Abstract
To investigate the agreement between available serologic tests for the detection of antibodies against Anaplasma phagocytophilum and Borrelia burgdorferi, 50 serum samples from horses of unknown clinical status and at low risk for infection were tested. In addition to a point-of-care enzyme-linked immunosorbent assay (pocELISA), the evaluated tests included 2 indirect fluorescent antibody tests (IFATs) for antibodies against A. phagocytophilum and an IFAT, an ELISA confirmed with Western blot, and the Lyme multiplex assay for antibodies against B. burgdorferi. For each pair-wise comparison between serologic tests, the difference in the proportion of seropositive results as well as kappa and the prevalence-adjusted, bias-adjusted kappa were calculated. The proportion of seropositive results differed significantly in each pairwise comparison of tests for detection of antibodies against A. phagocytophilum, and between the pocELISA and IFAT as well as between the pocELISA and Lyme multiplex assay for detection of antibodies against B. burgdorferi. Agreement based on kappa varied from poor to fair while agreement was improved when evaluating prevalence-adjusted, bias-adjusted kappa. Lack of agreement may be explained by differences in methodology between the evaluated tests, cross-reactivity or false-positive and false-negative tests. In addition to the limitations of serologic test interpretation in the absence of clinical disease, this data suggest that screening of horses for exposure to tick-borne diseases in nonendemic areas may not be warranted.
Keywords
Equine granulocytic anaplasmosis (EGA) caused by Anaplasma phagocytophilum and Lyme borreliosis (LB) caused by Borrelia burgdorferi are important tick-borne diseases in horses, and coexposure to both organisms has been reported in the United States. 16 In North America, both bacteria are transmitted by the black-legged tick (Ixodes scapularis), which occurs in the northeastern, midwestern, and southeastern United States, and the western black-legged tick (Ixodes pacificus), which occurs along the western coast of the United States.10,25 There are also an increasing number of established populations of both tick species in southern Canada,13,17 and an expansion of the geographic range of I. scapularis to the north and west as a consequence of climatic changes has been predicted.13,17 Cases of EGA1,3,21 and LB 2 have been reported in Canadian horses.
Diagnosis of clinical cases of EGA or LB is typically based on the manifestation of clinical signs, geographic location and likelihood of exposure to tick vectors, and detection of the infectious organism or host antibody response.4,16,24 The available serologic tests employ different test methodologies and target antigens (Table 1), which should be taken into account when attempting to achieve a diagnosis in a clinically suspect case. The use of serologic testing merely to determine exposure status is complicated by the inability to rely on clinical data; we previously observed that a point-of-care enzyme-linked immunosorbent assay (pocELISA) lacked agreement with a laboratory-based A. phagocytophilum indirect fluorescent antibody test (IFAT) and B. burgdorferi ELISA–Western blot (ELISA-WB) combination, respectively, when used for this purpose in a low-prevalence population. 19 Given that a perceived increase in disease risk in Canada may result in more frequent “screening” of horses for exposure, we expanded our investigation to all currently available serologic tests.
Comparison between methodology, time between infection and antibody detection, sensitivity, and specificity of serological tests for equine granulocytic anaplasmosis (EGA) and Lyme borreliosis (LB).*

pocELISA = point-of-care enzyme-linked immunosorbent assay; IFAT = indirect fluorescent antibody test; WB = Western blot; Osp = outer surface protein; NR = not reported. Superscript numbers correspond to references.
In experimentally infected horses.
In animals vaccinated with an Osp A vaccine.
Fifty equine serum samples that were originally submitted for equine infectious anemia testing in April 2013 were supplied by Prairie Diagnostic Services Inc. (Saskatoon, Saskatchewan, Canada) in accordance with the laboratory’s confidentiality rules, and the study was approved by the Animal Research Ethics Board at the University of Saskatchewan (protocol no. 20120015). The samples were divided into aliquots and frozen at −20°C until analysis. One aliquot (0.5 ml) was thawed at room temperature, centrifuged (3,000 × g for 10 min), and tested by one of the authors (G. Schvartz) using a pocELISA a according to the manufacturer’s instructions. Any color change in the sample spots was interpreted as a positive result. The remaining aliquots of each sample (1 ml each) were sent to 3 commercial diagnostic laboratories frozen and on ice packs. Testing for antibodies against A. phagocytophilum was done using IFAT1 b and IFAT2, c and results were interpreted as seropositive or seronegative based on each laboratory’s guidelines (Table 2). Testing for antibodies against B. burgdorferi was done using an ELISA-WB d combination, an IFAT, e and the equine Lyme multiplex assay f (Table 3).
