Abstract
To determine if ruminal hydrogen sulfide, urine thiosulfate, or blood sulfhemoglobin could be used as diagnostic indicators for sulfur-induced polioencephalomalacia, 16 steers (8 cannulated, 368 ± 12 kg; 8 unmodified, 388 ± 10 kg; mean ± standard error) were fed 1 of 2 dietary treatments. Diets consisted of a low sulfate (0.24% S; control) wheat midd–based pellet or the control pellet with sodium sulfate added to achieve a high-sulfate (0.68% S) pellet. As designed, intake did not differ (
Introduction
Ethanol production in the United States continues to expand at a rapid rate and has led to an increased supply of corn co-product feeds. During ethanol production, sulfuric acid is used for fermentor pH control and as a cleaning agent. The residual sulfate in these co-products can led to toxicity when fed to ruminants at high amounts. In the rumen, sulfate is reduced to sulfide by sulfate-reducing bacteria. Hydrosulfide has a pKa of 7.04, meaning that at a pH of 7 in the rumen, one-half of the sulfide will be in its gaseous form, hydrogen sulfide (H2S). As rumen pH decreases, the amount of sulfide present in its gaseous form increases. Increased ruminal H2S concentrations have been associated with the neurological disorder polioencephalomalacia7,9,11 (PEM). In addition to sulfur (S) toxicosis, this disorder can also be caused by lead poisoning, water deprivation, and thiamine deficiency. Currently, there are no antemortem diagnostic tests available that can be performed on tissues or fluids of an animal that quantify the risk of S-induced PEM (S-PEM) or allow veterinarians to differentiate between S-PEM and other causes of PEM. The objective of the current research was to determine if ruminal concentrations of H2S or urine thiosulfate could be used as diagnostic indicators for S-PEM.
Materials and methods
Procedures and use of animals for this experiment were approved by the Iowa State University Animal Care and Use Committee (8-09-6796-B). Sixteen steers (8 cannulated, 368 ± 12 kg; 8 unmodified, 388 ± 10 kg; mean ± standard error) were used in a 2 × 2 factorial design. Prior to the start of the trial, steers were paired by modification status and by weight. Steers within each pair were then randomly assigned to treatment. The treatment included low sulfate control (C) pellet (0.24% S) or a high sulfate (HS) pellet (0.68% S; Table 1). The sulfur level in the HS pellet was increased through the addition of sodium sulfate at 2.0% of pellet dry matter (DM). Eight steers (4 pairs per period) at a time were housed in stalls with individual feeders and waterers. Steers were adapted to the stalls and remained on a low sulfur diet for 4 days after which steers began adaptation to their treatment diets. During the first 7 days of adaptation to the new diet, the amount of diet offered was based on initial body weight. On day 1 of diet adaptation, steers were fed 1% of body weight and for the next 6 days the amount being fed was increased by 0.25% body weight each day. Starting on day 8, the HS-fed steers were fed 110% of the previous day’s intake while the C-fed steers were fed the average of the previous day’s intake of their HS counterpart. In addition to pellets, steers on both treatments were fed chopped smooth brome grass hay at 5% of their previous day’s pellet intake. Steers were fed hay at 0800 and fed pellets 15 min later. Steers had usually consumed all of the hay offered before the pellets were fed. Feed was sampled daily and composited on a weekly basis for determination of sulfur content. Feed samples were dried at 70°C to determine DM and then ground though a 2-mm screen. Dried and ground samples were then acid digested with trace mineral grade nitric acid, diluted to 10% nitric acid with deionized water, and analyzed for sulfur by inductively coupled plasma atomic emission spectrometry. a Orts were collected prior to feeding, weighed, and a subsample was dried at 70°C to determine DM that was not consumed each day.
Composition of pellets fed to steers in the current study.

Added to achieve cobalt, copper, iodine, manganese, selenium, and zinc levels at the National Research Council 13 recommended concentration in the diet and provide vitamin A at 4,400 IU/kg of the diet dry matter.
