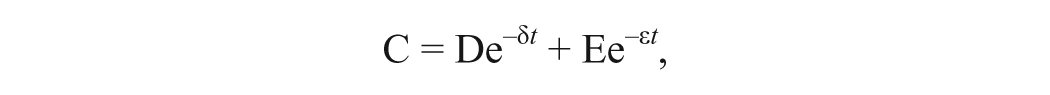Abstract
Sixteen of approximately 500 yearling steers died of acute selenium (Se) toxicosis after grazing on a Se-contaminated range for only a few days. Field studies and chemical analyses identified the predominant toxic plant as western aster (Symphyotrichum ascendens, previously Aster ascendens), which contained over 4,000 ppm Se (dry weight). Several dead animals that were necropsied had acute severe myocardial necrosis characterized by edema and myocyte swelling, with hypereosinophilia, clumping, and coagulation of myocardial proteins. Whole blood from 36 surviving steers was collected and analyzed, and 10 steers with elevated Se concentrations were selected for close monitoring and clinical evaluations. Each steer was weighed, and serum, blood, liver, skeletal muscle, and hair were regularly collected after removal from the Se-contaminated range. One animal that died 18 days after exposure was necropsied and exhibited severe multifocal myocardial fibrosis with extensive hepatic congestion, degeneration, and hemosiderosis. At 180 days postexposure, 2 of the 10 steers were euthanized, and tissue samples were collected. Both steers had rare, small fibrotic foci in their hearts. The Se elimination half-lives from serum, whole blood, liver, and muscle of the recovering steers were 40.5 ± 8.2, 115.6 ± 25.1, 38.2 ± 5.0, and 98.5 ± 19.1 days, respectively. The Se concentration in hair reached a peak of 11.5 ± 5.3 ppm at 22 days postexposure. The findings indicate that cattle are sensitive to acute Se toxicosis caused by ingestion of Se-accumulator plants, with myocardial necrosis as the primary lesion. Some poisoned animals may develop congestive heart failure weeks after the toxic exposure, and in the current study, Se was slowly excreted requiring a relatively long withdrawal time.
Introduction
Selenium (Se) is an essential element necessary for the activity of many enzymes and proteins. Selenium deficiencies in cattle result in white muscle disease.31,33 Excessive Se in forages, feeds, or supplements can result in either chronic or acute Se toxicosis. Chronic Se toxicosis is caused by consuming feeds or forages with high concentrations of Se over a period of weeks to months.12,13 Reports in the literature describe acute Se toxicosis caused by calculation errors in Se supplements,15,26 mistakes in formulation of feed rations, 32 and ingestion of Se-accumulator plants (Wyoming State Board of Sheep Commissioners: 1908, 10th annual report. Cheyenne, Wyoming). 6 Over 15,000 sheep died during the summers of 1907 and 1908 in a region north of Medicine Bow, Wyoming, and the losses were attributed to grazing of Se-accumulator plants, Xylorhiza glabriuscula (woody aster) and Astragalus bisulcatus (Gray’s vetch; Wyoming State Board of Sheep Commissioners: 1908, 10th annual report). In May 2003, several hundred sheep died from acute Se toxicosis after grazing in a Se-rich area in Idaho. 6 Selenium-accumulating plants such as A. bisulcatus, Astragalus praelongus, Aster spp., or Stanleya pinnata can store up to 10,000 ppm Se (dry weight),3,4,30 as selenate, Se-methylselenocysteine (MeSeCys), and γ-glutamyl-MeSeCys (γ-GMeSeCys).8,19
Plants that contain Se at concentrations of several thousand ppm are very toxic, as less than 0.5 kg of the plant may be lethal to cattle. However, there are many factors to consider when determining toxicity of Se, including the chemical form of Se, species of animal, age of animal, and nutritional status of animal. It was suggested that a median lethal dose (LD50) for sodium selenite orally dosed to ruminants was 1.9–8.3 mg Se/kg body weight (bw). 9 An oral dose of 9–20 mg Se/kg bw may be lethal to calves. 20 The LD50 of subcutaneous selenate injection is 1 mg Se/kg bw in lambs and 1.9 mg Se/kg bw in adult cattle. 9 A study published in 2011 showed that different forms of Se have different relative toxicities, as doses of 6 mg Se/kg bw of methylselenocysteine, selenomethionine, selenate, selenite, and western aster (Symphyotrichum ascendens, previously Aster ascendens) resulted in differences in the number of sheep that died in each treatment group as well the time of death after dosing. The order of toxicity was methylselenocysteine > selenomethionine > selenate = western aster > selenite. 5 The chemical forms of Se associated with some previously analyzed Se-accumulator plants (A. bisulcatus and S. pinnata) are selenate and MeSeCys. 8
Very little information is available in the literature about the clearance of Se from animals that suffer acute Se poisoning but appear to recover. The objectives of the current study were to 1) describe the clinical and histological changes of acute Se poisoning in cattle; 2) monitor the clearance of Se from blood, tissues, and hair of steers that had been acutely poisoned on Se-accumulator forages under natural conditions to determine the rate of Se clearance and the possible risk of contaminated animal products; and 3) better define the potential long-term effects of Se toxicity on the animals.
