Abstract
Accurate diagnosis of exposure to Actinobacillus pleuropneumoniae is important for maintaining negative farms. In the present study, the ability of a dual-plate complement fixation (CF) assay and 3 commercially available enzyme-linked immunosorbent assays (ELISAs; quad-plate ELISA-1, single-plate ELISA-2, and single-plate ELISA-3) in detecting serological evidence of A. pleuropneumoniae exposure was compared using serum samples of experimentally infected or vaccinated pigs, or field samples from the United States. Forty-two pigs were divided into groups of 2 pigs and were inoculated with 1 of 15 A. pleuropneumoniae strains representing all known serovars of A. pleuropneumoniae, or with Actinobacillus suis, or were vaccinated with a bacterin containing A. pleuropneumoniae serovar 1, 3, 5, or 7. Serum samples collected at the day of inoculation or vaccination and 7, 14, 21, and 28 days later were used to compare the assays. On samples from experimentally infected pigs, the dual-plate CF assay, quad-plate ELISA-1, single-plate ELISA-2, and single-plate ELISA-3 had sensitivities of 0.46, 0.74, 0.13, and 0.13 and specificities of 0.90, 1.0, 1.0, and 1.0, respectively. Vaccinated pigs were identified only by the dual-plate CF assay and the quad-plate ELISA-1. In addition, 90 serum samples with unknown A. pleuropneumoniae exposure collected under field conditions were tested with all assays. The agreement of the 4 assays on field samples was slight to fair. While several assays are available for demonstration of A. pleuropneumoniae exposure, differences in assay targets complicate test choices. Decisions on which assay or combination of assays to use depend on the specific reasons for running the assays.
Keywords
Introduction
Actinobacillus pleuropneumoniae is a primary respiratory pathogen and one of the leading causes of bacterial pneumonia in pigs globally. 30 In naïve herds, A. pleuropneumoniae is often associated with severe respiratory disease and high mortality rates. 18 Actinobacillus pleuropneumoniae can infect pigs of all ages, but clinical disease is mainly seen in 12–16-week-old pigs. This is suggestive of loss of protection due to waning of passively acquired antibodies, which can occur anytime between 5 to 12 weeks.6,8,46 The different A. pleuropneumoniae serovars vary in their ability to induce clinical disease; however, with some of the more virulent serovars, clinical signs may develop as quick as 4–12 hr postinfection. 30 Actinobacillus pleuropneumoniae immunoglobulin M (IgM) antibodies have been detected as early as 1 week after experimental infection. 25 Seroconversion and detectable levels of A. pleuropneumoniae IgG antibodies have been reported to appear between 1 to 3 weeks 7 or 2 to 4 weeks25,29 after experimental infection. The levels of IgG antibody typically increase 2 weeks after A. pleuropneumoniae vaccination 21 but titers vary depending on the type of adjuvant utilized. 21
Actinobacillus pleuropneumoniae is a Gram-negative, nonmotile, non–spore-forming, coccoid, or small rod-shaped bacterium that can be divided into 2 biovars: biovar 1 depends on nicotinamide adenine dinucleotide (NAD) for growth in vitro, whereas biovar 2 does not. 34 Currently, A. pleuropneumoniae of both biovars can be classified into 15 recognized serovars. 37 The serovar specificity resides mostly in capsular polysaccharides, but also in O-chain lipopolysaccharide (LPS) for some serotypes.1,2,11 Serology assays have been developed that detect antibodies directed against capsular polysaccharides and/or O-chain LPS to determine a humoral response against A. pleuropneumoniae.19,23
Actinobacillus pleuropneumoniae produces a number of toxins, any of which may be present in pathogenic strains. Presence of the RTX (repeats in the toxin) toxins is strongly correlated with virulence,2,3 and the main RTX toxins include ApxI, ApxII, ApxIII, and ApxIV. While all A. pleuropneumoniae serovars produce ApxIV, ApxI is produced by serovars 10 and 14; ApxII is produced by serovars 7, 12, and 13; ApxI and ApxII are produced together by serovars 1, 5, 9, and 11; and ApxII and ApxIII are produced together by serovars 2–4, 6, 8, and 15. 10 Antibodies against ApxI and ApxII may also be the result of infection with other Actinobacillus species such as A. suis. 10 However, ApxIV is thought to be highly specific for the species A. pleuropneumoniae. 38 Seroconversion to A. pleuropneumoniae traditionally has been determined by using the ability of antigen–antibody complexes to fix complement.22,31 A different approach to confirm seroconversion to A. pleuropneumoniae is to measure antibodies against selected RTX toxins. This principle is being utilized by several commercially available enzyme-linked immunosorbent assays (ELISAs) of which at least 1 specifically targets antibodies against ApxIV. 10
Assays targeting ApxIV 13 or long O-chain LPS 7 have been previously compared, and it was demonstrated that ELISAs utilizing different antigens or assay conditions had conflicting results, 12 which complicates determination of the true status of the pig. The objective of the current study was to directly compare 4 serological assays: a complement fixation (CF) assay, currently required for live animal movement into the People’s Republic of China, and 3 commercially available ELISAs for detection of antibodies against A. pleuropneumoniae in serum samples obtained from experimentally infected or vaccinated pigs (known exposure) and pigs under field conditions (unknown exposure).
Materials and methods
Experimental samples from pigs with known A. pleuropneumoniae exposure
Animals, housing, and experimental design
The experimental protocol was approved by the Iowa State University Institutional Animal Care and Use Committee (1-11-7071-S). Forty-two, 2–8-week-old pigs were obtained from a herd confirmed to be free of A. pleuropneumoniae by routine serology and bacteriology testing and without clinical signs of pneumonia. In the source herd, pigs are typically weaned at 2 weeks of age and put on a nursery feed ration. The pigs were transported to a biological safety level 2 facility at Iowa State University (Ames, Iowa) and kept in groups of 2 in 21 separate 2 m × 2.5 m rooms. Each room was equipped with a single nipple drinker, and pigs were fed a pelleted feed ration a that was free of antibiotics. Experimental vaccination or challenge was conducted after an acclimation period of 7 days in the research facility (3–9 weeks of age). Due to the small size of the source herd, the pigs ranged in age by 6 weeks upon arrival in the research facility. To address this, young pigs were assigned to the vaccinated groups and pigs 5 weeks and older were assigned to the challenged groups. After blocking by age, the pigs were randomly divided by gate cut into the groups and rooms. Thirty-four pigs were experimentally inoculated using 2 ml of 1 of 15 A. pleuropneumoniae strains corresponding to serovars 1–15 or A. suis intranasally at 5–9 weeks of age. Two pigs were used for each serovar, with the exception of serovar 4, which was used to infect 4 pigs. Eight pigs were vaccinated intramuscularly at 3 weeks of age and again 2 weeks later with 2 ml of 1 of 4 A. pleuropneumoniae bacterins produced in-house based on either serovar 1, 3, 5, or 7. To avoid cross-contamination between groups and to simplify the monitoring process for the observers, blocks of 3 groups of 2 pigs were infected at the same time. In addition, sentinel pigs, which were not part of the present study but belonged to other concurrent viral studies, where housed in adjacent rooms.
