Abstract
In the present study, a case of a spontaneously metastasizing seminoma in 9-year-old pet lionhead rabbit is described. The rabbit was presented with unilateral testicular enlargement and a palpable abdominal mass. Spiral computed tomography revealed the presence of an abdominal-pelvic mass in the region of the sublumbar lymph nodes. Testes and lymph nodes were collected, fixed in formalin, and submitted for histopathological examination. Microscopically, the normal architecture of the enlarged testis and lymph node was completely replaced by a diffuse malignant seminoma.
Keywords
A 9-year-old, 1.7 kg, pet lionhead rabbit presented with a 2-day history of weakness, anorexia, and diarrhea. On clinical examination, the left testicle was homogeneously and markedly enlarged. Moderate abdominal distension was observed, and an abdominal mass, painful at palpation, was detected. Based on clinical examination, a presumptive diagnosis of primary testicular neoplasia with abdominal lymph nodal metastasis was made. Blood samples were collected, and complete blood count and chemistry panel (urea, creatinine, glucose, total protein, albumin, globulin, alanine aminotransferase, alkaline phosphatase, sodium, potassium, and chloride) were performed. Hematologic and biochemical analyses were within normal limits for the species, with the only exception of the hematocrit value (HCT = 51%). Based on clinical signs and laboratory results, the rabbit was considered slightly dehydrated. The initial treatment included fluids (intravenous of ringer lactate a 10 mg/kg/hr for 24 hr), nutritional support with critical care oxbow b (oral administration of 50 ml every 6 hr), antiemetic therapy (metoclopramide c 0.5 mg/kg subcutaneous administration every 12 hr), and analgesic therapy with tramadol d (2 mg/kg subcutaneous injections every 12 hr) and meloxicam e (0.1 mg/kg oral administration every 24 hr). Because the abdominal mass was painful and caused marked discomfort, the owners elicited for surgery.