Material and methods
Clinical case and field study
During the first week of August 2009, 16 of approximately 500 mixed-breed yearling steers died within a 48-hr period after grazing forage from a pasture with a history of Se contamination. The 500-acre pasture included an approximately 5-acre reclaimed mine site. The reclaimed portion was waste from an inactive phosphate mine that was composed of seleniferous waste shale that had been mostly covered with soil. The reclaimed site historically supports various grasses and forbs, including some deep-rooted plants that have been documented to accumulate Se. However, during the past 20 years, the pasture had been safely grazed by cattle without a known case of poisoning. The steers were moved into the pasture on July 26, where they remained in a meadow, the largest portion of the pasture, until July 30, at which time the ranch manager indicated that the steers began to leave the meadow and move into the trees and the adjacent reclaimed area. One week after being put into the pasture, 12 steers were found dead. The remaining steers were gathered and moved from the pasture by evening, during which time 1 more steer died. Three additional steers died the following day, bringing the total to 16 steers in less than 48 hr. The ranch owners suspected larkspur (Delphinium spp.) poisoning as the cause of death and collected rumen contents, heart, lung, liver, and kidney from several of the dead animals for histological studies and chemical analyses.
Careful examination of the pasture found larkspur (Delphinium spp.) and lupine (Lupinus leucophyllus) plants, but there was little evidence that the steers had eaten significant amounts, and chemical analyses of these plants and animal tissues provided no evidence that they were responsible for the death of the steers. Various forbs and grasses were collected on the reclamation site for analysis. The most prominent plant that had been heavily grazed was western aster. Multiple samples of this plant were collected for Se analysis by inductively coupled plasma–mass spectrometry (ICP-MS). Chemical analysis of the plant samples provided strong evidence that the steers died from Se poisoning.
The surviving steers were moved to corrals, and blood samples were collected from those known to be in that portion of the pasture. These samples were analyzed for Se, and 10 of the steers with the highest whole-blood Se concentrations were purchased and transported to the Poisonous Plant Research Laboratory in Logan, Utah, where they could be closely monitored, repeatedly sampled, and weighed monthly. The animal handling and sampling were performed under an approved Utah State University Institutional Animal Care and Use Committee protocol.
The steers arrived at the laboratory 14 days after the initial exposure. The steers were fed alfalfa (containing less than 0.2 ppm Se) ad libitum. No mineral supplements were made available. The steers were in fair condition, although several were slow to come up to the manger and eat. One of these steers declined in condition, exhibited depression and respiratory distress, and died 72 hr after arriving at the laboratory. The steer was immediately necropsied, and samples were collected for histological and chemical studies.
Animal sampling
At the laboratory, samples were collected from the remaining 9 steers at 14, 30, 60, 90, 120, and 150 days postexposure to determine Se concentrations in serum, whole blood, hair, liver, and muscle. Hair samples were collected by clipping the hair just behind the shoulder on the right side of the steer. On the first day that hair was collected (day 14), 2 samples were collected. The hair samples were divided into day 0 and day 7 based on the fiber length and estimated time of Se exposure. The first hair sample included hair longer than 0.5 cm, and the sample was reported as hair from day 0. The “new growth” hair was shaved close to the skin after the longer hair (last 0.5 cm of the hair fiber) had been clipped, and this hair was reported as hair from day 7, which was the intermediate day over the time that the hair grew. The subsequent samples were the short hair that had grown since the previous clipping. The samples were shaved adjacent to the skin at the same location and reported as the intermediate day over the time period during which the hair grew. The liver samples were percutaneous biopsies (20–40 mg) collected using a 14-gauge biopsy needle a following techniques previously published. 22 Subsequent biopsies were directed slightly ventral or anterior to the initial site to avoid sampling previously damaged tissues. The muscle biopsies were collected via a small incision made over the semitendinosus muscle. The muscle sample (5 mm × 5 mm × 10 mm) was removed using sharp dissection and aseptic techniques with local anesthesia (5 ml of subcutaneous lidocaine). After biopsy, the cutaneous tissues were sutured closed. Subsequent biopsies were taken from the opposite leg and from locations just cranial to the previous sites. Postoperatively, the animals were treated with aspirin (two 240-g tablets orally) for postbiopsy analgesia.
Serum biochemistries and serum electrolyte analyses were performed using standard techniques with a biochemistry analyzer. b Reagents and methodology recommended by the manufacturers were used.
Selenium analyses
Generic Se analysis was performed following techniques previously described. 27 Briefly, 750 µl of the sample was introduced into a labeled 10-ml digestion tube. c An equal amount (750 µl) of trace metal–grade nitric acid d was added to the digestion tubes, and the caps were sealed. The tubes were then heated at 90°C for 2 hr without unscrewing the caps. After digestion, tubes were allowed to cool down, and contents were transferred to another trace metal–free tube. One milliliter of this digest was transferred into another trace metal–free tube containing 9.0 ml of ultrapure water to make up a 5% nitric acid matrix. After vortexing, the samples were analyzed using ICP-MS. e Quantification of Se was performed by the standard addition method, using a 4-point standard curve. A quality control sample (in similar matrix) was analyzed after every 5 samples, and analysis was considered acceptable if the Se concentration of the quality control sample fell within ±5% of the standard/reference value for the quality control.