Blood and rumen gas were sampled from all steers 8 hr postfeeding on days 7, 10, 14, 21, 24, and 28. For determination of H2S concentration in the rumen gas of the noncannulated steers, the skin of the left paralumbar fossa was clipped, scrubbed, and disinfected with isopropyl alcohol and povidone-iodine. A sterilized 16-gauge, 10.2-mm-long needle was introduced into the rumen gas cap through the left paralumbar fossa. Using a minimal amount of elastomer tubing, a 1-cc syringe was connected to a H2S detector tube. b A volumetric gas sampling pump b was used to pull 50 or 100 ml of gas though the detector tube. Ruminal gas samples from the cannulated steers were obtained through a modified cannula plug that contained a 1.4 cm in diameter plastic adapter that allowed insertion of a stainless steel sampling tube (5 mm in diameter by 40 cm long) without escape of rumen gas. The sampling tube was inserted into the rumen approximately 30 cm, and 25 ml of rumen gas was pulled through the tube via a syringe. Then the sampling tube was connected to the H2S detector tube via elastomer tubing and gas was pulled though the apparatus using the volumetric sampling pump. c After gas sampling, rumen fluid was collected from the cannulated steers using a suction strainer. Whole blood was collected via jugular venipuncture using 3-ml Vacutainer tubes d containing heparin, and was analyzed for total hemoglobin and sulfhemoglobin. 