Inoculum preparation and inoculation
The following A. pleuropneumoniae strains corresponding to serovars 1 (American Type Culture Collection [ATCC] 27088), 2 (ATCC 27089), 3 (ATCC 27090), 4 (4 pigs with ATCC 33378), 5 (ATCC 33377), 6 (VMRI-11-6-1985), 7 (WF 83), 8 (F384), 9 (F60), 10 (13039), 11 (56153), 12 (9499/84), 13 (N273 biovar 11 Fodor), 14 (3906 biovar 2), and 15 (HS143), or A. suis (ISU-2011030208) were used for inoculation. Confirmation of the serovar of the A. pleuropneumoniae strains used in this study was carried out with slide agglutination tests using rabbit hyperimmune sera against reference serovars 1–12 of A. pleuropneumoniae according to standard protocols utilized in the Veterinary Diagnostic Laboratory at Iowa State University. All isolates belonging to serovars 1–12 used in this study reacted with the correct antisera. Serovar 13–15 isolates were sourced from, and confirmed by, Dr. M. Gottschalk in Canada. In addition, due to slight discrepancies with the obtained results, A. pleuropneumoniae strain ATCC 33378 (serovar 4) was also sent to Dr. M. Gottschalk in Canada for further serovar confirmation with polyclonal and monoclonal antibodies. Each strain was grown on chocolate agar b at 37°C in a 5% CO2 chamber for 24 hr. Harvested bacteria were diluted in saline solution to McFarland 0.5. Serial dilutions were used to calculate the final concentration. 36 Initially, each animal was inoculated with 2 ml of one of the different bacterial strains, which contained 1 × 108 (serovars 1–3) colonies, by slowly dripping 1 ml of the inoculum into each nostril. Because severe clinical disease was observed after the first inoculations with serovars 1–3, the dose was reduced to 1 × 106 for all subsequent challenges using the remaining serovars.
Bacterin production and vaccination
Actinobacillus pleuropneumoniae strains corresponding to serovar 1, 3, 5, or 7 (same strains as named above expect that strain MG 131 was used for serovar 5) were used for bacterin production. For each strain, 3 bacterial colonies were diluted in 0.15 ml of PPLO (pleuropneumonia-like organism) broth b and incubated for 18 hr at 37°C. The resulting culture was added to 50 ml of PPLO broth. b The broth was incubated for 18 hr at 37°C, which was followed by inactivation with 2% formalin for 4 additional hours followed by subculture to assure successful inactivation. Next, 1.5 ml of a commercially available aluminum hydroxide adjuvant c was added to the broth. The resulting bacterins were incubated on chocolate agar b plates to verify successful inactivation. Each animal was vaccinated at day 0 and revaccinated 2 weeks later with 2 ml of the bacterin intramuscularly into the right neck area.
Clinical observations
After inoculation or vaccination, all pigs were examined daily for signs of illness such as lethargy, respiratory signs, inappetence, and lameness. In addition, rectal temperatures were measured on pigs experimentally inoculated with A. pleuropneumoniae every 3 hr for the first 24 hr after inoculation. Any animals experiencing severe clinical signs such as rectal temperatures above 41°C at 2 consecutive observation intervals, lethargy, severe respiratory distress (increased respiratory rates), vomiting, or bloody nasal discharge were immediately treated with 100 mg of ceftiofur d intramuscularly.
Sample collection and storage
Blood samples were collected at weekly intervals for a total of 4 weeks after inoculation or vaccination (day 0, 7, 14, 21, and 28) and used for evaluating the diagnostic performance of the serological assays. Blood was centrifuged at 3,220 × g for 10 min at 4°C, and the serum was aliquoted and stored at −20°C until testing.
Confirmatory assays
Because antimicrobial treatment may interfere with antibody production, 44 seroconversion to A. pleuropneumoniae was verified by additional assays in selected pigs. All samples from a pig collected after inoculation were excluded if there was no indication of seroconversion by all 4 assays utilized in this study and by the confirmatory assay(s). To confirm successful antibody production against A. suis or against A. pleuropneumoniae serovars 1, 5, 9, 10, 11, and 14, which all express the ApxI RTX toxin,16,33 an indirect ELISA was performed as previously described 42 with the following modifications. Microtiter plates e were coated with 100 μl of the ApxI recombinant polypeptide at a concentration of 1.5 µg/ml in phosphate buffered saline (PBS, pH 7.2) per well, and the plates were incubated overnight at room temperature. The concentration of the antigen was optimized in order to obtain the best discrimination between positive and negative samples. After 3 washes with PBS containing 0.05% Tween-20, the plates were blocked with 10% fetal bovine serum f for 2 hr at room temperature. After this, the serum samples, which were diluted 1:20 with PBS containing 10% fetal bovine serum, were added to each well and incubated for 45 min at 37°C. After a washing step, a 1:50,000 dilution of peroxidase conjugated goat anti-swine IgG g was added to each well, and the plates were incubated at 37°C for 30 min. Finally, the peroxidase reaction was visualized by using a tetramethylbenzidine–hydrogen peroxide solution h as substrate during a 15-min incubation step in the dark at room temperature. The reaction was terminated by adding 50 µl of 2.5 M sulfuric acid to each well, and the plates were read at 450 nm using a spectrophotometer. For additional confirmation of presence of antibodies in pigs infected with serovars 4, 13, and 14, the samples were tested by individual ELISA against these serovars as described for serovar 4 19 and serovar 13 35 or according to a laboratory-specific protocol for serovar 14 (M. Gottschalk, unpublished protocol). To determine the success rate of infection of pigs inoculated with serotypes 2, 3, 6–8, 12, and 15, indirect ELISA targeting antibodies against the ApxII RTX toxin 18 was performed with the same modifications as described for the ApxI RTX toxin assay.
