Abstract
Blood, tissue and oral fluid samples collected from experimentally infected animals and field cases were used to evaluate the safety, diagnostic sensitivity and specificity of Flinders Technology Associates (FTA) cards for Porcine reproductive and respiratory syndrome virus (PRRSV) reverse transcription polymerase chain reaction (RT-PCR) diagnostics. The analytical sensitivity of PRRSV RT-PCR from serum and oral fluids in FTA cards was reduced, although the virus could still be detected at concentrations of 101 and 103 TCID/ml, respectively. The sensitivity and specificity of PRRSV RT-PCR detection from serum, blood, and tissue samples in cards collected from experimentally infected animals were 100%. Sensitivity for oral fluids was 45% (95% CI: 19.97–73.01) compared to fresh. For field samples, sensitivity was 89% (95% CI: 77.35–95.63) and 100% (95% CI: 80.00–100) for serum and lung samples, respectively. The sensitivity was the same for samples stored in cards at room temperature or at 4ºC, and tested overnight or after 14 days. Cards inoculated with PRRSV-positive samples did not yield replicating virus after cell culture. In conclusion, FTA cards proved to be a safe, simple, and sensitive alternative method to transport serum, blood, and tissue samples for PRRSV RT-PCR diagnostics; however, a significant decrease in RT-PCR sensitivity should be expected from oral fluid samples.
Keywords
Porcine reproductive and respiratory syndrome virus (PRRSV) is one of the most important diseases affecting pigs worldwide. 1 Due to the devastating impact of PRRSV on swine production, 9 veterinarians require rapid, convenient, sensitive, and reliable diagnostic methods to properly identify and monitor the disease and prevent its spread to other farms. Flinders Technology Associates (FTA) cards offer a convenient alternative for the transport of infectious samples for molecular diagnostics. FTA cards contain a mix of chemicals that lyse cells and inactivate bacteria and viruses while preserving nucleic acids (Burgoyne LA, Kijas J, Hallsworth P, Turner J: 1994, Safe collection, storage and analysis of DNA from blood. Proceedings of the 5th International Symposium on Human Identification, Oct. 8–11. Abstract). 2 Samples embedded in FTA cards are suitable for molecular diagnostics but not for culture and isolation of infectious agents. Because samples are rendered noninfectious, the user is protected from any potential biohazards, and samples can be shipped in standard letter mail envelopes at room temperature. FTA cards can facilitate collection of samples in remote locations with the subsequent access to state-of-the-art diagnostic facilities, even those located in different countries.
FTA cards have been used for the transport, testing, and storage of infectious samples containing pathogens affecting human beings, plants, and animals, although testing sensitivity may be decreased. Among viruses affecting animals that have been used with FTA cards, there are reports of Rabies virus, Newcastle disease virus, Foot-and-mouth disease virus (FMDV), Swine influenza virus, and PRRSV.5,7,8,10,11 In general, most studies reported either slight decreased analytical sensitivity of reverse transcription polymerase chain reaction (RT-PCR) for RNA viruses spiked in vitro on FTA cards,5,8,10 or no difference in sensitivity in samples from experimentally infected animals.8,10 However, most of these studies used limited numbers of samples and validation had limited testing using samples from naturally infected animals. There is a need to conduct studies using a larger number of samples representing broader scenarios including field settings. In addition, there is limited information on whether sensitivity may change over time or with different storage temperatures.
A prior study showed that PRRSV could be detected in blood samples embedded in FTA cards 5 ; however, to the authors’ knowledge, no information is available on the sensitivity and specificity of PCR-based methods to detect PRRSV in tissue, serum, or oral fluids preserved on FTA cards or on how these cards may perform under field conditions. The overall objective of the current study was to assess whether FTA cards can be used as a reliable method for the submission and testing of swine samples for routine molecular PRRSV diagnostics. The specific objectives were to 1) evaluate the in vitro analytical sensitivity, stability, and safety of PRRSV-positive fluids embedded on FTA cards, 2) evaluate the sensitivity and specificity of PRRSV RT-PCR for tissues and fluids embedded on FTA cards from experimentally and naturally infected animals, and 3) evaluate collection and testing conditions with regard to storage time and temperature.