6 Sulfhemoglobin concentration was calculated using the extinction coefficient. 1
On day 24, H2S concentrations of the rumen gas of the cannulated steers were measured prior to feeding and at 2, 4, 6, 8, 10, 12, 16, 20, and 24 hr after feeding. At all time points except 8 hr after feeding, steers remained in their crate while gas samples were collected. At 8 hr after feeding, steers were moved to a chute for blood sampling, and gas samples were taken while the steers were in the chute. In-dwelling ruminal boluses
e
were inserted into the cannulated steers on day 12 of each period, and ruminal pH was measured every 15 min until the end of the trial. The mean pH of each hour was
Urine was collected between −2 and 0 hr prefeeding for AM sampling and between 7 and 9 hr postfeeding for the PM sampling on the days that ruminal H2S measures were taken. Urine was collected using stainless steel trays placed under the steer’s stalls and waiting for the steer to voluntarily urinate. Urine samples were frozen (−20°C) until analyzed for thiosulfate. Urine thiosulfate was detected using the bis(pentafluorobenzyl) disulfide method. 8 The concentration of urinary creatinine was measured using a commercially available kit f that utilized the Jaffe reaction.
During the trial, steers were monitored for symptoms of PEM in the morning prior to feeding and at 10 hr after feeding. On day 17, 1 steer fed the HS diet presented with signs of PEM (inappetence and blindness). A blood sample was taken from this steer and his pair-fed control, and both steers were subsequently removed from the study. The steer presenting with PEM was treated with dexamethasone (20 ml intravenously) and thiamine (20 ml intravenously) twice a day for 3 days and was given access to free choice hay. The steer fully recovered and was fed a low sulfur finishing diet. At harvest (approximately 6 months later) brain tissue was collected, and focal gliosis was observed.
All data were analyzed using the mixed procedure of SAS,
g
and the effects of period and steer were considered random. Hydrogen sulfide (8 hr), urine thiosulfate, and sulfhemoglobin data were analyzed as repeated measures and included the fixed effects of treatment, day, cannulation, and their interactions. Dry matter and sulfur intake data were analyzed as repeated measures and included the fixed effects of