplified product (5 µl) was mixed with 1.5 µl of bromophenol blue and electrophoresed in 1.5% agarose gels in 0.5×Tris-borate-EDTA buffer for 45 min at 175 volts. The DNA was stained with ethidium bromide, and the amplicons were identified based on the size of the product using an ultraviolet transilluminator. Samples that were positive for the wzx gene were subjected to the mPCR assays targeting stx1 and stx2 genes as previously described. 24
Out of 51 ground beef samples, 18 samples (35%) tested positive for the presence of at least 1 of the STEC O-groups, of which 11 belonged to O121, 6 to O45, and 1 to O145. Out of 16 ground pork samples, 6 tested positive for O121, 1 sample for O157, and 1 belonged to O103. Out of 16 ground chicken samples, 12 (75%) were contaminated; 7 samples tested positive for O157, 3 samples carried O45, and 2 samples exhibited the presence of O103 (Table 1). None of the samples from ground beef, pork, and chicken exhibited the presence of E. coli O26 or O111, or stx1 or stx2. Since Shiga toxin genes were absent, these were not considered as Shiga toxin-producing strains. Out of the total of 83 samples, 38 (45%) from ground beef, pork, and chicken examined were found to be contaminated with 1 of the following O-groups: O45 (n = 9), O103 (n = 3), O121 (n = 17), O145 (n = 1), and O157 (n = 8).
Incidences of Shiga toxin–producing Escherichia coli O-groups in samples from ground beef, pork, chicken, and game meat.*
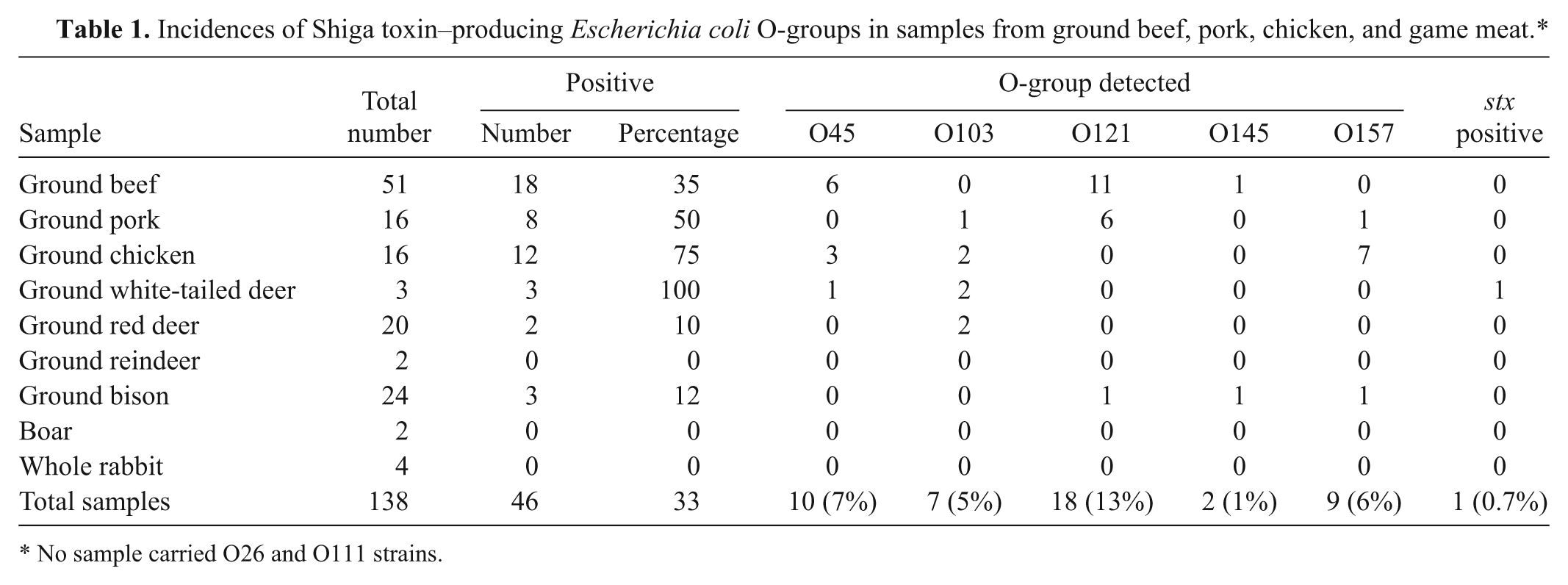
No sample carried O26 and O111 strains.