Reference guidelines for interpretation of serologic tests for equine granulocytic anaplasmosis as provided by the veterinary diagnostic laboratories.*
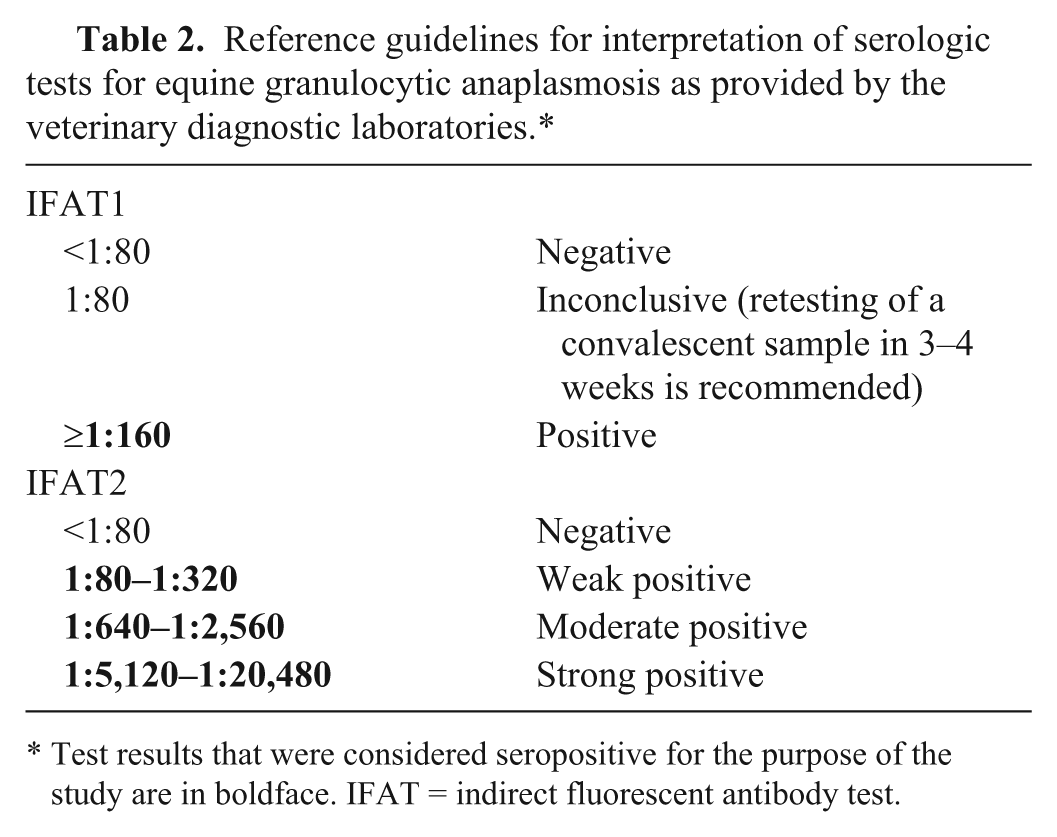
Test results that were considered seropositive for the purpose of the study are in boldface. IFAT = indirect fluorescent antibody test.
