Abstract
Interstitial pneumonia and lymphocytic bronchiolitis with interstitial emphysema is an unusual reaction pattern in fetal and neonatal calves. These changes often are thought to suggest a chronic bacterial infection acquired in utero, and an associated placentitis is expected. Viral agents can also be implicated. Specific known pathogens that can induce a similar inflammatory response include Ureaplasma diversum, Mycoplasmopsis bovis, Brucella abortus, bovine viral diarrhea virus, and bovine parainfluenza virus 3. We describe a series of 15 cases of interstitial pneumonia and lymphocytic bronchiolitis with interstitial emphysema in fetal and early neonatal (up to 3-d-old) beef calves collected over 10 y in Alberta, Canada. Where reported, calves appeared small, weak, and occasionally were dyspneic. On autopsy, lungs appeared diffusely voluminous with interstitial emphysema, bulla formation, and rarely mediastinal emphysema and pneumothorax. A few calves had additional features of bacterial infection, such as pericarditis. Placenta was not received. Histologic lung findings in the affected calves included alveolar septa expanded by mononuclear cells, sparse neutrophils and macrophages within alveoli, variable lymphocytic and histiocytic peribronchiolar cuffing, prominent lymphoid aggregates surrounding bronchioles, occasional vascular necrosis, and subpleural and interlobular emphysema. PCR testing and immunohistochemistry for the previously noted pathogens were negative, and the cause of this unique condition, although presumably infectious, remains unknown.
Calf morbidity and mortality during gestation, at parturition, and during the early neonatal period has detrimental effects on the welfare of affected animals and on the productivity of the beef herd.10,14,21 Lesions in the lung are a common finding at autopsy of these calves.6,12,19,21,24 Challenges in utero, during parturition, and in neonatal life can damage the respiratory tract, leading to changes that may affect the viability of the calf at the time or in the future. 25 Findings in calves of this age include pulmonary hemorrhage as a result of dystocia or trauma by the dam, acute respiratory distress syndrome associated with prematurity or fetal distress during parturition, asphyxiation and meconium aspiration as a result of anoxia and stress at calving, aspiration pneumonia associated with oral esophageal tube feeding, and pneumonia caused by infectious agents acquired in utero, during parturition, or in neonatal life.4,12,16,19,22
The reaction pattern of interstitial pneumonia and lymphocytic bronchiolitis with interstitial emphysema is unusual in this age group. These changes are commonly thought to suggest a chronic bacterial infection acquired in utero, and an associated placentitis is expected.3,7 Viral agents can also be implicated. Attributed pathogens that can induce a similar inflammatory response include Ureaplasma diversum, Mycoplasmopsis bovis, Brucella abortus, bovine viral diarrhea virus (BVDV), and bovine parainfluenza virus 3 (BPIV3).3,6,8 A syndrome encompassing interstitial pneumonia and emphysema with bronchus-associated lymphoid tissue hyperplasia has been described in mid-gestation to 1–2-d-old calves examined in multiple states across the United States, in which extensive ancillary testing for infectious agents was conducted and no cause found. 18 We describe a similar syndrome affecting fetal and early neonatal beef calves in Western Canada.
Materials and methods
Clinical details
Fifteen aborted or early neonatal (up to 3-d-old) beef calves had been submitted from 5 cow–calf ranches in Alberta to the University of Calgary, Faculty of Veterinary Medicine, Diagnostic Services Unit (UCVM-DSU; Calgary, Alberta, Canada) for diagnostic investigations between April 2013 and December 2024 (
Clinical details of 15 aborted or neonatal calves received for examination.
