Abstract
The hemagglutination inhibition (HI) assay is the standard diagnostic test for detection of antibodies to avian influenza viruses. It is well known that chicken serum does not require additional serum pretreatment to remove nonspecific inhibitors (NSIs). However, NSIs were recognized in certain Korean local breeds. In the present study, various treatments were compared to remove such NSIs. Heat treatment, red blood cell adsorption, and kaolin treatment did not remove NSIs effectively, and treatment with periodate only partly eliminated the NSIs. Receptor destroying enzyme (RDE) treatment appeared to effectively remove NSIs from chicken sera, regardless of breeds. It is proposed that RDE treatment should be included in the HI tests for serological diagnosis of avian
Infection of chickens with avian
The hemagglutination inhibition (HI) assay is a useful method among the various diagnostic methods available for detecting and measuring AIV subtype-specific antibodies. However, the HI test can be affected by the presence of nonspecific inhibitors (NSIs). The presence of NSIs against influenza virus in human and animal sera is well known,2,5,18-20 and there are many publications on serum treatments and reagents.4,12,21,22 Sera from avian species closer to turkeys and geese than to chickens can cause agglutination of chicken red blood cells (RBCs). Therefore, for use of the HI test on sera from those species, agglutination should first be determined and removed by adsorption with chicken RBCs. 23
In South Korea, intensive surveillance programs for the control of HPAIV and NAIV have been in operation since the first H5N1 subtype HPAIV outbreak in 2003. In such programs, the HI assay on chicken sera is used to detect antibodies naturally acquired from infection with NAIVs, but not to detect antibodies against the H5 and H7 subtypes of HPAIV. Because the government of Korea has adopted the eradication policy to control NAIV and HPAIV, it has not permitted vaccination against the H5 and H7 subtype viruses. Meanwhile, the inactivated H9 vaccine has been in use since 2007 6 because the H9N2 virus is endemic and has caused severe economic losses in the South Korean poultry industry following the first outbreak of LPAIV H9N2 in 1996.11,13,14