In order to determine the sensitivity and specificity in samples collected from experimentally infected animals, twelve 3-week-old pigs purchased from a PRRSV-negative farm were confirmed PRRSV negative by serology a and RT-PCR. b Upon arrival, pigs were individually identified and randomly housed in 2 separate rooms at the University of Minnesota animal research unit. Pig housing, care, and management procedures had been previously approved by the University of Minnesota Institutional Animal Care and Use Committee. Pigs were divided into infected (n = 8) and control (n = 4) groups. Pigs in the infected group were inoculated intramuscularly with 2 ml/pig of PRRSV isolate MN-184 at 10 5 TCID50/ml. Pigs in the control group received an injection of 2 ml of saline solution. Pigs were monitored daily for clinical signs of disease and sampled on a weekly basis. All pigs were blood sampled prior to inoculation and at 7, 14, 21, and 28 days postinfection (DPI). Additionally, oral fluids were collected from pigs in both groups (infected and control) 3 times per week according to procedures described previously. 12 Confirmation of PRRSV infection was carried out at 4 days postinoculation by serum RT-PCR. Samples from known positive (infected) and known negative (control) pigs were used to assess the collection, handling, shipping, and laboratory processing of samples using FTA cards. At each collection day, 1 control pig and 2 inoculated pigs were euthanized, necropsied, and sampled. At necropsy, blood and tissue (lung, tonsil, and superficial inguinal lymph nodes) samples were taken. One hundred µl of the fluid samples were transferred to the FTA cards c shortly after collection. A fragment of each tissue sample was transferred to the FTA card in the form of directly imprinting 1 cm2 of the tissue in the card, or by swabbing the interior of the tissue and then rubbing the swab d onto the card. Each collected sample was transferred to FTA cards in quadruplicates to be stored and processed after collection as follows: 4ºC/24 hr, 4ºC/14 days, 25ºC/24 hr, and 25ºC/ 14 days. In addition, serum, oral fluids, and tissue samples were also tested directly in the “fresh” form by RT-PCR.
In order to determine the sensitivity and specificity in field specimens, 75 blood serum samples and 62 lung samples from pigs submitted to the University of Minnesota Veterinary Diagnostic Laboratory (MN-VDL; St. Paul, Minnesota) were selected. Serum and lung samples were inoculated into FTA cards as described above and tested for PRRSV by RT-PCR. Fresh samples were also tested for comparison. Upon arrival at MN-VDL, all samples were kept at 4ºC until they were tested. Samples were processed within 24 hr of arrival.
Two different punching protocols were used to obtain the FTA card discs for the RNA elution. The first protocol produced 3 discs, each obtained with the 2-mm Harris punch e as recommended by the manufacturer of the elution solution. f The total punched surface was equivalent to 2.4% of the FTA card sampling area. The second protocol produced one 6-mm disc, equivalent to 7.6% of the FTA card sampling area, which was obtained with an office paper punch. g The Harris punch was disinfected using 99% ethanol solution, dried using disposable wipes, and used to punch 2 blank cards before punching each sample. The office punchers were disinfected using 1:100 disinfectant h in a 10-min bath and autoclaved for 10 min at 200ºC. The office punchers were also compared to a 6-mm Harris punch. To validate the disinfection protocol and to rule out the possibility of cross contamination between samples, 1 negative control (card embedded with 100 μl of saline solution) was punched after every fifth PRRSV-positive control sample for a total of 10 negative controls. Once the disinfection protocol was validated, 1 negative control was punched after every tenth test sample.
RNA from the FTA card was eluted using RNA rapid extraction solution f as recommended by the manufacturer. RNA extraction, RT-PCR, and quantitative (q)PCR were performed at the MN-VDL following routine protocols. Briefly, after the elution step, RNA was further purified and concentrated using high throughput magnetic bead–based technology. f The RT-PCR protocol used was a real-time, TaqMan-based multiplex protocol that detects both North American and European strains of PRRSV and that was developed and standardized at the MN-VDL. 3 PRRSV qPCR plate setup was performed using a commercial RT-PCR kit. i The qPCR reaction was conducted using a real-time PCR system. j The products obtained from the RNA extraction step of 4 selected FTA serum samples from experimentally infected animals (7, 14, 21, and 28 dpi) and 1 FTA field lung sample were submitted to the University of Minnesota Biomedical Genomics Center for PRRSV open reading frame-5 sequencing (utilizing Sanger sequencing through a 96 capillary DNA analyzer k ).
