Abstract
Neurolymphomatosis is a very rare form of nervous system infiltration by lymphoma that can affect cranial and peripheral nerves and spinal nerve roots. The clinical appearance can mimic autoimmune or paraneoplastic neuropathies. To date, only 2 cases of neurolymphomatosis have been reported in the veterinary literature (1 dog and 1 cat). A case of neurolymphomatosis in a 5-year-old female Domestic Shorthair cat is reported. Two, whitish, bosselated, non-symmetric masses (1 cm × 1.2 cm × 0.5 cm) that incorporated almost all cranial nerves and semilunar ganglia occupying the basisphenoid depression were histologically composed of a proliferation of monomorphic lymphocytes. These lymphoid cells were positive for CD3 (T-cell lymphoma). Nested polymerase chain reaction detected feline leukemia provirus. Fragment analysis of feline T-cell receptor (TCR) gene rearrangements evidenced an oligoclonal pattern with few peaks of similar height. The integration of pathologic with biomolecular findings adds to the information concerning the role of Feline leukemia virus on TCRγ rearrangements in cases of feline lymphoma.
A 5-year-old, spayed, female Domestic Shorthair cat from a cattery was presented at the Veterinary Teaching Hospital of the Department of Veterinary Medical Sciences (Alma Mater Studiorum University of Bologna, Bologna, Italy) with a history of acute monoplegia of the left forelimb and jaw paralysis. The cat displayed a severely depressed mental status, tetraparesis, and plantigrade stance of the pelvic limbs. Other findings included decreased conscious proprioception in pelvic limbs, reduced withdrawal reflex in all 4 limbs, and multiple cranial nerves deficits. Neuroanatomical localization was consistent with syndrome associated with multifocal lesions.
Antemortem clinicopathological evaluation included complete blood count, clinical chemistry panel, serum electrophoresis, Feline immunodeficiency virus (FIV) and Feline leukemia virus (FeLV) serology, and cerebrospinal fluid (CSF) analysis. Ancillary findings consisted of a severe increase in creatine kinase (10256 U/l, reference [ref.] interval: 91–326 U/l), increased transaminase (alanine aminotransferase 857 U/l, ref. interval: 22–45 U/l; aspartate aminotransferase 145 U/l, ref. interval: 21–41 U/l), and a slight increase in bile acids (9.2 μmol/l, ref. interval: 0–7.6). Other findings were unremarkable. Serologic test for FeLV was positive. Complete blood count parameters were within the reference interval. Samples of CSF were collected; severe pleocytosis (526 cell/µl; ref. range: 0–8 cell/µl) and elevated protein concentration (97.5 mg/dl; ref. range 0–30 mg/dl) were observed. Cytological specimens obtained from centrifuged CSF samples stained with May-Grünwald/Giemsa showed high cellularity and were composed mainly of medium to large (36–40 μm) monomorphic lymphocytes with high nucleus-to-cytoplasm ratio, diffuse chromatin pattern, and prominent nucleoli. One to 2 mitotic figures were observed at high magnification (40×). On the basis of these findings, a diagnosis of lymphoma was made. Due to the poor prognosis, the cat was euthanatized, and a complete necropsy was performed.