A detailed total body evaluation with spiral computed tomography (CT) was performed just before surgery. The pet rabbit was premedicated with glycopyrrolate f (0.05 mg/kg) and butorphanol g (0.3 mg/kg) subcutaneously, followed 20 min later by intravenous administration of propofol h (5 mg/kg) and midazolam i (0.25 mg/kg). The rabbit was then intubated, and anesthesia was maintained with 1% or 2% of isoflurane. j Spiral computed tomography k revealed the presence of a large, homogeneous, well-defined abdominal lobulated mass in close proximity to both iliac arteries and veins and ureters. After intravenous administration of nonionic iodinated contrast medium at a dose of 600 mg iodine/kg l the mass showed low and heterogeneous uptake (Fig. 1). A diagnosis of sublumbar lymph nodes enlargement was made.
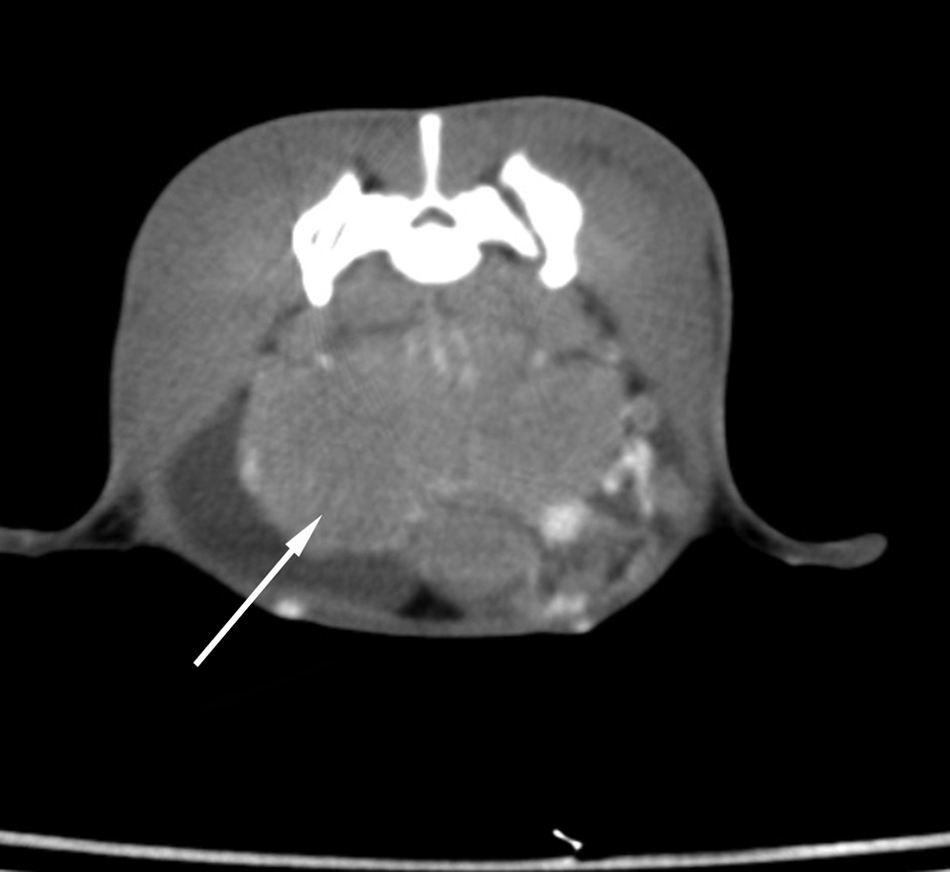
Postcontrast spiral computed tomography scan of a rabbit with seminoma metastatic to the sublumbar lymph nodes obtained at the level of lumbosacral region: the mass (arrow) showed a poor contrast enhancement (WW300, WL50).
Spiral CT scans obtained at the level of the scrotum revealed an enlargement of the left testis that appeared heterogeneous. The postcontrast acquisitions on the testis showed an irregular uptake due to the presence of necrotic and hemorrhagic areas (Fig. 2). This pattern was consistent with germinal tumor of the left testis. No metastatic lesions in the thorax and in the other districts were detected. At surgery, a large, hemorrhagic, firm mass was strictly adhered to the iliac vessels. A complete resection of the mass was extremely hazardous for the vessel wall integrity, and the owners therefore elected for intraoperative euthanasia. A complete necropsy was declined, but both testes and the abdominal mass were collected for histopathology. Grossly, the right testis appeared normal while in the left testis, a lobulated, irregularly bulging, white to gray neoplastic nodule compressing and effacing the adjacent parenchyma was present. The abdominal mass, similarly to the testicular nodule, was multilobulated, white, firm, and with a rough surface.