Reference guidelines for interpretation of serologic tests for equine Lyme borreliosis as provided by the veterinary diagnostic laboratories.*
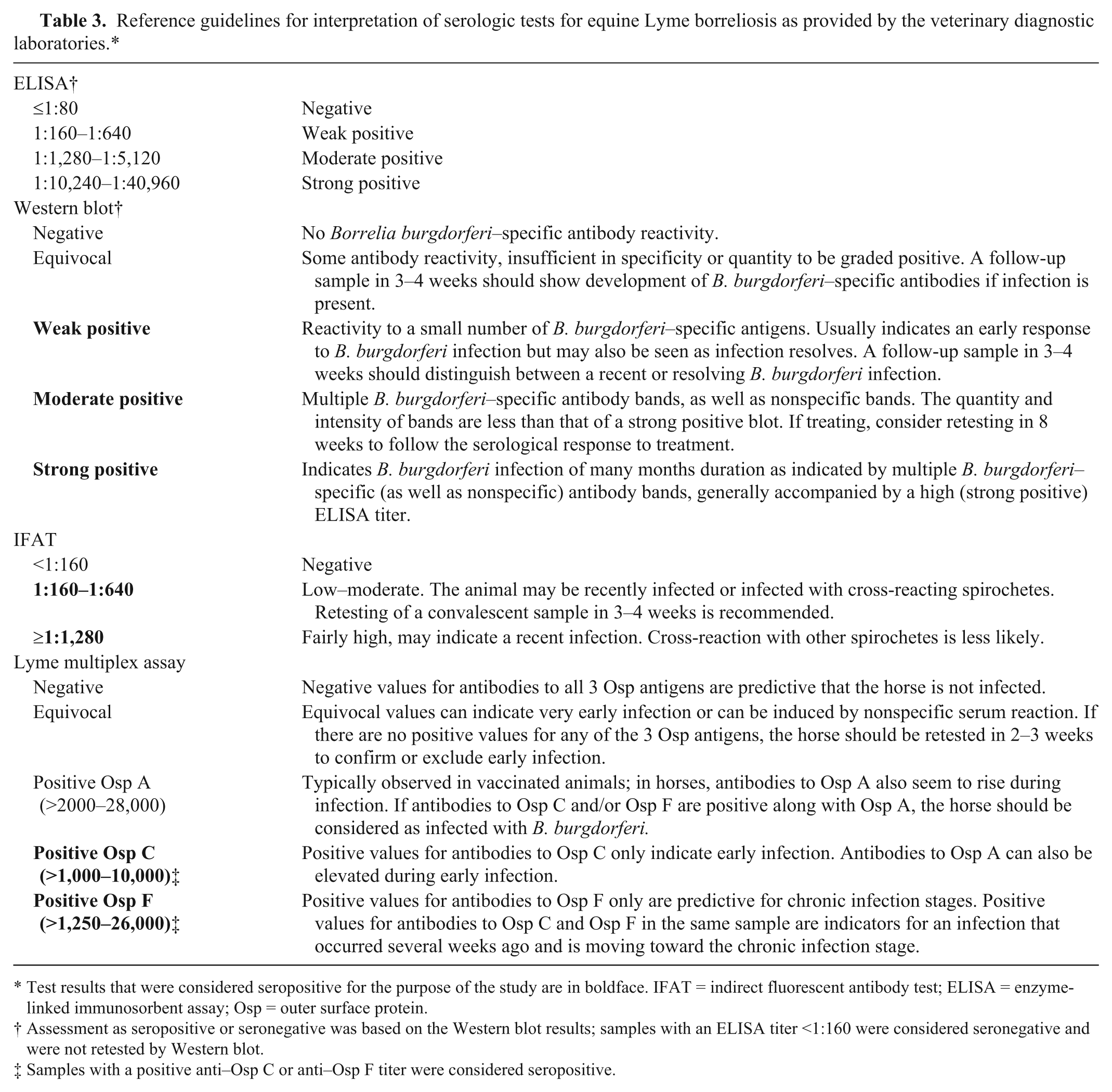
Test results that were considered seropositive for the purpose of the study are in boldface. IFAT = indirect fluorescent antibody test; ELISA = enzyme-linked immunosorbent assay; Osp = outer surface protein.
Assessment as seropositive or seronegative was based on the Western blot results; samples with an ELISA titer <1:160 were considered seronegative and were not retested by Western blot.
Samples with a positive anti–Osp C or anti–Osp F titer were considered seropositive.