Gross changes were not evident within abdominal and thoracic cavities. After removal of brain and cerebellum, 2 solid, whitish, irregularly bosselated, non-symmetric masses, both measuring 1 cm × 1.2 cm × 0.5 cm, were observed to occupy the basisphenoid depression and incorporate almost all cranial nerves (with the exception of optic nerves) and semilunar ganglia (Fig. 1); a bulbous thickening of the proximal tract of first thoracic spinal roots and ganglia was also evident. Representative tissue samples of central nervous system (CNS; cerebrum, cerebellum, spinal cord) and abdominal and thoracic organs (liver, kidney, spleen, lymph nodes, lung, stomach, and intestine) were formalin fixed and paraffin embedded; sections (4 µm) were stained with hematoxylin and eosin. Histologically, the masses at the ventral aspect of the skull were formed by a monomorphic, highly cellular proliferation of round cells, ranging from 25 to 30 µm, with a low amount of amphophilic cytoplasm, and hyperchromatic, round to oval, often indented nuclei with a high nucleus-to-cytoplasm ratio (Fig. 2). Nucleoli were prominent, round, and central. Mitotic rate was 5–6 per high power field. An infiltration of these neoplastic cells within peripheral nerves, extending along the branches of the cervical muscles, was present. Along with the lymphomatous infiltration in cranial nerves, spinal roots, and ganglia, as well as in myofibers of the cervical region, there was massive degeneration and single cell necrosis of ganglion neurones, severe degeneration of myelin fibers with axonal swelling and severe compression atrophy of myofibers. The CNS sections evidenced multifocal leptomeningeal thickening by neoplastic cells and monomorphic perivascular cuffing within the neuropil of the brain stem and cervical spinal cord. The other organs sampled did not show any significant histologic lesions.
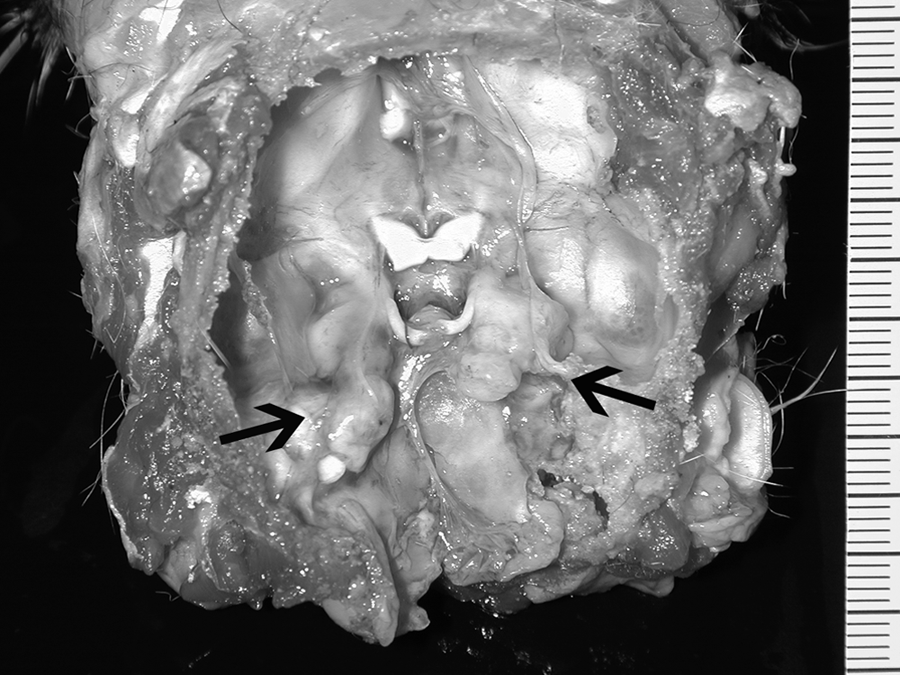
Skull base; cat. Two whitish irregularly bosselated, not symmetric masses (arrows) occupied the basisphenoid depression incorporating cranial nerves and ganglia.