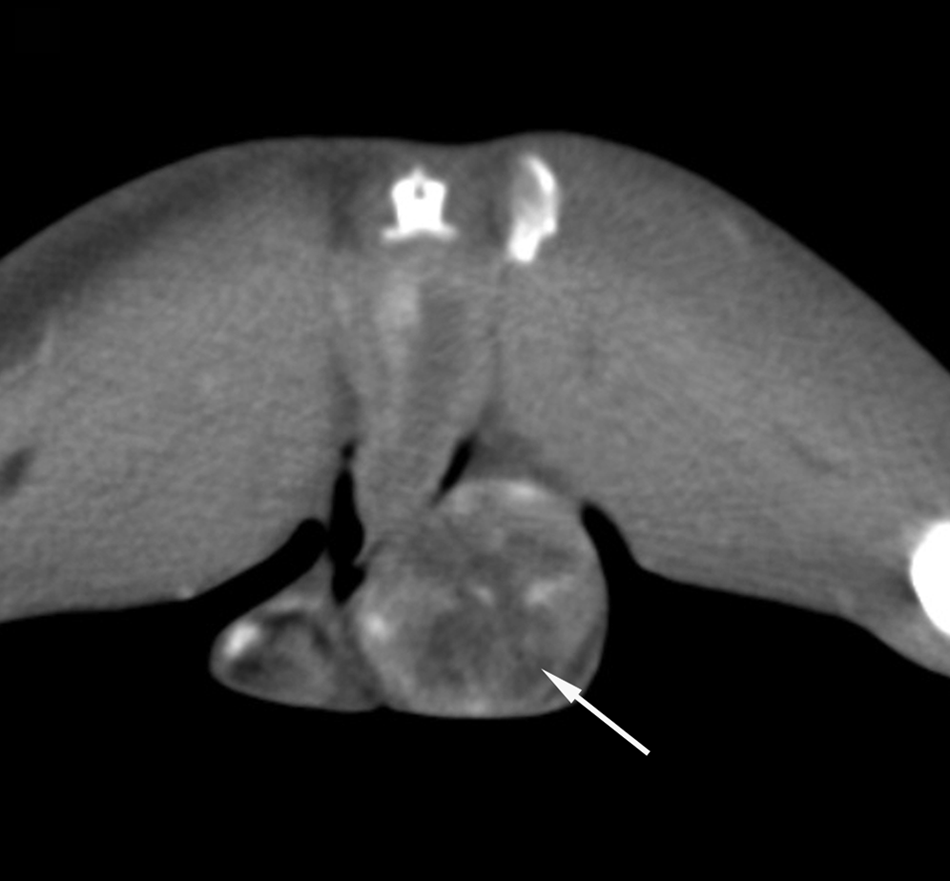
Postcontrast spiral computed tomography scan of a rabbit with testicular seminoma obtained at the level of the testis: a severe and heterogeneous contrast enhancement is recognizable (arrow). No abnormalities of tomographic pattern are present in the right testis (WW300, WL50).
Tissue samples were processed by conventional methods and embedded in paraffin. For each sample, 5 µm-thick sections were obtained and stained with hematoxylin and eosin.
Histologically, the right testis was moderately atrophic. In the left testis, more than 95% of testicular parenchyma was substituted by an expansive, unencapsulated, poorly demarcated, densely cellular neoplasm, multifocally invading the capsule and the epididymis, composed of sheets of round, large cells separated by scant fibrovascular stroma (Fig. 3). Neoplastic cells were round, 20–30 µm in diameter, with distinct cell borders, high nuclear-cytoplasmic ratio, scant amount of eosinophilic cytoplasm, and large, round, centrally located nuclei with vesicular chromatin pattern and 1 or 2 prominent, centrally located, magenta nucleoli. Anisokaryosis and anisocytosis were prominent, and mitotic figures ranged from 3 to 13 per high power field (400×). Scattered lymphocytic aggregates and multifocal foci of necrosis were also present. In addition, neoplastic emboli were evident in the lumina of blood and lymphatic vessels within the neoplasia. The sublumbar abdominal lymph node architecture was almost completely replaced by an expansive neoplasm composed of the aforementioned neoplastic cells separated by abundant fibrovascular stroma (Fig. 4). Based on anatomic location, extension of the masses, and gross and histological features of the neoplastic lesions, the diagnosis of unilateral diffuse malignant testicular seminoma with sublumbar lymph node metastasis was made.
