Abstract
Diagnosis and control of Foot-and-mouth disease virus (FMDV) requires rapid and sensitive diagnostic tests. Two antibody enzyme-linked immunosorbent assay (ELISA) kits, Ceditest® FMDV-NS for the detection of antibodies against the nonstructural proteins of all FMDV serotypes and Ceditest® FMDV type O for the detection of antibodies against serotype O, were evaluated under African endemic conditions where the presence of multiple serotypes and the use of nonpurified vaccines complicate serological diagnosis. Serum samples from 218 African buffalo, 758 cattle, 304 goats, and 88 sheep were tested using both kits, and selected samples were tested not only in serotype-specific ELISAs for antibodies against primarily FMDV serotype O, but also against other serotypes. The FMDV-NS assay detected far more positive samples (93%) than the FMDV type O assay (30%) in buffalo (P < 0.05), with predominant antibodies against the South African Territories (SAT) serotypes, while the seroprevalence was generally comparable in cattle with antibodies against serotype O elicited by infection and/or vaccination. However, some districts had higher seroprevalence using the FMDV type O assay indicating vaccination without infection, while 1 cattle herd with antibodies against the SAT serotypes had far more positive samples (85%) using the FMDV-NS versus the FMDV type O (10%), consistent with the latter test’s lower sensitivity for antibodies against SAT serotypes. Based on the current investigation, the FMDV type O ELISA may be limited by the presence of SAT serotypes. The FMD NS assay worked well as a screening test for antibodies against all FMDV serotypes present in Uganda; however, as long as nonpurified vaccines are applied in the region, this test cannot be used to differentiate between vaccinated and infected animals.
Introduction
Foot-and-mouth disease (FMD) is a highly contagious viral disease of cloven-hoofed animals including domestic ruminants, pigs, and more than 70 wild animal species.1,21 Foot-and-mouth disease virus (FMDV; order Picornavirales, family Picornaviridae, genus Aphthovirus) contains 7 serotypes, namely O, A, C, Asia1, Southern African Territories (SAT)1, SAT2, and SAT3, all with a number of subtypes. Infection with 1 serotype does not prevent infection with other serotypes. 8 Different serotypes have distinct geographical distributions and epidemiology; however, they are clinically indistinguishable. 22
The epidemiology of FMD in East Africa is complex with 5 (O, A, SAT1, SAT2, and SAT3) out of the 7 serotypes being continuously present. 22 Serotype C was last diagnosed in Kenya in 2004 18 while serotype Asia1 has never been diagnosed on the African continent. 22
Previously, serological diagnosis of FMDV implied running up to 7 different tests for antibodies against each of the relevant serotypes; however, more broadly directed antibody tests detecting antibodies against all 7 serotypes of FMDV have recently been offered by a number of commercial companies, including an enzyme-linked immunosorbent assay (ELISA) kit for detection of antibodies against all 7 serotypes of FMDV marketed by Prionics AG, Switzerland: the Ceditest® FMDV-NS a (hereafter, NSP test). The same company has marketed a screening test for detection of antibodies against serotype O, the Ceditest® FMDV type O ELISA kit b (hereafter, SP-O test), which has such broad specificities for antibodies against the Eurasian serotypes of FMDV (O, A, C, and Asia1) 9 that it has been speculated that this test could work as a pan-FMDV antibody test.