Field samples from pigs with unknown A. pleuropneumoniae exposure
A total of 90 serum samples were selected from routine submissions to the Iowa State University Veterinary Diagnostic Laboratory. The samples came from pigs of different ages including newborn pigs prior to colostrum uptake (n = 5), suckling pigs less than 21 days of age (n = 11), nursery pigs less than 56 days of age (n = 34), finisher pigs less than 25 weeks of age (n = 15), and adult pigs older than 25 weeks (n = 25). The samples originated on 18 different farms located in 5 U.S. states (Colorado, Illinois, Iowa, Oklahoma, and Pennsylvania). The A. pleuropneumoniae status of the 18 herds was unknown. From each submission, 1–5 samples were randomly selected. Prior to testing, the samples were stored at −20°C.
Serological assays
Dual-plate complement fixation assay
The dual-plate CF assay was performed as previously described using 100 μl of serum for each sample.22,31 Titration was conducted by using sample dilutions from 1:4 to 1:128. The dual-plate CF assay was performed simultaneously for serovars 1, 5, and 7 (CF-1-5-7) on the same plate or for serovar 3 (CF-3) on the other plate (Table 1). The results were read after 24 hr subjectively by a technician and expressed as reciprocal of the highest dilution of serum capable of producing red blood cell settling in the bottom of the well. Samples were considered positive if the titer was equal to or greater than 1:8.
Prevalence of Actinobacillus pleuropneumoniae antibody–positive animals on sera collected at days 7, 14, 21, and 28 after experimental inoculation.*
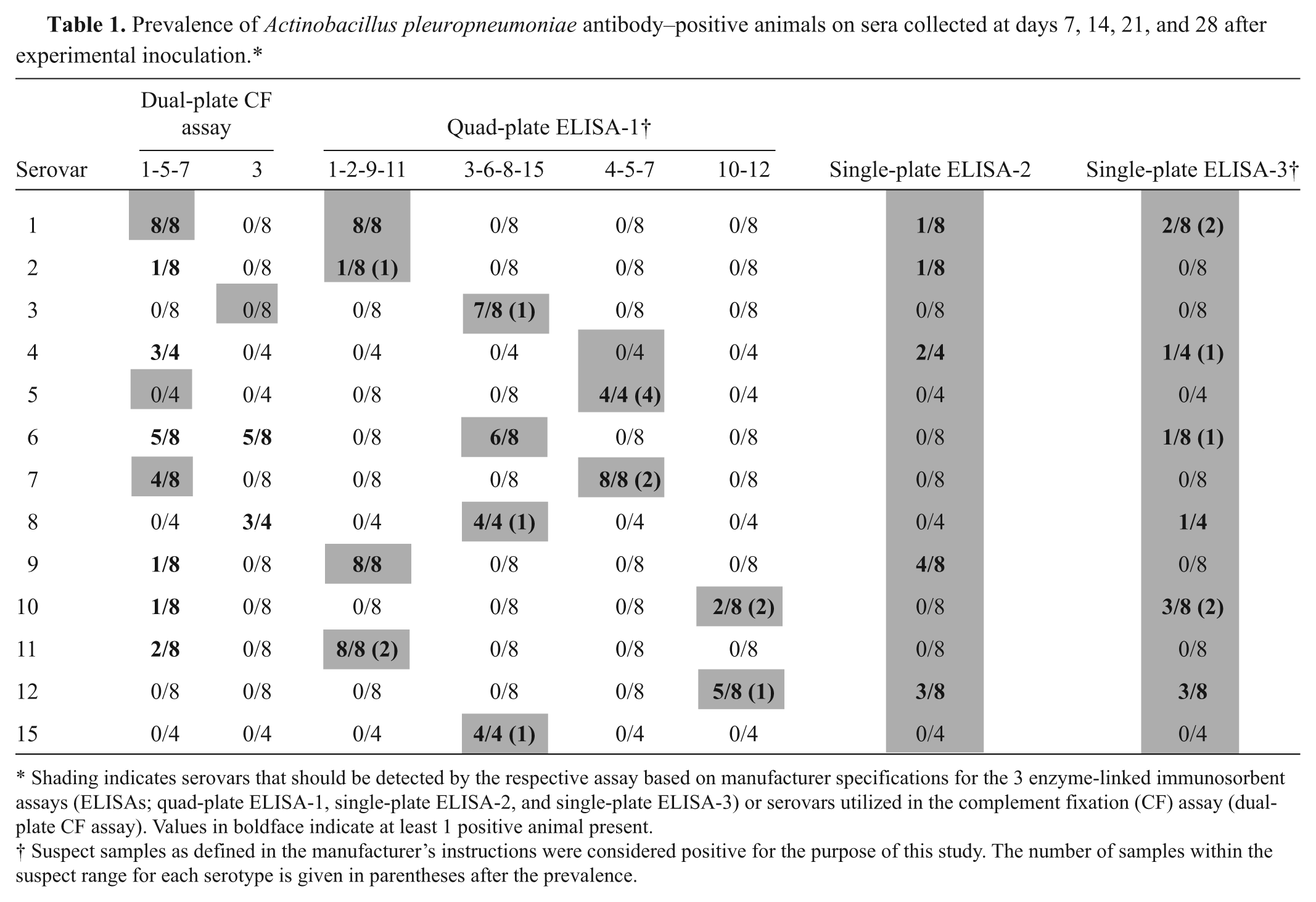
Shading indicates serovars that should be detected by the respective assay based on manufacturer specifications for the 3 enzyme-linked immunosorbent assays (ELISAs; quad-plate ELISA-1, single-plate ELISA-2, and single-plate ELISA-3) or serovars utilized in the complement fixation (CF) assay (dual-plate CF assay). Values in boldface indicate at least 1 positive animal present.
Suspect samples as defined in the manufacturer’s instructions were considered positive for the purpose of this study. The number of samples within the suspect range for each serotype is given in parentheses after the prevalence.
