Abstract
Just prior to an international polo event, 21 horses from one team exhibited clinical signs of central nervous system disturbance, hyperexcitability, sweating, ataxia, tachycardia, dyspnea, pyrexia, and rapid death. The suspected cause of this peracute onset of illness and death included intentional contamination of feed or iatrogenic administration of performance-enhancing drugs resulting in a severe adverse reaction. Six horses were submitted to the Bronson Animal Disease Diagnostic Laboratory for necropsy and toxicological examination. The clinical signs and sudden death, the similarity to earlier work by the lead author of selenium toxicosis in calves, as well as published reports, prompted investigators to focus on selenium testing. Sixty-four hours following receipt, the laboratory detected toxic selenium concentrations in the tissues of these animals. Following further investigation of the case by regulatory officials, it was determined that all affected horses had received an intravenous injection of a compounded “vitamin/mineral” supplement just prior to the onset of signs. The compounded supplement contained toxic levels of selenium. The present report illustrates the in-depth laboratory investigation of the cause of acute death in 6 polo ponies due to selenium toxicosis. In addition to solving this high profile case, the toxic levels of selenium found in livers (6.13 ± 0.31 mg/kg wet weight), kidneys (6.25 ± 0.3 mg/kg wet weight), and sera (1.50 ± 0.11 µg/ml) of these affected animals may provide important diagnostic criteria for future interpretations of selenium concentrations in tissues of horses.
Selenium is essential for the normal functioning of most organ systems including the heart, muscle, liver, and kidneys in both animals and human beings, as well as many plants and some species of bacteria. 1,5,8–10,15,16,22,23 Selenium plays an important role in the cellular antioxidant defense mechanism through selenium-dependent enzyme systems and selenium-binding proteins. 1,8,13 Glutathione peroxidase (GSH-Px) was the first functional selenium-dependent enzyme identified and is considered to play a major role in inhibiting peroxidative cellular damage from free radicals and reactive oxygen species. 6,8,16,19,20 Selenium-dependent GSH-Px catalyzes the reduction of hydrogen peroxide and several organic hydroperoxides by oxidation of reduced GSH to oxidized glutathione (GSSG), which is converted back to reduced GSH by GSH reductase for use by the antioxidant defense system. 3,8,9,13,19,20 Four glutathione peroxidases and a range of selenoproteins have subsequently been identified indicating multiple biochemical functions for selenium. 1,9,13 The biological roles of most have yet to be determined. 1,18 Several roles for selenium in the normal functioning of the immune and cardiovascular systems, and in the prevention of some types of cancer, have been reported by investigators. 6,8,10
Plants are the main source of dietary selenium by uptake of selenium from the soil. 23 As a result of geographic variations in the concentration of selenium in the soil, animals and human beings from low selenium regions do not receive adequate selenium in the diet and may be deficient; those from seleniferous regions may be exposed to excess selenium and develop chronic selenium toxicosis. 5,6,9,10,15,22,23 It is known that many biological functions and processes are affected by selenium deficiency, and the associated selenium responsive disorders can be alleviated by the administration of selenium compounds. 9,13 Acute selenium intoxication may result from accidental overdosage by administration of selenium compounds by parenteral or oral route; toxicosis also occurs from environmental contamination and nutritional overdose. 6,9,10,21,23 The current report describes acute selenium toxicosis in polo ponies that were taking part in the U.S. Polo Championships on April 19, 2009, in which toxic selenium concentrations were detected in tissues and sera collected from these animals.
Although several attending veterinarians at the event provided emergency treatment utilizing various medications, 20 of 21 polo ponies nevertheless died on their origin premises just prior to the event, en route to the event, or soon after unloading at the event. The 1 additional horse that developed clinical signs was transported to a veterinary clinic where it died later that evening. No other horses, other than those intended for participation in the event, on the origin premises or at the event premises, showed any clinical signs. Specific history of treatment prior to the onset of clinical signs was not available although there were unconfirmed reports that the affected horses had been given an unspecified treatment just prior to the event and the onset of clinical signs. Clinical signs were observed in 2 mares 1.5 hr after injection and therefore, these 2 horses were not transported to the event. Two horses died en route to the event 2.5 and 3.0 hr after injection, respectively. Most horses had signs on arrival at the polo event facilities. In all, 20 polo ponies died within 6 hr of injection.