During HPAIV surveillance in 2009, suspect HI test results were obtained on chicken sera from 5 Korean native chicken farms. The sera were retested following kaolin and heat treatment, and tested positive for the H5N1 and H9N2 AIVs, with a higher titer than before treatment. Additional samples for virus isolation were collected and tested. Although H9N2 virus infections were identified on 2 of the farms, no H5 subtype virus was isolated. The positive results of the HI test against H5 antigen were thus false positives, which provided the impetus for the present study to compare various treatments of serum from different breeds of chickens to obtain the accurate HI titers.
Four groups of poultry sera (H5-positive layer, H9-positive layer, breeder, and Korean native chicken) were tested. The Ck L-I group (
Hemagglutination inhibition (HI) titers (mean ± standard error) of serum samples from various chicken breeds subjected to 5 treatments for removal of nonspecific inhibitors of agglutination.*
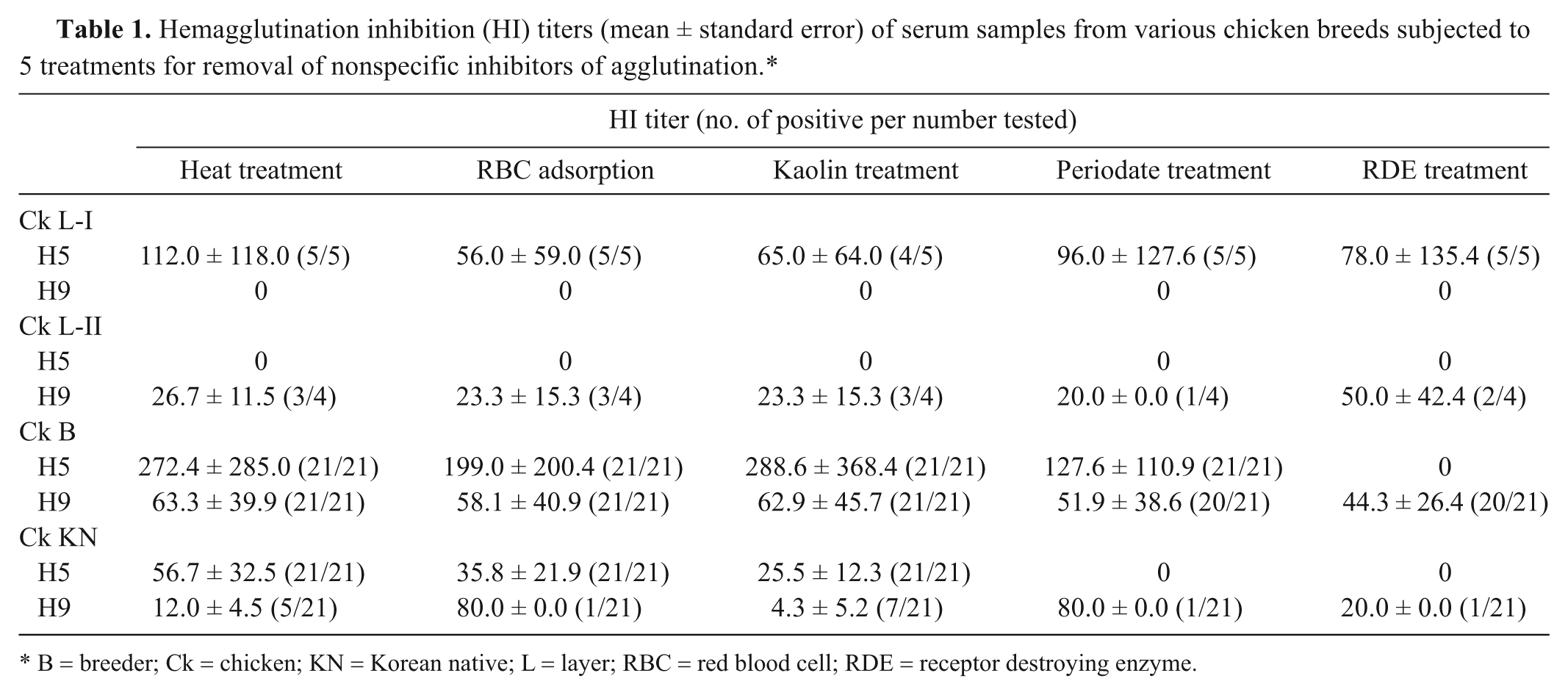
B = breeder; Ck = chicken; KN = Korean native; L = layer; RBC = red blood cell; RDE = receptor destroying enzyme.
A/chicken/Korea/310/01 (H9N2) and A/chicken/Korea/IS/06 (H5N1) viruses were inactivated and used as antigens for the HI test. Formalin-inactivated antigens were provided by the Avian Disease Division, National Animal Disease Control Project of the Ministry of Food, Agriculture, Forest and Fisheries of the Republic of Korea. In brief, the viruses were grown in the allantoic fluid of 9- to 11-day-old specific pathogen–free embryonated chicken eggs a in an incubator at 37°C for 72 hr. The viruses were inactivated by the addition of 0.1% formalin b (v/v) and kept at 20°C for 12 hr. The inactivation was confirmed by 2 blind passages in specific pathogen–free embryonated chicken eggs.
To evaluate the effect of the removal of NSIs, the following treatments were used: heat treatment, RBC adsorption, kaolin treatment, periodate treatment, and receptor destroying enzyme (RDE) treatment. Heat treatment was performed by inactivating undiluted serum at 56°C for 30 min and then adding to 9 volumes of phosphate buffered saline (PBS). For removal of NSI by adsorption of serum with chicken RBC, 1 volume of serum and 1 volume of 5% chicken RBC suspension were mixed for 1 hr. Eight volumes of PBS were added and centrifuged at 3,000 ×
For periodate treatment, 1 volume of serum was treated with 1 volume of 0.4% trypsin d (w/v in saline) at 56°C for 30 min. After cooling to room temperature, 3 volumes of 0.01 M potassium periodate e (KIO4) was added and incubated for 15 min at room temperature. Then, KIO4 was inactivated by adding 3 volumes of 1% glycerol f (v/v in normal saline) and 2 volumes of PBS for 1 hr at 4°C resulting in a 1:10 dilution of periodate-treated serum.