The NSP test is a blocking ELISA that detects antibodies against the nonstructural 3ABC protein of FMDV in infected animals, independent of the serotype that induced the antibodies. 19 Like other commercially available test kits for antibodies against the nonstructural proteins (NSPs) of FMDV, the NSP assay has important applications in differentiating infected from vaccinated animals. 19 Conversely, the SP-O assay is based on the structural proteins (SPs) of FMDV serotype O and can be used in cattle, sheep, pigs, and goats. 9 These 2 test kits have been successfully used for investigating FMD outbreaks and for control programs.9,16,17
The NSP ELISAs are widely applied in the diagnosis of FMD and have been evaluated in Europe 5 and to a lesser extent in Africa.6,7 In a direct comparison of 6 NSP ELISAs, the NSP test had comparative sensitivities of 98.9% and 96.6% in field sera from Israel and Zimbabwe, respectively, while diagnostic specificity was 97.2% in nonvaccinated animals and 99.5% in experimental animals that had received 1 vaccination. 5 In Africa, using Hui–Walter latent class modeling and comparing with a combination of neutralization test results for all 3 SAT serotypes of FMDV, it has been demonstrated that the NSP test is a useful tool for screening buffalo infected with multiple SAT serotypes of FMDV with estimated sensitivity of 87.7% and specificity of 87.3%. 6 Using the same modeling methodology but without comparing to neutralization test results, estimated sensitivity and specificity of 96.6% and 90.9%, respectively, have been found in Zebu cattle kept under extensive rangeland conditions in Cameroon. 7 Although only limited studies of performance of these tests have been carried out among sheep and goats, lower sensitivity has been reported particularly in sheep. 13
The SP-O test has mainly been used and evaluated in Europe and has been found to identify antibodies against serotypes O, A, C, and Asia1 in cattle, with higher sensitivity for antibodies against serotype O. 9 The aim of the present study was to evaluate the performance of the SP-O and NSP tests in Ugandan cattle, sheep, goats, and African buffalo (Syncerus caffer). In addition, the SP-O test was compared to a solid phase blocking ELISA (SPBE) for detection of antibodies against FMDV serotype O. 4
Materials and methods
Serum samples were collected from Ugandan African buffalo, cattle, goats, and sheep. Two hundred eighteen samples from African buffalo were collected from Lake Mburo, Kidepo Valley, Murchison Falls, and Queen Elizabeth National Parks as described elsewhere.2,3 In addition, 449 cattle sera were collected in July 2006 during an extensive postoutbreak (PO) sampling in a number of districts in the western and southwestern regions of Uganda and 309 sera were collected during an attempted random sampling (RS) within Kasese and Bushenyi Districts in 2007.14,15 Three hundred four goat sera and 88 sheep sera were collected from the same farms or villages during these sampling trips. 4
Testing for antibodies against FMDV was done using the NSP a and SP-O b tests according to the manufacturer’s instructions. Optical density values (OD) were determined using a dual wavelength ELISA reader c at 620 nm and 450 nm. Results were expressed as OD% (ODP: ODsample in percent of ODnegative controls), 4 and cutoffs were set at 50%.
Selected samples, mostly positive for antibodies against NSPs, were screened at dilution 1:5 using an in-house SPBE for antibodies against FMDV serotype O (SPBE-O) with a cutoff of 50% as previously described. 4 The selected samples included 174 buffalo sera, 126 cattle sera from the 2006 PO sampling, and 81 cattle sera from the 2007 RS sampling. The selected cattle sera constituted 28–80% of sera from each of the 18 herds with a history of recent clinical signs of FMD in 2006 and 17–100% of sera from 16 randomly sampled farms without clinical signs of FMD in 2007. For small ruminants, all samples positive in the NSP test and/or the SP-O test from both 2006 and 2007 were further tested using the SPBE-O. In addition, 20 sera from 1 herd (Ks12) 15 were examined in SPBEs for antibodies against all 7 serotypes of FMDV as previously described. 4 Differences between groups were tested for statistical significance using chi-square, and differences between seroprevalence obtained with the NSP and SP-O tests were analyzed using McNemar test in a spreadsheet program. d
Results
Antibodies against NSPs, and SPs of FMDV serotype O, were detected in African buffalo, cattle, goat, and sheep samples (Table 1). Seroprevalence of antibodies against NSP was much higher in African buffalo (93%) compared to cattle (48%), sheep (28%), and goats (9%), while cattle had the highest number of positives in the SP-O test (52%) compared to buffalo (30%) sheep (24%), and goats (7%).