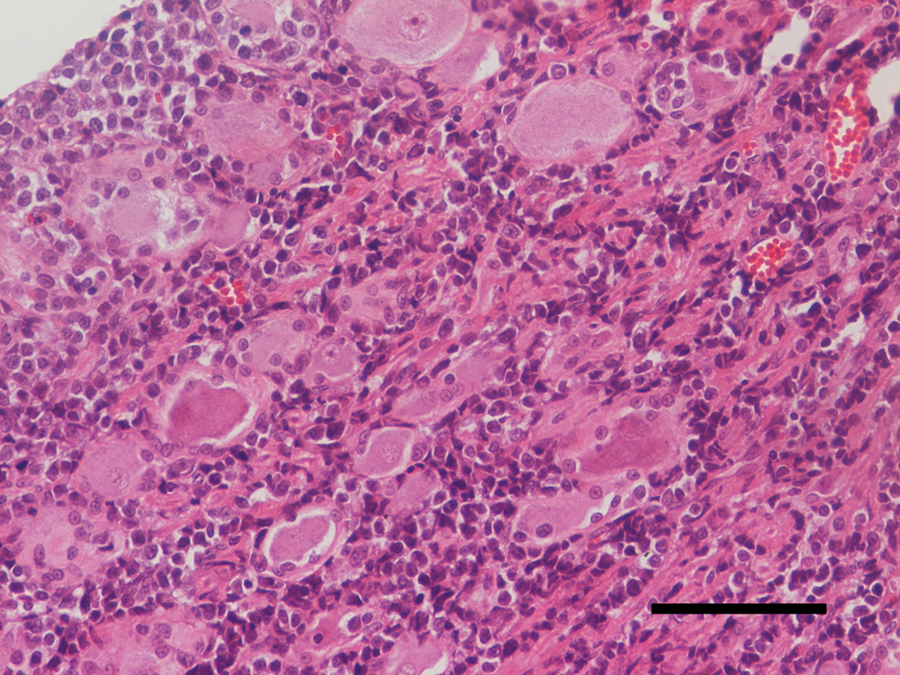
Histology of the masses; cat. A monomorphic population of neoplastic lymphocytes invaded the interstitium causing a severe ganglion cells atrophy and degeneration. Hematoxylin and eosin. Bar = 400 µm.
Immunohistochemistry was performed on serial sections of the CNS masses with a modified avidin–biotin peroxidase complex method a employing CD3 and CD79a b as primary antibodies. Positive control included a normal canine lymph node, while homologous non-immune sera replaced the primary antibodies on negative control preparations. Neoplastic lymphocytes revealed exclusively a CD3-positive T-cell phenotype. A diagnosis of neurolymphomatosis was made. In order to ascertain the clonality of immunoglobulin (Ig)/T-cell receptor (TCR) gene rearrangements as well as to confirm the presence of integrated FeLV provirus, DNA was purified from snap-frozen tissues and subjected to polymerase chain reaction (PCR). c
The GeneScanning d assay used primers targeting the feline TCR gamma chain (TCRγ) gene: fwd_TCRYFEL (5′-FAM AAGAGCGAYGAGGGMGTGTP-3′) and r_TCRYFEL (5′-CTGAGCAGTGTGCCAGSACC-3′). 8 Polymerase chain reaction assays were carried out in a 25-μl final volume. The PCR mixture was composed of 10 µl of 2.5× PCR master mix without SYBR Green, c 300 nM of each primer, and 4 μl of template brought to 25 µl with molecular biology grade water. Negative PCR controls were included in each PCR run.
A touchdown cycling protocol was used, which included an initial denaturation at 94°C for 2 min, 10 cycles at 94°C for 30 sec as denaturation step, and 68°C for 30 sec for annealing/extension, followed by 30 cycles at 94°C for 30 sec as denaturation step and 60°C for 30 sec for annealing/extension. A final extension step of 30 min at 60°C was used to avoid the plus-A artifact. The PCR reactions were carried out in duplicate.
Feline leukemia virus provirus was detected using a nested PCR as previously described. 3 All the PCR reactions were carried out in duplicate.
On the basis of clinical findings, the current case has yet to receive a diagnosis of lymphoma; because information about the TCR in cat lymphomas are relatively few it is interesting to flank to the classic data the fragment analysis (GeneScanning) of feline TCR gene rearrangements that evidenced an oligoclonal pattern with few peaks of similar height (Fig. 3). In the present case, the meaning of the oligoclonal pattern is uncertain, but can represent a reduced diversity of the TCR repertoire caused by either antigen- or neoplastic-driven evolution of T-cell subclones.
