Abstract
A case of infiltrative gastric adenocarcinoma in a 5-year-old female chinchilla (Chinchilla lanigera) is described. The animal died after a 5-day period of lethargy and anorexia. Gross examination revealed increased gastric volume. The gastric wall was thickened and contained numerous firm, white nodules on the serosal surface. The lumen was obliterated by a firm mass and large amount of white, mucous secretion. Numerous white, irregular plaques and nodules were observed on the mesentery, visceral surface of the diaphragm, renal capsule, and uterine serosa (implantation metastases). Histopathological examination revealed an infiltrative gastric adenocarcinoma composed of neoplastic cells organized in a gland-like pattern interspersed with connective tissue. Neoplastic cells infiltrated the lamina propria, submucosa, and muscular layers, and were positive for cytokeratin. Detection of Helicobacter spp. in the gastric mucosa failed using Warthin–Starry and Steiner silver stains or immunohistochemistry, but a Helicobacter-specific nested polymerase chain reaction followed by sequencing was positive for Helicobacter pylori and showed more than 99% similarity to the 16S ribosomal RNA gene.
Keywords
Although cases of gastric cancer in human beings have declined since the early 20th century, this form of neoplasia is still a health concern in human medicine and is the second most common cause of cancer-related death worldwide.9,16 In veterinary medicine, gastric neoplasms are uncommon to rare and primarily affect dogs. 7 Chinchillas (Chinchilla lanigera) have been farmed commercially for their pelts for a long time, but are currently increasing in importance as a pet species. 20 Neoplasms have not been consistently described in chinchillas. 19 Single descriptions of neoplasms include neuroblastoma; carcinoma; lipoma; hemangiosarcoma (unspecified locations) 3 ; multicentric lymphosarcoma involving the liver, spleen, and kidneys 14 ; hepatic carcinoma 15 ; uterine leiomyosarcoma; pulmonary lymphosarcoma; pulmonary adenocarcinoma 6 ; lumbar osteosarcoma 19 ; and salivary gland carcinoma. 20 The current study describes an infiltrative gastric adenocarcinoma in a 5-year-old female chinchilla.
The chinchilla died in the owner’s care after a 5-day period of lethargy and anorexia. The carcass was thin, was depleted of fat tissue, and displayed moderate muscle atrophy. Gross examination revealed a markedly thickened gastric wall. The gastric serosa was irregular and covered by numerous, firm, white plaques and small nodules (Fig. 1). The gastric lumen was almost completely obliterated by a firm, white, transmural mass. Numerous ulcers of 1–10 mm in diameter were observed in the mucosal surface. A copious amount of opaque white mucoid substance was observed within the gastric lumen. Multiple white, irregular plaques were observed covering the mesentery, visceral surface of the diaphragm, renal capsule, and uterine serosa. The liver was swollen and diffusely yellow. Samples from several tissues were immersed in formalin and processed, embedded in paraffin, cut at 3-µm sections, and stained with hematoxylin and eosin. Sections from the stomach were also stained with alcian blue, and Warthin–Starry and Steiner silver stains. Histopathological examination of the gastric mass revealed a poorly demarcated, nonencapsulated, highly infiltrative, transmural proliferation of neoplastic epithelial cells. Numerous gland-like nests lined by 1–4 layers of neoplastic cells supported by prominent mature and immature connective tissue stroma (predominantly in the muscular layer) were constantly observed. Neoplastic cells were cuboidal to columnar with moderate amount of eosinophilic and finely fibrillar cytoplasm. Numerous cells had large intracytoplasmic vacuoles. Nuclei were round to oval and contained finely stippled chromatin. Some neoplastic glands were dilated and filled with faintly basophilic, amorphous, alcian blue–positive substance (mucus). This finding was consistent with intestinal metaplasia. Other glands were dilated and filled with necrotic epithelial and/or inflammatory cells. Neoplastic cellular infiltrates were arranged in small groups or isolated cells containing abundant, intensely eosinophilic cytoplasm and squamous metaplasia with formation of keratin pearls. Overall, the neoplastic cells exhibited discrete anisokaryosis and anisocytosis, which were more prominent in areas with squamous metaplasia. Rare mitoses were seen. A moderate, surface-oriented, lymphoplasmacytic, and neutrophilic inflammatory infiltrate was observed throughout the neoplasm. In other areas, lymphoid aggregates were present in the lamina propria. Numerous fibrin thrombi were seen in blood vessels. Multifocal ulceration and infiltration by large numbers of neutrophils were present in the mucosal surface. Severe hepatocellular vacuolation also was observed.