Comparative evaluation of Ceditest® FMDV-NS (NSP) and Ceditest® FMDV type O (SP-O) enzyme-linked immunosorbent assay for the detection of antibodies against nonstructural and structural proteins of Foot-and-mouth disease virus in livestock and wildlife species in various districts and national parks in Uganda.
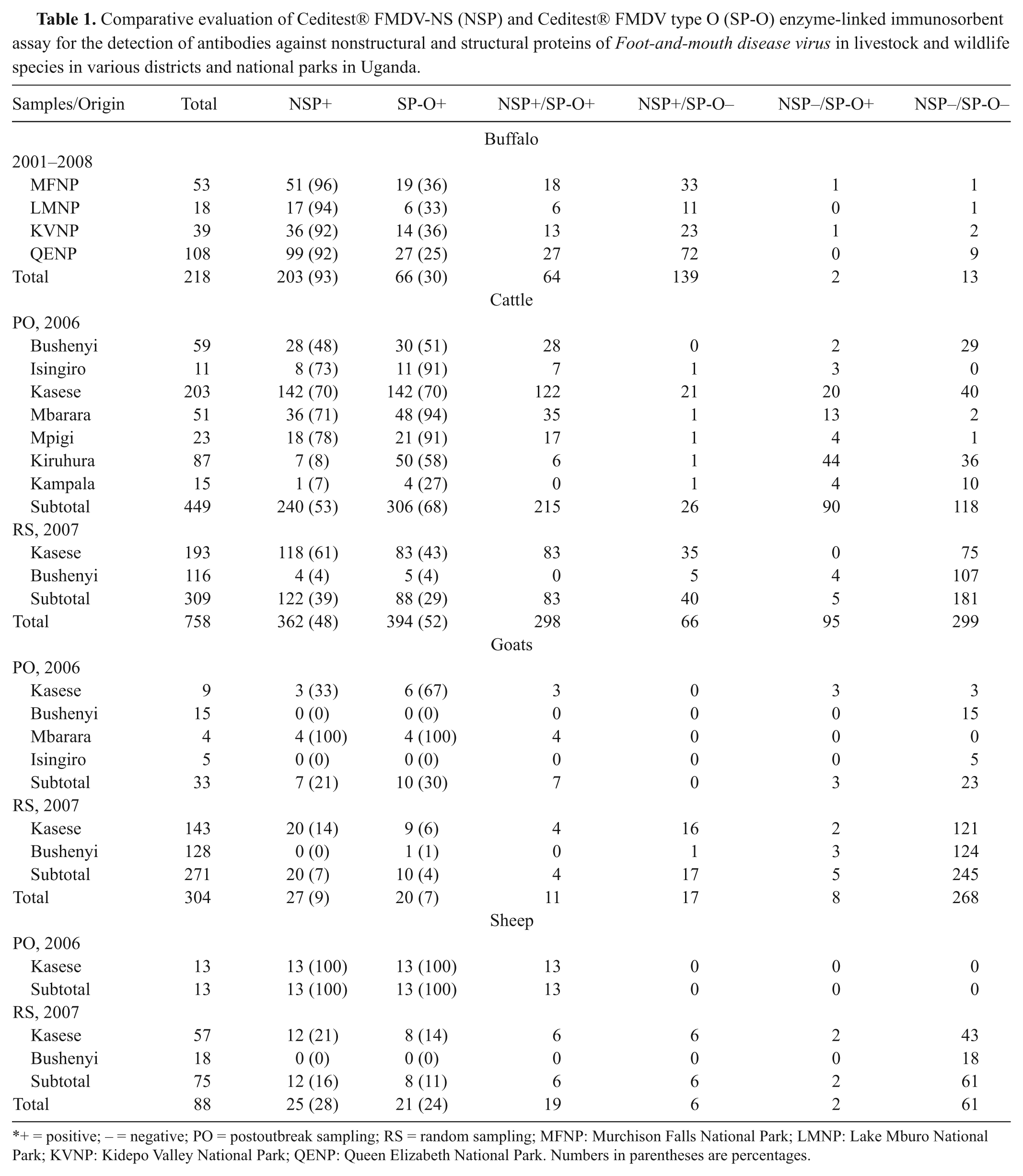
+ = positive; − = negative; PO = postoutbreak sampling; RS = random sampling; MFNP: Murchison Falls National Park; LMNP: Lake Mburo National Park; KVNP: Kidepo Valley National Park; QENP: Queen Elizabeth National Park. Numbers in parentheses are percentages.