Three methods were used to assess agreement between pairs of serologic tests. The proportion of positive test results was compared using a 2-tailed McNemar test, and P < 0.05 was considered significant. The overall agreement beyond chance was assessed using Cohen’s kappa with its 95% confidence interval (CI), and the prevalence-adjusted, bias-adjusted kappa (PABAK) with its 95% CI was calculated to adjust kappa for low prevalence of the disease 5 or significant differences in the proportion of positives. The magnitude of kappa was interpreted as described for inter-rater agreement with kappa ≤0 indicating poor, 0.01–0.2 slight, 0.21–0.4 fair, 0.41–0.6 moderate, 0.61–0.8 substantial, and 0.81–1 indicating almost perfect agreement. 20
When testing for antibodies against A. phagocytophilum, all 50 samples were seronegative by the pocELISA, 6 samples were seropositive by IFAT1, 15 samples were seropositive by IFAT2, and only 4 samples were seropositive using both IFATs (Table 4). A significant difference in the proportion of positives was found in each pairwise comparison (Table 5). As kappa may not provide useful additional information in cases where the proportion of positives differs, 9 it was not surprising that kappa indicated poor agreement between the pocELISA and each IFAT, and fair agreement between the 2 IFATs (Table 5). The PABAK values indicated better agreement and differed from kappa, based on the lack of overlap of the 95% CI, in the comparisons between the pocELISA and each IFAT (Table 5). Overlap between the rather large CI in the comparison between IFAT1 and IFAT2 may have been due to a limited sample size. While the expected low prevalence of exposure in the tested population may warrant adjustment of kappa, it should be mentioned that some authors have cautioned against the use of PABAK. 20
Samples (out of 50) testing positive for antibodies against Anaplasma phagocytophilum by 1 or more serologic tests.*
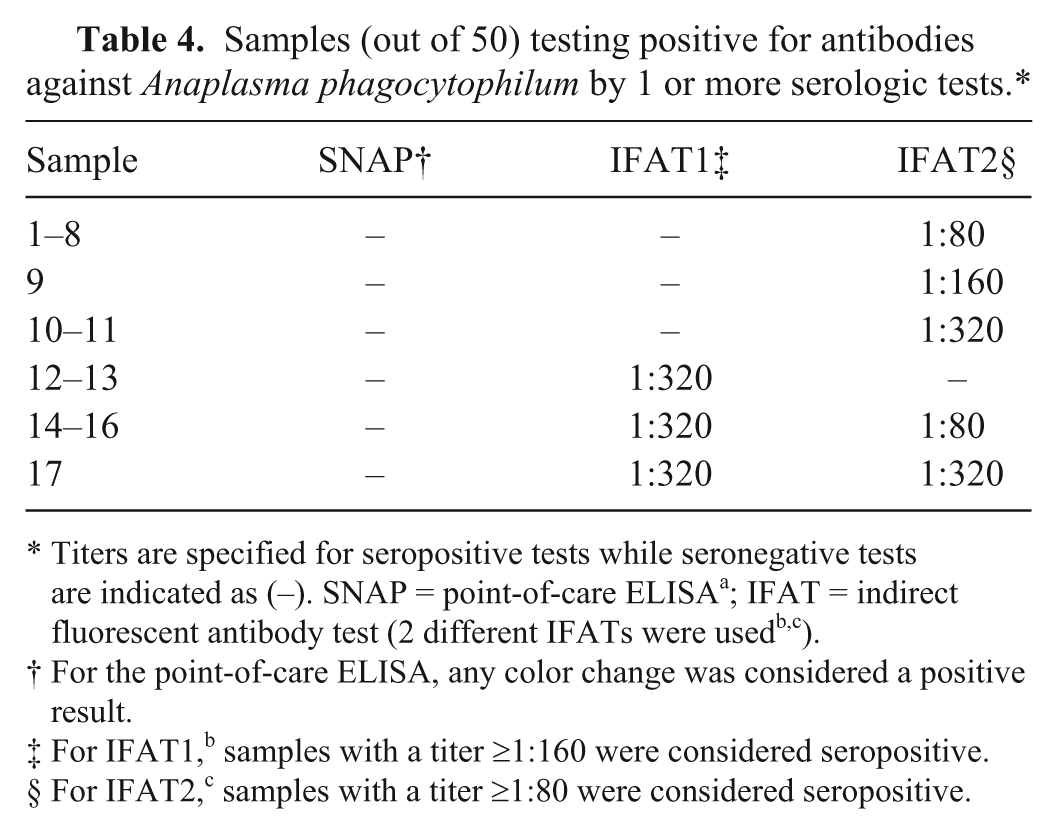
Titers are specified for seropositive tests while seronegative tests are indicated as (–). SNAP = point-of-care ELISA a ; IFAT = indirect fluorescent antibody test (2 different IFATs were usedb,c).
For the point-of-care ELISA, any color change was considered a positive result.
For IFAT1, b samples with a titer ≥1:160 were considered seropositive.
For IFAT2, c samples with a titer ≥1:80 were considered seropositive.