For game meat, all white-tailed deer (Odocoileus virginianus) samples (n = 3) were contaminated with strains belonging to O45 (n = 1) and O103 (n = 2), while 2 samples out of 20 (10%) from ground red deer (Cervus elaphus; n = 2) exhibited the presence of O103. Ground bison (Bison bison) samples (n = 3) tested positive for the presence of O145 (n = 1), O121 (n = 1), and O157 (n = 1). Reindeer (Rangifer tarandus; n = 2), boar (Sus scrofa; n = 2), and rabbit (Oryctolagus cuniculus) samples (n = 4) were negative for the O-groups tested. A total of 8 game meat samples out of 55 (14.5%) were found to be contaminated with at least 1 of the STEC O-groups (Table 1). Only 1 sample from white-tailed deer belonging to O45 exhibited the presence of the stx1 gene. No game meat samples belonged to groups O26 or O111.
Although numerous outbreaks of diseases caused by the top 6 STEC O-groups have been reported, it has been harder to identify the STEC O-groups as compared to E. coli O157 because of the lack of differentiating culture media or detection methods. Therefore, the burden of illness in human beings due to STEC O-groups may be higher than reported. 14 While the serogroups are important for determining potential pathogens, the presence of virulence attributes, such as stx1 and stx2 and the locus of enterocyte effacement (eae), are important parameters for pathogenicity of the strains. 19 In addition, the virulence factors associated with these strains are linked to either plasmids or phages. Consequently, they are likely to be subject to horizontal gene transfer between the species as exhibited by dissemination of plasmids in E. coli between pigs, farm workers, and the environment. 10
It has been challenging to determine the prevalence of the STEC O-groups in food, as there are only a few methods known for confirming the O-groups. The traditional method for detecting the O-groups is serotyping, which is labor-intensive and may provide equivocal results. A mPCR assay that can detect the presence of the top 7 STEC O-groups, including O157, has been reported. 9 Other laboratories also have developed PCR assays for detecting the O-groups.5,12,21 Because the detection limit of the mPCR used 9 in the current study was 10 colony-forming units and the meat samples were enriched, the mPCR assay utilized should have detected the targeted serogroups, although the strains were not isolated using standard culture methods. It has been reported that rifampicin decreases stx1 and stx2 expression at both the transcription and protein release levels, while novobiocin may prevent optimal growth of STEC O111.12,27 It has been ascertained that novobiocin (8 µg/ml) in the modified TSB medium can slow the growth of STEC O111 strains and inhibit non-O157 STEC strains to a larger extent than E. coli O157:H7 isolated using 20 µg/ml of novobiocin.12,34 It is important to remember that false-negative results can occur when testing for non-O157 STEC in food.12,34
Escherichia coli O157:H7 is associated with most cases of disease outbreaks, and hamburgers have been a major source of foodborne infections. 15 Wide ranges of prevalence of E. coli O157, from 0.01% to 54.2%, in beef in the United States and worldwide have been reported. 17 The prevalence of E. coli belonging to the top 6 STEC O-groups in beef and other meats has not been extensively studied. Based on previous research, non-O157 STEC were found to be more common than STEC O157 in beef, with a range of 1.7–62.5% in beef products. 17 The prevalence of non-O157 STEC in packing plants and retail beef sold in the United States also varies considerably,1,6,17 with a range of 10–30% in imported and domestic boneless beef trim used for ground beef. 7 In the present study, although 35% of ground beef samples exhibited the presence of 1 of the STEC O-groups, predominantly O121, none of the strains carried the stx gene and, therefore, would not be considered as adulterant. In Uganda, 70% of the STEC strains belonging to different serotypes from cattle and diarrheic children were eae positive, and 2 of the strains from human beings were related to cattle isolates. 22 Meat contamination with STEC O157 and non-O157 strains in Casablanca, Morocco, was found to be 0.9%. 4 In pork, the prevalence of STEC is lower and is estimated to be 18% in the United States and 15.3% in Canada. 14 However, STEC from pigs are not known to cause disease in human beings. 14 In the current investigation, while 50% of the pork samples exhibited STEC O-groups (mostly O121, O45 [1 sample], and O157 [1 sample]), none of them carried the stx gene and were, therefore, nonpathogenic. Poultry products appear to carry little risk, with prevalence rates of STEC in these products at less than 1%.25,28 At retail locations in the United States, 6.7% of turkey samples and 12.1% of poultry samples were STEC positive, 29 possibly due to postproduction cross-contamination during handling and packaging. Of the poultry samples in the present study, 81% carried the STEC O-groups, mainly O157, O45, and O103, although none of them were stx positive.