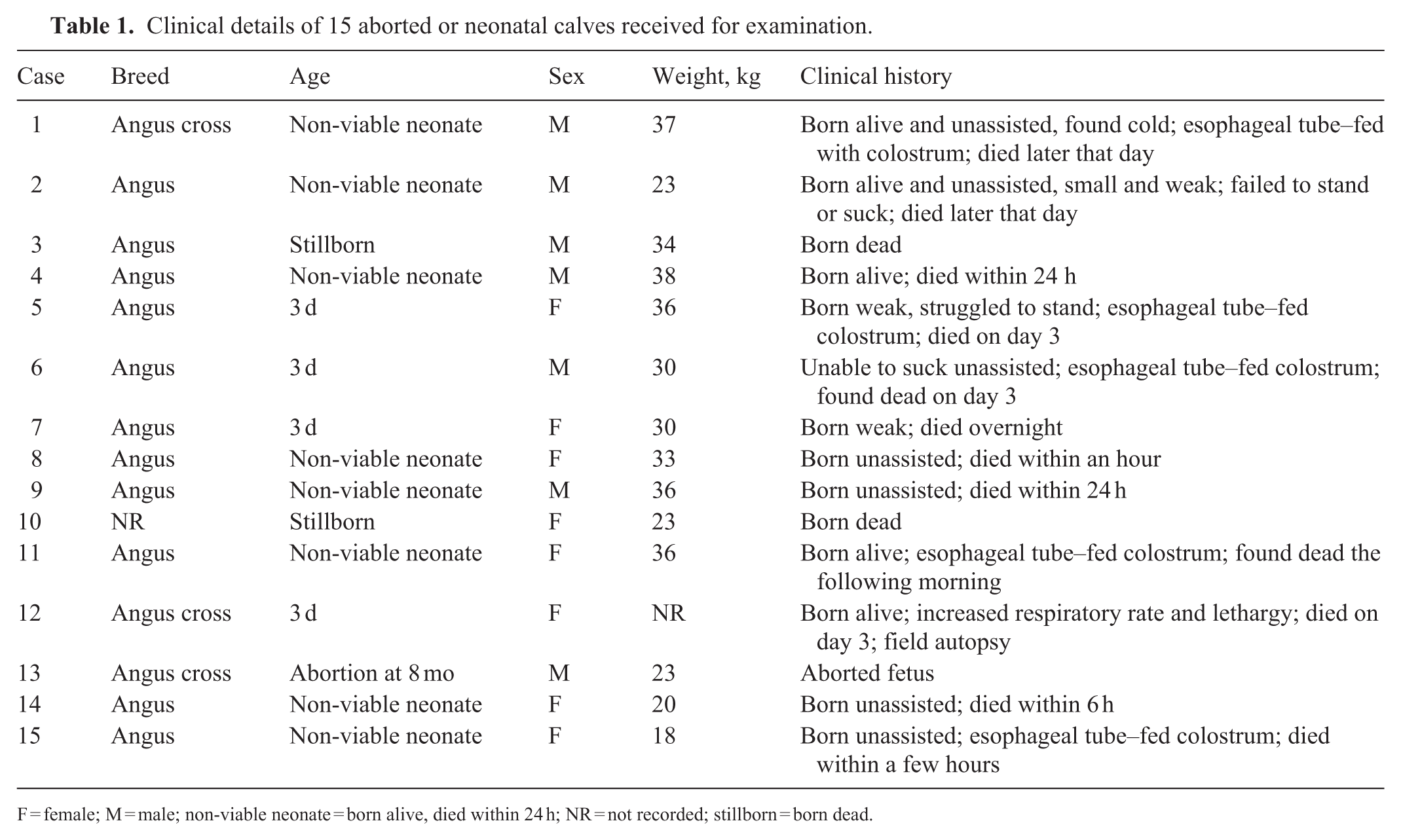
F = female; M = male; non-viable neonate = born alive, died within 24 h; NR = not recorded; stillborn = born dead.
Pathology
Fourteen autopsies were conducted by UCVM-DSU veterinary pathologists, and one was conducted in the field by the clinic veterinarian. Fresh tissues, including fetal fluid and fetal stomach contents (fetal and stillborn calves only), ear notch, lung, liver, kidney, spleen, and brain, were collected for bacteriology and virology. Tissue samples, including eyelid, tongue, parotid salivary gland, thyroid gland, thymus, lung, heart, diaphragm, liver, kidney, adrenal gland, urinary bladder, spleen, ileum, colon, lymph node, skeletal muscle, brain, pituitary gland, and umbilical artery, were fixed in 10% neutral-buffered formalin and processed for histopathology, special histochemical stains, and immunohistochemistry. Bacteriology was performed at the UCVM-DSU; samples were submitted to Prairie Diagnostic Services (PDS; Saskatoon, SK, Canada) for molecular testing.