Agreement between pairs of serologic tests for antibodies against Anaplasma phagocytophilum and Borrelia burgdorferi using 50 equine serum samples from a low-prevalence population.*
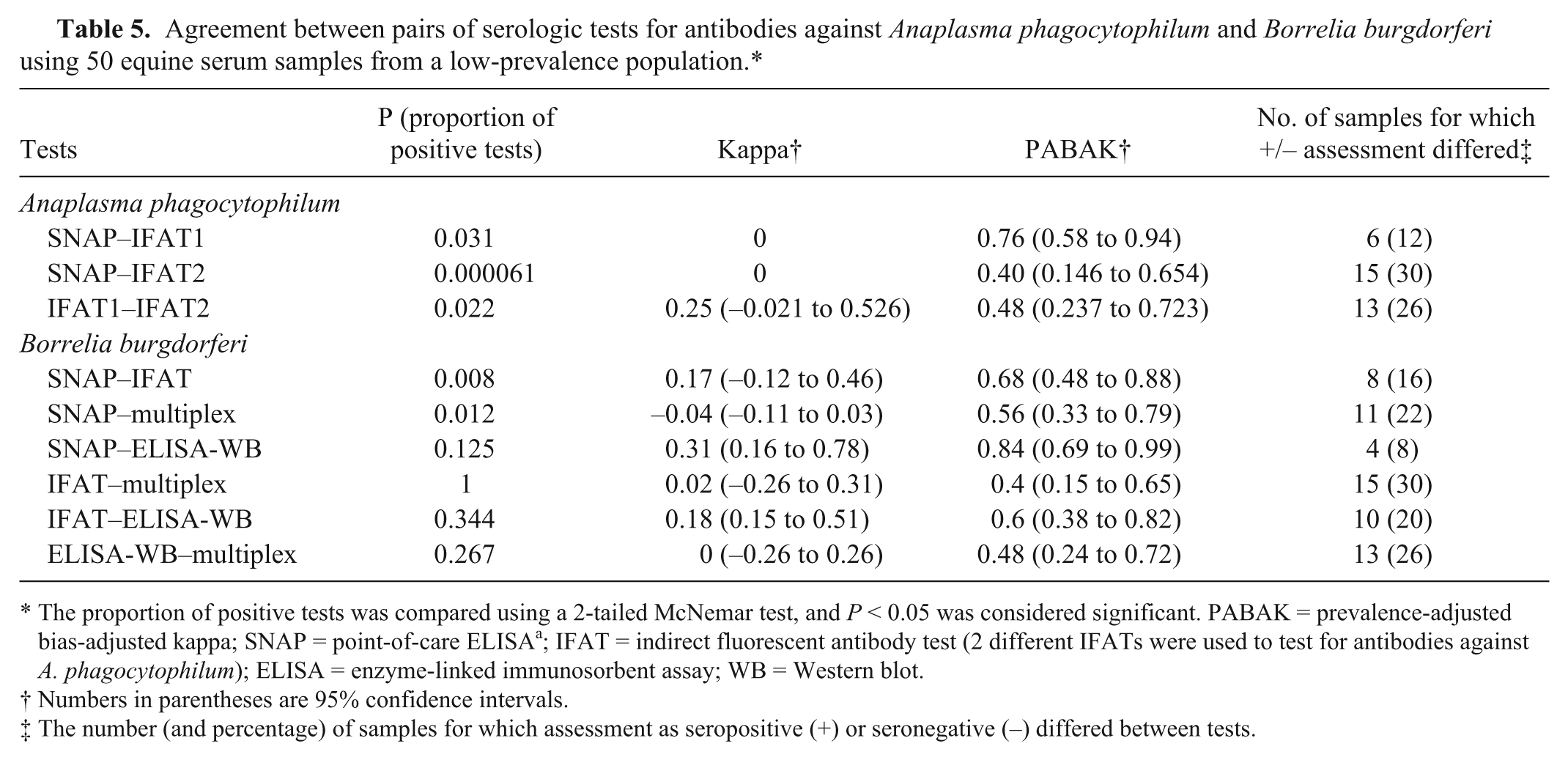
The proportion of positive tests was compared using a 2-tailed McNemar test, and P < 0.05 was considered significant. PABAK = prevalence-adjusted bias-adjusted kappa; SNAP = point-of-care ELISA a ; IFAT = indirect fluorescent antibody test (2 different IFATs were used to test for antibodies against A. phagocytophilum); ELISA = enzyme-linked immunosorbent assay; WB = Western blot.
Numbers in parentheses are 95% confidence intervals.
The number (and percentage) of samples for which assessment as seropositive (+) or seronegative (–) differed between tests.