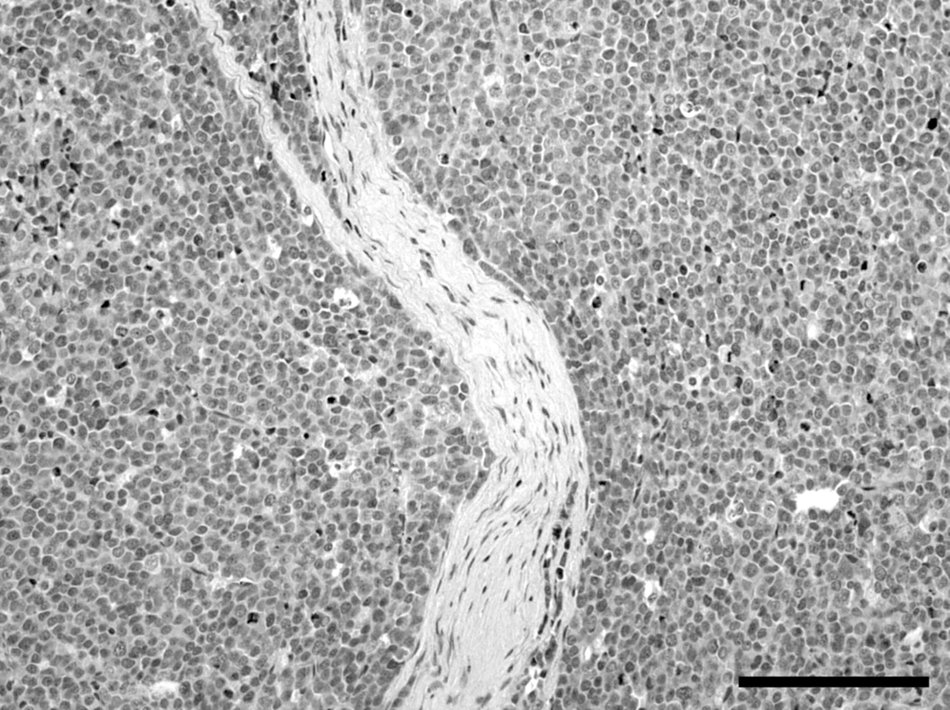
Testis with diffuse seminoma from a rabbit. Seminoma is composed of sheets of large round cells separated by scant amount of fibrovascular stroma. Hematoxylin and eosin stain. Bar = 375 µm.
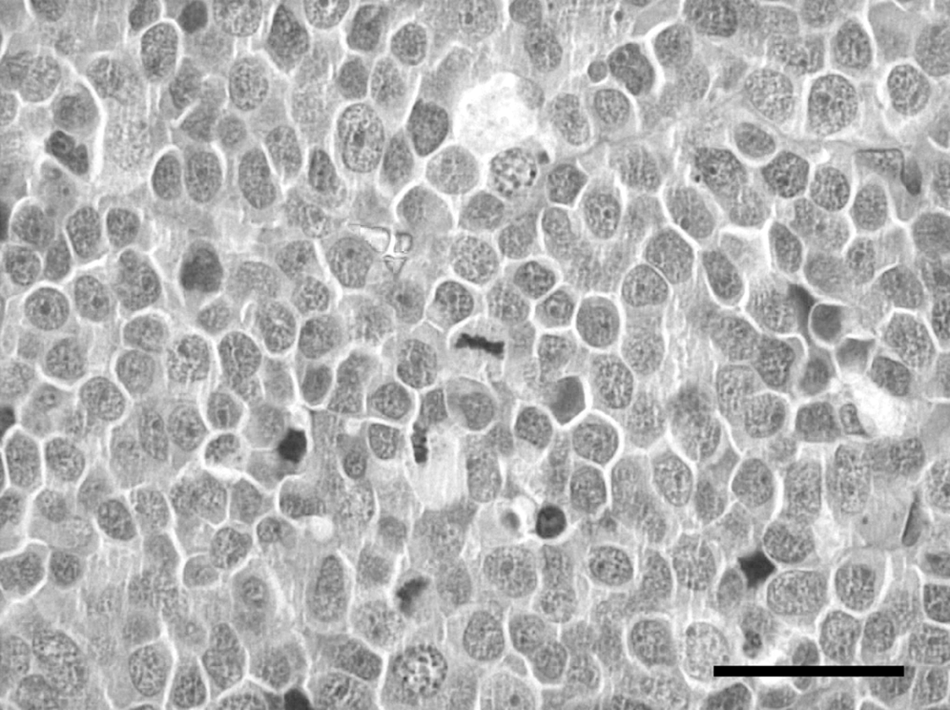
Sublumbar lymph node with metastatic seminoma from a rabbit. Prominent anisokaryosis, anisocytosis, and several mitoses are present. Hematoxylin and eosin stain. Bar = 100 µm.