Quad-plate ELISA-1
A commercial indirect ELISA i composed of 3 individual plates, each capable of detecting serovars to certain groups (serovars 1, 2, 9, and 11; serovars 3, 6, 8, and 15; and serovars 4, 5, and 7) was utilized. Additionally, a separate complementary commercial indirect ELISA kit j sold as a different kit, but produced by the same manufacturer and capable of detecting A. pleuropneumoniae antibodies to serovars 10 and 12, was included (Table 1). As these 2 kits were produced to complement each other, the results of both kits were combined. The plates for these 2 assays were coated with LPS of certain serovars. 24 A total of 20 μl of each serum sample was used and diluted 1:200. The quad-plate ELISA-1 results were expressed as corrected sample-to-positive (S/P) ratio. Samples were considered to be positive if the S/P ratio was equal to or greater than 0.5 for all plates except for the serovar 10 and 12 plate where the positive cutoff was 0.6 or greater. Samples with results below the positive cutoff but equal to or greater than 0.33 (serovars 1, 2, 9, and11), 0.30 (serovars 3, 6, 8, and 15), 0.35 (serovars 4, 5, and 7), or 0.40 (serovars 10 and12) were considered to be suspect according to the kit instructions provided by the manufacturer. For the purpose of the present study, all suspect samples were considered positive.
Single-plate ELISA-2
A commercial indirect assay k based on ApxI and surface transferring-binding protein 2 (Tbp2) of A. pleuropneumoniae was utilized. According to the manufacturer, this assay is capable of using a sample volume of 5 μl at a dilution of 1:200 for the detection of infections caused by A. pleuropneumoniae. Samples were considered positive if their relative index value was greater than 20.
Single-plate ELISA-3
A commercial indirect assay l based on a recombinant ApxIV antigen was utilized. According to the manufacturer, this assay is effective when using a sample volume of 15 μl at a dilution of 1:10 for the detection of infections caused by all A. pleuropneumoniae serovars (Table 1), but it does not detect vaccine induced A. pleuropneumoniae–specific antibodies. Samples were considered positive if their S/P ratios were equal to or greater than 50. According to the manufacturer’s instructions supplied in the kit, samples were considered to be suspect if the results were below the positive cutoff but equal to or greater than 0.40. For the purpose of the current study, all suspect samples were considered positive.
Statistical analysis
Sensitivity and specificity were calculated for each assay using the samples of known exposure. A kappa statistic was calculated for paired tests using dichotomized data obtained from samples with unknown exposure. Values for kappa range from −1 to 1, where −1 indicates agreement worse than expected by chance, 0 equals agreement no better than expected by chance, and 1 equals complete agreement. 43 The following arbitrary standards for the strength of agreement were used: ≤0 = poor, 0.01–0.2 = slight, 0.21–0.4 = fair, 0.41–0.60 = moderate, 0.61–0.80 = substantial, and 0.81–1 = almost complete. 26 Statistical analysis was performed using a commercial software package. m
Results
Experimental samples from pigs with known A. pleuropneumoniae exposure
Sentinel pigs
All sentinel pigs remained free of clinical signs consistent with bacterial septicemia, respiratory disease, and A. pleuropneumoniae infection, and none had gross lesions suggestive of bacterial pneumonia. Blood samples obtained from 4 of these pigs were collected weekly for 56 days, the serum samples were tested by all assays described in the present study, and all serum samples were negative by all assays (data not shown).
Clinical disease
Pigs in 8 of the 17 groups inoculated with A. pleuropneumoniae (2/2 pigs infected with serovar 1–3, 8, or 9; 2/4 pigs infected with serovar 4; and 1/2 pigs infected with serovars 10 and 11) developed mild to severe clinical signs and were treated with antibiotics. None of the vaccinated pigs or the pigs infected with A. suis displayed clinically disease.
Confirmatory assays and exclusion of pigs
The serotype 4 isolate was positive when tested with antibodies against the long-chain LPS, cross-reacting with serovars 4 and 7, negative when tested with serovar 7–specific monoclonal antibodies against the capsular polysaccharide, and positive when tested with a serovar 4–specific coagulation assay further confirming its serovar origin. 27 The presence of anti-ApxI IgG antibodies was detected in experimentally infected pigs on days 7, 14, 21, and 28, after A. pleuropneumoniae infection with serovars 1 (2/2 pigs), 5 (1/2 pigs), 9 (2/2 pigs), 10 (2/2 pigs), 11 (2/2 pigs), and 12 (2/2 pigs), and with A. suis (2/2 pigs). Anti-ApxI IgG antibodies were not detected in 2 out of 2 pigs infected with serovar 14. Serotype-specific antibodies were not detected in serum samples of pigs inoculated with serotype 4, 13, or 14 on any day postinfection. Seroconversion to ApxII was detected in pigs experimentally infected with serovar 2 (2/2 pigs), 3 (2/2 pigs), 6 (2/2 pigs), 7 (1/2 pigs), 8 (1/2 pigs), 12 (2/2 pigs), and 15 (1/2 pigs). The serotype 7 pig that was ApxII negative showed seroconversion to A. pleuropneumoniae by 2 other assays (dual-plate CF assay, quad-plate ELISA-1), and the ApxII result was considered as a false negative. Due to lack of any detectable antibodies by all assays, the following numbers of pigs were considered noninfected and excluded from the A. pleuropneumoniae serovar 4 (3/4), 5 (1/2), 8 (1/2), 13 (2/2), 14 (2/2), and 15 (1/2) groups. The final numbers of experimentally infected pigs used for the comparison study were as follows: A. pleuropneumoniae serovars 1 (n = 2), 2 (n = 2), 3 (n = 2), 4 (n = 1), 5 (n = 1), 6 (n = 2), 7 (n = 2), 8 (n = 1), 9 (n = 2), 10 (n = 2), 11 (n = 2), 12 (n = 2), and 15 (n = 1), and A. suis (n = 2).
Dual-plate CF assay
Among the samples from pigs experimentally infected with A. pleuropneumoniae, 28 were considered true positive and 210 were considered negative. Negative samples included true-negative serum samples from pigs prior to infection (n = 34), serum samples from pigs infected with A. suis (n = 8), and heterosubtypic samples from pigs infected with serovars other than 1, 3, 5, or 7 (n = 168). Some samples were accounted for twice as the samples were run on 2 plates. Both pigs challenged with serovar 1, none of the pigs challenged with serovar 3 or 5, and 1 of the 2 pigs infected with serovar 7 were correctly identified as positive by the dual-plate CF assay (Table 1). Positive identification occurred as early as 7 days after inoculation (Table 2), and the overall sensitivity was 0.46. However, among correctly identified true-positive animals, the same pig was not always recognized as positive on consecutive bleeding days. All samples collected prior to infection or from pigs infected with A. suis were negative. However, cross-reactivity was observed involving several serovars resulting in an overall specificity of 0.90. Specifically, the CF-1-5-7 assay cross-reacted with serovars 2, 4, 6, and 9–11, whereas the CF-3 assay cross-reacted with serovars 6 and 8 (Table 1). Vaccinated animals were recognized as antibody positive as early as day 14 in 75% (6/8) of the pigs; 25% (2/8) were positive on day 21 and 62.5% (4/8) were positive on day 28. Interestingly, on day 28, 1 pig vaccinated with a serovar 3–based bacterin had a low titer (1:4) on the CF-1-5-7 assay but not on the CF-3 assay and was therefore considered as false positive. The overall sensitivity for vaccinated samples was 0.40.