When testing for antibodies against B. burgdorferi, 31 samples were seronegative by all tests, and none of the samples was seropositive based on all 4 tests (Table 6). The proportion of positives differed significantly when comparing the pocELISA to the IFAT or the Lyme multiplex assay, which was compatible with poor to slight agreement indicated by kappa (Table 5). In these pairs, the PABAK values were again higher than the kappa values based on the lack of overlap of the 95% CI. The proportion of positives did not differ significantly in the other 4 pairwise comparisons; however, kappa values were also low and indicated, at best, fair agreement between the pocELISA and ELISA-WB. While the PABAK values indicated better agreement in these pairs, there was no real difference to kappa because the 95% CI overlapped. For all comparisons, it should be noted that the described categories of kappa were originally established to assess agreement between clinical evaluators and may not be stringent enough for a comparison between diagnostic laboratory tests. With the possible exception of the comparison between the pocELISA and the ELISA-WB combination for antibodies against B. burgdorferi, the differences in the proportion of positive test results along with kappa and PABAK values suggest a lack of agreement between the evaluated tests in the context of our study. It is important to emphasize that samples for our study originated from horses of unknown clinical status in a population with an expected low prevalence of disease. We were interested in a classification of samples as seropositive or seronegative (i.e., we compared tests with regard to their assessment of the exposure status of a horse). Our study does not allow us to draw conclusions regarding the diagnostic usefulness of the evaluated tests in cases where disease is suspected based on clinical signs.
Samples (out of 50) testing positive for antibodies against Borrelia burgdorferi by 1 or more serologic tests.*
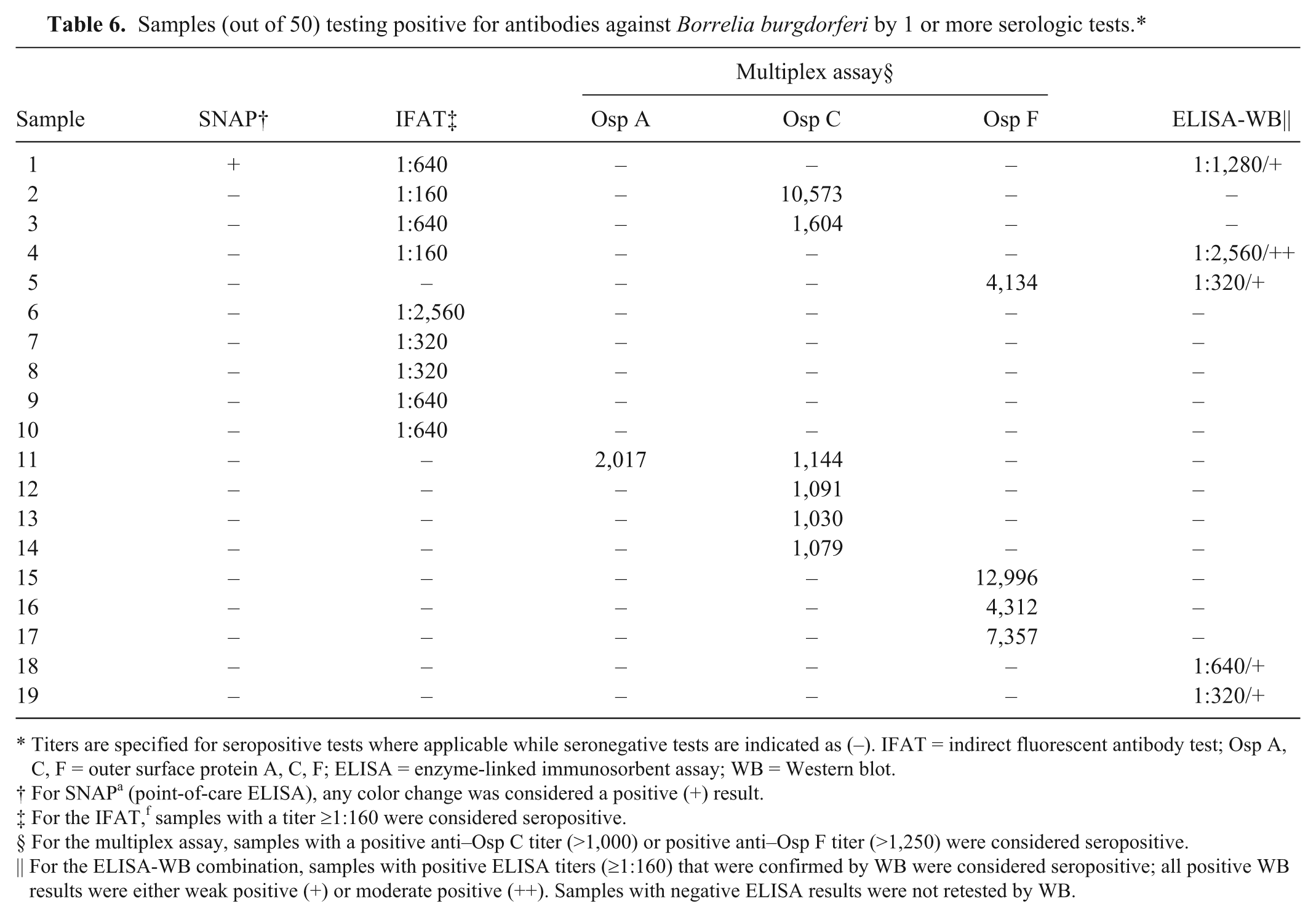
Titers are specified for seropositive tests where applicable while seronegative tests are indicated as (–). IFAT = indirect fluorescent antibody test; Osp A, C, F = outer surface protein A, C, F; ELISA = enzyme-linked immunosorbent assay; WB = Western blot.