Clinical signs included severe central nervous system symptoms, with hyperexcitability, ataxia, sweating, tachycardia, dyspnea, pyrexia, and sudden death. The rapid death of otherwise healthy ponies taking part in an international polo match led to speculation of poisoning, possibly by injection or via tampering with feed. Considerations were given to the possibility of “concocted” performance-enhancing drugs given to the ponies prior to the match. Six pony cadavers were submitted to the Bronson Animal Disease Diagnostic Laboratory (BADDL; Kissimmee, Florida). Samples of kidney, liver, fat, heart, and brain tissue, and stomach contents were collected for toxicological examination. Samples from the liver, kidneys, urinary bladder, tonsils, retropharyngeal lymph nodes, larynx, lung, spleen, pancreas, heart, skeletal muscles, trachea, adrenal glands, thyroid and parathyroid glands, esophagus, stomach, duodenum, jejunum, cecum, colon, cerebrum, and cerebellum were collected and preserved in 10% buffered formalin for histopathologic examination. Serum and urine samples, collected prior to death, as well as feed and hay samples, were also submitted. The possibility of criminal activity in the death of the ponies and associated insurance considerations amplified the need for a thorough and comprehensive laboratory evaluation to determine the cause of death. The BADDL followed chain of custody procedures to ensure that all samples were accounted for and that sample results were properly reported.
Complete postmortem examinations were performed on the 6 ponies. Grossly, the skeletal muscles of the thoracic limb, pelvic limb, neck, thorax, and diaphragm revealed multifocal and coalescing areas of acute hemorrhage. The severity and extent of hemorrhage varied greatly, with some muscles showing