Plant digests
Plant samples were dried and ground to pass through a 0.5-mm screen. The ground plant material (0.50 g) was placed into a labeled 30-ml digestion tube. c Trace metal–grade nitric acid d (9.0 ml) was added to the digestion tubes. The tubes were then heated at 90°C for 2 hr with the caps loose on the tubes. After digestion, tubes were allowed to cool, and contents were increased to 10 ml, by adding nitric acid, and then transferred to another trace metal–free tube. Five hundred microliters was transferred into another trace metal–free tube containing 9.5 ml of ultrapure water to make up a 5% nitric acid matrix. After vortexing, the samples were analyzed using ICP-MS. e
Specific tissue digests
Liver, kidney, and semitendinosus muscle were digested as previously described. 27 Hair samples were carefully washed and dried to remove any foreign material. Hair (0.300 g) was introduced into a labeled 30-ml digestion tube. c Trace metal–grade nitric acid d (8.0 ml) was added to the digestion tubes. The tubes were then heated at 90°C for 2 hr with the caps loose on the tubes. After digestion, tubes were allowed to cool, and contents were increased to 10 ml, by adding nitric acid, and then transferred to another trace metal–free tube. Five hundred microliters was transferred into another trace metal–free tube containing 9.5 ml of ultrapure water to make up a 5% nitric acid matrix. After vortexing, the samples were analyzed using ICP-MS. e
Selenium data analysis
The 2-component mathematical model that best fit and described the kinetics of Se in the blood and serum was derived for the individual steers using the curve stripping method. The kinetic evaluations were performed using PK solutions 2.0 software. f The Se concentrations of the whole blood, serum, liver, and semitendinosus muscle from each steer were individually plotted as log concentrations versus time curves. The biexponential equation representing the curve was represented by the following formula:
where C was the concentration of Se in serum or whole blood at a given time (t), D and E designated the Y-axis (t = 0) intercepts, and δ and ϵ represented the rate constants for disposition/distribution and elimination, respectively. Curve stripping was performed to calculate the intercepts and rate constants. Each curve was stripped in the following order: 1) elimination phase and 2) disposition/distribution phase. Stripping involved mathematical subtraction of each term from the remaining expression to isolate and determine the individual linear constants associated with each phase.
Results
Selenium concentrations detected in collected tissues from 3 animals that died near the reclamation site are shown in Table 1. Plants collected from the reclamation site were later analyzed for Se by ICP-MS, and the results are reported in Table 2. The plants were identified by Katie Lott of the Poisonous Plant Research Laboratory Herbarium and Dr. Stanley L. Welsh at the Stanley L. Welsh Herbarium at Brigham Young University. Voucher specimens for the different plants were given accession numbers (see Table 2) and deposited in the Poisonous Plant Research Laboratory Herbarium in Logan, Utah. The western aster, heavily grazed in the localized reclaimed area, contained more than 4,000 ppm Se and was present in large enough quantities to be lethal. Fifty meters from the reclamation site, Se concentrations in nearly all of the plants, including the western aster, were less than 15 ppm. Some deep-rooted clover up to several hundred meters from the site had slightly higher concentrations of 28 ppm Se (Table 2). All water samples tested from streams in the meadow and from the pond below the reclaimed site were ≤0.005 ppm Se.
Tissue and serum selenium concentrations from steers that were grazing on high selenium forages growing on a reclaimed mine site in Idaho.*

NA = no specimen available.
Selenium concentrations of plants growing on a reclaimed mine site in Idaho.*
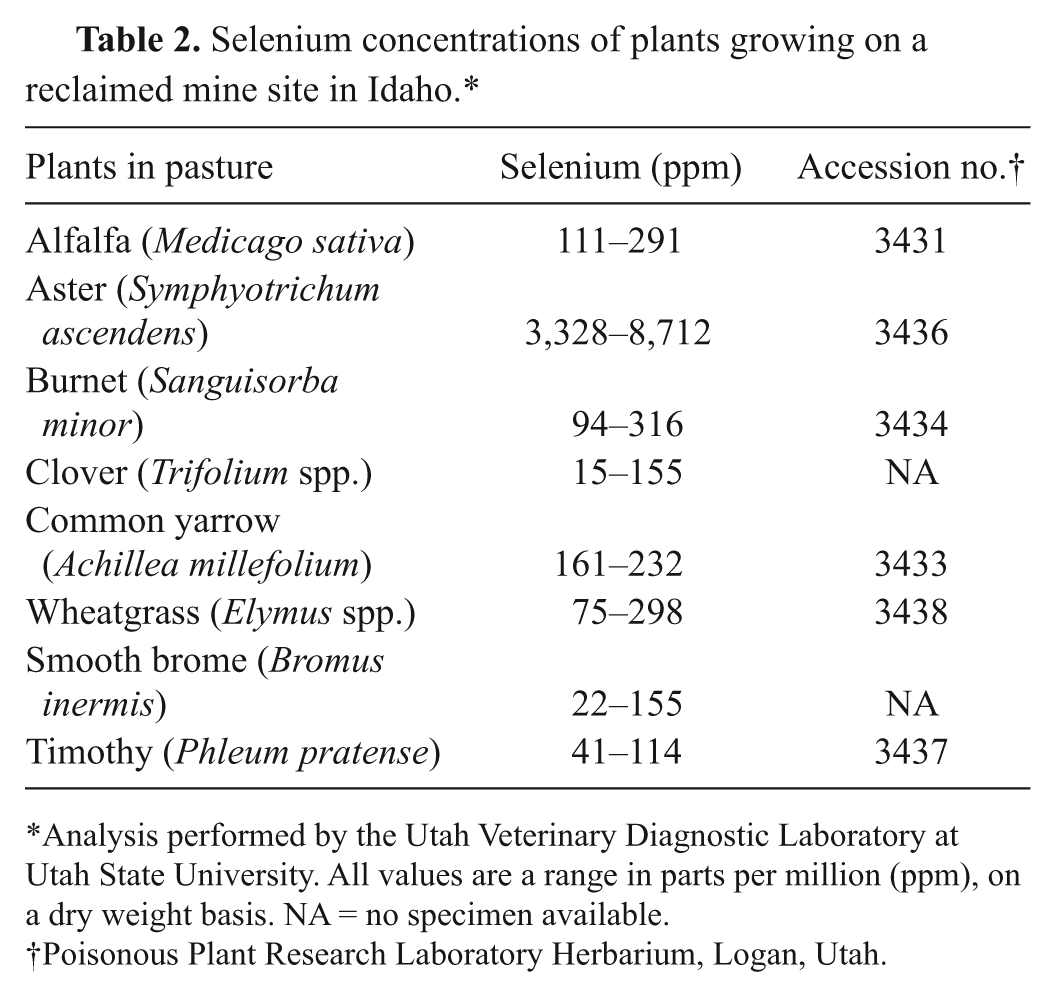
Analysis performed by the Utah Veterinary Diagnostic Laboratory at Utah State University. All values are a range in parts per million (ppm), on a dry weight basis. NA = no specimen available.