For SNAP a (point-of-care ELISA), any color change was considered a positive (+) result.
For the IFAT, f samples with a titer ≥1:160 were considered seropositive.
For the multiplex assay, samples with a positive anti–Osp C titer (>1,000) or positive anti–Osp F titer (>1,250) were considered seropositive.
For the ELISA-WB combination, samples with positive ELISA titers (≥1:160) that were confirmed by WB were considered seropositive; all positive WB results were either weak positive (+) or moderate positive (++). Samples with negative ELISA results were not retested by WB.
The observed lack of agreement has several potential explanations that are based on differences in the tests’ methodology. First, the evaluated tests may differ in their ability to differentiate “active infection” from the serologic response following exposure to a pathogen. The test for antibodies against B. burgdorferi as part of the pocELISA, for example, is based on the VlsE antigen, which is only expressed during active infection,7,8 while the whole-cell IFAT and ELISA-WB combination do not explicitly make this differentiation. The ability of the Lyme multiplex assay to differentiate antibodies against outer surface protein (Osp) A, C, and F reportedly allows the differentiation of early and chronic infection as well as an immune response to vaccination. 22 Interestingly, the only sample that was positive by pocELISA yielded an equivocal result for Osp F in the Lyme multiplex assay and, according to our study definitions, was interpreted as testing seronegative by the latter test. Although a previous study in horses described a strong correlation between a positive pocELISA result and detection of antibodies against Osp F in the Lyme multiplex assay, 23 the fact that results of all other tests were positive suggests that the Lyme multiplex assay yielded a false-negative result for this sample. While we cannot exclude the possibility that some seropositive tests resulted from extra-label vaccination with canine vaccines, as is reported anecdotally in some horses, we are not aware that vaccination against Lyme borreliosis is a common occurrence in the population from which samples were derived.
Second, the timing of sample collection with respect to pathogen exposure may affect test results. Antibodies against A. phagocytophilum in horses can reportedly be detected by whole-cell IFAT for up to 2 years, 15 while the time frame for detection by pocELISA is not reported. In contrast to our results, however, the pocELISA showed good agreement with an IFAT in an asymptomatic group of horses in a previous study. 24 As Osp C of B. burgdorferi is only expressed when the spirochete is transferred to a mammalian host, 4 antibodies to this antigen may be detected only transiently and from as early as 3 weeks following infection, 23 while anti-VlsE antibodies may not be detected until 1–2 weeks after Osp C antibodies are detected.14,23 An early antibody response may therefore explain negative results by pocELISA for samples that tested positive by whole-cell IFAT and Lyme multiplex assay, both of which detect antibodies against Osp C. Varying sensitivities for Osp C antibodies may explain differences between the latter 2 tests, and this may be supported by the low anti-Osp C titers in the Lyme multiplex assay. Unfortunately, the antigens used in the WB, in the ELISA-WB combination, are not public knowledge. Previously reported B. burgdorferi antigens that are used for WB testing of equine samples include Osp A, Osp B, Osp C, Osp F, VlsE, and flagellin,16,23 all of which are similar to the antigens that the U.S. Centers for Disease Control and Prevention (CDC) recommends for the testing of human samples. 6
Third, some serologic tests, specifically whole-cell assays, may be more prone to cross-reactivity. Cross-reactivity is arguably of greater concern in samples with low titers, and, interestingly, a reduced relative sensitivity of the pocELISA in samples with low IFAT titers for antibodies against A. phagocytophilum was previously demonstrated. 24 False-positive results due to nonspecific reactivity with antibodies against flagellin or heat shock proteins may also occur with the B. burgdorferi whole-cell IFAT. 16 In this context, it is interesting to note that Borrelia miyamotoi and Borrelia kurtenbachii have been previously reported to infect I. scapularis in Canada. 18 Two-tier approaches are intended to increase the specificity of testing; however, our observation that the proportion of positives did not differ significantly between the B. burgdorferi IFAT and ELISA-WB combination may support nonspecific reactivity in the latter test when used in nonendemic areas or as a screening test alone.6,16 The CDC does not recommend the 2-tier approach for screening purposes in humans. 6