treatment, day, and their interactions. Serial samples of H2S were analyzed as repeated measures and included the fixed effects of treatment, hour, and their interactions. Ruminal pH data were analyzed as repeated measures and included the fixed effects of treatment, day, hour, treatment-by-day, and treatment-by-hour. Significant differences were defined as
Results
Dry matter and sulfur intake
As designed, DM intake of C-fed (6.08 kg−1·hd−1·day) did not differ (
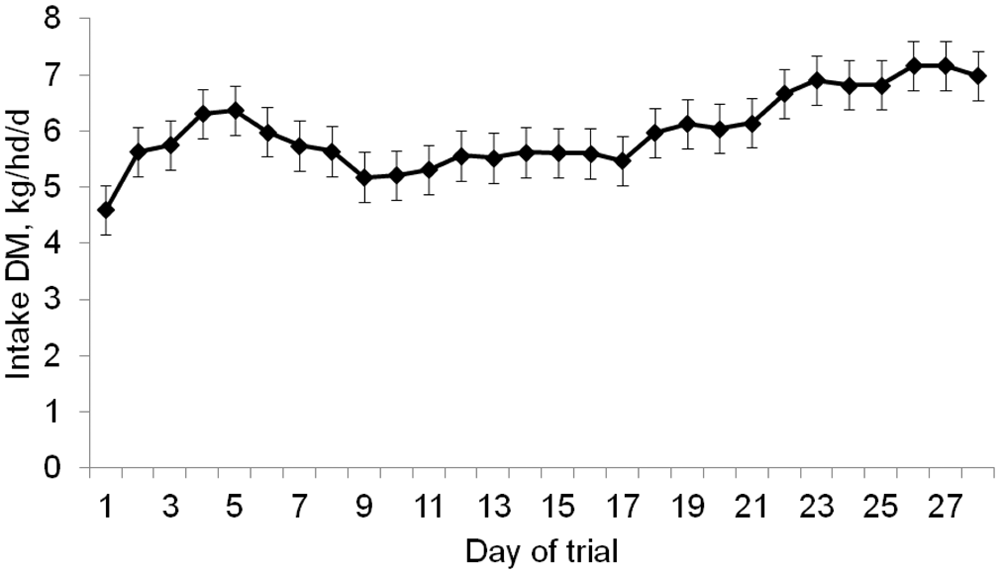
Mean daily DM intake of steers (pooled across modification status of steers;
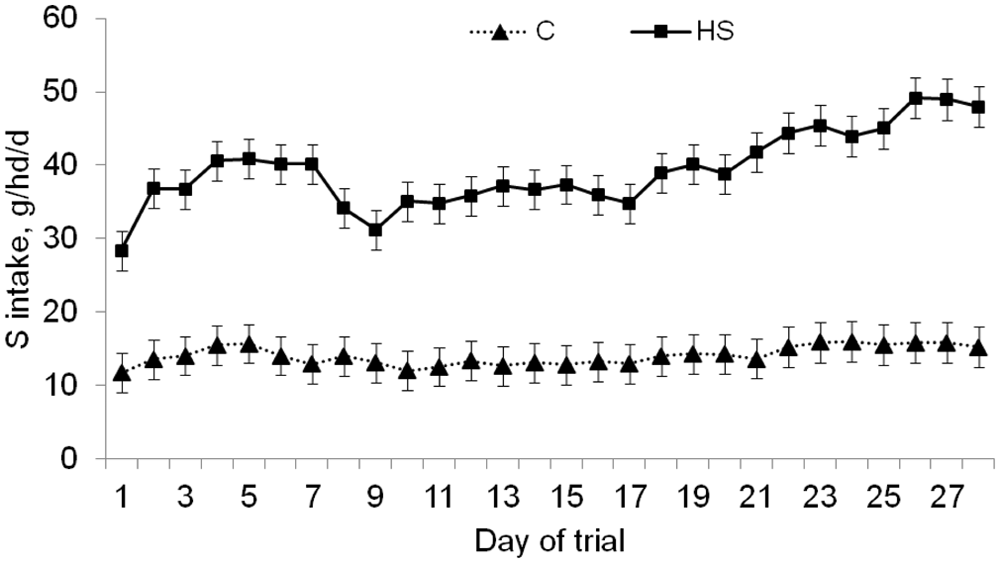
Daily sulfur (S) intake (in grams) of steers (pooled across modification status of steers;
Rumen hydrogen sulfide
There was no effect (
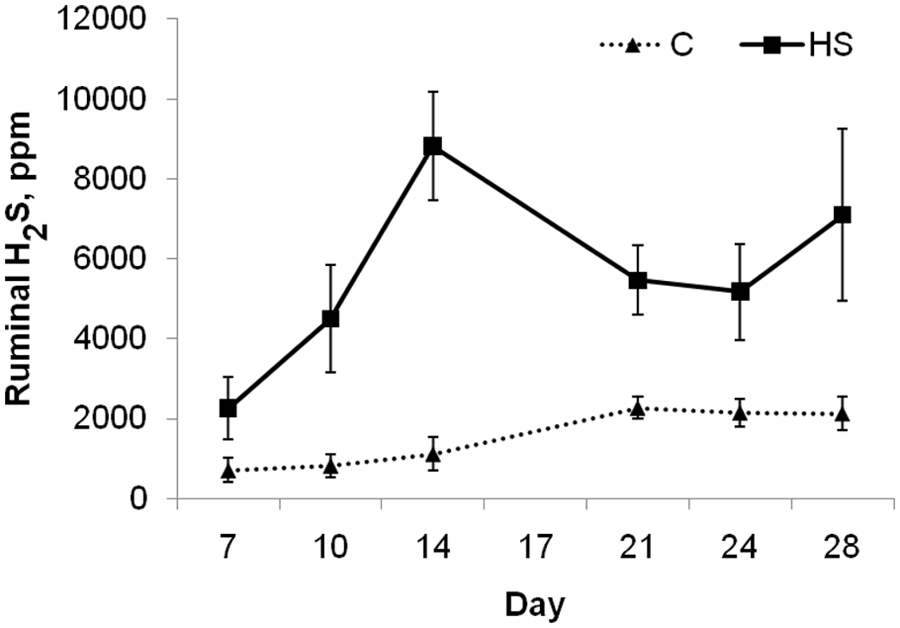
Ruminal hydrogen sulfide (H2S) concentrations (pooled across modification status of steers;
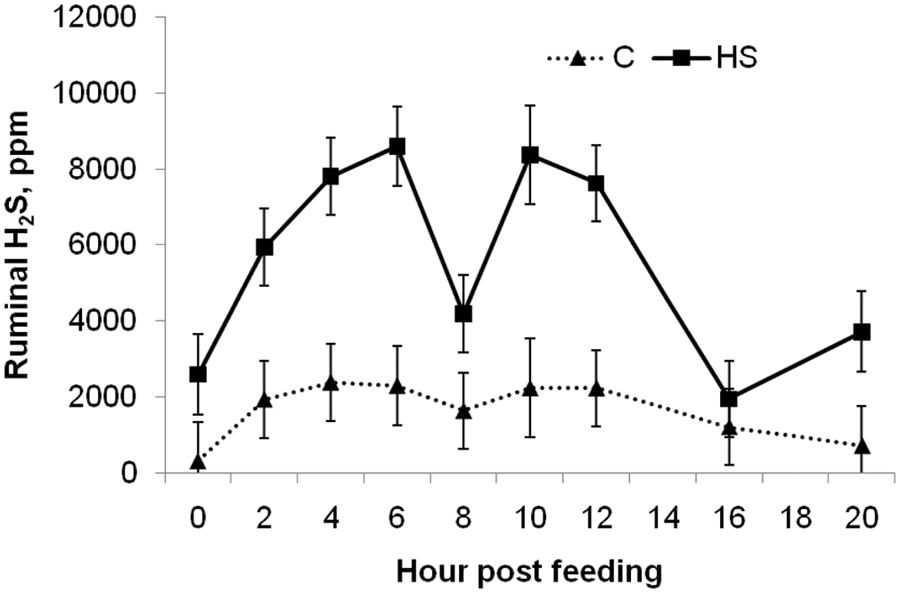
Ruminal hydrogen sulfide (H2S) concentrations on day 24 during a 24-hr period for cannulated steers fed either control (C; 0.24% sulfur) or high sulfur (HS; 0.68% sulfur) diet (treatment by hour;
Ruminal pH
There were significant (
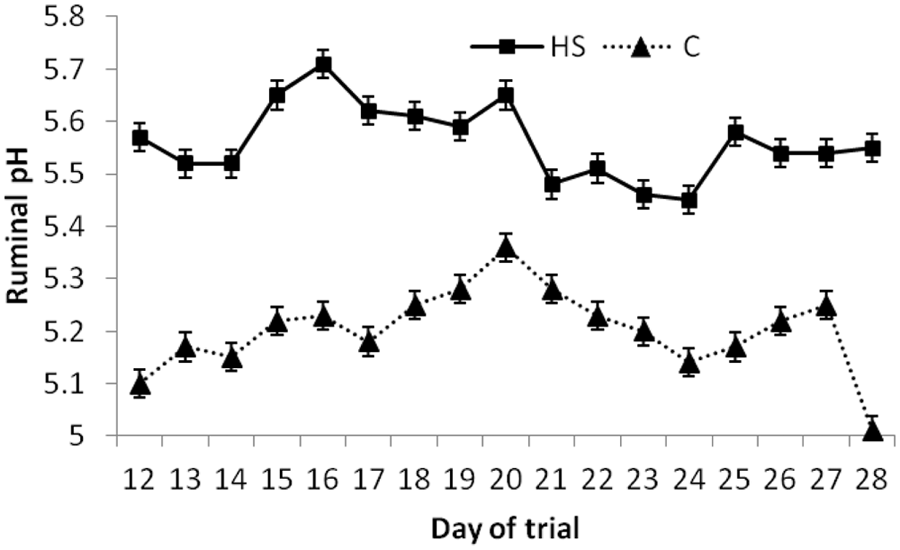