Bacteriology
From 9 cases, fresh samples of abomasal fluid, lung, kidney, liver, and/or spleen were processed by the UCVM-DSU bacteriology laboratory for culture and susceptibility testing. In brief, tissue samples were seared with a heated spatula using a glass bead sterilizer (VWR). A sterile scalpel blade was used to incise the seared area and scrape tissue from the cut surface for direct inoculation. All samples were plated onto Columbia blood agar (Oxoid) and incubated aerobically at 35°C with the addition of 5% CO2, and onto MacConkey agar (Oxoid) incubated at 35°C in ambient air. Additionally, lung samples were plated onto chocolate enriched agar (Oxoid) and incubated under the same conditions as the blood agar. All inoculated plates were incubated for a minimum of 72 h and checked daily at 18–24 h intervals for growth.
Bacterial identification was performed (MALDI Biotyper Sirius benchtop spectrometer; Bruker). MBT Compass software (Bruker) was used to match individual bacterial spectra against the MBT Compass reference library database. Bacterial identification criteria were as follows: scores of 2.00–3.00 = high-confidence species identification; 1.70–1.99 = low-confidence species identification; 0.00–1.69 = no organism identification.
Molecular analysis
Fresh lung samples submitted to PDS were tested by real-time PCR (rtPCR) to detect bovine coronavirus (BCoV), bovine alphaherpesvirus 1 (BoAHV1), BPIV3, bovine respiratory syncytial virus (BRSV), BVDV, influenza D virus (IDV), M. bovis, and U. diversum. Nucleic acid extracts were amplified (AgPath-ID one-step RT-PCR kit; Applied Biosystems, ThermoFisher) and read (CFX96 rtPCR detection system; Bio-Rad).
Each 25-μL reaction contained 5 μL of nucleic acid extract and 20 μL of a mix of 2× AgPath-ID one-step RT-PCR master mix, 25× primer and probe mix (specific to each target pathogen; cellular 18S rRNA as an internal control), and RNase/DNase-free water (ThermoFisher). The CFX Maestro software was used to analyze the amplification data. Ct values were recorded for each target, and positive and negative controls were included in each run to ensure assay validity.
Detection and preliminary identification of Mycoplasma and Campylobacter species were performed using conventional PCR assays (C1000 Touch thermal cycler; Bio-Rad). Any amplified PCR products were submitted to Macrogen (South Korea) for Sanger sequencing. The resulting sequences were analyzed using BLASTn to determine species identity by comparing to known NCBI GenBank sequences.
Immunohistochemistry
Formalin-fixed, paraffin-embedded tissues were submitted to PDS for BVDV and M. bovis immunohistochemistry (IHC). BVDV IHC staining was performed on an automated staining platform (Agilent). Epitope retrieval consisted of applying proteinase K (Agilent) for 5 min, followed by a 30-min incubation of the primary mouse monoclonal antibody (Mouse anti-BVD clone 15c5; Idexx) at a dilution of 1:1,000. Binding of the primary antibody was detected using horse anti-mouse immunoglobulins (Vector) and an avidin-biotin immunoperoxidase complex reagent (Vector), with 3,3′-diaminobenzidine tetrahydrochloride (DAB; Agilent) as the chromogen.
M. bovis immunohistochemical staining was conducted on an automated staining platform (Autostainer Plus; Agilent). Epitope retrieval consisted of applying proteinase K (Autostainer Plus) for 10 min, followed by a 30-min incubation of the primary mouse monoclonal antibody (Mouse anti–Mycoplasma bovis; H. Ball, VSD, N. Ireland) at a dilution of 1:800. Binding of the primary antibody was detected using horse anti-mouse immunoglobulins (Vector) and an avidin-biotin immunoperoxidase complex reagent (Vector), with DAB (Dako liquid DAB+ substrate chromogen system; Agilent) as the chromogen.