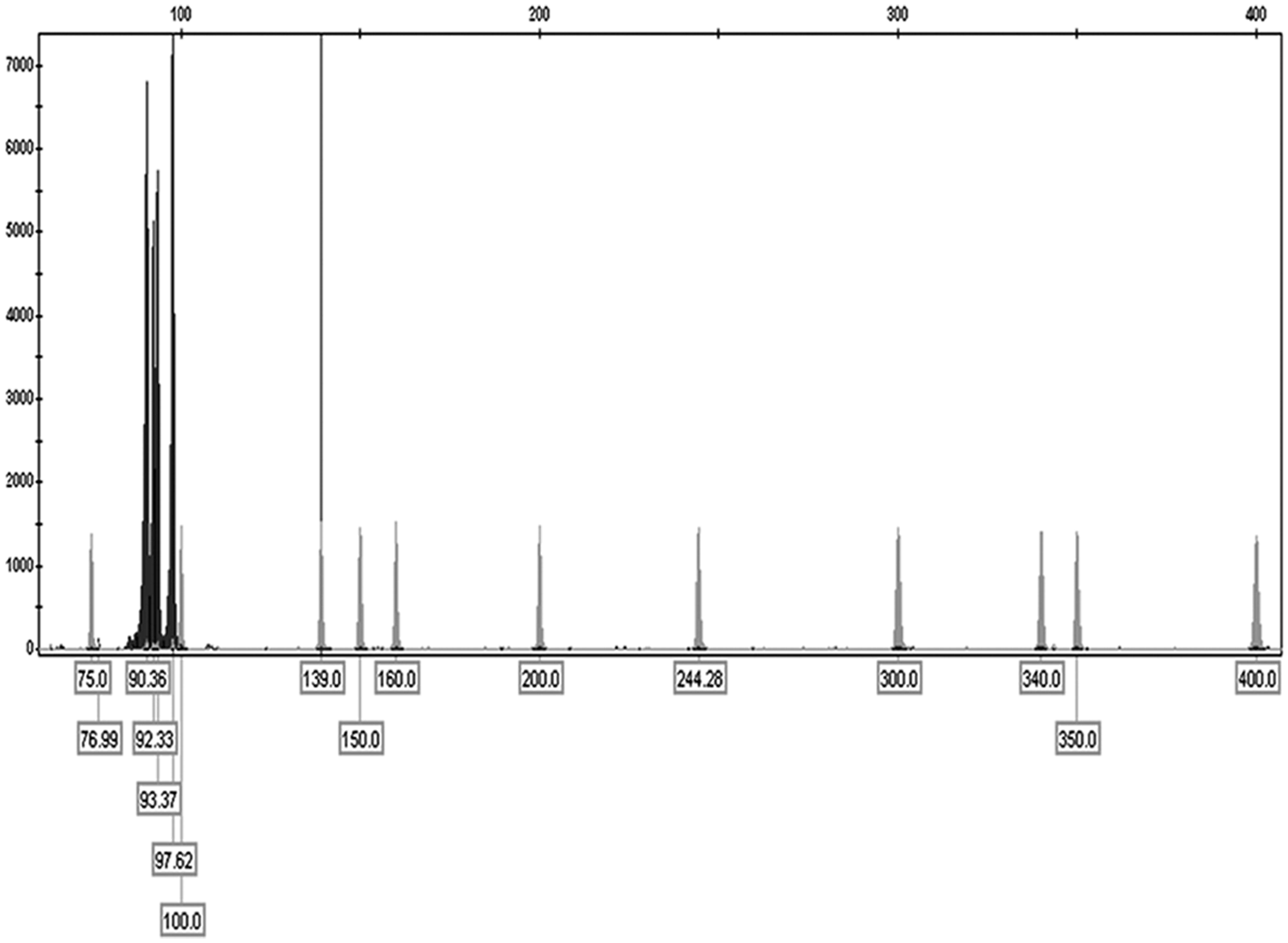
Fragment analysis (GeneScanning d ) of feline T-cell receptor gene rearrangements evidenced an oligoclonal pattern with few peaks of similar height.