Average daily ruminal pH for cannulated steers fed either control (C; 0.24% sulfur) or high sulfur (HS; 0.68% sulfur) diet (treatment by day;
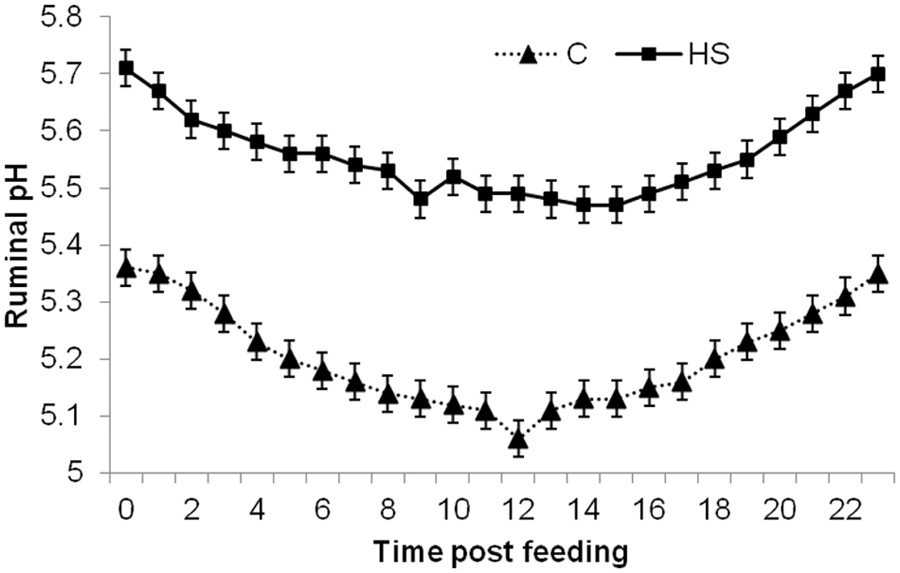
Diurnal ruminal pH of cannulated steers fed either control (C; 0.24% sulfur) or high sulfur (HS; 0.68% sulfur) diet (treatment by hour;
Thiosulfate
There was a treatment-by-time of sampling interaction (
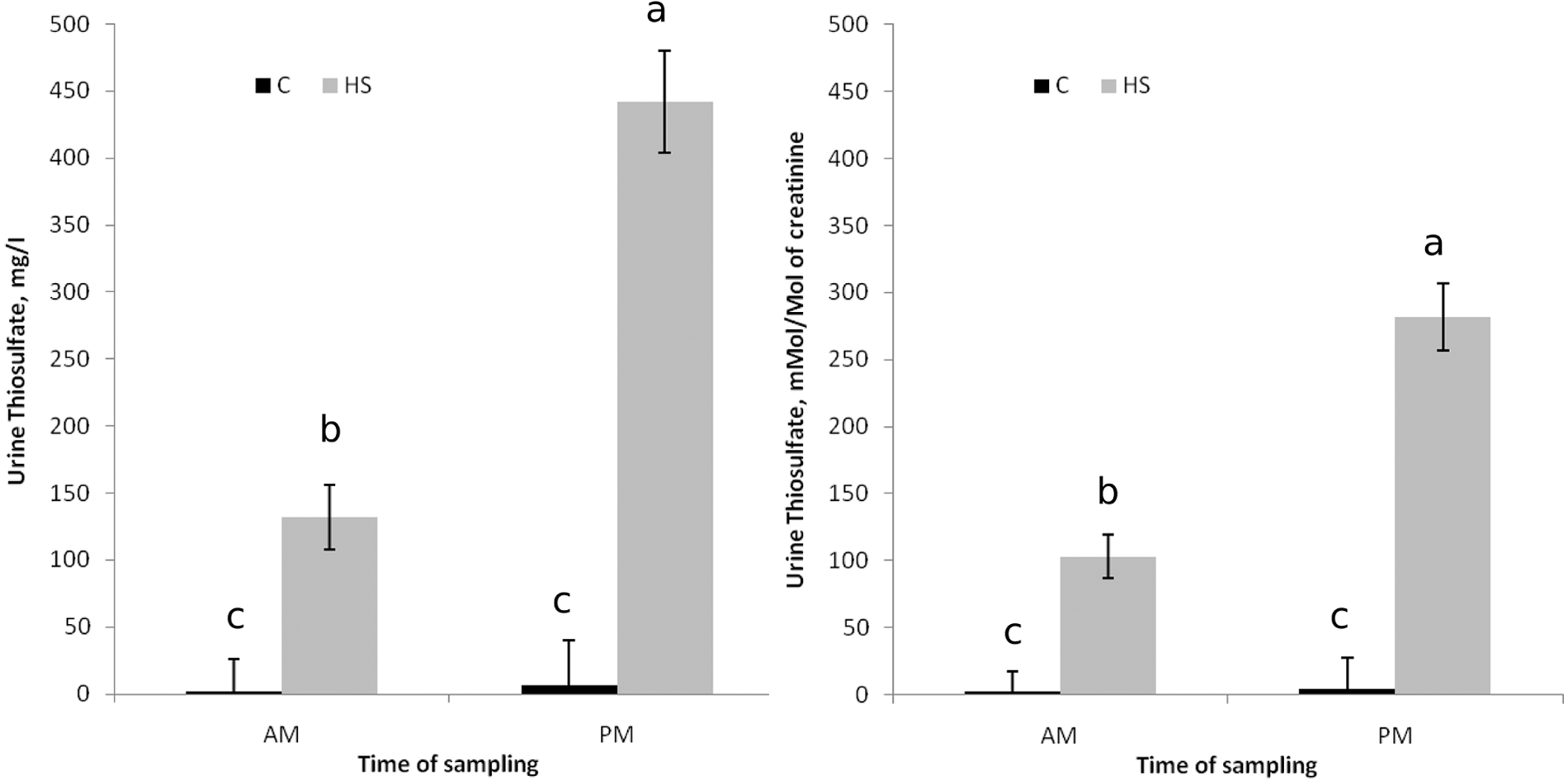
Urine thiosulfate concentrations (pooled across modification status of steers [
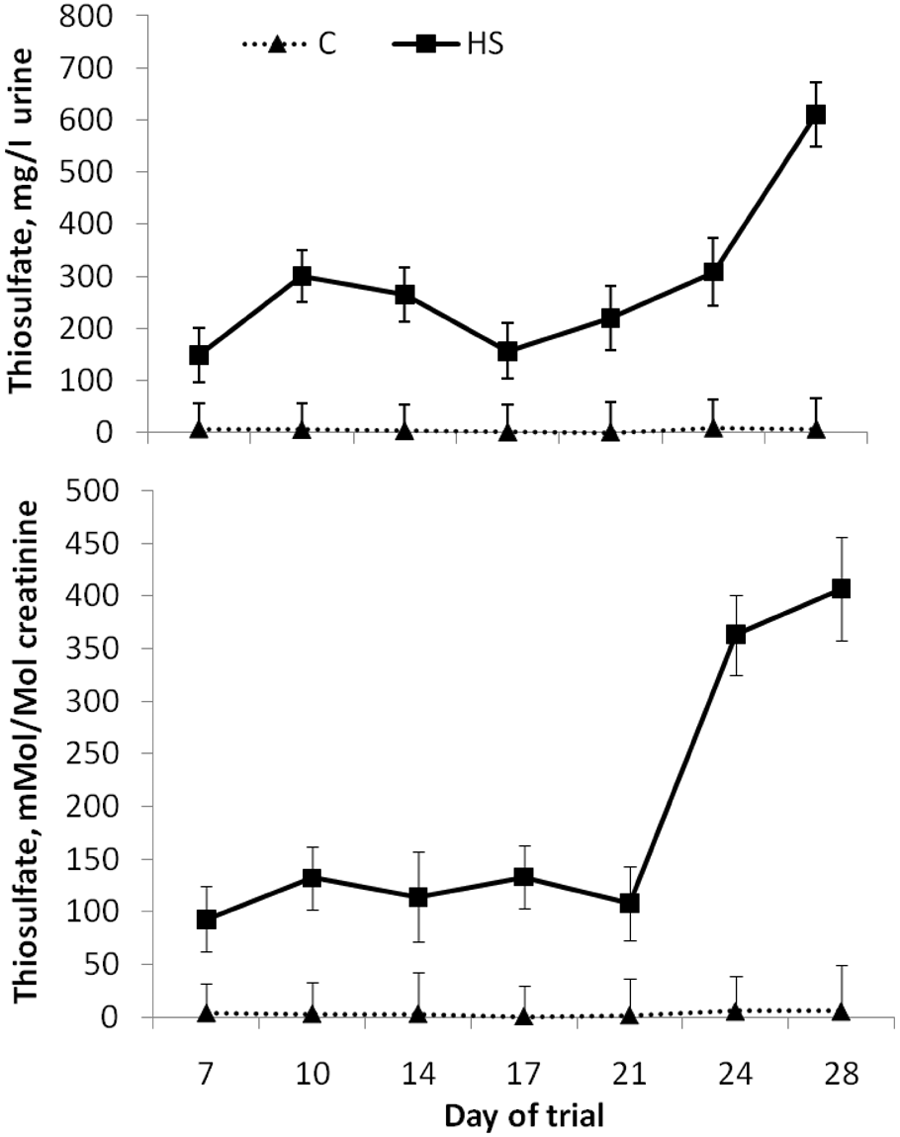
Urine thiosulfate concentrations (pooled across modification status of steers [
Sulfhemoglobin
Total hemoglobin did not differ (
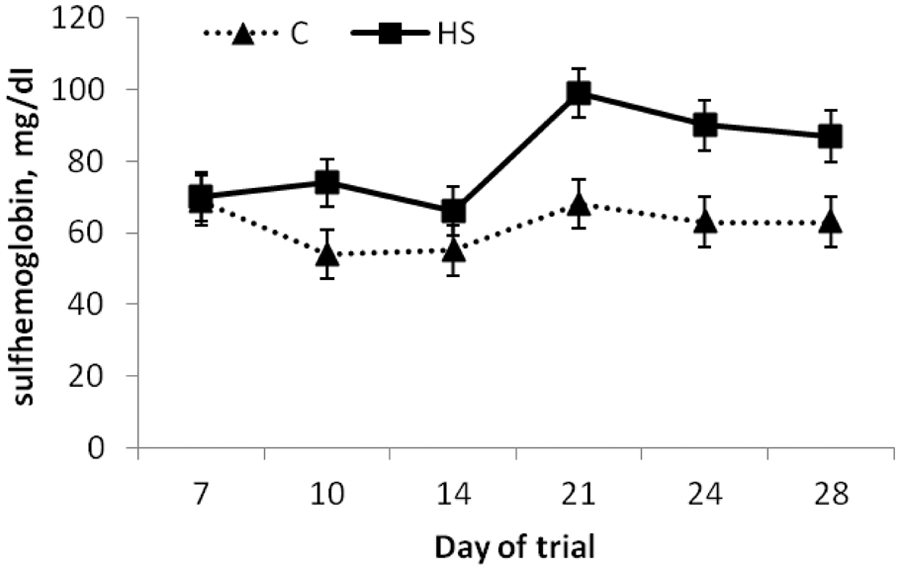
Blood sulfhemoglobin concentrations (pooled across modification status of steers;
Discussion
Polioencephalomalacia was first reported in 1956 and was described as a disorder of the nervous system. At one time, PEM was thought to be caused exclusively by thiamine deficiency. However, PEM is now known to be associated with several types of metabolic or toxic alterations, including thiamine deficiency, acute lead poisoning, sodium toxicosis, and ruminal generation of high sulfides.
In 1976, a form of PEM associated with a diet high in molasses (0.47% S) and not associated with altered thiamine status was described.