Results
Gross pathology
On autopsy, the lungs were variably voluminous or atelectatic having failed or partially failed to collapse (Fig. 1); negative thoracic pressure was not found in case 1, suggesting pneumothorax (Suppl. Table 1). Eleven of the 15 cases had marked interlobular and subpleural emphysema (Fig. 1A, 1B); 7 of 15 cases had large bullae in the caudal lung tissue (Fig. 1C), expanding the mediastinum (Fig. 1D), and extending across the surface of the pericardial sac (Fig. 1A). Case 7 had fibrinous pericarditis and fibrinous arthritis (Suppl. Table 1). Other organs in all calves appeared relatively unremarkable.
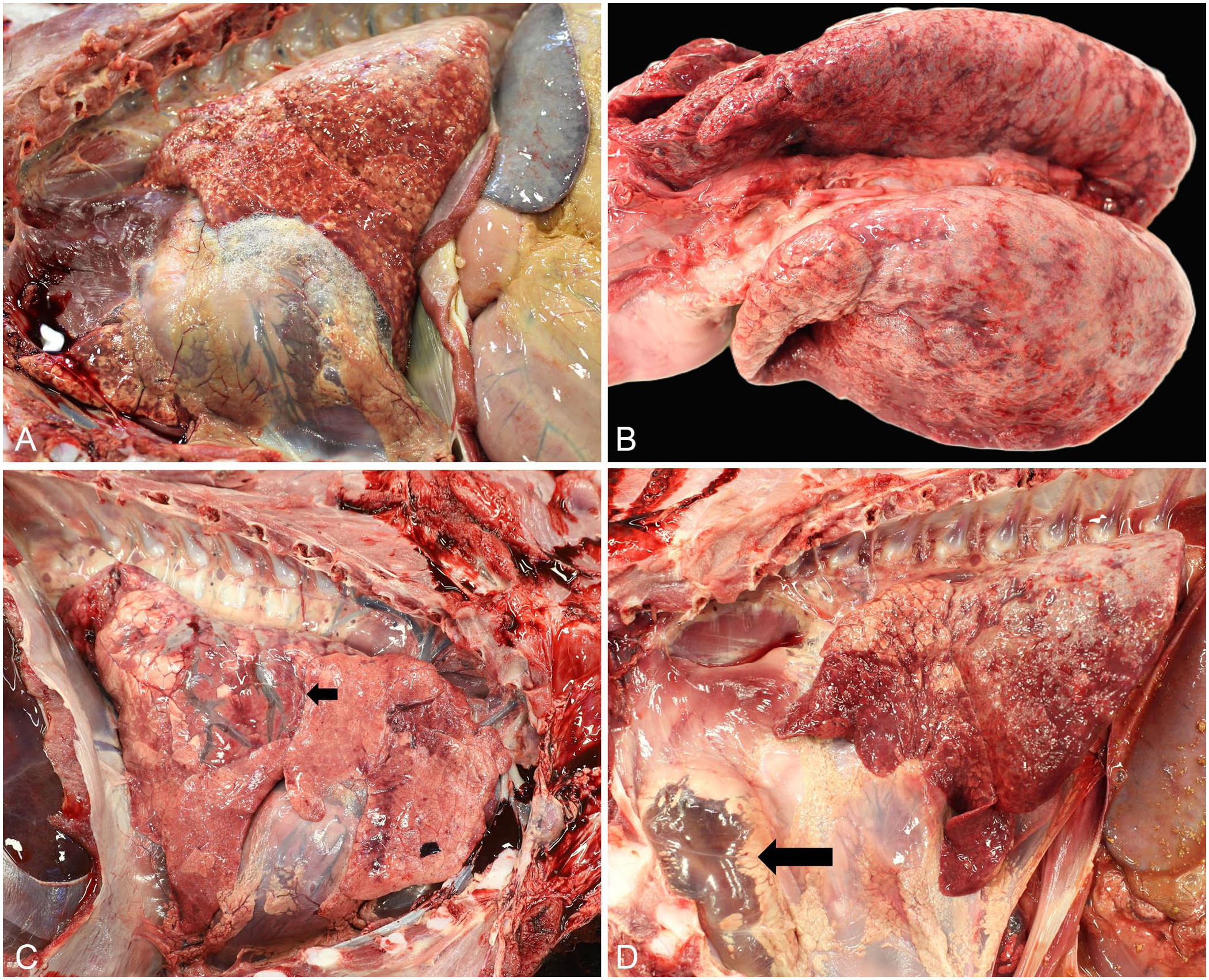
Gross pathology of interstitial pneumonia and emphysema in fetal and neonatal calves.