In order to determine analytical sensitivity (limit of detection), eight 10-fold dilutions of a pure culture of PRRSV strain MN-184 containing 10 5 TCID50/ml were spiked into PRRSV-negative serum, oral fluid, and porcine alveolar macrophage (PAM) cells cultured in Dulbecco modified Eagle medium (DMEM) supplemented with 10% fetal calf serum (FBS) and antibiotic cocktail. One hundred twenty-five microliters of each dilution from each of the 3 samples (pure culture, serum, and oral fluids) was inoculated onto FTA cards, while another aliquot was saved and stored at 4ºC for direct testing. FTA cards were stored overnight at room temperature. RNA extraction and RT-PCR was performed for all samples as described above. Duplicates of the FTA cards inoculated with dilutions of PRRSV-positive culture media, serum, and oral fluids were left at room temperature for 14 days. These samples were then processed and tested by RT-PCR. The safety of samples embedded in FTA cards was determined by inoculating 4 FTA cards with 100 μl of a pure PRRSV culture of isolate MN-184 at 10 5 TCID50/ml and storing them at room temperature for overnight testing. The cards were then resuspended in viral culture DMEM media supplemented with 10% FBS and tested for viral growth on PAM and MARC (cloned African green monkey kidney cell line)-145 cell lines.
The results were statistically analyzed using a paired t-test to compare differences in threshold cycle (Ct) values between fresh and FTA card samples. The kappa coefficient was used to estimate the diagnostic agreement between RT-PCR results from samples tested in fresh or embedded in FTA cards. Kappa coefficients were considered “excellent” when above 0.75, “good” when from 0.4 to 0.75, and “poor” when below 0.4. 4
Results from the current study showed reduction in the in vitro analytical sensitivity of PRRSV detection by RT-PCR from serum and culture media in FTA cards. Analytical sensitivity was reduced 100 times compared to detecting the virus directly from serum or culture media. However, viral RNA in FTA cards could still be detected at concentrations of 10 1 TCID50. There was no loss of sensitivity in testing the FTA cards at 24 hr compared to 14 days of storage. The analytical sensitivity of oral fluids embedded on FTA cards and stored for 14 days was also reduced approximately 100 times compared to testing the spiked samples directly. However, a 10,000-fold reduction was observed in oral fluid samples stored in FTA cards for 24 hr. No virus growth was observed after 5 days in MARC-145 or PAM cell cultures from cards that had been inoculated with virus for 24 hr.
Results from the experimental setting indicated that serum, blood, oral fluids, and tissues collected from the control group (noninfected animals) were PRRSV RT-PCR negative at all time points when tested as fresh or after the 4 FTA card treatments. Therefore, the specificity for RT-PCR from FTA cards was 100%. All serum, blood, and tissue samples collected from the PRRSV-infected group were PRRSV RT-PCR positive at all time points resulting in a sensitivity of 100% (95% confidence interval [CI]: 73.78–100). Sensitivity for cards tested with storage at 4ºC or 25ºC, or at 24 hr or 14 days postinoculation was the same and also estimated at 100% (95% CI: 73.78–100). Sensitivity of PRRSV RT-PCR was lower for oral fluid samples embedded in FTA cards compared to fresh samples (Table 1), and overall sensitivity was estimated at 45.45% (95% CI: 19.97–73.01). Differences in sensitivity increased as the infection progressed in time. Sensitivity was 71.43% (95% CI: 35.23–93.53) between 1 and 16 DPI, but decreased to zero between 19 DPI until the termination of the study at 26 DPI.