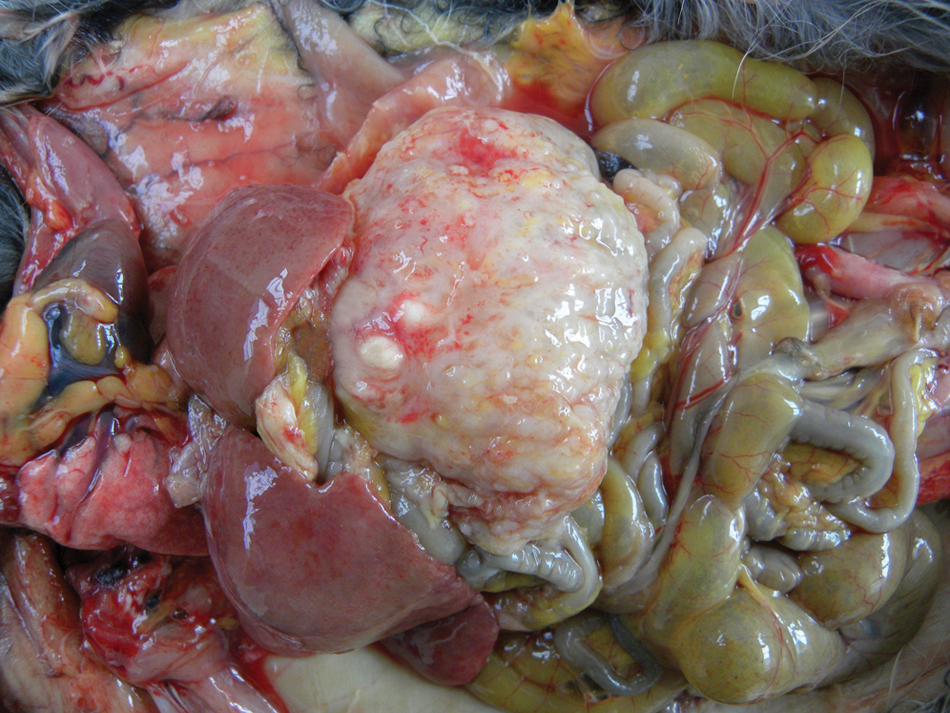
Gastric adenocarcinoma in a chinchilla (Chinchilla lanigera). There is marked thickening of the gastric serosa, which displays numerous variably sized nodules. The liver is moderately enlarged and pale due hepatic lipidosis.