Histopathology
The predominant histopathologic changes in the lungs of the 15 affected calves included expansion of alveolar septa by mononuclear cells (n = 7), a few neutrophils and macrophages within alveoli with scant fibrin (8), multinucleate giant cells within alveoli (4), mononuclear peri-bronchiolar cuffing with variable lymphoid aggregates (15), and marked subpleural and interlobular emphysema (11; Suppl. Table 1,
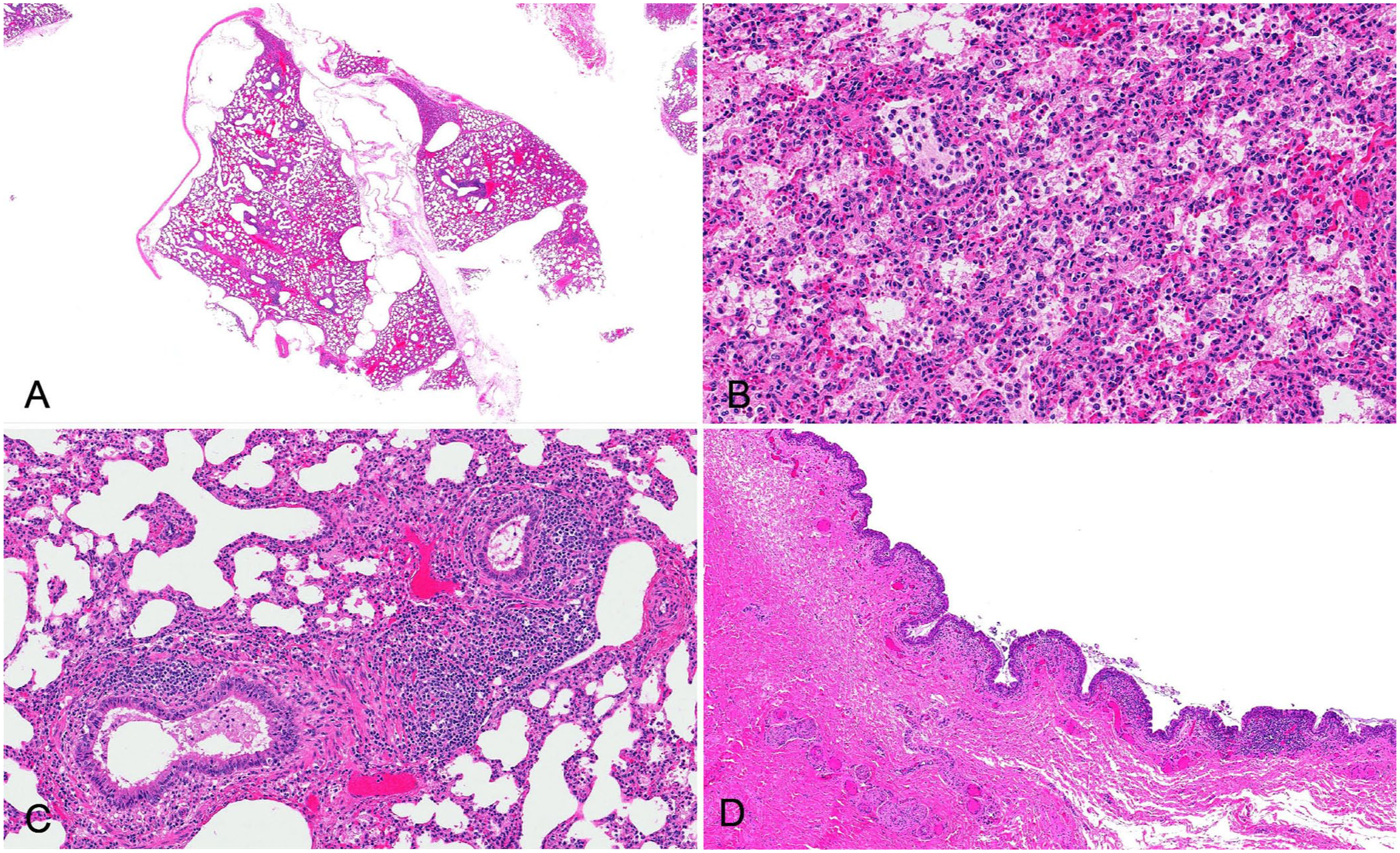
Histopathology of interstitial pneumonia and emphysema in fetal and neonatal calves. H&E.