In buffalo, for both tests, there was no significant difference in the seroprevalence of FMDV antibodies between national parks (P = 0.758 and P = 0.413 for the NSP and SP-O tests, respectively). Thus, the data from all buffalo samples were pooled, and statistical analysis of these results confirmed that the NSP test detected significantly more seropositive buffalo than the SP-O test (P < 0.005) in all the 4 investigated national parks.
The results were less clear in cattle because the NSP test detected significantly more positive samples than did the SP-O test in samples from the 2007 RS, while it detected significantly fewer samples than did the SP-O test in the 2006 PO sampling (P < 0.001 for both comparisons). The difference in 2007 was caused by more positive samples in the NSP test than in the SP-O test in the Kasese District (P < 0.001), while in 2006, it was caused by more positive samples in the SP-O test than in the NSP test in Mbarara (P = 0.013) and Kiruhura (P < 0.001) districts. Moreover, the 2 tests had different outcomes for approximately 20% of the samples from Kasese District in 2006, but this difference was not statistically significant since the samples that were only positive in one of the tests were distributed equally between the 2 tests (and thus not significantly different, P = 0.859). The remaining district samples (Bushenyi, Isingiro, Mpigi, and Kampala in 2006, and Bushenyi in 2007) had comparable results with only a few samples differing between the 2 tests.
With regard to sera from goats and sheep, the differences between the results obtained with the 2 tests were either inconclusive or insignificant apart from those of goats in 2007 where the NSP test found more positive animals than did the SP-O test (P = 0.0105).
When comparing the results obtained in dilution 1:5 in the SPBE-O test to results in the SP-O test, there was generally good agreement between the results obtained with the 2 tests in cattle, sheep, and goats in the 2006 PO sampling, while the results were more scattered in the 2007 RS and in buffalo, where the SPBE-O screening ELISA identified more positive cattle and buffalo samples than did the SP-O test (Table 2).
Comparative screening results for Foot-and-mouth disease virus using the Ceditest® FMDV type O (SP-O) enzyme-linked immunosorbent assay (ELISA) and the solid phase blocking ELISA (SPBE-O) in buffalo, cattle, and small ruminants in Uganda.

NSP = Ceditest® FMDV-NS (Cedi Diagnostics B.V., Leystad, The Netherlands [currently produced and marketed as Priocheck® FMDV-NS by Prionics AG, Schlieren-Zurich, Switzerland]. + = positive; − = negative. The samples investigated by SPBE-O were all SP-O positive.
In the 2006 PO sampling, on-farm seroprevalence in the NSP and SP-O tests were generally comparable 14 ; however, 1 out of 22 cattle herds (Ks12) with a history of recent clinical signs of FMD had much higher prevalence of antibodies against FMDV in the NSP test than in the SP-O test. 14 Analysis of serotype-specificity of FMDV antibodies of 20 heads of cattle from this herd using SPBEs for all 7 serotypes revealed that these cattle exclusively had antibodies against the SAT serotypes (Table 3). Most of these 20 samples had low antibody titers against serotypes SAT2 and SAT3; however, 1 animal had a titer of 80 for antibodies against SAT2, 3 animals had titers of 80–160 against SAT3, and 1 animal had a titer of 640 against SAT1. This herd was regularly grazed in Queen Elizabeth National Park.