Lyophilized RDE g (50 units) was dissolved completely in 20 ml of sterile saline. One volume of serum was treated with 3 volumes of RDE solution. The mixture was incubated overnight (18–20 hr) in a 37°C water bath. The RDE was inactivated by the addition of 3 volumes of a 2.5% sodium citrate h solution (w/v in distilled water) for 30 min at 56°C. Last, 3 volumes of PBS were added to obtain a 1:10 dilution of RDE-treated serum.
The HI assay was performed as outlined in the World Organization for Animal Health (OIE) manual. 23 Serum samples were serially diluted in rows of a V-bottomed, 96-well plate up to the eighth well using a 2-fold dilution series. The lowest dilution used was 1:10. A 1% chicken RBC suspension (v/v in PBS) was used. The reciprocal of the highest serum dilution that completely inhibited erythrocyte agglutination was assigned as the HI titer.
In the Ck L-I group, 5 sera that were treated by heat, RBC adsorption, kaolin, periodate, and RDE tested positive to A/chicken/Korea/IS/06 (H5N1) virus, while none of the serum samples tested positive to A/chicken/Korea/310/01 (H9N2) virus (Table 1). The Ck L-II sera tested positive to the H9N2 virus regardless of serum treatments but did not test positive for H5N1 virus. The 21 Ck B group serum samples tested positive with high titer values to the H5 virus even though these samples were treated by heat, RBC adsorption, kaolin, and periodate. Only the RDE-treated sera tested negative to H5 virus. The Ck B group sera treated by 5 methods all tested positive to H9 virus with a constant titer. In the Ck KN group sera, heat treatment, RBC adsorption, and kaolin treatment did not change the positive result to H5 virus; however, periodate and RDE effectively eliminated false-positive results for H5N1 virus antibodies. Meanwhile, the HI result for the Ck KN sera was positive for H9 virus with all 5 treatment methods.
The results show that the occurrence of NSIs in chicken sera varies according to chicken breeds. The sera of layer chickens (Ck L-I and Ck L-II groups) contained antibodies against H5 and H9 viruses, respectively, and the sera of breeder and Korean native chicken (Ck B and Ck KN groups) yielded false-positive results for the H5N1 virus, demonstrating a NSI response to virus. Heat treatment, RBC adsorption, and kaolin were not affective in eliminating NSIs from chicken sera; however, periodate and RDE treatments eliminated false-positive result for the A/chicken/Korea/IS/06 (H5N1) virus in the sera of the Ck KN group. Only RDE treatment eliminated NSIs in the sera of the Ck B group.
Both the Ck B and Ck KN groups tested positive for the H9N2 antigen, but the percentage of sera in the Ck B group that tested positive was higher and more consistent than that of the Ck KN group, regardless of the serum pretreatments (Fig. 1). It seems that the H9-positive antibodies of the breeder chickens were induced by the H9 vaccine and that the H9-positive antibodies of Korean native chicken were naturally acquired from an infection with H9 AIV. Since 2007, most of the layer and breeder chickens in South Korea have been vaccinated with H9 vaccines, but Korean native chicken farms rarely used this vaccine.