Combined (all plates for assays composed of more than 1 plate) sensitivity for Actinobacillus pleuropneumoniae antibody–positive animals on sera collected at days 7, 14, 21, and 28 after experimental inoculation.*
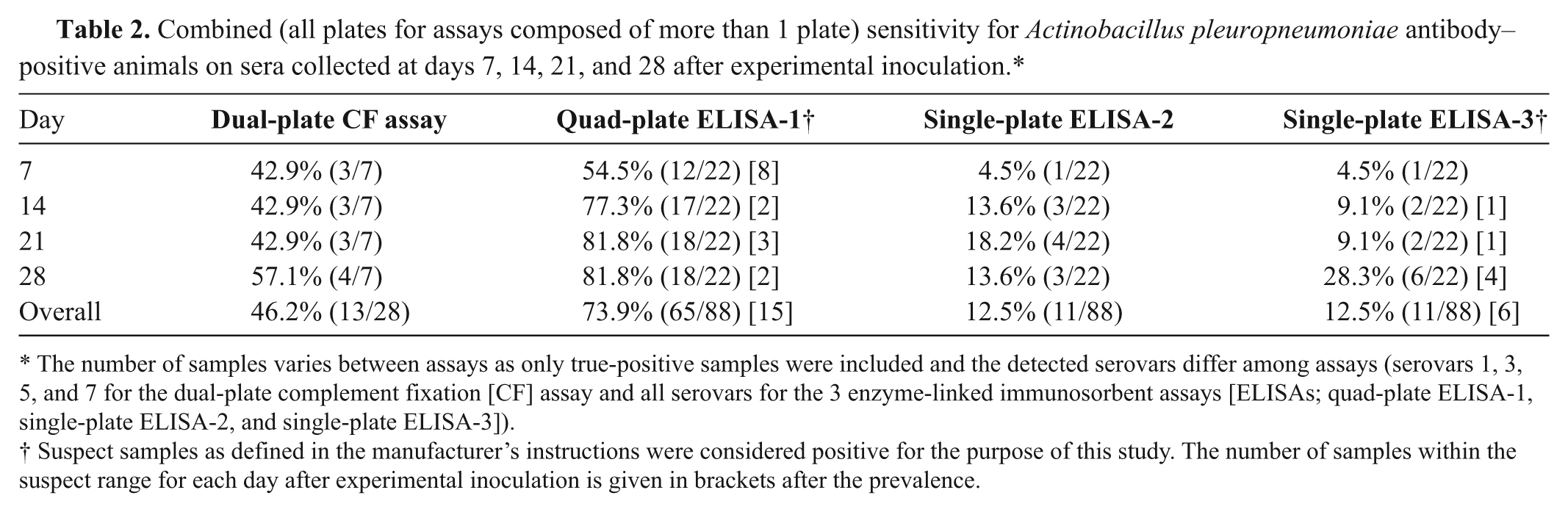
The number of samples varies between assays as only true-positive samples were included and the detected serovars differ among assays (serovars 1, 3, 5, and 7 for the dual-plate complement fixation [CF] assay and all serovars for the 3 enzyme-linked immunosorbent assays [ELISAs; quad-plate ELISA-1, single-plate ELISA-2, and single-plate ELISA-3]).
Suspect samples as defined in the manufacturer’s instructions were considered positive for the purpose of this study. The number of samples within the suspect range for each day after experimental inoculation is given in brackets after the prevalence.
Quad-plate ELISA-1
Among the samples from pigs experimentally infected with A. pleuropneumoniae, 88 were considered true positive and 346 were considered negative. Negative samples included true-negative serum samples from pigs prior to infection (n = 34), serum samples from pigs infected with A. suis (n = 8), and heterosubtypic samples from pigs infected with serovars other than each of the 4 individual plates was specified to detect; some samples were accounted for 3 times as the samples were run on 4 plates. First detection of positive pigs infected with a certain serovar ranged from day 7 (serovars 1, 3, 5, 7–9, 11, 15) to 21 (serovar 10). In all cases, once a positive pig was detected it continued to be quad-plate ELISA-1 positive for the subsequent bleeding days, which indicates reliable detection. One of the 2 pigs infected with serovar 2 or 10, and the pig infected with serovar 4 were not detected by the quad-plate ELISA-1. However, all these pigs except the pig infected with serovar 4 had increased S/P ratios on the expected respective individual plate compared to their day 0 result and negative control pigs, indicating a weak immune response in these pigs. The overall sensitivity on samples from experimentally infected pigs was 0.74 (Table 2), while the specificity was 1. When suspect samples were considered negative, the sensitivity decreased to 0.57. Cross-reaction was not observed with A. suis. Vaccinated pigs were initially detected on day 14 (37.5%, 3/8 pigs; 1 pig within the suspect range), and 50% of them (4/8; 1 pig within the suspect range) were positive on days 21 and 28. Specifically, the 2 pigs vaccinated with serovar 1 and 1 pig vaccinated with serovar 3 had a recognizable antibody response by day 14, and 1 pig vaccinated with serovar 5 seroconverted by day 21. Among the remaining vaccinated animals, all had increased S/P ratios compared to noninfected pigs on their appropriate individual plates, serovar group 3, 6, 8, and 15 or serovar group 4, 5, and 7. However, these S/P ratios were below the cutoff value for suspect animals by day 28.
Single-plate ELISA-2
Among the samples from pigs experimentally infected with A. pleuropneumoniae, 88 were considered true positive and 42 were considered negative. Negative samples included true-negative serum samples from pigs prior to infection (n = 34) and serum samples from pigs infected with A. suis (n = 8). Single-plate ELISA-2 correctly identified 1 pig infected with serovar 9 by day 7, which remained seropositive until day 28; 1 pig infected with serovar 4, which was positive on days 21 and 28; 1 pig infected with serovar 12, which was positive on days 14, 21, and 28; and individual pigs infected with serovar 1 or 2 were found to be positive on day 14 or 21, respectively (Table 2). Cross-reactivity with A. suis was not observed. The overall sensitivity on pigs experimentally infected with A. pleuropneumoniae was 0.13 (Tables 1, 2), and the specificity was 1. None of the vaccinated pigs were detected with the single-plate ELISA-2.