Serological analysis for Foot-and-mouth disease virus (FMDV) of a cattle herd (Ks12) in the Kasese District of Uganda with high seroprevalence in the Ceditest® FMDV-NS enzyme-linked immunosorbent assay (ELISA) and low seroprevalence in the Ceditest® FMDV type O ELISA.*
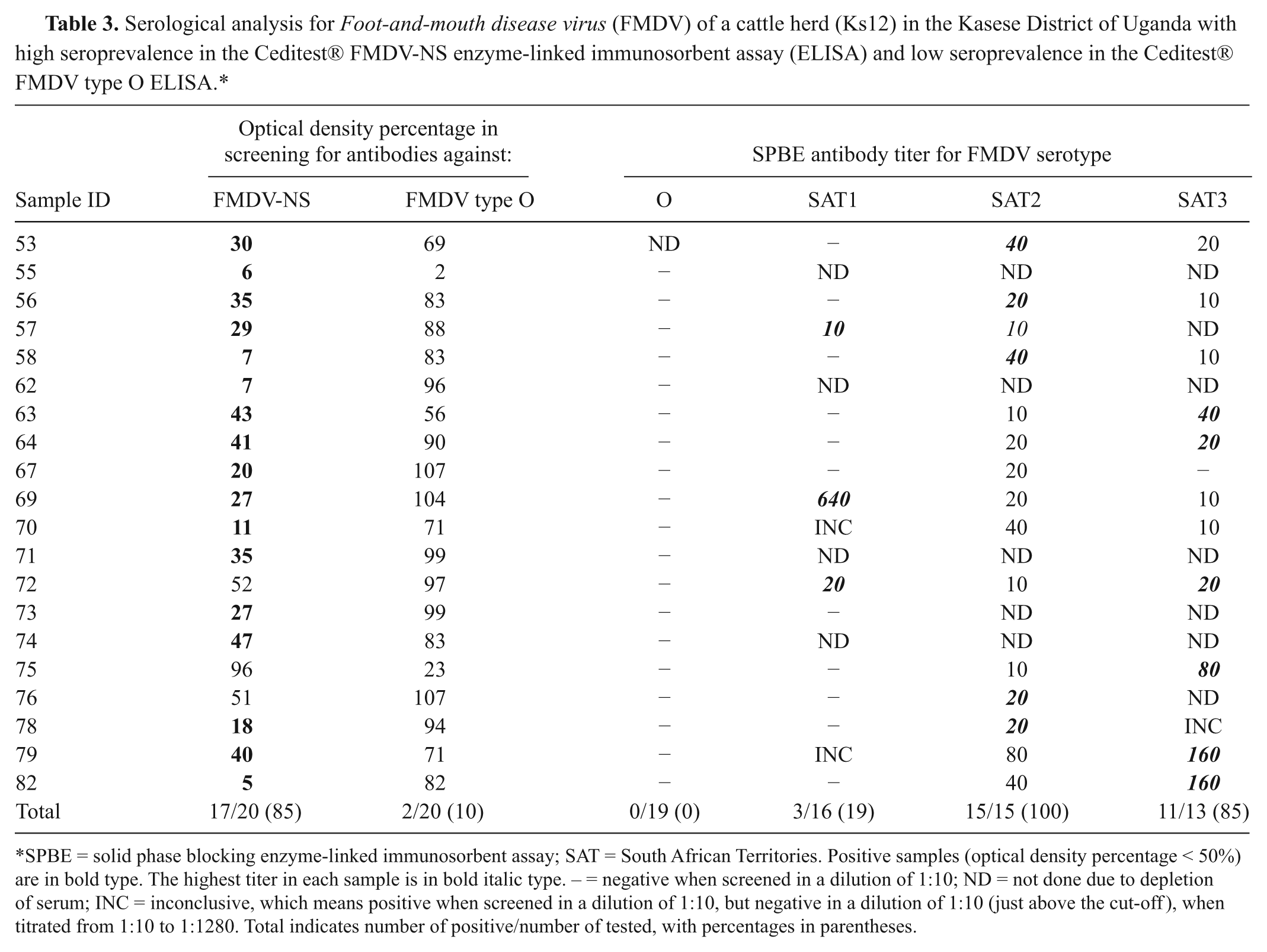
SPBE = solid phase blocking enzyme-linked immunosorbent assay; SAT = South African Territories. Positive samples (optical density percentage < 50%) are in bold type. The highest titer in each sample is in bold italic type. − = negative when screened in a dilution of 1:10; ND = not done due to depletion of serum; INC = inconclusive, which means positive when screened in a dilution of 1:10, but negative in a dilution of 1:10 (just above the cut-off ), when titrated from 1:10 to 1:1280. Total indicates number of positive/number of tested, with percentages in parentheses.