acute, petechial, multifocal, and coalescing hemorrhage, while in other muscles the hemorrhage was more extensive (Fig. 1). Mild to severe lesions were also observed in the hearts, with the myocardium and subendocardium being mostly affected. The hearts appeared irregularly reddened with alternating pale and marbled appearances. Other gross finding in the hearts included ecchymotic hemorrhages of the epicardium and endocardium. In all ponies, the colon and cecum appeared glossy while the intestinal mucosa depicted variations of hyperemia, petechiae, and suffuse hemorrhage. Other gross lesions included mild to marked laryngeal hemorrhages and edema, pulmonary hyperemia, and profuse tracheal froth (Table 1).
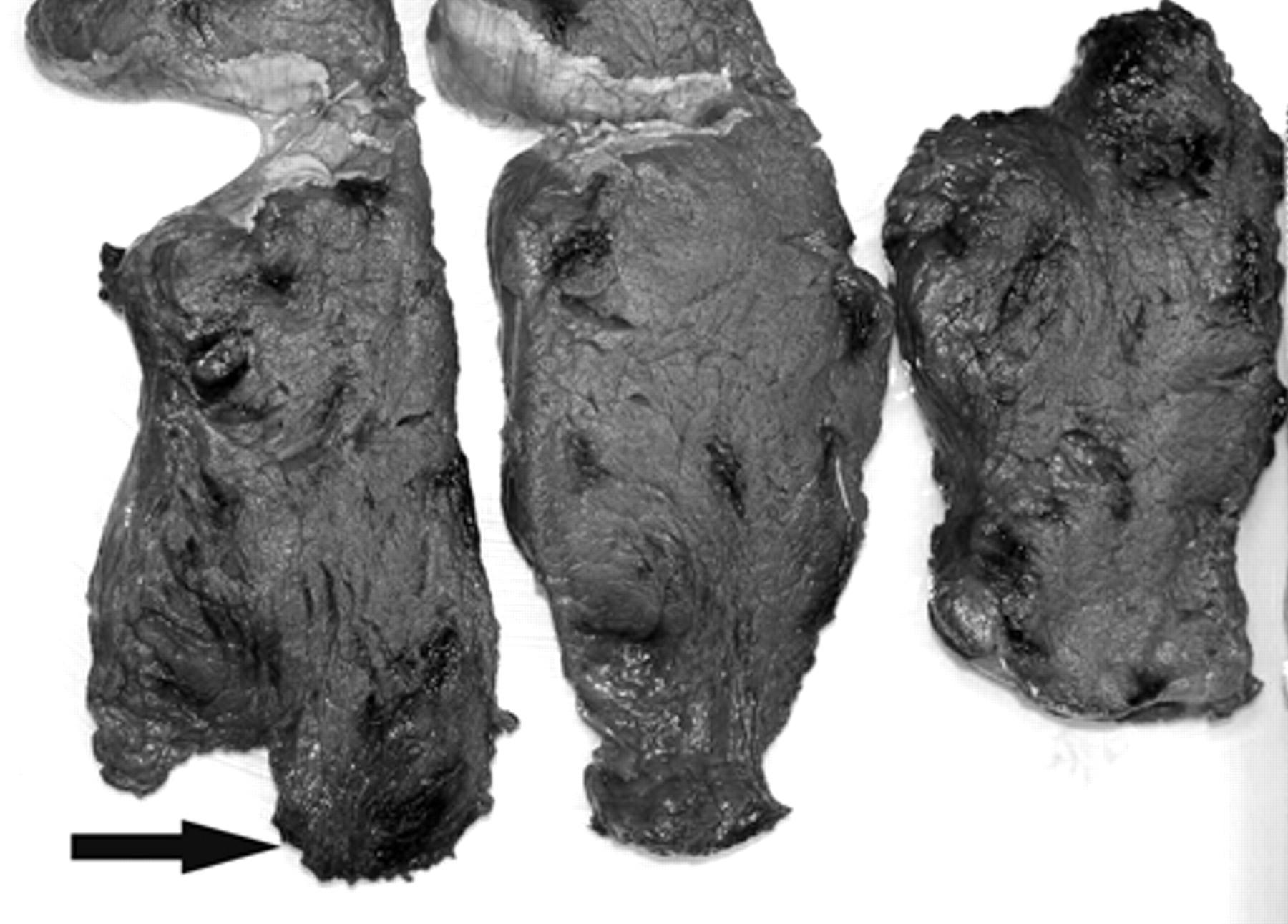
Superficial gluteal muscle; horse. Multifocal-to-multifocal and coalescent acute hemorrhages in muscle tissue (arrow).
Gross and microscopic distribution of prevailing lesions in different organs in 6 polo ponies examined in the current study.

Microscopically, the skeletal muscles depicted large areas of multifocal, coalescing, interfascicular hemorrhages, in addition to multiple foci of acute myofiber degeneration with intracytoplasmic vacuolation. Hemorrhage also effaced the normal architecture of the diaphragm. In the hearts, hemorrhages were predominantly subendocardial and subepicardial, multiple, linear, and occasionally extended deeply between myocytes. Myocardial lesions included myofiber degeneration and hypercontraction, myocytolysis, and calcification (Figs. 2, 3). In the endocardium, lesions were prominent adjacent to and within papillary muscles. Marked hyperemia and hemorrhage with severe, diffuse submucosal edema and discrete lymphocytic and eosinophilic infiltrate were present in the cecum and colon. Hemorrhage in the larynx was multifocal.