A previous case described macroscopic and histological findings that were very similar to those found in the cat described herein, in particular the infiltration within cranial nerves and roots by neoplastic lymphocytes. 1 In the previous study, 1 the mass from which the lymphomatous proliferation probably started migrating within the roots and Gasserian ganglia, was detected into the pharyngeal region. In the case described herein, it is not possible to hypothesize any site of a nodal origin of lymphoma as, both at the macroscopic and microscopic level, the investigated organs and tissues (with the exception of cranial nerves, cervical spinal roots, and muscles, brain, and meninges) were not affected by the lymphomatous infiltration.
The findings in the current study are in accordance with other reports describing human primary leptomeningeal lymphoma, of B- or T-cell origin, as a rare condition, although secondary leptomeningeal lymphoma is relatively common. In a survey that followed 100 primary CNS lymphoma patients, only 5 had T-cell tumors, and all of the patients presented with leptomeningeal involvement as a sole manifestation of CNS lymphoma; all patients also demonstrated involvement of cranial nerves, and some also of peripheral nerves, indicative of neurolymphomatosis in 4 of them. 6
Feline peripheral nervous system tumors are uncommon, and lymphomas resulting in peripheral nervous system complications are rare, both in animals1,2 and human beings.4,5,9 Central and peripheral nervous system lymphomas in human beings, cats and avian species are often associated with neurotropic and oncoviral infections (herpesvirus, retrovirus). 2 Feline leukemia virus is the most lymphomagenic retrovirus; FeLV-associated lymphomas tend to be of T-cell lineage and include neurolymphoma. 7 In the current case, integrated feline leukemia provirus was detected by PCR in snap-frozen samples of the neoplastic masses and thus confirmed the presence of the integrated provirus within the neoplastic tissues.
On the basis of the selective lymphomatous involvement of the cranial nerves and spinal roots, this condition has been named neurolymphomatosis. Neurolymphomatosis in a cat bearing a peripheral nervous system B-cell lymphoma and in a dog with B-cell lymphoma are reported in the veterinary literature.2,10
In the current case, the reason for the peripheral neuropathy was probably direct nerve infiltration, rather than a paraneoplastic syndrome; moreover, a direct demyelinating effect of retrovirus could also be considered, reinforcing the direct effect of FeLV on tissues. In the present case, it was advisable to confirm the presence of the viral genome with PCR.
Leptomeningeal spread and neurolymphomatosis appeared to be common manifestations of T-cell lymphoma in a small cohort human study; such an appearance can mimic other neurological conditions, such as pseudotumor cerebri, vasculitis, or mononeuritis multiplex, and therefore represent a diagnostic challenge. 5 The leptomeningeal and perivascular CNS involvement in association with multiple cranial nerves by lymphomatous proliferation in the current case can explain the neurologic signs and permitted to formulate a neurological multifocal diagnosis.
The case described herein contributes to the knowledge of feline neurolymphomatosis, which should to be included among the other neurological entities belonging to the neoplastic category. The integration of pathologic with biomolecular findings adds to the information concerning the role of FeLV on TCRγ rearrangements in cases of feline lymphoma.
Footnotes
Acknowledgements
The authors have contributed equally to the drafting of the paper.
a.
LSAB Kit, Dako Denmark A/S, Glostrup, Denmark.
b.
Dako Denmark A/S, Glostrup, Denmark.
c.
EP-gradient S thermal cycler, Real MasterMix; Eppendorf AG, Hamburg, Germany.
d.
ABI Prism 310 Genetic Analyzer, Life Technologies Europe BV, Monza, Italy.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