12
The brain and liver thiamine status of cattle that developed PEM did not differ from those in normal cattle. This was one of the first studies to suggest that HS diets could induce PEM independent of thiamine deficiency. Then, in the early 1980s, it was noted that in several cases, cattle with PEM had been consuming rations containing high concentrations of gypsum or other sulfate salts (used as intake limiters). In 1982, cattle from 72 herds in Missouri that were fed rations with either high SO4 (
In the late 1980s and early 1990s, PEM was experimentally induced by feeding HS diets to both cattle7,15 and sheep. 10 In one study, 15 14 Holstein steers were fed a pelleted semipurified diet that was low in long fiber and high in carbohydrates and contained 0.3% sulfur. Thiamine deficiency was not induced by the diet. In contrast, steers consuming the HS diet had higher rumen and plasma thiamin content compared to baseline levels taken when steers were fed an alfalfa diet. The thiamine pyrophosphate effect on erythrocyte transketolase activity was also unaltered. However, clinical signs of PEM were seen in 9 steers and brain lesions were found in 12 steers.
In the late 1990s, a positive association between elevated sulfide levels in the rumen gas cap and the onset of PEM in steers was demonstrated.7,11 In one study,
7
Holstein steers (
Dianion sulfide (S2−) is the main metabolite of sulfate metabolism in the rumen (Bray AC, Till AR: 1975, Metabolism of sulphur in the gastro-intestinal tract.
In the current study, the ruminal pH of HS-fed steers was consistently greater than C-fed steers despite both treatment groups consuming the similar amounts of DM. The increased ruminal pH in HS-fed steers was most likely due to the use of sodium sulfate (a neutral salt; pH 7) to increase the sulfur in the diet.
There was a day effect for ruminal H2S with peak ruminal concentrations occurring on day 14 of the trial. This did not correlate to the highest dietary intakes or the lowest ruminal pH. Other studies10,12 have shown a peak in ruminal H2S and incidence of PEM between 1 and 4 weeks after starting a HS diet, suggesting that cattle are the most susceptible to acute sulfur toxicosis during the first 4 weeks after transitioning to a HS diet. This may relate to microbial population or metabolism changes that may be occurring during this dietary transition period.
The effect of time of day of sampling did appear to correlate to ruminal pH. The peak in H2S concentrations that occurred in HS-fed steers 4–12 hr after feeding was partially explained by the decrease in ruminal pH that occurred during this period. The H2S concentration of C-fed steers did not statistically differ throughout the day; however, numerically they had a similar pattern to those fed a HS diet. The fact that H2S concentrations and pH measures were not well correlated in C-fed steers is not totally unexpected as their pH only ranged from 5.3 to 5.1 within a single day. At a pH of 5.3, over 95% of sulfide present will be in the form of H2S and thus a further decrease to 5.1 would not be expected to cause a significant increase in the conversion of HS− to H2S.
Despite the fact that H2S concentration was significantly elevated (>4,000 mg/l of ruminal gas) in HS-fed steers, only 1 case of PEM was observed. This steer had a H2S concentration of 12,000 mg/l in its ruminal gas on day 14 two days prior to the onset of symptoms. However, 3 other steers reached or exceeded this concentration during the study but did not show signs of PEM. Therefore, a threshold concentration of H2S above which PEM would be induced could not be found. The reason why 1 animal succumbed to PEM while others did not is unknown but may be a result of differences in eructation frequency or pattern. Researchers 4 introduced 300 ml of H2S gas into the rumen of mature sheep and caused PEM-like symptoms within 15 sec to 2 min. However, time of onset differed depending on timing after feeding and frequency of eructation. Assuming a rumen gas cap volume of 2.8 liters for mature sheep, the researchers would have introduced the equivalent concentration of 107,000 mg/l of HS in the rumen. 2
In a previous study, 7 it was suggested by the authors that PEM was associated with H2S concentrations above 2,000 mg/l. In the current study, 42% and 79% of gas samples taken 8 hr postfeeding from C-fed and HS-fed steers, respectively, had H2S greater than 2,000 mg/l. However, only 1 case of PEM was diagnosed. One of the differences between the current study and the previous study4 is the time at which samples were taken postfeeding. The results of serial sampling taken throughout the day in the present study showed that time of sampling can have major impacts on the H2S values obtained. The sampling protocol was to take samples 8 hr postfeeding whereas the previously mentioned study 4 was not consistent on time of sampling but typically took samples in the morning (D. H. Gould, personal communication, 2010). The apparent separation of H2S concentrations between treatments in the current study occurred at 4,000 mg/l, with 65% of gas samples from HS-fed steers being greater than 4,000 mg/l, while no samples from C-fed steers were above 4,000 mg/l. Therefore, ruminal H2S concentrations at 6–8 hr postfeeding above 4,000 mg/l would indicate consumption of high levels dietary sulfur.
The diurnal pattern of H2S may make it difficult to use H2S concentration as a definitive diagnostic tool in the field. Use of this tool is further complicated by the fact that animals with PEM tend to stop eating and drinking, resulting in decreases in H2S concentrations. It may be possible to overcome this difficulty by taking the samples from nonsymptomatic pen mates, at a consistent time after feeding (6–8 hr), as an indicator of possible exposure. More research is needed to further develop the criteria for diagnosis of S-PEM based on ruminal H2S concentration.