Immunohistochemistry
In case 2, IHC for BVDV was negative (1 case tested). IHC for M. bovis was negative in 9 cases (Suppl. Table 2).
Bacteriology
Mixed growths of low numbers of bacteria, often including Escherichia coli, were found most often on bacteriology, suggesting postmortem overgrowth or contamination. Cases 5 and 6 both had E. coli cultured from 3 different organs. A light pure growth of Bacillus licheniformis was found on culture of the lung from case 12.
Molecular testing
Nine calves tested negative for U. diversum; 6 calves tested negative for BoAHV1, BRSV, and BVDV; and 5 tested negative for BPIV3, BCoV, and IDV on PCR testing (Suppl. Table 2). One calf tested negative on PCR for Neospora caninum and Leptospira spp. Another calf was negative for Coxiella burnetii on PCR testing. One case was positive for Mycoplasma spp. on PCR, but the species was not identified; 2 cases were negative. Four calves were negative for M. bovis on PCR. Case 7 was positive for Campylobacter upsaliensis and C. vulpis.
Discussion
Our case series is of a unique syndrome of interstitial pneumonia and lymphocytic bronchiolitis with interstitial emphysema of unknown cause in fetal and early neonatal (up to 3-d-old) beef calves in Western Canada. The lymphocytic bronchiolitis and mononuclear interstitial infiltrate were advanced given the age of the calves, suggesting that the lung lesions developed in utero. The fibrin exudation and vascular changes appear to be more acute, and it is difficult to determine if they are part of the same process, part of a secondary terminal septicemia, or potentially a result of neonatal pulmonary hypertension. 23 The extensive emphysema makes this syndrome a distinct entity on gross examination and is likely the result of previous lung damage, which led to increased respiratory effort as the calf started to breathe. As in other reports,18,23 we did not identify a specific infectious cause.
The age of calves affected ranged from fetuses aborted at 7–8-mo gestation, up to calves 3-d-old, 1 d older than the cases reported by others. 18 Cases in calves >2-d-old could be accurate or, given the extensive systems of cow–calf ranches in Alberta, the timings of calving and discovery of the carcasses could have falsely extended the age affected. All calves were of the Angus breed. Although a genetic association cannot be entirely excluded, Angus is the most popular beef breed in Canada and the reaction pattern also makes this less likely. 2 Some calves were small at birth, described as weak, a few were dyspneic, and many failed to suck and required tubing with colostrum. These findings were consistent with those of others. 18 Many calves were described as born unassisted; however, it is not known to what degree parturition was uneventful or if any difficulties occurred.