Reverse transcription polymerase chain reaction results from oral fluids collected from experimentally infected pigs.*
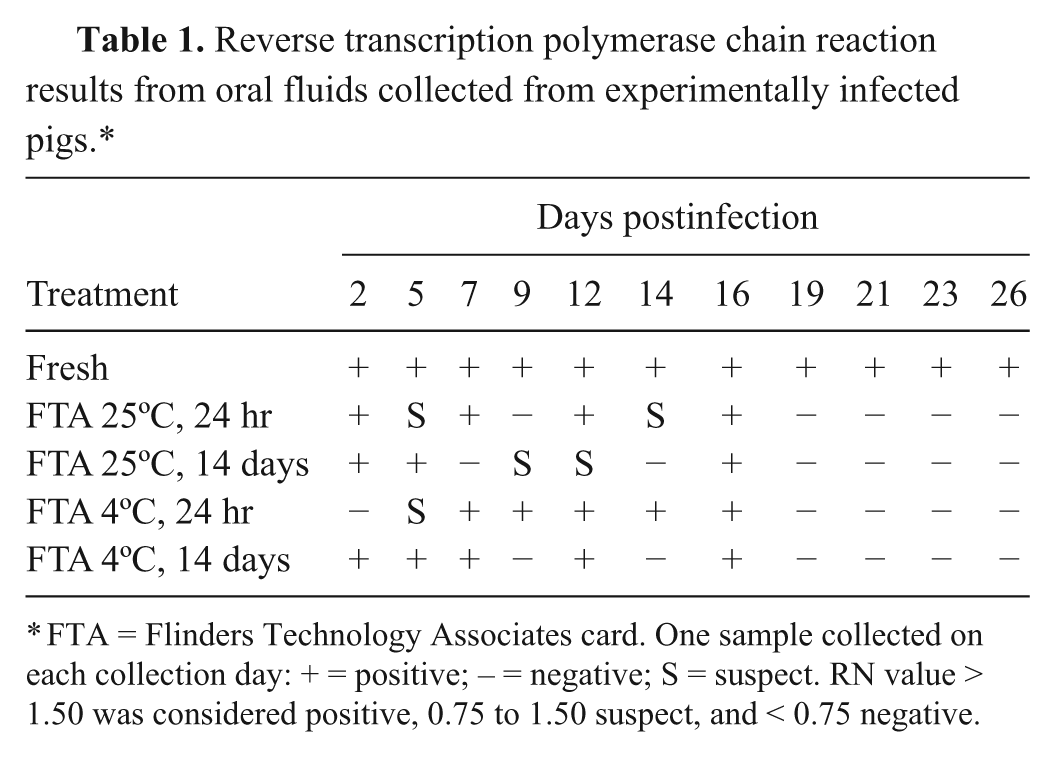
FTA = Flinders Technology Associates card. One sample collected on each collection day: + = positive; – = negative; S = suspect. RN value > 1.50 was considered positive, 0.75 to 1.50 suspect, and < 0.75 negative.
The RT-PCR results from field samples are shown in Table 2. Sensitivity of detecting PRRSV from FTA cards compared to detecting PRRSV directly from the fresh serum and lung samples was 88.89% (95% CI: 77.35–95.63) and 100%, respectively. The specificity for both field serum and lung samples was 100% compared to the fresh results. The overall diagnostic agreement between testing the field samples directly or from the FTA cards was 93.24% (95% CI: 85.83–97.38) and 100% (95% CI: 80.00–100) for serum and lung samples, respectively; the kappa coefficient of agreement was 0.86 (95% CI: 0.78–0.94) and 1.00 (95% CI: 0.96–1.0), respectively.
Results from number of field sera and lung samples tested directly, or in Flinders Technology Associates (FTA) cards, by Porcine reproductive and respiratory syndrome virus quantitative polymerase chain reaction.