Discussion
Interpretation of the higher seroprevalence of antibodies against NSPs than against SPs of FMDV serotype O in the investigated buffalo sera should take into consideration that when titrated, these sera had higher seroprevalences of antibodies against the FMDV SAT serotypes than against serotype O.2,3 Thus, these data suggest that the SP-O test may be much less sensitive for antibodies against the SAT serotypes of FMDV circulating in Uganda than the NSP test. It may be speculated that the low sensitivity of the SP-O test in buffalo could be species-related; however, this test is a blocking ELISA, which is generally not species-specific. Moreover, the similar finding in a cattle herd (Ks12) of much higher seroprevalence in the NSP test and in the SPBEs for antibodies against the SAT serotypes than in both the SP-O and SPBE-O tests supports that the low sensitivity in buffalo was not species-related.
In cattle, the high seroprevalence in both tests in most districts in 2006 were found during a major outbreak of FMDV serotype O in the region in 2006, which was preceded and followed by vaccination campaigns with nonpurified trivalent vaccines containing FMDV serotypes O, SAT1, and SAT2. Moreover, the majority of the sampled herds exhibited clinical evidence of exposure to FMDV and/or reported previous vaccination, and a serotype O virus was isolated from 2 probang samples collected from this outbreak. 15 In agreement with this, all herds with clinical evidence of FMDV infection, except the 1 Kasese herd described in Table 3 (Ks12), had high seroprevalence in the SP-O test and had high titers of antibodies against serotype O in the SPBE-O ELISA. 15 The higher seroprevalence registered with the SP-O test in Mbarara, Kiruhura, and Kampala districts is consistent with vaccinated animals that have not recently been infected.
The Kasese herd (Ks12; Table 3) with much higher seroprevalence in the NSP test (85%) than in the SP-O test (10%) had serological evidence of exposure to SAT serotypes, while all tested samples were negative in the SPBE-O ELISA. Thus, this result supports the preceding conclusion from the buffalo data that the SP-O test is a less suitable tool for screening antibodies against FMDV than the NSP test when antibodies against the Ugandan SAT serotypes of FMDV are prevailing.
The 2007 RS differed from the 2006 PO sampling in that the 2 districts under study had very different levels of seroprevalence in both the NSP and SP-O tests. Moreover, Bushenyi District had very low seroprevalence and had, except for a small, quickly contained outbreak in 2006 (sampled in the 2006 PO sampling), been free from FMD for a prolonged time, 14 while Kasese District had high seroprevalence reflecting a situation with frequent reports of FMD outbreaks and vaccinations.
Thirty-five of the Kasese 2007 samples (18%) were positive only in the NSP test (Table 1), and half of these samples came from a herd with higher titers of antibodies against the SAT serotypes than against serotype O (K1), 14 confirming the results from Ks12 sampled in 2006, while the remaining 18 samples were dispersed among 10 of the 16 sampled herds. 14
The NSP test, like other commercially available test kits for antibodies against the NSPs of FMDV, have important applications in differentiating infected from vaccinated animals, 19 but this usage may be complicated by nonpurified FMDV vaccines inducing antibodies against the NSPs.10,14,20 Such vaccines including FMDV serotypes O, SAT1, and SAT2 were applied in the investigated regions in 2005, 2006, and 2007, and it is thus not possible to definitively conclude if the antibodies detected in the NSP test were elicited by vaccination and/or infection.