We were surprised to find disagreement even between tests that employ similar methodology, such as between the 2 IFATs for antibodies against A. phagocytophilum or between the Lyme multiplex assay and ELISA-WB combination. In contrast to our results, a previous study 23 reported complete agreement between WB and the Lyme multiplex assay when testing sera from horses with unknown history of infection with B. burgdorferi. The criteria and cutoff values we chose to categorize samples as seropositive or seronegative may have influenced the agreement between tests. Although follow-up testing may have been helpful in some instances, it was not possible in the context of our study. It is worth mentioning that an increase in the cutoff values for the B. burgdorferi IFAT to >1:1,280 and for the ELISA-WB to a moderate positive WB would have resulted in worse agreement of each test with the pocELISA and worse agreement between the 2 tests, effectively reducing agreement based on kappa to less than that expected by chance alone (data not shown).
In addition to methodological considerations, we consider it likely that some of our results represent false positives. False-positive results may explain why individual samples showed rather strongly positive results in 1 test but were negative in several or all other tests, and are supported by an expected low-positive predictive value of serologic tests in this to date low-prevalence population of horses.
In summary, our results show that available serologic tests lacked agreement when used to assess the exposure to A. phagocytophilum or B. burgdorferi of horses from a low-prevalence population. We, therefore, caution against the use of serologic “screening” in the absence of clinical suspicion and recommend that methodological details of the available tests as well as the potential for cross-reactivity and false positive results are taken into account. As stated earlier, our study is not suitable to draw any conclusions with regard to sensitivity and specificity of the employed tests in a clinical context. While there may be insufficient information to unequivocally support individual serologic tests as the “gold standard” in horses, previous studies reported high sensitivity and specificity of available serologic tests7,23,24 when known positive and negative samples were tested. Previous reports in other populations of horses also stated much better agreement between diagnostic tests.7,12,23,24
Footnotes
Acknowledgements
We thank Prairie Diagnostic Services Inc. (Saskatoon, Saskatchewan, Canada) for providing the samples for this study.
Authors’ contributions
G Schvartz drafted the manuscript. G Schvartz and KL Lohmann contributed to acquisition, analysis, and interpretation of data. T Epp contributed to analysis and interpretation of data. HJ Burgess contributed to acquisition and interpretation of data. NB Chilton contributed to interpretation of data. G Schvartz, T Epp, HJ Burgess, NB Chilton, and KL Lohmann substantially contributed to conception and design; critically revised the manuscript; gave final approval; and agree to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
SNAP 4Dx Plus ELISA, IDEXX Laboratories Inc., Westbrook, ME.
b.
Anaplasma phagocytophilum 12-well IFA substrate slides, Fuller Laboratories, Fullerton, CA. Tested at the Michigan State University Diagnostic Center for Population & Animal Health, Lansing, MI.
c.
Anaplasma phagocytophilum IFA substrate slide, VMRD Inc., Pullman, WA. Tested at the Connecticut Veterinary Medical Diagnostic Laboratory, Storrs, CT.
d.
Lyme ELISA and Lyme Western blot, Connecticut Veterinary Medical Diagnostic Laboratory, Storrs, CT.
e.
Borrelia burgdorferi 12-well FA substrate slides, VMRD Inc., Pullman, WA. Tested at the Michigan State University Diagnostic Center for Population & Animal Health, Lansing, MI.
f.
Equine Lyme multiplex assay, Animal Health Diagnostic Center, Cornell University, Ithaca, NY.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Equine Health Research Fund at the Western College of Veterinary Medicine, University of Saskatchewan (grant 414214) with in-kind support from IDEXX Laboratories (Westbrook, Maine).