Poisonous Plant Research Laboratory Herbarium, Logan, Utah.
Histological studies of submitted tissues from steers that died within 48 hr of exposure showed that heart tissue had focally severe myocardial necrosis characterized by swelling and edema of many myofibers (Fig. 1). Many of the myofibers were degenerative with marked hypereosinophilia and focal clumping of myofiber proteins. The adjacent connective tissue was edematous with minimal infiltrates of lymphocytes and macrophages. Many myocardial Purkinje cells were also swollen and vacuolated with peripheral edema and infiltrates of lymphocytes and macrophages. The livers from these animals were congested with dilation of many of the central veins. Adjacent to the central veins, many of the hepatocytes were degenerative and necrotic with marked cytoplasmic vacuolation and nuclear pyknosis. The other tissues (kidney and spleen) were congested.
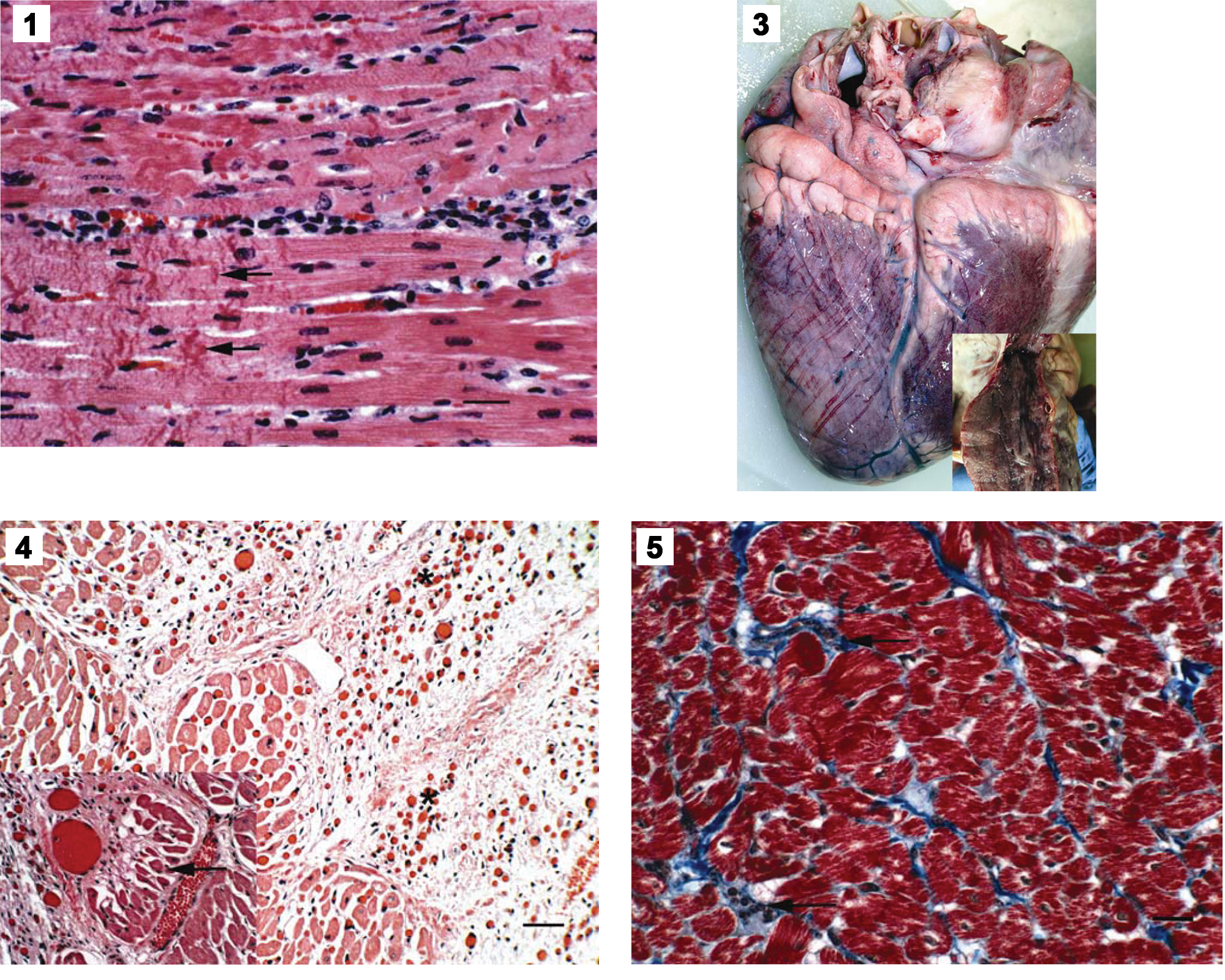
Photomicrograph of acute myocardial necrosis from a steer that was exposed to high selenium forages on a reclaimed mine site in Idaho. Notice the extensive myofiber hypereosinophilia, as well as coagulation and banding of myofiber proteins (arrows). Hematoxylin and eosin. Bar = 30 µm.