Single-plate ELISA-3
Among the samples from pigs experimentally infected with A. pleuropneumoniae, 88 were considered true positive and 42 were considered negative. The number of negative samples includes true-negative serum samples from pigs prior to infection (n = 34) and serum samples from pigs infected with A. suis (n = 8). Seroconversion was first detected by day 7 for serovar 10 (1/2 pigs), by day 14 for serovar 12 (1/2 pigs), and by day 21 for serovar 1 (1/2 pigs; Table 2). In addition, on day 28, individual pigs infected with serovars 4, 6, and 8 were also identified as being A. pleuropneumoniae positive. Cross-reaction was not observed with A. suis. The overall sensitivity on pigs experimentally infected with A. pleuropneumoniae was 0.13 (Tables 1, 2), and the specificity was 1. When suspect samples were considered negative, the sensitivity decreased to 0.06. None of the vaccinated pigs were detected with the single-plate ELISA-3.
Field samples from pigs with unknown A. pleuropneumoniae exposure
Based on recommended cutoff criteria and depending on the assay used, 4.4–46.7% of the pigs were positive. Antibodies to A. pleuropneumoniae were not detected in newborn pigs with any of the assays. Prevalence rates in the other age groups are summarized in Figure 1. Specifically, the quad-plate ELISA-1 indicated 1 finisher pig and 5 adult pigs as positive for the serovar 3-6-8-15 cluster, 3 adult pigs were positive for the serovar 1-2-9-11 cluster, 2 adult pigs were positive for the serovar 4-5-7 cluster, and 5 adult pigs were positive for serovar cluster 10-12. The agreement between the dual-plate CF assay and the quad-plate ELISA-1 (0.07 ± 0.12), single-plate ELISA-2 (0.05 ± 0.05), or single-plate ELISA-3 (0.01 ± 0.06) was slight. Similarly, the agreement between single-plate ELISA-3 and the quad-plate ELISA-1 (0.01 ± 0.06) or single-plate ELISA-2 (0.19 ± 0.10) was also slight, whereas the agreement between quad-plate ELISA-1 and single-plate ELISA-2 was fair (0.28 ± 0.17). Care must be taken by comparing agreements among assays on positive or negative samples, as the CF assay only detects 4 serovars and so it is expected to have more negative results with this assay compared to the others. Overall, all 4 assays agreed on the negative status of 37 of the 90 samples and the positive status of 1 of the 90 samples. Interestingly, the positive sample that all the assays agreed on was positive for the serovar 10-12 cluster by quad-plate ELISA-1, which should not have been detected by the dual-plate CF assay.
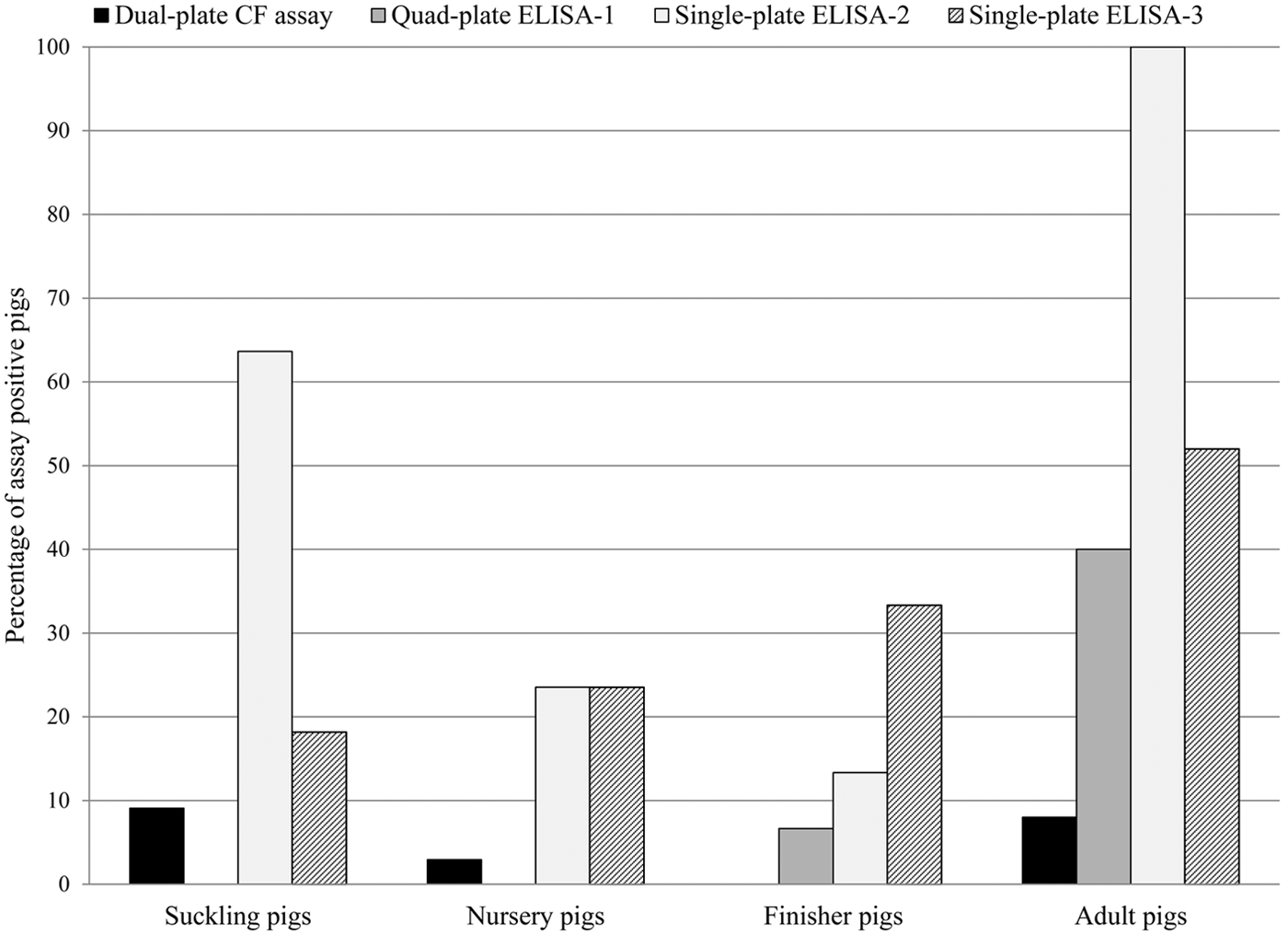
Percentages of Actinobacillus pleuropneumoniae antibody–positive serum samples detected by 4 different antibody assays obtained from pigs of 5 different age groups with unknown A. pleuropneumoniae exposure collected from 18 U.S. farms. Suckling pigs = 1–20 days of age (n = 11); nursery pigs = 21–55 days of age (n = 34); finisher pigs = 56–175 days of age (n = 15); adult pigs = older than 175 days (n = 25). The assays included a complement fixation (CF) assay and 3 enzyme-linked immunosorbent assays (ELISAs). For quad-plate ELISA-1 and single-plate ELISA-2, suspect samples as defined in the manufacturer’s instructions were considered positive for the purpose of this study.