The gross pathology was often striking in our cases, with extensive interstitial emphysema creating grossly obvious crepitus over the surface of the lung. Although emphysematous change because of autolysis can appear similar, all but one of our cases were in a good state of postmortem preservation, and autolysis was not considered to be a contributing factor. Cattle have well-developed interlobular septa that separate the lung tissue into lobules, resulting in a lack of collateral ventilation. 11 High intra-alveolar pressure caused by increased respiratory effort (which was observed clinically in one calf) then results in interstitial emphysema. 5 The pneumothorax seen in one case was likely the result of rupture of an emphysematous bulla. 19
Causes of interstitial pneumonia in older calves and adult cattle include viral insults (e.g., BRSV, BPI3V), pneumotoxins, migrating parasites, and immune responses (including that associated with septicemia). 9 BRSV infection can also result in the development of interstitial emphysema. 17 Although terminal sepsis may have contributed to the interstitial lesions in a number of our cases, it was not possible to consistently identify a cause in all cases. Prominent peri-bronchiolar lymphoid tissue is not present in healthy perinatal calves; hence, this infiltrate suggests antigenic stimulation, which is seen in aborted bovine fetuses associated with U. diversum, BVDV, BPIV3, M. bovis, and chronic bacterial infections; an accompanying placentitis can be expected.8,15 These pathogens were not consistently found in our cases, although one case was positive for Mycoplasma spp. on PCR. Multinucleate cells were occasionally found within alveoli, which has been associated with perinatal M. bovis, BPIV3, and Tritrichomonas foetus infections,8,20 but these pathogens could not be implicated in our cases. Mixed growth was the most common bacteriology finding, suggesting postmortem contamination or overgrowth; 2 calves had E. coli cultured from 3 organs, which does raise the possibility of a primary or secondary colisepticemia. One calf was positive for Campylobacter spp., and this animal also had pericarditis, which was a likely sequela. Campylobacter abortions tend to be acute processes. The presence of lymphocytic bronchiolitis and interstitial emphysema in this case suggests a concurrent underlying and more chronic insult. 13 B. licheniformis was found on lung culture of another calf, but the lack of placenta for examination made it difficult to assess the significance of this finding, and a concomitant infection remains possible. 1
Others 23 have described a premature calf that died after a few days, with diffuse interstitial pneumonia and peri-airway lymphoid proliferation. However, similar to our case series, no infectious agents were found, and mild ongoing antigenic stimulation was considered a possibility. The possibility that an infectious agent caused the lesions in our series remains feasible, as the insult could have occurred historically, with the agent cleared by the time of testing. The ancillary testing conducted also may not have been sufficiently extensive, or a novel agent could be involved. It would seem logical to assume the lesions described could be associated with placentitis, and a substantial limiting factor in the investigation of each calf was the lack of placenta submitted to the laboratory. For the older calves, this is logical, but for the fetal and stillborn calves it reflects the reality of sample availability and submission in extensive beef cattle management systems. Other limitations of our investigation include incomplete sets of molecular testing, reflecting the reality of diagnostic investigations in production animals. Although PCR testing was done for BoAHV1, panherpesviral testing was not performed, and the involvement of another herpesviruses, such as bovine gammaherpesvirus 6, is possible. 6 Complete evaluation of the clinical presentation of these calves, including the details of parturition and viability of the calves immediately after birth, were unknown because of the extensive management systems. Maternal health and disease status also were unknown.
We suggest a case definition of an interstitial pneumonia syndrome, with mononuclear infiltrate expanding alveolar septa, lymphocytic bronchiolitis, and interstitial (interlobular and subpleural) emphysema in fetal or newborn calves up to 3-d-old. A novel pathogen remains a possible cause, and future research might use metagenomic sequencing. Our case series describes observations of a syndrome not described previously in the peer-reviewed literature, to our knowledge. We retrieved no cases of interstitial pneumonia and lymphocytic bronchiolitis with interstitial emphysema in fetal and neonatal beef calves in a search of Google, PubMed, and Web of Science using the search terms “bovine AND beef AND neonate AND interstitial AND pneumonia AND lymphocytic bronchiolitis AND interstitial emphysema”. Our study offers a resource for diagnosticians who may see similar cases, and highlights opportunities for future research.
Supplemental Material
sj-pdf-1-vdi-10.1177_10406387251410557 – Supplemental material for Interstitial pneumonia and lymphocytic bronchiolitis with interstitial emphysema of unknown cause in Western Canadian fetal and neonatal beef calves
Supplemental material, sj-pdf-1-vdi-10.1177_10406387251410557 for Interstitial pneumonia and lymphocytic bronchiolitis with interstitial emphysema of unknown cause in Western Canadian fetal and neonatal beef calves by Dayna Goldsmith, Katie Waine, Carolyn Legge, Erin Zachar, Beverly Morrison, Maria Bravo Araya, Vanessa Boone, Lindsay Rogers and Jennifer L. Davies in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank the producers and veterinarians for submitting these cases to the laboratory. Appreciation goes to the UCVM-DSU necropsy and histology technicians, administrative and auxiliary staff for their involvement in processing these tissues, and Melencio Nicolas for photography. We also thank the laboratory team at Prairie Diagnostic Services for molecular testing and immunohistochemistry.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Our investigations were funded in part by the producers, with extensive financial support from the UCVM Veterinary Outbreak Investigation Service (VOIS).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