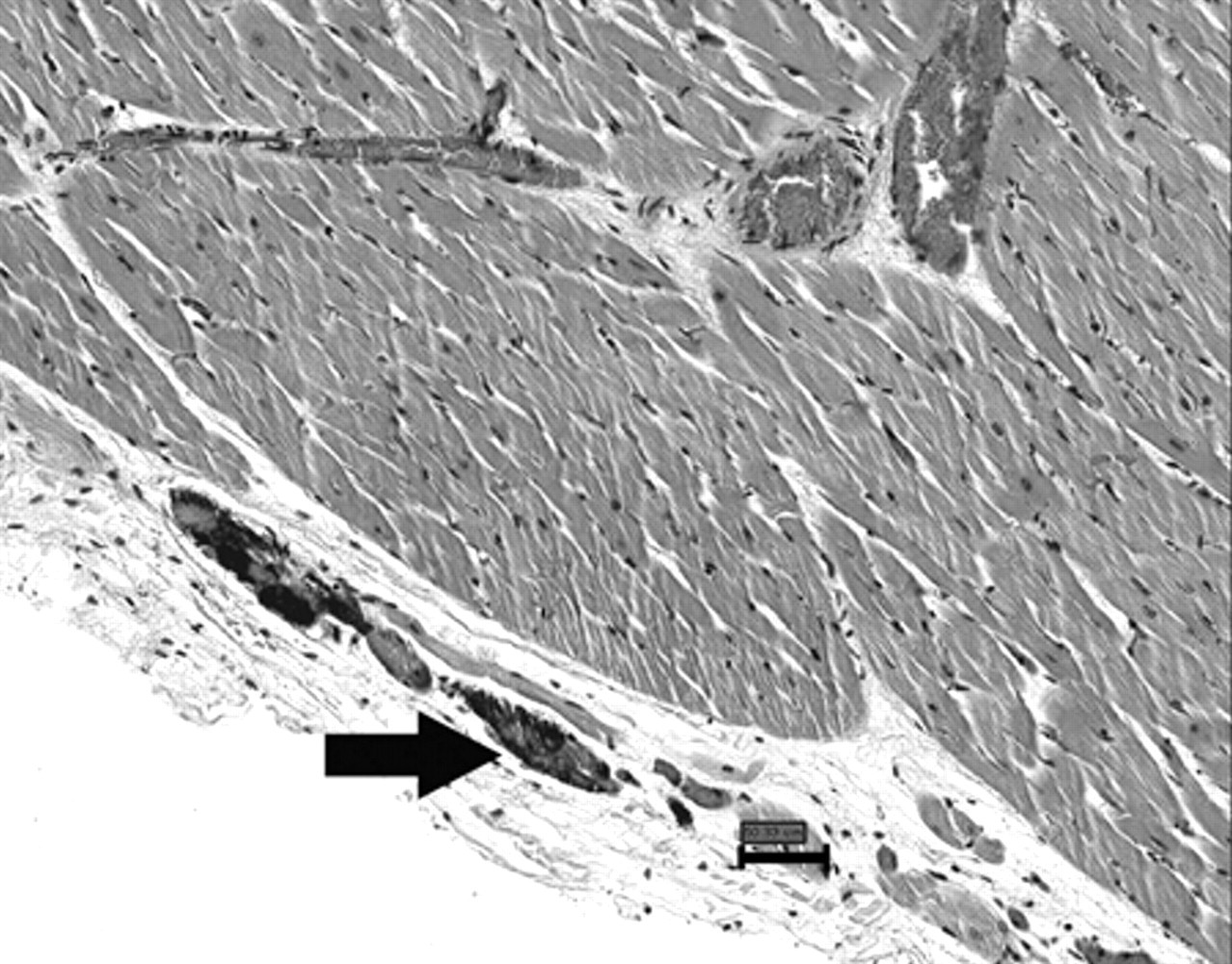
Heart; horse. Endocardial calcification (arrow). Hematoxylin and eosin. Bar = 50.33 µm.
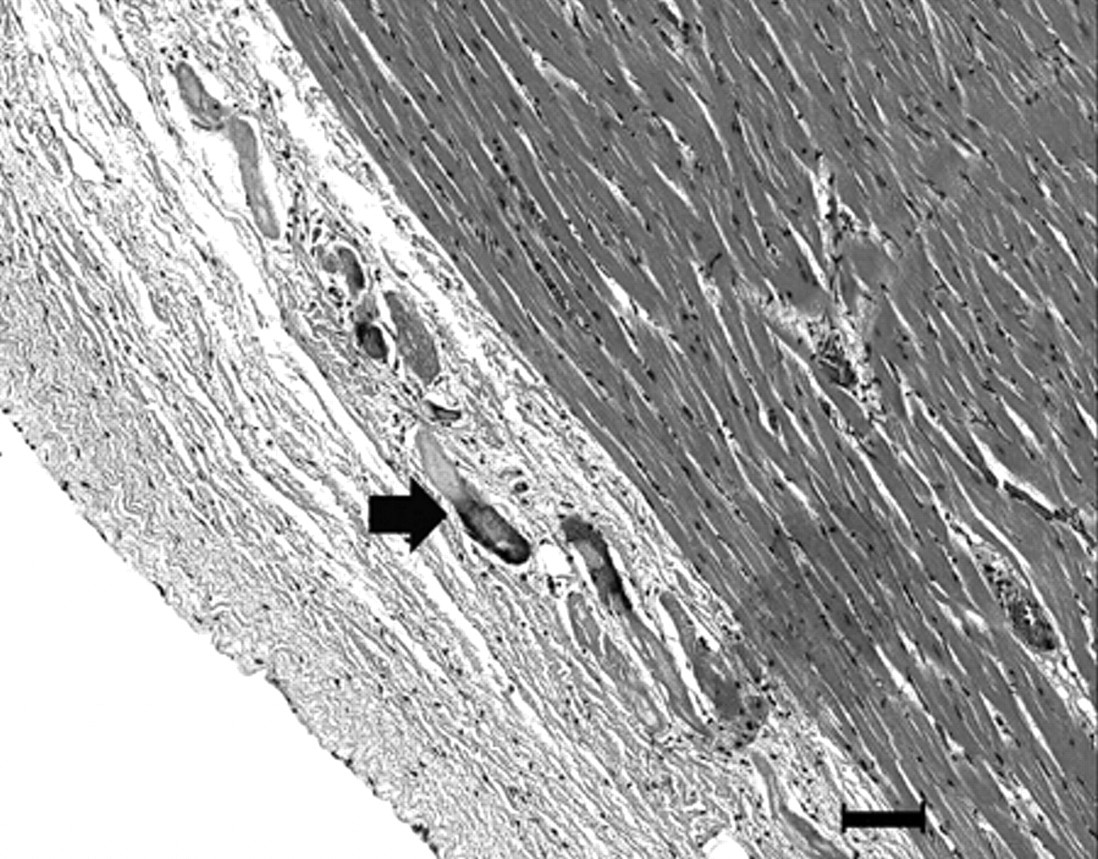
Heart, left ventricle; horse. Epicardial and subepicardial degeneration and calcification (arrow). Hematoxylin and eosin. Bar = 100 µm.
The gross and histopathological findings encountered in the heart were consistent with hemorrhage and acute ischemic myocardial necrosis. The lesions in the left heart were more prominent, possibly because of greater intramyocardial tension during systole and increased likelihood of ischemia. The lesions observed in the skeletal muscle, particularly the diaphragm, and those in the heart were consistent with microangiopathy.
The challenge for the Toxicology laboratory was how to deal with this high profile, high visibility case in a timely manner while ensuring that the necessary quality controls were followed. Since the BADDL Toxicology laboratory could not perform all the comprehensive tests required, it was immediately decided to split the samples with the California Animal Health and Food Safety Laboratory System (CAHFS; University of California, Davis, California) for additional toxicology testing. Stomach contents, liver, kidney, brain, serum, urine, feed, and hay were analyzed by the CAHFS toxicology laboratory for a comprehensive list of toxicants. Gas chromatography–mass spectrometry (GC-MS) screens were performed to evaluate the samples for the presence of a large number of pesticides, drugs, plant toxins, and other organic compounds. Liquid chromatography–mass spectrometry techniques were used to analyze specimens for ionophore antibiotics and certain drugs. Metal concentrations in tissues were determined by inductively coupled plasma–atomic emission spectroscopy (ICP-AES).
The BADDL had previously encountered a similar case where 15 calves died within 3–7 days after they were worked in cow pens. The calves had been injected with a “vitamin/mineral mix.” Liver samples from 2 calves submitted to the BADDL showed toxic concentrations of selenium more than 5 times higher than the reference range for calves (i.e., 7.0 mg/kg wet weight, reference range in liver of new born calves: 0.30–1.20 mg/kg 17 ). Experimentally induced selenium intoxication can cause sudden death in several species including lambs, pigs, cattle, and horses. 4,7,12 In addition, clinical signs observed in the polo ponies were similar to those seen in these experimentally induced toxicosis. With this background information, selenium overexposure was a likely etiology in the sudden onset of illness and deaths of the polo ponies and provided sufficient ground to first focus on selenium.