Serum and whole-blood Se concentrations of the 9 steers that were monitored are reported as a mean (± standard deviation [SD]) in Figure 2A. Of the time points sampled, peak Se concentrations in both the serum and whole blood were at 14 days postexposure. Whole blood had a peak Se concentration of 1.071 (± 0.254) ppm with a range of 0.726–1.451 ppm. The distribution half-life of Se in whole blood was 9.67 (± 3.60) days, whereas the Se elimination half-life was 115.6 (± 25.1) days with a range of 82.1–145.7 days. Peak Se concentration in serum was 0.744 (± 0.179) ppm with a range of 0.552–1.016 ppm. The distribution and elimination half-lives of Se in serum were 6.80 (± 4.43) and 40.5 (± 8.2) days, respectively. The normal reference range of Se in bovine whole blood and serum is 0.20–1.20 ppm and 0.08–0.30 ppm, respectively. 20 However, in the area where these steers were located, normal Se concentrations in whole blood of cattle are typically between 0.15 and 0.30 ppm as animals in this area frequently are marginally Se deficient.
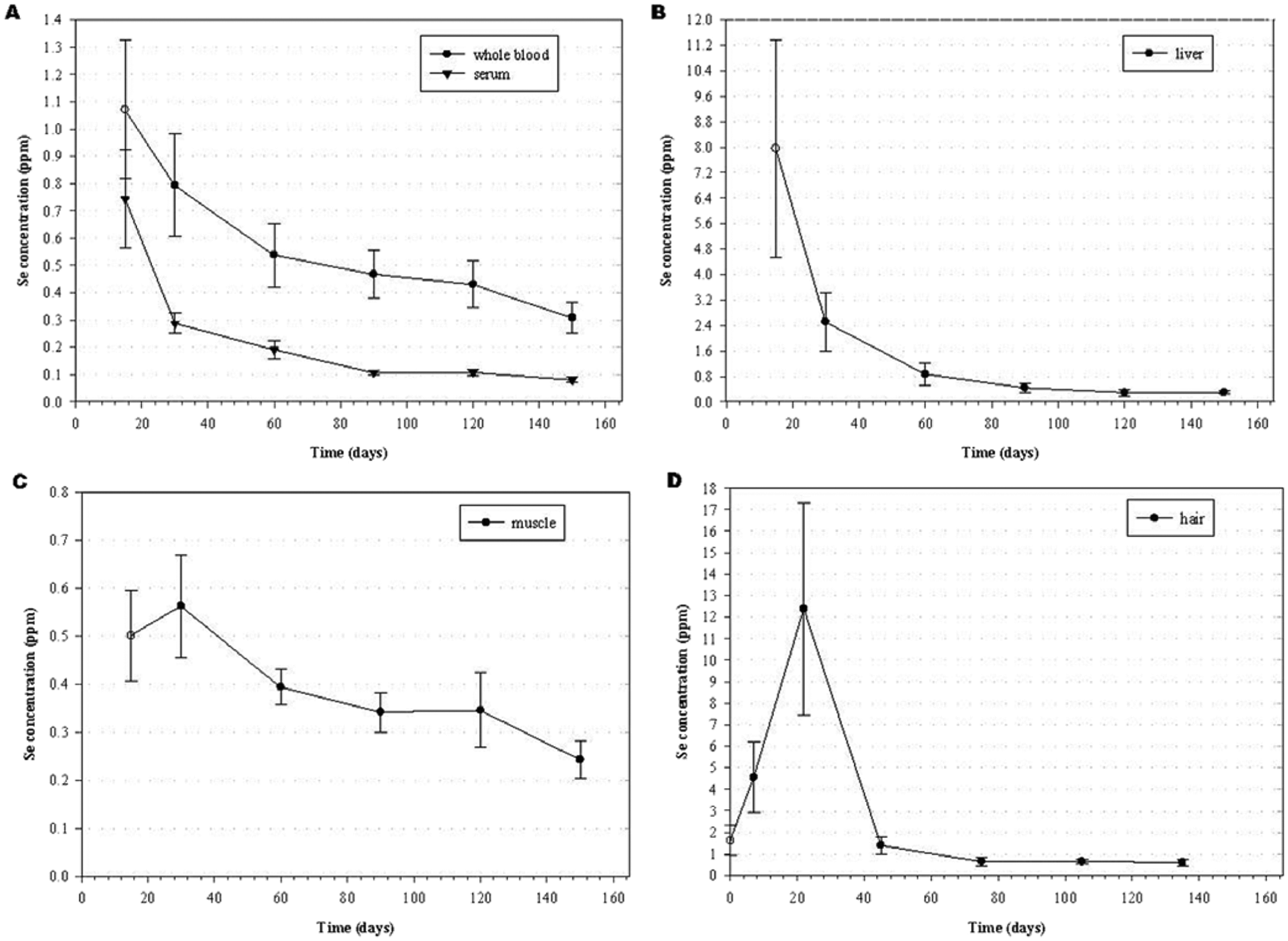
Mean (± standard deviation [SD]) selenium concentrations (ppm) in (
Selenium concentrations in the liver at various time points are also reported as a mean (± SD) in Figure 2B. Highest Se concentrations in the liver were observed at the first time point in which samples were taken, which was 14 days postexposure. The mean Se concentration at 14 days postexposure was 7.958 (± 3.411) ppm (wet weight [ww]) with a range of 3.634–13.257 ppm (ww). Three steers had liver Se concentrations greater than 11.500 ppm (ww). The distribution half-life of Se in the liver was 6.19 (± 1.30) days with a range of 4.10–8.67 days. The elimination half-life of Se in the liver was 38.2 (± 5.0) days with a range of 30.7–46.6 days. Normal reference range of Se concentration in bovine liver is 0.25–0.50 ppm (ww). 20 The liver Se concentrations in all steers in the current study had returned to within the normal range by 120 days postexposure. By day 150, 2 of the steers had become marginally Se deficient (0.235 and 0.246 ppm [ww], respectively) according to Se concentrations in the liver.