In wild game, prevalence rates of 1–15% for non-O157 STEC from fecal samples from deer have been reported in Japan.2,13 The occurrence of STEC strains in feces from sheep and goats was found to be 23% and 26%, respectively, in a study conducted in Wisconsin in the United States. 18 In Spain, out of 243 wild ruminant samples analyzed, 23.9% were found to be positive for STEC. 30 In Argentina, 50.8% fecal samples from nondomesticated mammals in a zoo were STEC positive. 20 In a study in Germany, STEC strains were frequently present in meat samples from sheep (11.1%), wild game (9.9%), cattle (4.5%), pork (0.68%), and dairy products (0.6% and 1.8%). 23 Prevalence of E. coli O157:H7 in a northern province of South Africa was reported to be 67.7% in pigs and pork meat, 27.7% in cattle and beef meat, and 0.77% in human beings. 3 In a 2012 study on the presence of STEC strains in red and roe deer, chamois, and ibex, 11 strains out of 52 exhibited the presence of the top 8 STEC O-groups (the top 6, O91, and O113). 16
In the current study, samples from red deer and white-tailed deer exhibited the presence of O45 and O103, and meat from bison was contaminated with O121, O145, and O157. Only 1 white tailed-deer sample was contaminated with E. coli O45 carrying the stx gene. Limitations were encountered related to diversified sample acquisition, leading to difficulties in designing sampling strategies that adequately represented the population of interest. Given this nonideal scenario, the sample collection in the present study represented a compromise associated with availability. 32 The knowledge generated from the study, which showed a high incidence of STEC O-groups in unregulated meat, suggests further scrutiny and testing may be necessary to avoid foodborne disease outbreaks due to consumption of game meat.
Footnotes
Acknowledgements
The project was conducted at the Pennsylvania State University, E. coli Reference Centre. Kudakwashe Magwedere and Huu Anh Dang contributed equally to the project. Many thanks to Professor Louw C. Hoffman from Stellenbosch University for his assistance in the application process of the Borlaug fellowship (K Magwedere).
a.
Rocky Mountain Natural Meats LLC, Henderson, CO.
b.
Ardeng Rabbit, Fountain Inn, SC.
c.
D’Artagnan Inc., Newark, NJ.
d.
Sigma-Aldrich, St. Louis, MO.
e.
Cook Laboratory Products, Pico Rivera, CA.
f.
Masterpure DNA Purification kit, EPICENTRE Biotechnologies, Madison, WI.
g.
Qiagen Multiplex PCR kit, Qiagen Inc., Valencia, CA.
h.
Eppendorf AG, Hamburg, Germany.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors acknowledge receiving funding for the project from USDA Foreign Agricultural Services, Cooperative Agreement nos. 58-3148-0-136 (E Mills and CN Cutter) and 58-3148-0-217 (C DebRoy).