Immunohistochemical analysis of the neoplasm was performed as follows: Sections were dewaxed and rehydrated and endogenous peroxidase activity was blocked with H2O2 3% in distilled water. Antigen retrieval was performed by microwaving (10 min at full power) in Tris–ethylenediamine tetra-acetic acid (pH 9.0). Sections were incubated at 37°C for 60 min with the primary antibodies for pancytokeratin a (polyclonal; dilution 1:2,000) and vimentin a (polyclonal; dilution 1:100). The secondary reagent was biotinylated antibody b followed by application of a streptavidin–peroxidase conjugate. b Substrate development was accomplished with 3,-3’diaminobenzidine (DAB). c Tissue sections were counterstained lightly with hematoxylin and then coverslipped. All neoplastic cells exhibited strong cytoplasmic labeling for pancytokeratin (Fig. 2). A diagnosis of infiltrative gastric adenocarcinoma was based on histopathology and immunohistochemistry (IHC). Because Helicobacter spp. are commonly incriminated in the development of gastritis and gastric neoplasms in human beings and other species and because, in the authors’ experience, Helicobacter sp. is often observed in the gastric mucosa of chinchillas submitted to routine necropsy, a search for Helicobacter organisms in the gastric mucosa was performed using special silver stains, IHC, and polymerase chain reaction (PCR). Immunohistochemical staining for H. pylori was performed as previously described for the other antibodies, except for the use of citrate buffer (pH 6.0) for antigen retrieval. The primary antibody for H. pylori was a polyclonal product that was diluted 1:1,000 prior to use. d Nested PCR followed by genetic sequencing was used to detect the Helicobacter-specific 16S ribosomal RNA (rRNA) gene. 18 DNA was extracted from a gastric fragment with a commercial DNA kit. e The outer primer pair (C70 and B37) was used to generate 16S rRNA amplicons of approximately 1,500 bp. The nested inner primers (C97 and C05), which are specific for the Helicobacter gene, amplified a fragment of 1,200 bp. After purification, f DNA sequencing was done with a commercial sequencing kit g using the sequencing primers B34, B35, B36, C01, C31, and X91 in an automated sequencer g according to the manufacturer’s instructions. Nucleotide sequences were obtained from both strands and analyzed using the CAP3 Sequence Assembly Program (http://pbil.univ-lyon1.fr/cap3.php) and the homology search program BLASTn (http://www.ncbi.nlm.nih.gov/nuccore/JN595861). The alignment analysis of the sequences with previously deposited sequences of the H. pylori genome was conducted with the MultAlin Interface Page Program (http://multalin.toulouse.inra.fr/multalin/).
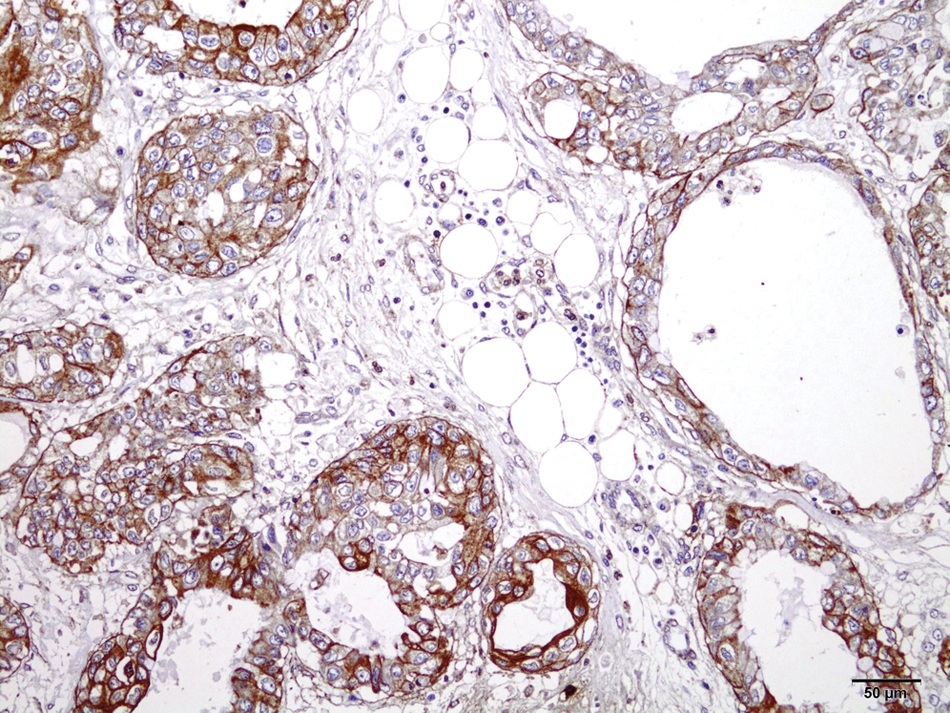
Gastric adenocarcinoma in a chinchilla (Chinchilla lanigera). Positive staining in most neoplastic epithelial cells with antipancytokeratin antibody. Streptavidin–biotin–peroxidase method. Counterstained with Harris hematoxylin. Bar = 50 µm.