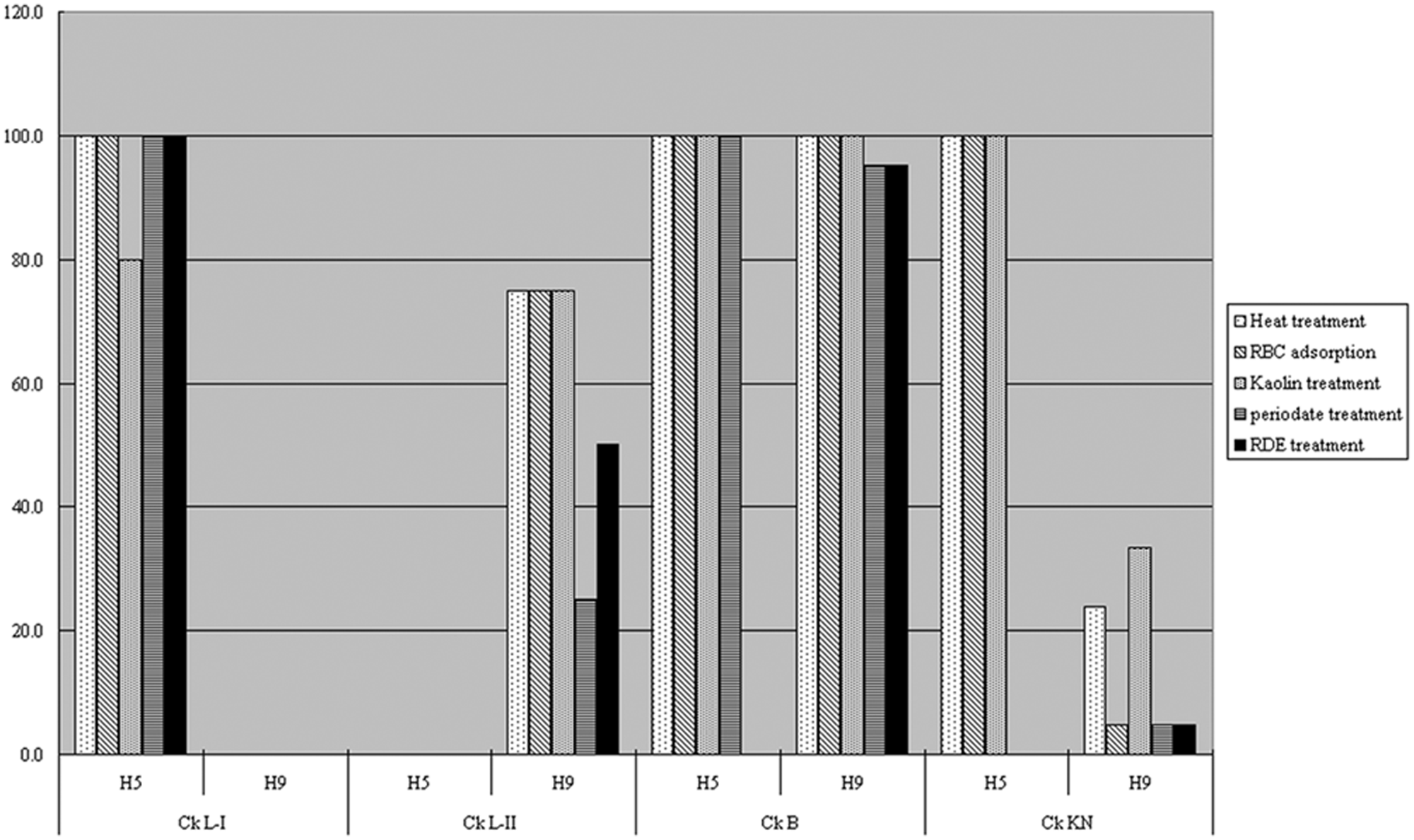
Effect of various treatments on hemagglutination inhibition test results from 4 groups of chicken sera tested against A/chicken/Korea/IS/06 (H5N1) and A/chicken/Korea/310/01 (H9N2) avian
In an additional experiment, heat treatment, periodate treatment, and RDE treatment were compared using field sera samples. A total of 3,347 samples from 169 flocks were collected from Korean native chicken farms in 2009, and the HI assay was performed on these samples to determine the presence of antibodies to H5 and H9 antigens (Table 2). For the heat-treated sera, 17.8% of 169 flocks and 2.4% of 3,347 sera were positive against H5N1 virus, but the percentage of positive samples was lower (2.4% and 0.2%, respectively) after periodate treatment. All RDE-treated sera tested negative against the H5N1 virus. The percentage of samples positive for H9 virus antibodies was reduced when the sera were treated by RDE. The lower positive rate after RDE treatment may represent the birds naturally infected with H9 virus on farms. The RDE treatment was an effective way of removing NSIs during HI tests for detecting H5 and H9 antibodies from the Korean native chicken sera.
