Abstract
Equine sarcoids are the most common tumor of horses. Bovine papillomavirus (BPV) has been suggested as the cause of sarcoids. Studies have shown that BPV is present in swabs or biopsies from nonsarcoid-bearing equine skin. Skin biopsies from a variety of different conditions and normal skin from horses with no reported history of sarcoids were examined by polymerase chain reaction (PCR) for the presence of BPV, which was found in all different types of skin conditions as well as normal skin. Forty-one out of 86 skin biopsies from horses without sarcoids were found to contain BPV DNA. Laser microdissection, followed by DNA amplification through both PCR and isothermal loop-mediated amplification, was performed on these 41 biopsies and on 70 additional BPV-positive sarcoid biopsies to localize the virus. Location of BPV DNA was different between sarcoid and nonsarcoid groups. Nonsarcoid skin biopsies were more likely to have BPV within intact or inflamed epidermis than sarcoids (P = 0.016 and P = 0.007, respectively). Areas of inflammation within the dermis and epidermis were more likely to contain BPV than in noninflamed areas (P = 0.008 and P = 0.009, respectively). Bovine papillomavirus was also found in the epidermis of all types of sarcoids examined, more frequently in occult sarcoids than in fibroblastic and nodular types (P = 0.03 and P = 0.01, respectively). Results suggest that BPV is commonly found in normal and inflamed equine skin, and it is likely an important predisposing factor in the development of sarcoids.
Introduction
Bovine papillomavirus (BPV) has been associated with the development of the most common skin tumor of horses, the equine sarcoid. 11 Such association has been made based on the detection of BPV DNA in equine sarcoids, and its absence in other equine skin tumors. 19 Given this association, BPV has been suggested as the cause of equine sarcoids. However, some studies have found BPV DNA in the skin of a small number of horses with nonsarcoid-related inflammatory skin conditions, but the significance of such a finding is unknown.2,12,29
The frequency with which BPV can be detected in nonsarcoid skin conditions has not been determined in a larger sample of equine tissues. In addition, it is unclear where BPV is located histologically in these biopsies from unaffected horses. If the same BPV can be commonly found in nonsarcoid skin conditions of horses, and it is present in the same histologic locations in these conditions as in equine sarcoids, then the hypothesis that BPV causes equine sarcoids may be called into question.
Previous studies have utilized polymerase chain reaction (PCR) to amplify the BPV DNA present in samples to allow for its identification, but this technique does not identify where within the sample BPV is located.3,9,17,22,23,25 Studies have demonstrated the location of BPV in sarcoids using in situ hybridization,14,25 but while this technique allows for the visualization of BPV, it does not amplify the BPV genome to any significant degree when compared to PCR and is, therefore, a much less sensitive technique for identification of DNA when it is present in small quantities. 17 Laser microdissection (LM) allows for the very precise excision of a cell or cells from a sample of interest. The excised cell(s) become the template for DNA amplification allowing an accurate determination of where in a tissue sample the DNA of interest is located.
Numerous techniques exist to amplify DNA, including PCR, which has frequently been used in the study of equine sarcoids. More recently, another technique, isothermal loop-mediated amplification (LAMP), has been developed,20,21 and has not only been purported to be as sensitive as PCR for the detection of DNA, but is potentially faster and less expensive.26,27
Historically, BPV DNA had only been found within the neoplastic fibroblasts of equine sarcoids and not within the epidermis. However, a 2010 study 6 examined a small number of sarcoids using LM and found BPV DNA in the keratinocytes of the epidermis and that its presence varied depending on the type of sarcoid examined. While only 1 clinical type of sarcoid was identified and examined, the authors of the 2010 study suggested that BPV DNA location might change as sarcoids progress through different clinical types and that further research was required to see if results are repeatable and applicable to different clinical types of sarcoids.
The current study consisted of 3 parts. Part A was conducted to determine the frequency with which BPV DNA could be detected in nonsarcoid skin conditions of horses and whether it was the same type and has the same partial gene sequence as that found in sarcoids. Part B used BPV-positive sarcoid biopsies and BPV-positive nonsarcoid skin condition biopsies from part A and utilized LM in combination with PCR and LAMP to determine if differences existed in the location of BPV in BPV-positive sarcoids and nonsarcoid skin conditions of horses. In addition, the viability of LAMP as a DNA amplification technique in formalin-fixed, paraffin-embedded (FFPE) equine tissue dissected by LM was examined. The information gleaned from parts A and B helped clarify the role of BPV in the development of equine sarcoids. In part C of the study, the location of BPV DNA in a variety of different clinical types of sarcoids was determined to discover if BPV location varies between types and between more and less clinically aggressive types.