Analysis of selenium was conducted using a graphite furnace atomic absorption spectrometer (GFAAS). a Liver and kidney samples (0.5 g each) were digested with 10 ml of 70% nitric acid using a microwave digester. The digested samples were then diluted to 50 ml with deionized water and analyzed by GFAAS using standard additions technique. Serum samples were diluted and analyzed by GFAAS. The liver selenium concentrations of all 6 ponies were highly elevated (Table 2), ranging from 5.1 to 7.3 mg/kg wet weight (reference range in liver: 0.30–1.00 mg/kg wet weight 17 ). The kidney selenium concentrations were also elevated (Table 2) and ranged from 5.2 to 7.2 mg/kg wet weight (reference range in kidney: 0.70–2.00 mg/kg wet.weight 17 ). Liver selenium concentrations above 2.5 mg/kg wet weight are considered to be toxic in horses. 17 Kidney selenium concentrations greater than 2.0 mg/kg wet weight are observed in acute intoxications. 17 The serum selenium concentrations were also elevated (Table 2), ranging from1.3 to 1.9 µg/ml (reference range in serum of adult horses: 0.14–0.25 µg/ml 17 ). Livers, kidneys, and sera from unaffected horses were also analyzed for comparison (Table 2).
Selenium concentrations in tissue and sera of 6 ponies injected with toxic doses of a compounded “selenium-vitamin mix”.*
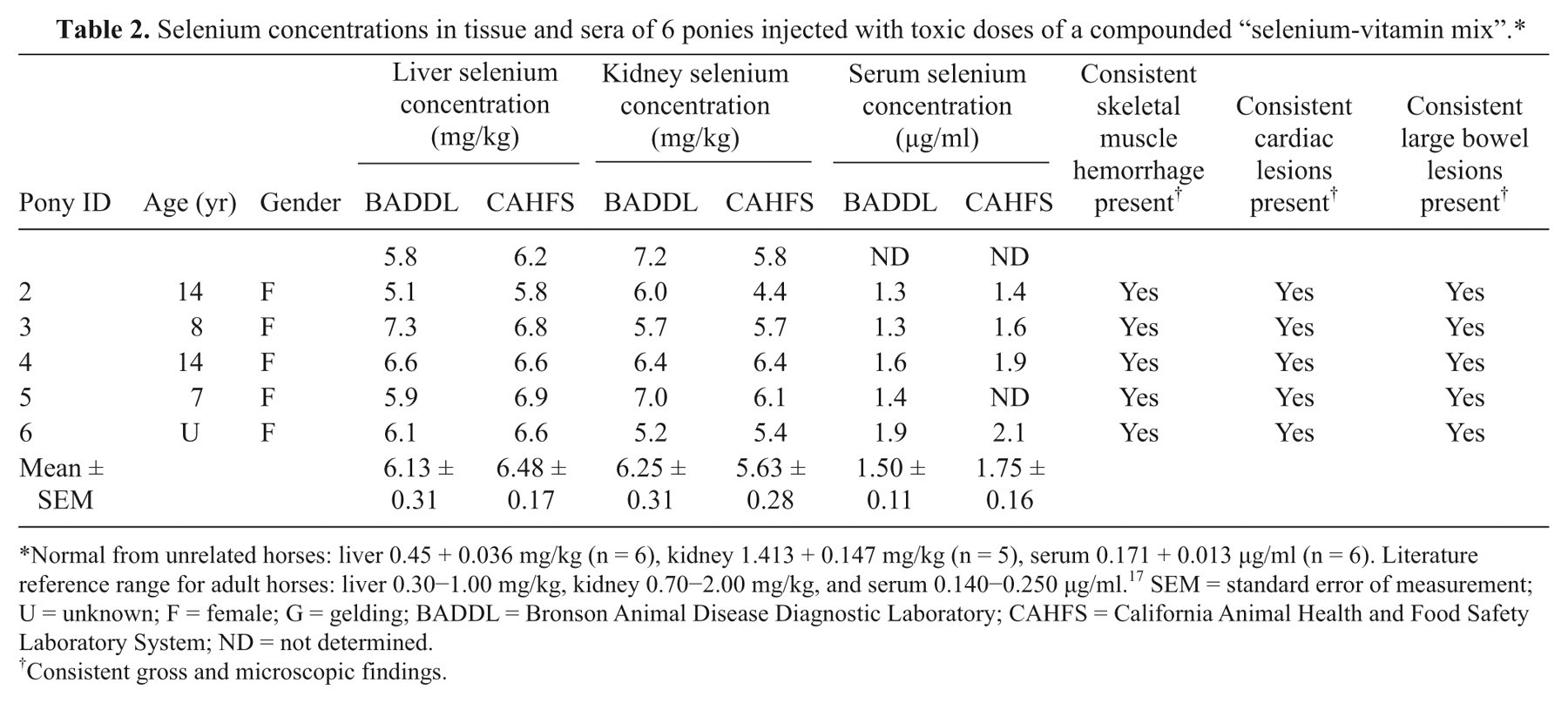
Normal from unrelated horses: liver 0.45 + 0.036 mg/kg (n = 6), kidney 1.413 + 0.147 mg/kg (n = 5), serum 0.171 + 0.013 µg/ml (n = 6). Literature reference range for adult horses: liver 0.30−1.00 mg/kg, kidney 0.70−2.00 mg/kg, and serum 0.140−0.250 µg/ml. 17 SEM = standard error of measurement; U = unknown; F = female; G = gelding; BADDL = Bronson Animal Disease Diagnostic Laboratory; CAHFS = California Animal Health and Food Safety Laboratory System; ND = not determined.