Discussion
Actinobacillus pleuropneumoniae continues to be a pathogen of high concern in the swine industry worldwide. In the current study, 3 commercially available ELISAs, all based on different antigens, and the dual-plate CF assay were directly compared using samples of known (n = 134) and unknown (n = 90) A. pleuropneumoniae exposure. Each of the 4 assays used employs a different detection method. Complement fixation (dual-plate CF assay) is based on measuring the ability of any present antigen–antibody complexes to fix complement. 4 Both antibody isotypes, IgM and IgG, are capable of fixing complement 40 and therefore this assay is thought of as being useful in the early stages of infection.45,48 Detection of antibodies of the IgG class against long-chain LPS, which differ for certain serovar groups, is the basis of quad-plate ELISA-1, which is composed of 2 separate kits. Kit 1 (3 plates) is capable of detecting antibodies to serovars 1–9, 11, and 15, and kit 2 (1 plate) is capable of detecting antibodies to serovars 10 and 12. If combined, as in the current study, quad-plate ELISA-1 should detect infections with all A. pleuropneumoniae serovars except serovars 13 and 14. Detection of anti-IgG antibodies against certain RTX toxins is utilized by single-plate ELISA-2, which detects antibodies against ApxI, and single-plate ELISA-3, which detects antibodies against ApxIV. While ApxIV is expressed by all A. pleuropneumoniae strains, 39 ApxI expression is limited to strains 1, 5, 9–11, and 14.16,33 To detect strains that do not express ApxI, single-plate ELISA-2 also utilizes detection of antibodies against the surface protein Tbp2.
The present study has the following limitations, which are important for interpreting results. The variation of pig age at vaccination or challenge could account for differences in immune responses. To address this concern, the younger pigs were used for vaccination, and pigs 5 weeks and older were used for the experimental challenge. In addition, as only 1 or 2 pigs per group were used for most serovars, individual differences in the humoral immune response may have affected the results. Negative control animals were not included; however, the absence of cross-contamination between groups was confirmed with the obtained serotype-specific serology results. Also, the McFarland standard, a subjective measure of turbidity, was utilized to estimate the concentration of bacteria in the inocula, meaning that the actual count may have varied substantially and influenced immune responses. Finally, although bacterial isolates used for the bacterin production were grown in a similar way with a similar initial bacterial population, the bacterial concentration present in each dose of the bacterin was not determined prior to inactivation. Therefore, the low number of positive samples from vaccinated pigs could be due to a low sensitivity of the assays to postvaccination seroconversion, to vaccine composition, or to performance characteristics of the assays.
In the current study, pigs that developed severe clinical disease after inoculation were treated with an antimicrobial to avoid severe illness and death. Previously, it has been shown that pigs experimentally inoculated with A. pleuropneumoniae serovar 2 and injected with enrofloxacin after onset of clinical sign had evidence of interference with antibody production to A. pleuropneumoniae. 44 In the present study, ceftiofur was used for treatment of clinically affected pigs, and it is possible that the antigenic load in treated pigs was reduced, resulting in a reduced or even absent humoral immune response. A previous report on the evaluation of ELISAs for A. pleuropneumoniae also showed that antibodies against some serovars could not be detected. Specifically, pigs infected with serovars 5 and 12 did not seroconvert, and polymerase chain reaction performed on tonsils at termination of that earlier study was negative. 7 Therefore, it is possible that the animals in the present study remained uninfected perhaps due to a lower dose than required to induce infection with these particular serovars, explaining the lack of detection of antibodies. In the present study, successful infection and lack of interference by the antimicrobial treatment was confirmed by assays targeting antibodies against ApxI and ApxII or serotype-specific assays. To avoid the problem of a lack of a humoral immune response due to antimicrobial treatment or low antigenic load of the inoculum, all samples from pigs with no evidence of seroconversion by any of the study assays and by additional confirmatory assays were excluded.
All pigs infected with serovars that are known to express ApxI (1, 5, 9–11, and 14) were anti-ApxI positive with the exception of both pigs inoculated with serovar 14. The same pigs were also negative by all other assays. As the serovar 14 isolate (strain 3906) used in the current study has been previously characterized and is known to express ApxI,33,41 this may indicate problems with the inoculum production (viability, antigenic load) or inoculation procedure. Interestingly, both pigs infected with serovar 12 were seropositive for ApxI in the study. This can most likely be considered as false positive results as production of ApxI has not been reported for serovar 12 isolates.14,15 It could also be a cross-contamination from a different group. In particular, cross-contamination with serovar 10 is a possibility as the serovar 12 pigs were positive on the quad-plate ELISA-1 10-12 plate and the 2 groups were in the research facility at the same time (but in different rooms and building wings). Pigs infected with serovars 2, 3, 6–8, 12, and 15 were tested for presence of ApxII antibodies, and seroconversion to ApxII was confirmed in 11 of the 14 pigs in these groups.