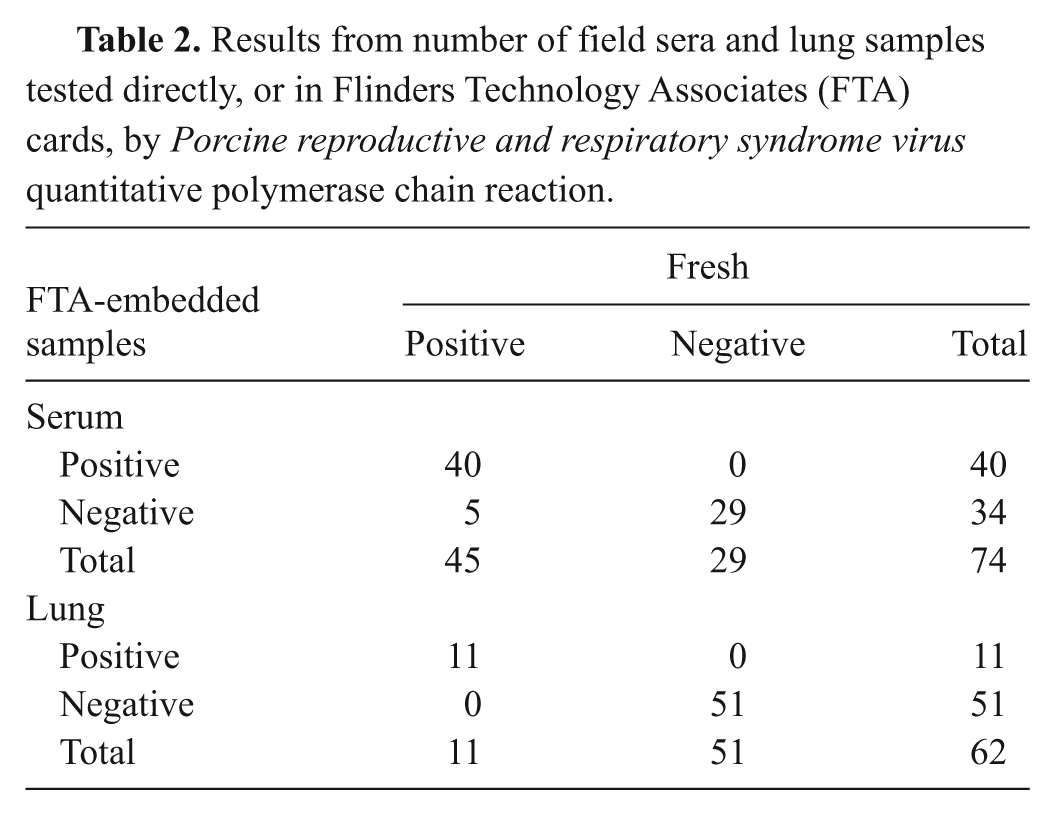
The mean Ct values for the PRRSV qPCR direct testing of field serum samples was 27.38 with a standard error (SE) of 0.95. The mean Ct values of FTA-embedded samples was 32.90 with SE of 0.84. The Ct values for FTA samples were significantly higher than were their paired fresh samples (p < 0.001). Therefore, the sensitivity of fresh samples for PRRSV qPCR was significantly better than that of FTA card–embedded samples. Regarding the field lung samples, mean Ct values for the PRRSV qPCR for the fresh and FTA samples were 26.08 (SE: 2.49) and 25.16 (SE: 2.26), respectively, and differences were not statistically significant (p = 0.47). Therefore, sensitivity of PRRSV qPCR for lung field samples was similar when samples were tested fresh or after embedded onto FTA cards. The PRRSV sequencing was successful from the only lung sample attempted but not from 8 serum samples attempted. The sequence obtained from the FTA card was compared to the sequence obtained from the corresponding fresh sample and the percentage of identity was 100%.
Comparison of the punching protocols resulted in an analytical sensitivity of 1 TCID50/ml log better when using the single 6-mm disc compared to the three 2-mm discs (results not shown). There was no difference in the analytical sensitivity comparing the 6-mm Harris punch to the 6-mm office punch. However, the protocol with Harris punch yielded more cross-contamination results than the protocol with the office punch (results not shown). To overcome the cross-contamination, an additional step of disinfecting the puncher using 99% ethanol solution followed by drying using disposable wipes before punching the 2 blank cards was added. After this additional step, the cross-contamination issue was resolved, although this protocol was complex to implement in the laboratory. Overall, the 6-mm office puncher was preferred to the 6-mm Harris puncher.
The present study showed that analytical sensitivity for RT-PCR from FTA cards decreased approximately 100 times compared to testing the samples directly. A decrease in the analytical sensitivity of this magnitude has been described in previous studies.5,8,10 This decrease can be explained in part by the differences in the amount of sample used in the PCR reaction between testing the sample directly or when using the FTA card published protocols. While 100 µl of sample was used when conducting the PCR directly from the fresh sample, only a portion of the FTA card (a 6-mm punch) equivalent to 5.7% of the sample was used in the PCR reaction. For oral fluids, the equivalent of 3.3% was used for the FTA card punch. Further evaluation is needed to determine the efficiency of the PCR reactions when using multiple punched discs.