Selenium concentration in the semitendinosus muscle of the steers at various time points is reported as a mean (± SD) in Figure 2C. Selenium concentrations in semitendinosus muscle increased between days 14 and 30 to reach peak Se concentrations of 0.562 (± 0.107) ppm (ww) at 30 days postexposure. At 30 days postexposure, the range of Se in the muscle was 0.554–1.026 ppm (ww). The elimination half-life of Se in the semitendinosus muscle was 98.5 (± 19.1) days, which was much longer than the elimination half-life of Se in the liver and serum, which was 38.2 (± 5.0) and 40.5 (± 8.2) days, respectively. The elimination half-life of Se in semitendinosus muscle ranged from 59.0–125.9 days and was inversely related to weight gain. The inverse relationship to weight gain is likely due to increased protein turnover and a dilution effect of increasing muscle mass. At 150 days postexposure, Se concentrations remained nearly twice the high end of the normal Se reference range found for bovine muscle of 0.07–0.15 ppm (ww). 20
Selenium concentrations in hair were determined from hair sampled at 14, 30, 60, 90, 120, and 150 days postexposure and are reported as the mean (± SD) at 0, 7, 22, 45, 105, and 135 days in Figure 2D. The concentrations are reported as the mean (± SD) on the intermediate day over which the hair was grown because Se concentration in hair collected on a particular day is an average of Se in hair grown over a specific time period. For example, hair collected on day 60 was hair grown between days 30 and 60, and therefore Se concentration in the hair is reported as the mean Se concentration in the hair on day 45. Mean Se (± SD) concentration in the hair furthest from the skin on day 14 was 1.613 (± 0.686) ppm and was reported as the mean (± SD) Se concentration in the hair on day 0. The average Se concentration in the hair collected nearest the skin on day 14 was 4.547 (± 1.636 ppm) and was reported as the mean (± SD) Se concentration on day 7. The mean (± SD) Se concentration on day 22 contained the peak Se concentration in samples collected and was 11.555 (± 5.346) ppm with a range of 4.049–21.445 ppm. By day 45, Se concentration in the hair sampled was lower than Se concentration in hair samples reported for day 0. Average Se concentration in hair of cattle is reported 20 to be 0.50–1.32 ppm, with a toxic range of 1.40–45.00 ppm on a dry weight basis.
The heart from the steer that died at the laboratory was dilated and pale with marked dilation, small hemorrhages, and hyperplasia of the epicardial lymph vessels (Fig. 3). The myocardium was soft with prominent pale streaking (Fig. 3 inset). The liver was enlarged with prominent lobular congestion or hemorrhage seen grossly as a nutmeg-like pattern. Histologically, there was focally extensive myocardial fibrosis that entrapped prominent proliferative cardiac vessels (Fig. 4). Occasionally, the fibrous connective tissue entrapped bundles of myocardium (Fig. 4 inset). The centrilobular portions of the liver were markedly disrupted and expanded with replacement of hepatocytes with dilated, blood-filled sinusoids. Hemosiderin-filled macrophages were common. The central veins had thickened fibrous walls and were blood filled. The remaining hepatocytes in mid-lobular and periportal zones were swollen and vacuolated. The lung was edematous with marked interlobular emphysema and edema. One small portion of lung had a focally extensive fibrous bronchoalveolitis characterized by vascular thrombosis with focal alveolar and bronchiolar accumulations of fibrin, proteinaceous fluid, and small numbers of neutrophils and lymphocytes. The adjacent pneumocytes were hyperplastic. The spleen and kidney were congested.
Biochemical changes of interest and animal weight gains are presented in Table 3. Several steers had mildly increased serum creatinine kinase activities and elevated troponin I concentrations on day 14 postexposure. These activities returned to normal, and all were within normal activities and concentrations by day 150. The amount of weight gained by the steers from day 15 to day 150 postexposure ranged from 44.5 to 98.9 kg. The steers that gained the least amount of weight during the 5 months following exposure were euthanized and necropsied at the end of the study to determine any possible long-term effects from the acute Se exposure.