The differences between the performances of the NSP test and the SP-O test in small ruminants followed the results in cattle, but were not statistically significant due to low numbers of positives. These variations were most likely results of the presence of antibodies against FMDV serotype O after the outbreak of FMDV serotype O during 2006. 4 The much higher seroprevalence in sheep and goats in the 2006 PO sampling than in the 2007 RS, combined with the higher seroprevalence in cattle in 2007, may indicate faster waning of antibodies in these species than in cattle as previously reported,11,12 but could also be a consequence of the common practice of not vaccinating small ruminants.
The results of the SPBE-O test matched those of the SP-O test in cattle sampled in 2006 (PO sampling), while the first test was more sensitive in cattle sampled in 2007 RS and in buffalo that tested positive for antibodies against SAT serotypes. In small ruminants, these 2 tests were also more in agreement in the 2006 PO sampling than in the 2007 RS. The results from buffalo clearly demonstrate that the SPBE-O when used in dilution of 1:5 is less specific with regard to identifying serotype than the SP-O test. The data, taken together with reported predominance of antibodies against SAT serotypes,2,3 indicate that the SP-O test may not be very efficient for identification of antibodies against the SAT serotypes of FMDV, and thus cannot be recommended as a pan-FMDV antibody test.
Like other commercially available test kits for antibodies against the NSPs of FMDV, the NSP test has important applications in differentiating infected from vaccinated animals, 21 but this usage may be complicated by detection of antibodies against FMDV NSPs elicited by nonpurified FMDV vaccines,10,21 which are applied in some sub-Saharan African countries including Uganda. This was also the case in the samplings in the current study, where nonpurified trivalent vaccines had been used in the area in vaccination campaigns in cattle in 2005, 2006, and 2007. Thus, in the presented analysis of cattle samples from 2006 and 2007, it is impossible to conclude whether the identified antibodies were induced by vaccination and/or infection. However, the NSP test can still be applied for mass screening to identify animals with antibodies against FMDV whether these are vaccine-derived or elicited by infection. 20
In conclusion, though the SP-O test is of low specificity toward antibodies against the Eurasian FMDV serotypes 9 and it has been speculated that this test could be used as a pan-FMDV antibody test, this usage cannot be recommended in populations where the SAT serotypes of FMDV are prevalent, as exemplified by the SAT-infected buffalo and the cattle herd exposed to 1 or more of the SAT serotypes, only. The SP-O test can be used in combination with the NSP test when investigating FMD outbreaks caused by serotype O; however, it will not be possible to use the NSP test to differentiate between vaccinated and infected animals as long as nonpurified vaccines are applied in the region.
Footnotes
Acknowledgements
Chrisostom Ayebazibwe, Frank Norbert Mwiine, and Sheila Nina Balinda equally contributed to this research. The authors thank the Ministry of Agriculture Animal Industry and Fisheries, Uganda, for allowing us to undertake this study.
a.
Ceditest® FMDV-NS, Cedi Diagnostics B.V., Leystad, The Netherlands (currently produced and marketed as Priocheck® FMDV-NS by Prionics AG, Schlieren-Zurich, Switzerland).
b.
Ceditest® FMDV-type O, Cedi Diagnostics B.V., Leystad, The Netherlands (currently produced and marketed as Priocheck® FMDV-type O by Prionics AG, Schlieren-Zurich, Switzerland).
c.
Thermo Labsystems Multiskan Ascent® Photometric plate reader, Thermo Fischer, Slangerup, Denmark.
d.
Microsoft® Office Excel® 2007, Microsoft Corp., Redmond, WA.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Danish International Development Agency under the Livestock Wildlife Diseases in East Africa Project (grant no. P104.Dan.8.1.316).