Gastric adenocarcinomas in human beings are classified into 2 major types: intestinal or diffuse. 10 The tumor described herein falls in the latter category as it consists of recognizable well-differentiated glands and chronic follicular gastritis. 10 In human beings, intestinal-type adenocarcinomas are more common and usually follow a multistep, progressive process characterized by superficial gastritis, atrophic gastritis, intestinal metaplasia, dysplasia, and neoplasia. 2 Studies have shown that human intestinal-type gastric adenocarcinoma is more frequently associated with H. pylori infection,1,11 although this might be only a reflection of the different methods of detection of the microorganism in such tumors. 12
Gastric adenocarcinomas have been associated with Helicobacter spp. infection in ferrets 4 and a Syrian hamster. 13 In addition, a case of gastrointestinal adenocarcinoma was reported in a cougar infected by Helicobacter-like organisms and other spirochetes. 22 Experimental infection by H. pylori has shown to induce gastric adenocarcinoma in Mongolian gerbils 21 and C57BL/6 mice. 5 Helicobacter sp. infection was evaluated in the chinchilla in the current study by routine histopathology and IHC. Because the degree of infection might vary according to the number of areas examined in the stomach, 8 a sample of gastric tissue also was submitted for nested PCR analysis. The sequencing of more than 98% of the 1,200-bp amplicon showed more than 99% similarity to 16S rRNA gene of H. pylori (BankIt1474861 1199-10-LPB JN595861)
The role of Helicobacter spp. in the development of gastric cancer is still an unresolved issue, 12 but studies have shown overwhelming evidence that favors a link between infection and development of cancer. 9 Helicobacter sp. infection has been detected in 40% of the stomachs of the approximately 252 necropsied chinchillas routinely necropsied at the authors’ laboratory in 2010 and 2011, the diagnosis being based on both the urease rapid test and IHC. Several infected chinchillas displayed varying degrees of gastritis, lymphoid hyperplasia, intestinal metaplasia, and/or dysplasia (unpublished data). The absence of the bacterium in the immunohistochemical analysis in the present case possibly indicates that despite its potential role in the carcinogenesis, the bacterium was no longer colonizing the mucosa at the time of diagnosis, similar to what is described in human cases of H. pylori–induced gastric carcinomas. 17
In conclusion, the present study describes an infiltrative gastric adenocarcinoma in a chinchilla, which shared some characteristics with intestinal-type gastric adenocarcinomas found in human beings. Although neoplasms have been rarely described in chinchillas, the frequency of neoplasia in this species will probably increase in the future because of the increasing popularity of this animal as a pet, its long life span, and more routine use of medical veterinary services for geriatric rodents. 20
Footnotes
a.
Dako North America Inc., Carpinteria, CA.
b.
LSAB+ System-HRP, Dako North America Inc., Carpinteria, CA.
c.
Sigma-Aldrich, St. Louis, MO.
d.
Dako Denmark A/S, Glostrup, Denmark.
e.
QIAamp DNA Mini kit, Qiagen GmBH, Hilden, Germany.
f.
Wizard SV Gel and PCR Clean-Up System, Promega Corp., Madison, WI.
g.
Big Dye Terminator v3.1 Cycle Sequencing Kit, ABI PRISM Big Dye Terminator Cycle Sequencing Ready Reaction Kit, ABI Prism 3130; Applied Biosystems, Foster City, CA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the Brazilian agency National Council of Scientific and Technologic Development (CNPq), Grant 473493/2010-1 and through a fellowship from the Brazilian Coordination for the Improvement of Post Secondary Education (CAPES) within the National Post-Doctoral Program (PNPD).