The dual-plate CF assay was initially developed to detect A. pleuropneumoniae serovars 1, 3, 5, and 7, but some cross-reactivity with A. suis may occur. 30 In addition, although the dual-plate CF assay has been the standard test for many years, the relative low sensitivity of the assay, based on results from the present study, is a major concern. In the current study, as expected, the dual-plate CF assay was capable of detecting A. pleuropneumoniae–specific antibodies to serovars 1, 3, 5, and 7, but also cross-reacted with serovars 2, 4, 6, and 8–11 in animals infected experimentally. To some extent, cross-reactions between serovar 1 and serovars 9 and 11, serovar 7 with serovar 4, and between serovar 3 and serovars 6 and 8 were to be expected, due to similarities in the LPS composition. 18 The limited sensitivity and specificity of the dual-plate CF assay compared to the ELISAs in this study is not surprising as similar observations have been described by others.20,28,32 Although a wide range of cross-reactivity was expected, certain cross-reactions such as with serovars 2 and 10 were not anticipated. Besides similarities of LPS between strains resulting in cross-reactivity, another reason for unexpected cross-reactivity could be activation of the complement cascade 47 by the innate response against Gram-negative bacteria LPS thereby interfering with the dual-plate CF assay. Among field samples, the dual-plate CF assay only recognized 4.4% of the samples as positive, resulting in slight agreement with quad-plate ELISA-1 (12.2% positive) and single-plate ELISA-2 (46.7% positive). Of note, the dual-plate CF assay is currently the only test acceptable to confirm that pigs are free of A. pleuropneumoniae for export to China.
Quad-plate ELISA-1 was developed to detect antibodies against all A. pleuropneumoniae serovars except 13 and 14 by using 4 individual ELISA plates with antigens based on the long-chain LPS of certain serovar groups. Although the 4 ELISA plates are currently being sold in 2 separate kits, the results from both kits were combined for the purpose of this study. A study published in 2011 7 showed that by using a combination of 7 individual ELISAs specific for serovar group 2, 5, 10, and 12 or serovar group 1, 9, and 11, serovar group 3, 6, and 8, and serovar group 4 and 7, produced by the same manufacturer as the quad-plate ELISA-1, the majority of pigs experimentally infected with serovar 1, 3, 7, 10, or 15 were identified, which is similar to the present study with these serovars. In the current study, quad-plate ELISA-1 was able to accurately detect A. pleuropneumoniae–specific antibodies against serovars 1–12, with the exception of serovar 4. The quad-plate ELISA-1 also detected A. pleuropneumoniae antibodies in vaccinated pigs, including serovars 1, 3, and 5 but not serovar 7. Under the study conditions, this ELISA had the highest sensitivity on samples from experimentally infected pigs among all assays tested but the need to run 4 different plates represents a disadvantage.
Single-plate ELISA-2 was developed to detect antibodies against the strongly hemolytic and cytotoxic ApxI exotoxin15,16 and heterogeneous surface Tbp2. 17 In the present study, single-plate ELISA-2 was capable of correctly identifying pigs infected with serovars 1, 2, 4, 9, and 12. Although serovars 2, 4, and 12 do not secrete ApxI toxin, positive identification of pigs infected with these serovars was likely due to similarities in the tbp2 gene of which similar restriction patterns between the serovars have been documented. 9 The high percentage (47.6%) of field samples of unknown exposure status classified as positive was likely due to cross-reaction with other bacteria producing ApxI such as A. suis as previously suggested. 10 In addition, there is a high sequence homology (approximately 70%) between the tbpB genes found in A. pleuropneumoniae and A. suis. 5 Interestingly, none of the samples from the pigs experimentally infected with A. suis were recognized as positive by single-plate ELISA-2 even though the serum samples contained anti-ApxI antibodies as determined by an in-house ApxI ELISA.
Single-plate ELISA-3 was developed to detect antibodies against the ApxIV exotoxin, which is expressed by all serovars of A. pleuropneumoniae only after infection of pigs.10,39 Single-plate ELISA-3 correctly identified 12.5% (11/88) of the samples from the experimentally infected pigs with increasing detection rates as time after inoculation progressed. This rather low detection rate was somewhat unexpected but could be due to antibodies against ApxIV being produced later than antibodies against LPS components of A. pleuropneumoniae. Among field samples, 31.1% (28/90) were identified as positive with a trend of higher identification rates with increasing pig age as expected.
In conclusion, with samples from experimentally infected pigs, the sensitivity and specificity of the 3 commercially available ELISAs exceeded the detection capability of the dual-plate CF assay. While testing samples with single-plate ELISA-2 and ELISA-3 was simple as both assays required only 1 plate, the specificity and sensitivity of these 2 assays were reduced when compared to the 4 assays of quad-plate ELISA-1. On field samples, the overall agreement between tests was slight to fair. Because information on the A. pleuropneumoniae status of the 18 farms where field samples were obtained was not available, none of the positive results can be attributed to specific reactions. Moreover, the mixed detection results for antibodies in young piglets could be due to passively derived antibodies, rather than cross-reactivity following infection of the piglets. Because different assays are based on different detection antigens, antigenic marker selection (e.g., LPS, toxins, others) should also be considered in the decision-making process on which assay to use. The results of the current study indicate that none of the kits tested demonstrated an overall sensitivity greater than 74% (on an individual base), although specificities of up to 100% were observed. Therefore, when using serology, especially when trying to classify a farm as “infection free,” a substantial number of samples must be tested, especially in farms with low prevalence of infection.
Footnotes
Acknowledgements
The authors thank Curt Thompson for assistance with the preparation of the bacterial strains for inoculation. The authors are grateful to Dr. Han-Sang Yoo at Seoul National University for kindly providing the ApxI plasmid used in this study.
a.
Nature’s Made, Heartland Co-op, Cambridge, IA.
b.
Remel, Thermo Fischer Scientific Inc., Waltham, MA.
c.
Rehydragel HPA Adjuvants, General Chemical, Parsippany, NJ.
d.
Excede, Pfizer Animal Health Inc., Madison, NJ.
e.
Nunc, Thermo Fisher Scientific Inc., Waltham, MA.
f.
Gibco, Invitrogen Corp., Carlsbad, CA.
g.
Jackson ImmunoResearch Laboratories Inc., West Grove, PA.
h.
KPL Inc., Gaithersburg, MD.
i.
Swinecheck mix-APP 1-2-9-11, 3-6-8-15 and 4-5-7 antibody test kit, Biovet Inc., Quebec, Canada.
j.
Swinecheck mix-APP 10-12, Biovet Inc., Quebec, Canada.
k.
CIVTEST SUIS APP, Laboratories Hipra S.A., Girona, Spain.
l.
IDEXX APP-ApxIV Ab Test, IDEXX Laboratories Inc., Westbrook, ME.
m.
JMP 9.0.0 software, SAS Institute Inc., Cary, NC.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was internally funded by the Veterinary Diagnostic Laboratory at Iowa State University.