Thiosulfate is one of the major metabolites of sulfide and is formed by its oxidation, and it has been suggested that urine thiosulfate could be used as a biological indicator of exposure to H2S in human beings. 5 Thiosulfate in urine has been found 2 to be stable at room temperature for 8 hr and at 4°C for at least 24 hr. After 24 hr, the recovery for thiosulfate at 25°C and 4°C was 67.9% and 95.8%, respectively. 3 Therefore, it was hypothesized that thiosulfate concentration in urine may be useful as a diagnostic tool in ruminants. In the current study, urine thiosulfate seemed to differ greatly, depending on the time of day of sampling. However, urine thiosulfate was considerably greater for HS-fed than C-fed steers in both the morning and evening sampling. Normalizing the urine thiosulfate concentrations to creatinine did not seem necessary as it did not significantly affect the interpretation of the data.
Like ruminal H2S, the apparent diurnal pattern and the fact that animals with PEM tend to stop eating and drinking may make urine thiosulfate difficult to use as a diagnostic indicator of S-PEM. Further research is needed to determine how long urinary thiosulfate remains elevated after an animal stops consuming high dietary sulfur. Two days prior to the onset of symptoms the steer that exhibited signs of PEM did not have extremely high concentrations of urine thiosulfate in the PM. In fact, this animal had the lowest concentration (101 mg/l) for that time-point when compared with that of other steers receiving HS (866, 582, and 342 mg/l). Therefore, urine thiosulfate may not be useful in predicting likelihood of sulfur toxicosis.
Taking urine samples of nonsymptomatic pen mates 6–9 hr after feeding, however, may be useful in determining if there has been consumption of high concentrations of sulfur. Urine from C-fed steers did not have thiosulfate concentrations above 50 mg/l; most samples (100% of AM and 92% of PM) were below 30 mg/l, with the majority (94% of AM and 77% of PM) being less than 15 mg/l. Whereas the majority (60% of AM and 81% of PM) of urine samples from HS-fed steers had thiosulfate concentrations above 15 mg/l; most (30% of AM and 76% of PM) of the urine samples from HS-fed steers had concentrations above 100 mg/l. Therefore, urinary thiosulfate concentrations above 50 mg/l would seem to indicate consumption of a high amount of dietary sulfur.
Absorbed sulfide can convert functional hemoglobin to sulfhemoglobin that cannot carry oxygen. 4 Thus, formation of sulfhemoglobin may cause hypoxia and thus cause brain cell dysfunction and death though oxygen deprivation. In a previous study 4 in which steers were fed a high sulfate PEM-inducing diet, sulfhemoglobin concentration were not increased. It was suggested by the authors of the previous study that this was most likely due to the short feeding period of only 17 days. Many cases of PEM, however, have been demonstrated within 9–15 days after changing to a HS diet. In the current study, sulfhemoglobin concentrations were increased in HS-fed compared to C-fed steers, but this increase was likely not great enough to cause the onset of hypoxia (sulfhemoglobin was only 0.58% of total hemoglobin). The majority (77%) of blood samples from C-fed steers had less than 70 mg/dl of sulfhemoglobin whereas the majority (59%) of samples from HS-fed steers had sulfhemoglobin concentrations above 70 mg/dl. Sulfhemoglobin of the steer that exhibited signs of PEM was only 78 mg/dl two days prior to onset of symptoms. Given that sulfhemoglobin concentration was not substantially elevated in the steer that exhibited signs of PEM and the time-consuming nature of this assay, concentration of sulfhemoglobin may not be the best choice for a diagnostic tool for sulfur toxicosis.
A threshold concentration of ruminal H2S above which S-PEM will be induced could not be determined in the current study. It seems that factors other than just ruminal H2S concentration affect individual animal susceptibility to S-PEM. Timing of sampling relative to feeding affected ruminal H2S and urine thiosulfate concentrations, suggesting that time of sampling should be considered when comparing data among studies and diagnosing clinical cases of S-PEM. However, both ruminal H2S and urine thiosulfate concentrations may be useful for differentiation of S-PEM from other causes of PEM. Currently, differentiation between S-PEM and other causes of PEM is determined by estimates of the total sulfur intake from water and feed. However, it is often difficult to monitor water intake and/or know the true composition of the diet being consumed. Sampling urine for thiosulfate or rumen gas for H2S concentration of nonsymptomatic pen mates 4–8 hr after feeding may allow for a better assessment of sulfur exposure.
Footnotes
a.
Optima 7000, Perkin Elmer, Waltham, MA.
b.
Kitagawa America, Pompton Lakes, NJ.
c.
Model 8014-400B, Kitagawa, Pompton Lakes, NJ.
d.
BD, Franklin Lakes, NJ.
e.
Well Cow Limited, Roslin, Midlothian, United Kingdom.
f.
Urinary creatinine assay kit, Cayman Chemical Co., Anna Arbor, MI.
g.
SAS version 9.2, SAS Institute Inc., Cary, NC.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Iowa Office of Energy Independence through the Iowa Power Fund.