There was no difference in PRRSV RT-PCR sensitivity between 24 hr or 14 days of storage, which is similar to results published previously.13,14 A decrease in sensitivity using cards was not observed in serum, blood, or tissues collected from experimentally infected animals, but sensitivity was slightly lower when field serum samples were used (100% [95% CI: 73.8–100] vs. 88.9% [95% CI: 77.4–95.6]). Differences in sensitivity between experimental and field samples can be explained by the disease status at the time of sample collection. Experimentally infected pigs were acutely infected at the time of sample collection with significant viral loads distributed throughout the tissues. In contrast, stage of infection was unknown for the field samples selected for the study. Other factors such as immune status, age, presence of coinfections, and differences among PRRSV strains may have also played a role. In the present study, a single highly virulent, highly transmissible PRRSV strain was used in the experimental infection, which may have caused an overestimation of the sensitivity. Strain variability should have been captured while using the field samples, which may explain the slightly lower sensitivity.
Field samples tested as fresh that had qPCR Ct values near the cutoff value resulted in qPCR-negative results when the samples were transferred to FTA. In contrast, a drop in sensitivity was not observed when field lung samples were tested. A previous study reported extended detection of PRRSV RNA in lungs from experimentally infected pigs even when the viremic phase of infection had been completed. 6 It is reasonable to speculate that the likelihood of detecting PRRSV RNA in lungs is higher than that of serum, which could result in the lungs being a more suitable sample for FTA card testing. In fact, PRRSV open reading frame-5 was successfully sequenced from a field lung sample impregnated onto FTA cards, but it could not be sequenced when the cards were impregnated with serum.
A decrease in sensitivity was also observed in oral fluids in FTA cards. It is concluded that sensitivity from oral fluids in FTA cards was very low and unacceptable to replace direct testing and that overall storage conditions did not have a significant effect on PCR sensitivity. Therefore, additional research is needed to improve the performance of oral fluids in FTA cards. Further research is also needed to evaluate FTA cards for transporting and testing semen samples. Semen was not included in the present study because semen submissions for PRRSV testing at the MN-VDL is limited to only 2% of all samples received, compared to 50% serum, 24% blood swabs, 15% oral fluids, and 9% tissue samples (Rovira, 2011, unpublished observations), indicating the limited use of semen for PRRSV monitoring in North America.
In summary, FTA cards proved to be a safe, simple, and sensitive alternate method to transport serum and tissues of infected animals for PRRSV molecular diagnostics. Use of FTA cards for oral fluids warrants further investigation and is not recommended at this time.
Footnotes
Acknowledgements
The authors would like to acknowledge Michele Confeld and Tracy Seda from the University of Minnesota Veterinary Diagnostic Laboratory for their technical assistance and laboratory work, and Dr. Enrique Mondaca from Boehringer Ingelheim Vetmedica for the insightful discussions.
a.
HerdChek® PRRS 2XR Antibody ELISA, IDEXX Laboratories, Westbrook, ME.
b.
TaqMan RT-PCR kit, Applied Biosystems, Foster City, CA.
c.
FTA® Indicating Classic cards (catalog no. WB 120206), Whatman International Ltd., Maidstone, United Kingdom.
d.
BBL Culture swab (catalog number 220099), BD, Franklin Lakes, NJ.
e.
Catalog no. WB 100040, Whatman International Ltd., Maidstone, United Kingdom.
f.
Ambion®, Applied Biosystems, Foster City, CA.
g.
803 CR¼” round economy punch, McGill Inc., Marengo, IL.
h.
Synergize, Preserve International, Reno, NV.
i.
One-step RT-PCR kit (catalog no. 104485), Qiagen Inc., Valencia, CA.
j.
7500 Real-Time PCR System, Applied Biosystems, Foster City, CA.
k.
ABI 3730xl, ABI BigDye Terminator version 3.1, Applied Biosystems, Foster City, CA.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The National Pork Board funded this study through grant number 09-220.