Consistent gross and microscopic findings.
The selenium results obtained by BADDL were replicated by CAHFS (Table 2). Following the determination of toxic selenium concentrations, it was reported that the affected animals had been given a compounded vitamin and mineral supplement at the origin premises just prior to transport to the event.
Further supporting evidence for the diagnosis of acute selenium toxicosis came from the results of additional toxicological analyses by CAHFS. No toxic organic compounds were detected in the stomach contents by GC-MS analysis; brain cholinesterase activities were within normal ranges. Feed samples and stomach contents were negative for monensin, lasalocid, salinomycin, and narasin. Trace elemental analysis of sera showed no significant abnormalities of iron, magnesium, sodium, copper, zinc, calcium, phosphorus, or potassium. The stomach contents did not contain toxic plant alkaloids. Drug screening of sera from 2 of 5 horses revealed lidocaine, methocarbamol, trimethoprim, prednisolone, dexamethasone, and ketoprofen. Methocarbamol, trimethoprim, prednisolone, and dexamethasone were detected in the 3rd horse. Serum from the 4th horse contained lidocaine, methocarbamol, trimethoprim, dexamethasone, and tripelennamine. Methocarbamol dexamethasone, detomidine, and phenylbutazone were detected in serum from the 5th horse. The urine of the 5th horse was also positive for lidocaine, methocarbamol, dexamethasone, detomidine, phenylbutazone, and furosemide. Drugs detected were all believed to have been administered by attending veterinarians following the onset of signs in an attempt to treat the animals. The exact drugs used were not disclosed. No significant findings of bacterial or viral infections were noted. Gross postmortem and microscopic histopathology findings were consistent with acute selenium toxicosis. 4,5,12
The deaths of all ponies within a few hours of receiving a “vitamin/mineral mix” injection, the consistent clinical signs, the histopathological findings, and the toxic selenium concentrations in tissues and sera strongly suggested that the horses died from an intravenous administration of an acutely toxic selenium preparation. The concentration of selenium in the “vitamin/mineral mix” was disclosed recently after original submission of the manuscript. In an attempt to replicate the formula for Biodyl, a vitamin mineral supplement by Merial, the compounding pharmacy incorrectly converted 100 mg to 10 g instead of 0.1 g, resulting in a 100 times higher concentration of selenium than in the Biodyl (1 mg/ml). According to Merial, the recommended dosage given intravenously to an adult horse is 20 ml. Considering the calculation error, each polo horse received 2 g of selenium as sodium selenite, compared to the intended dose of 20 mg selenium. A dose of 2 g selenium for an adult horse weighing 400–500 kg corresponds to 4–5 mg/kg body weight, a lethal dose for most animals. One study showed that the parenteral lethal dose of selenium for horses is 1.49 mg/kg body weight. 14 The clinical signs reported herein were similar to those described for many species including horses and human beings. 2,4,5,7,11,12,14,15 Other investigators have reported that intramuscular administration of 1–2 mg/kg selenium as sodium selenite to calves resulted in death in less than 12 hr following injection. 11 Intravenous administration of 1–2 mg/kg sodium selenite was fatal within 10 hr in swine, and 1 mg/kg was fatal in a steer in 8 hr. 4 Oral administrations of large doses of sodium selenite to horses, mules, cattle, and swine resulted in death within 6–72 hr. 12 The level of selenium detected in the horses tested, the rapid death following intravenous injection of the “vitamin/mineral mix,” and the admission of error in compounding by the entity that prepared the formulation confirms selenium toxicity to be the cause of death. Selenium has a very narrow margin of safety. In sheep, the onset of clinical signs of toxicosis is reported to be from 1 to 2 hr after intramuscular injection of selenium with a median lethal dose of 0.7 mg/kg, while at 0.6 mg/kg no mortality occurred, showing a very narrow margin between lethal and nonlethal doses. 2 While the actual toxic dose of selenium may vary widely between species and within the same species, the effects appear to be fairly similar. Studies suggest that selenium toxicity is dependent on several factors including the susceptibility of the animal, efficacy of elimination, amount of selenium administered, and interaction with dietary constituents. 11 Stress due to exercise, transportation, and other underlying conditions may play an important role in susceptibility of individual animals. 11