Seminomas are typical of older animals and are subdivided into intratubular and diffuse form, based upon their histological appearance. Histologically, judging the malignant potential of these tumors is difficult or impossible, and the only reliable indicator of malignancy is the presence of metastases. 4
In domestic animals, malignant metastasizing seminomas are considered rare lesions and have been reported in dogs,9,15–17 stallions, and mules.4,13 In the majority of these reports, the sublumbar lymph nodes were consistently affected. Sublumbar lymph nodes receive afferent lymphatic vessels from the testes and represent in animals, as in human beings, the most common site of metastases for testicular tumors.4,6
In the rabbit, spontaneous testicular tumors are not commonly observed. The most frequently reported testicular neoplasia in this species is the interstitial cell tumor.3,8,19 Simultaneous interstitial cell tumor and seminoma12,18 or unilateral/bilateral seminoma1,2 have been more rarely reported, and granular cell tumor 7 and teratoma 10 are considered very rare lesions.
A single case of experimental metastasizing seminoma has been previously described. 11 The primary lesion, confined to the testis, occurred spontaneously in an adult laboratory rabbit. The neoplasia was then experimentally induced in a recipient rabbit by inoculation of the neoplastic cells into the seminiferous tubules of the recipient animal, in which it became generalized.
In the current case, there was a single, large, peritoneal metastatic lesion, extensively effacing sublumbar lymph node architecture. Surgery is usually the therapy of choice for rabbit neoplasia. Chemotherapy is a therapeutic option for treatment of seminoma in different species (i.e., canine and human). Rabbits have been treated with chemotherapeutics mostly for experimental purposes,5,14 but no data are currently available in the veterinary literature on chemotherapy treatment for spontaneous tumors. Based on studies on the toxicity of chemotherapeutic agents in experimental rabbits, 5 most veterinarians do not recommend chemotherapy as a therapeutic choice for neoplasia in pet rabbits.
In the rabbit in the current report, morphologic features appeared similar to the human, equine, and canine metastasizing seminoma, and, as reported in these species, the histopathology of the intra-abdominal metastases was similar to the primary testicular tumor. Numerous neoplastic emboli within vessels were observed. In 2 previous studies, presence of neoplastic cells within vessels of the pampiniform plexus 1 and of the testicular capsule 12 were observed, but in both cases, despite the evidence of vascular invasion, no metastatic lesion was reported 1 year and 7 months, respectively, after the orchiectomy.
Testicular neoplasia is uncommon in lagomorphs, and testicular seminoma has been rarely reported. Neutering rabbits has becoming a common and safe procedure that is recommended by most veterinarians in order to reduce difficult behavior and decrease the risk of diseases of the reproductive system. Nevertheless, as in present case, there is still strong resistance from owners, especially in some countries, to the neutering practice of young rabbits.
The spontaneous tumor incidence reported in the literature indicates that age is a determinant factor in rabbit testicular neoplasia. It is conceivable that age can also influence the frequency of metastatic disease, as in other species. As rabbits are becoming common pet animals, and their life expectation is consequently longer, in the future veterinarians can most likely be expected to deal more frequently with testicular tumor and related metastatic disease in their routine practices.
Footnotes
Acknowledgements
The authors thank Mr. Marco Colombo for photographic support.
a.
Ringer lactate, Altaselect S.r.l., San Giovanni Lupatoto (Vr), Italy.
b.
Oxbow Animal Health, Murdock, NE.
c.
Vetpramide, Bayer S.p.A., Milano, Italy.
d.
Altadol, Formevet S.r.L, Milano, Italy.
e.
Metacam, Boehringer Ingelheim Italia S.p.A., Milano, Italy.
f.
Robinul, MIKART Inc., Atlanta, GA.
g.
Dolorex, Intervet Italia S.r.l., Aprilia (LT), Italy.
h.
Rapinovet, Intervet Italia S.r.l., Aprilia (LT), Italy.
i.
Midazolam IBI, Ibi Istituto Biochimico Italiano Giovanni Lorenzini S.p.A., Aprilia (LT), Italy.
j.
Isoflo, Abbott Italia S.r.l., Campoverde di Aprilia (LT), Italy.
k.
PQ2000S, Philips, Monza, Italy.
l.
Visipaque 320®, GE Healthcare, Milano, Italy.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship and/or publication of this article.