Animal weights and select biochemical values from steers that were exposed to high selenium forages from a reclaimed mine site in Idaho.*
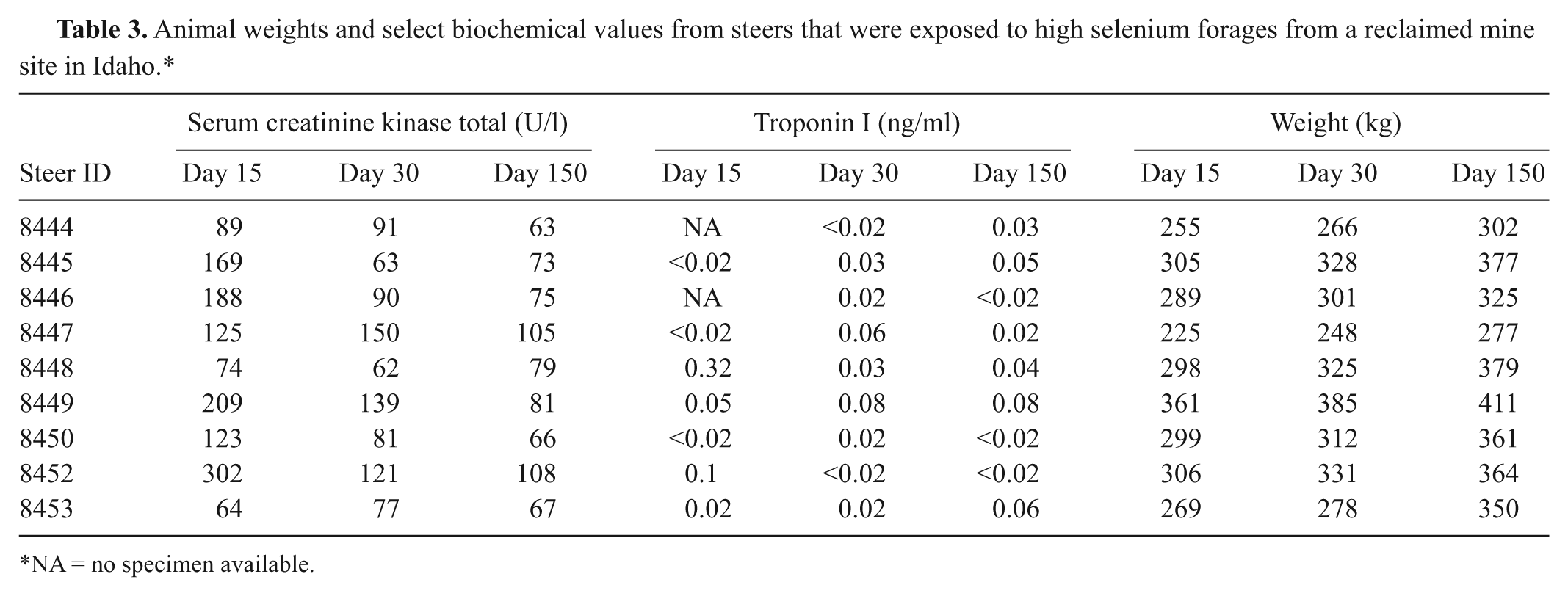
NA = no specimen available.
The livers of the 2 steers that were euthanized on day 150 had fibrous tracts and rare small (1–2 cm) abscesses along the biopsy needle tracts in the peripheral capsule of the liver. The skeletal muscle biopsy sites were also fibrotic and scarred. No additional gross lesions were observed. Histologically, the myocardium of both animals had rare small foci of fibrosis with minimal lymphocytic inflammation (Fig. 5). No significant lesions were detected in the other sections of lung, liver, spleen, kidney, lymph node, or gastrointestinal tract.
Discussion
The appearance of clinical signs of acute Se poisoning occurs as early as 8–10 hr after exposure; however, signs may be delayed for up to 36 hr postexposure.7,18,21 The appearance of clinical signs of the poisoned steers in the present study was also very sudden and similar to previous reports, including lethargy, head down, droopy ears, respiratory distress, gaunt appearance, excessive salivation, tachycardia, stilted gait, tetanic spasms, and/or death. Clinical signs tended to progress quickly after the first observations of poisoning appeared. Most animals that are going to die from acute Se poisoning will do so within 72 hr. However, lethality was observed for an extended period (18 days) after acute exposure, and evidence of extensive fibrose tissue damage to the heart was found.
The amount of Se-accumulator plants ingested by the steers is unclear. However, it has been reported that lambs dosed with western aster at 2 mg Se/kg have shown significant clinical signs of poisoning, whereas a dose of 4 mg Se/kg of western aster has been lethal (Wilhelm A: 2010, Investigation of the toxicity and toxicokinetics of selenium from the accumulator plant Symphyotrichum spathulatum (Western Mountain Aster) in sheep. Master’s Thesis, Department of Animal, Dairy and Veterinary Science, Utah State University Merrill-Cazier Library, Logan, UT). The authors of the current study expect that approximately the same doses are lethal in cattle. It is reported that acute oral Se poisoning occurs with sudden exposure of Se ranging from 2.2 to greater than 20 mg/kg bw across species.16,17,23 The relative acute toxicity of Se-containing compounds is dependent on their solubility, with poorly soluble selenides and elemental Se being much less toxic than soluble selenates, selenites, and organic Se. 18 The LD50 for oral selenite has been estimated to be 1.9–8.3 mg/kg bw in ruminants, 9 but other references suggest it to be 9–20 mg/kg bw. 20 In a previous study, an oral dose of 1.1 mg/kg in mature cattle resulted in illness and death, whereas a dose of 0.55 mg/kg seemed to have little effect on the same group of cattle. 14 Injectable Se is more acutely toxic than oral, with intramuscular LD50 of 0.5 mg/kg in lambs. 1 Subcutaneous LD50 of Se is 1 mg/kg in lambs and 1.9 mg/kg in adult cattle. 9
The elimination kinetics differed, depending on the tissue of analysis. The whole-blood elimination half-life of 115 days interestingly approximates the red blood cell half-life in cattle of 130 days. 29 This would be expected in that increased red blood cell–incorporated seleno-proteins, following high Se exposure, would be retained in the cells until removed from circulation. The liver and serum half-lives were quite similar (2–3 times faster than the muscle elimination), indicating that either could be equally used to follow primary clearance of excessive Se. A long elimination half-life of skeletal muscle is somewhat expected. Turnover of the muscle tissue proteins, which could release incorporated seleno-amino acids, is expected to be very slow. In fact, some of the tissue decrease in Se content is likely due to the dilution effect of continued muscle mass growth. These rates of Se elimination are for cattle that were acutely poisoned by Se, and the rates may be different for cattle that are chronically poisoned or are poisoned by other forms of Se.