Serum selenium concentrations correlated well with the toxic selenium dosage administered and toxic tissue levels presented in the present report. Reports have shown a good correlation between blood selenium concentrations and toxic tissue levels following intramuscularly administered toxic doses of selenium in sheep and calves. 2,11 Combined with clinical findings, serum selenium determination is useful for diagnosis of acute or chronic selenium poisoning as well as determination of nontoxic concentrations. 15,21 Urinary selenium measured in the 1 polo pony that died in the clinic was high at 2.4 µg/ml and correlates well with the tissue and selenium levels. Urinary concentration of selenium could be a useful indicator of selenium toxicity. 21 Urinary excretion is known to play a major role in the elimination of selenium and the maintenance of selenium homeostasis particularly at excessive selenium concentrations. 5,13,15
The precise mechanism of selenium toxicity is not clearly understood. Several mechanisms have been proposed by many investigators. It has been proposed that selenium toxicity might be caused by the interaction of selenium with the disulfide and thiol groups of proteins thereby inhibiting enzyme activity. 10 Furthermore, it has been reported that selenium compounds interact with thiols and generate free radical species inducing DNA damage as well as interfere with integrity and/or function of DNA repair proteins. 10 Many investigators have speculated that toxicity of selenium is associated with the generation of reactive oxygen species, which may promote DNA oxidation and that GSH promotes these reactions. 9,10,18 Physiologic and toxic doses of selenium may be metabolized through the same metabolic pathway which includes reduction, methylation, and incorporation into selenoenzymes. 5 The metabolic pathway of selenium can generate superoxide, hydrogen peroxide, and oxyradicals. 5,10 At higher concentrations, overproduction of hydrogen selenide (H2Se) and other auto-oxidizing selenium metabolites may produce oxygen radicals and cause DNA strand breaks. 5,10,18
The acute selenium toxicosis in the polo horses presented in the current paper highlights the importance of using selenium supplements, in accordance with Food and Drug Administration (FDA) requirements, from reputable manufacturing companies that adhere to good manufacturing practices. While it may be advisable to establish a well-controlled regimen of selenium supplementation to prevent selenium deficiency and associated diseases, the present study also highlights the importance of monitoring blood concentrations of selenium before and after specific treatment.
The current study represents the first report of acute selenium toxicosis in horses due to a compounding error. The authors were fortunate to be able to obtain confirmatory and correlating indices that were supportive of the diagnosis based on the clinical signs exhibited, necropsy, and histopathological lesions, and the toxic levels of tissue and serum selenium. The present study also represents the first published report that establishes levels of acute tissue selenium intoxications in horses following parenteral injections. The documentation of the serum and tissue concentrations of selenium in these polo ponies may also be useful for future suspect cases of intoxication in horses. The potential for stricter monitoring and controls for compounding pharmacies by the FDA in Florida and elsewhere is anticipated.
Footnotes
Acknowledgements
The authors would like to acknowledge the Florida Department of Agriculture and Consumer Services, Division of Animal Industry; Victor Alzona and BADDL staff; and Drs. Hailu Kinde, Scott Stanley, Robert Poppenga, and the CAHFS staff for their technical support and invaluable assistance.
a.
Perkin Elmer Inc., Waltham, MA.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