Hemagglutination inhibition assay with H5 and H9 antigens using Korean native chicken sera (collected from 2009 avian influenza monitoring) subjected to 3 treatments for removal of nonspecific inhibitors of agglutination.*
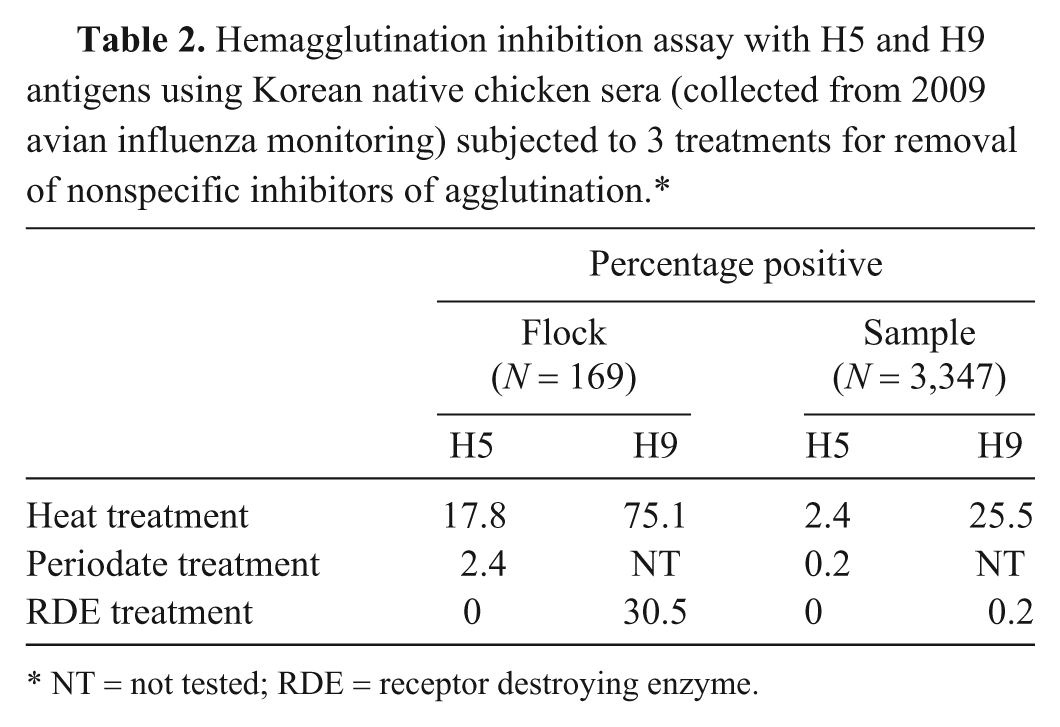
NT = not tested; RDE = receptor destroying enzyme.
Although the sera from the majority of avian species do not contain NSIs and do not need to be treated prior to testing, 16 the results of the present study indicate that the serum of some chicken breeds may contain NSIs, which adversely affect the specificity of the HI test. Therefore techniques are needed to eliminate the NSIs. The heat-labile factor, one of the NSIs in serum, is known to be destroyed by heating at 56°C for 30 min. 7 Nonspecific agglutination of the chicken erythrocytes used in the HI test can be removed by pretreatment of the serum with chicken erythrocytes. 15 Kaolin adsorbs NSIs but may not completely inactivate them. 4 Periodate acts by oxidizing cis-hydroxyl groups on carbohydrates, and RDE cleaves N-acetylneuraminic acid residues from sialyloligosaccharides of α-2 macroglobulin. 19 The results of the present study suggest that of the 5 suggested treatments, only RDE treatment appeared to effectively remove NSIs from all chicken breeds studies and should be the preferred treatment for the HI assay of AIV.
Footnotes
Acknowledgements
The authors thank Dr. Youn-Jeong Lee at the Avian influenza laboratory of the Avian Disease Division for providing the H5-positive serum used in this research.
a.
SPF ECEs, Hy-Vac, Adel, IA.
b.
Formalin, Sigma-Aldrich Korea Ltd., Yongin, South Korea.
c.
Kaolin, Sigma-Aldrich Korea Ltd., Yongin, South Korea.
d.
TPCK-trypsin, Thermo Scientific, Rockford, IL.
e.
Potassium periodate, Sigma-Aldrich Korea Ltd., Yongin, South Korea.
f.
Glycerol, Sigma-Aldrich Korea Ltd., Yongin, South Korea.
g.
RDE, Denka Seiken Co. Ltd, Tokyo, Japan.
h.
Sodium citrate, Sigma-Aldrich Korea Ltd., Yongin, South Korea.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) declared that they received no financial support for their research and/or authorship of this article.