The average skeletal muscle content of background beef in the United States is 0.22 mg Se/kg, with a 95% upper confidence limit of 0.39 mg Se/kg, 24 and different cuts of meat had similar Se concentrations when taken from the same steer. 10 Steers grazed on the reclaimed mine site in southeastern Idaho for 9 weeks and then placed in a feedlot for 22 weeks had 0.74 mg Se/kg, with an upper 95% confidence limit of 0.86 mg Se/kg (Talcott P, Kinery J, Knight J, Wright W: 2000, Skeletal muscle selenium content of treatment steers included in the Idaho Mining Association’s Beef Study for Southeast Idaho Selenium Project: a worst-case analysis of confined animals on reclaimed mine land. A final report to the United States Department of Agriculture’s Food Safety and Inspection Service. March). The peak Se concentration (0.562 ± 0.107 ppm [ww]) in the muscles of these steers occurred 30 days postexposure. The risk of contaminating food supplies with beef from cattle similar to these that live after receiving a large acute dose of Se is very low. Assume that if 8 oz. (0.2270 kg) of beef is consumed per day, which is 4 times the national average (U.S. Environmental Protection Agency (USEPA): 1996, Exposure Factors Handbook, vols. I–III. EPA/600/8-89/043. USEPA, Washington, DC. Available at http://cfpub.epa.gov/ncea/cfm/recordisplay.cfm?deid=12464#Download. Accessed on November 29, 2011), from the steer with the highest Se content (0.70 ppm), then the daily Se intake from beef would be 158 µg Se. The human recommended intake is 55–70 µg/day, and the highest tolerable intake level has been recommended at 400 µg/day. 11 No adverse toxic effects of Se have been reported in people consuming less than 1,000 µg,33,34 and in fact, as much as 300 µg/day may be needed to reduce the risk for certain cancers. 2
Cattle that are exposed to toxic doses of Se will generally die within the first 36 hr from heart failure with acute myocardial necrosis. Purkinje cell involvements suggest these early deaths are likely due to altered electrophysiology and arrhythmias. However, a small number (1/15 or approximately 7–8%) may have enough myocardial damage so that myocardial fibrosis and subsequent heart failure develop in subsequent weeks. These animals may be identified as they have increased serum creatinine kinase activities and elevated troponin I concentrations. Animals whose biochemical analyses return to normal within 4–6 weeks are likely to recover.
The histologic myocardial lesions are similar to those that have been described in other cases of Se poisoning.15,25,27 However, in the animals examined in the present study, the lesions were confined to the heart, and the subsequent effects of heart failure were observed. Lambs dosed with selenite and selenomethionine had severe pulmonary lesions characterized by pulmonary alveolar vasculitis with pulmonary edema and hemorrhage.27,28 There were no vascular lesions in the lungs of the steers observed in the current study, and the steers did not die of pulmonary edema as has been described in some acute poisonings.26,27 This is similar to other reports in which mild pulmonary congestion and alveolar edema were found in only 1 of the calves that died acutely after parenteral injection of a single dose of sodium selenite. 15
Future losses in production due to decreased weight gain are unlikely. Of the steers that were monitored, those that gained weight more slowly had minimal myocardial changes. Their decreased weight gains were most likely due to animal variation in production as these changes are small and probably not physiologically important. One of the steers was visibly more nervous than the other steers and likely did not consume as much feed. However, more research is needed to better characterize the progression of such lesions and definitively determine their long-term impact on animal production.
One of the most interesting results of the present study was the concentrations of Se obtained in the hair from acute exposure to Se. It is reported 20 that the average Se concentration in hair of cattle is 0.50–1.32 ppm, with a toxic range of 1.40–45.00 ppm on a dry weight basis. The average peak Se concentration (11.555 ± 5.346 ppm) in the current case was obtained 22 days postexposure. The 5 steers with the highest hair Se concentrations had values of 21.45, 17.39, 14.79, 12.91, and 12.69 ppm, respectively. Because the hair that was furthest from the skin when the first samples were taken on day 15 and reported as day 7 was less than 2.578 ppm on each steer and the hair that was grown during the next 15 days contained much higher concentrations, an acute exposure to Se can at least be partially diagnosed by sampling the hair 3–4 weeks postexposure.
Management recommendations to avoid future losses of livestock included fencing off a 5-acre portion of the 500-acre pasture to allow safe utilization. Range managers were cautioned to maintain fences and avoid accidentally leaving gates open to avoid further accidental exposure. Landowners should also be mindful of potential problems on their range. In the current case, the pasture had been used for nearly 20 years without problems; however, the risk was nonetheless present, so when conditions were right, poisoning of livestock was a result. Contaminated sites should be monitored, and when risks are present with forages that may be toxic, the areas should be fenced to restrict access of livestock.
In conclusion, the findings indicate that cattle are extremely sensitive to Se poisoning from Se-accumulating plants. The primary change of acute selenosis in cattle was found to be myocardial necrosis with subsequent heart failure seen as passive congestion and centrilobular hepatic congestion with hepatocellular degeneration and necrosis. A small percentage of animals that do not die immediately may, if myocardial damage is significant, develop myocardial fibrosis and subsequent heart failure. In addition, Se is slowly excreted, thus requiring long withdrawal times. Therefore, acute Se poisoning can be diagnosed by sampling hair 3–4 weeks postexposure in cattle that recover.
Footnotes
Acknowledgements
The authors thank Katie Lott, Edward L. Knoppel, Joseph Jacobson, Harold Winger, and Mary Hubbard for their assistance with this research, as well as Drs. Jean Hall and Merl Raisbeck for their review of this manuscript. This research was supported by USDA/ARS and the Utah Agriculture Experiment Station, Utah State University.
a.
Care Fusion, McGaw Park, IL.
b.
Hitachi 7180, Hitachi High Technologies Inc., Pleasanton, CA.
c.
Oak Ridge Teflon digestion tube, Nalge Nunc International, Rochester, NY.
d.
Fisher Scientific, Pittsburg, PA.
e.
ELAN 6000, PerkinElmer, Shelton, CT.
f.
PK Solutions, Montrose, CO.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.