Materials and methods
Detection of Bovine papillomavirus in nonsarcoid conditions of equine skin
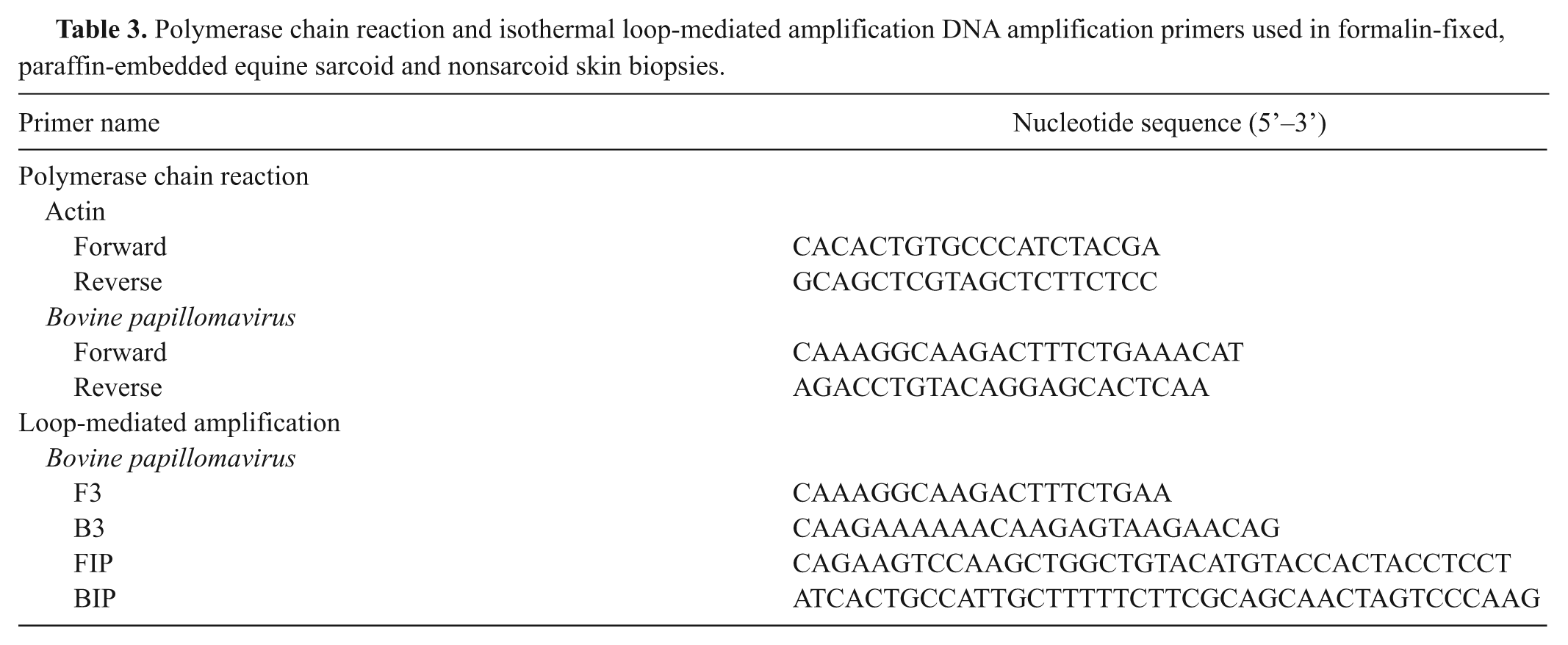
Surgical equine skin biopsies of non-neoplastic conditions diagnosed during the 12-year period between January 1, 1986, and December 31, 2007, were identified by using computer-based record searches at the Department of Veterinary Pathology at the University of Saskatchewan and Prairie Diagnostic Services in Regina and Saskatoon, Saskatchewan, Canada. From this series of collected records, samples of inflammatory skin conditions of a variety of different types were selected. The conditions were chosen based on their frequency of occurrence or clinical similarity to equine sarcoids (Table 1). Normal skin samples were selected from horses with no reported history of sarcoids and that had morphologically and histologically normal skin biopsies taken as control samples in cases of nonsarcoid inflammatory skin conditions. In total, the 86 most recent cases were chosen (Table 2). From these cases, the FFPE tissue blocks from these samples were utilized for PCR assay, as previously described. Briefly, 100-µm- thick sections were cut from each FFPE block using a microtome, which was cleaned using acetone between samples, and blank paraffin blocks were cut every 5 samples for use as negative controls. The single primer set that amplifies a 244–base pair (bp) sequence spanning portions of the E2 gene, the intergenic spacer region, and a small portion of the E5 gene of BPV type 1 (BPV-1), or a similarly located 248-bp sequence of BPV-2, was utilized (Table 3). To confirm that the amplified product was BPV, the 17 most strongly positive PCR samples were nucleotide sequenced.
Nonsarcoid equine skin condition categories assessed for the presence of Bovine papillomavirus (BPV), brief descriptions, and rationale for their use.

The number and percentage of equine skin biopsies that were polymerase chain reaction positive for Bovine papillomavirus (BPV) DNA grouped by inflammatory skin condition type as well as normal skin.*
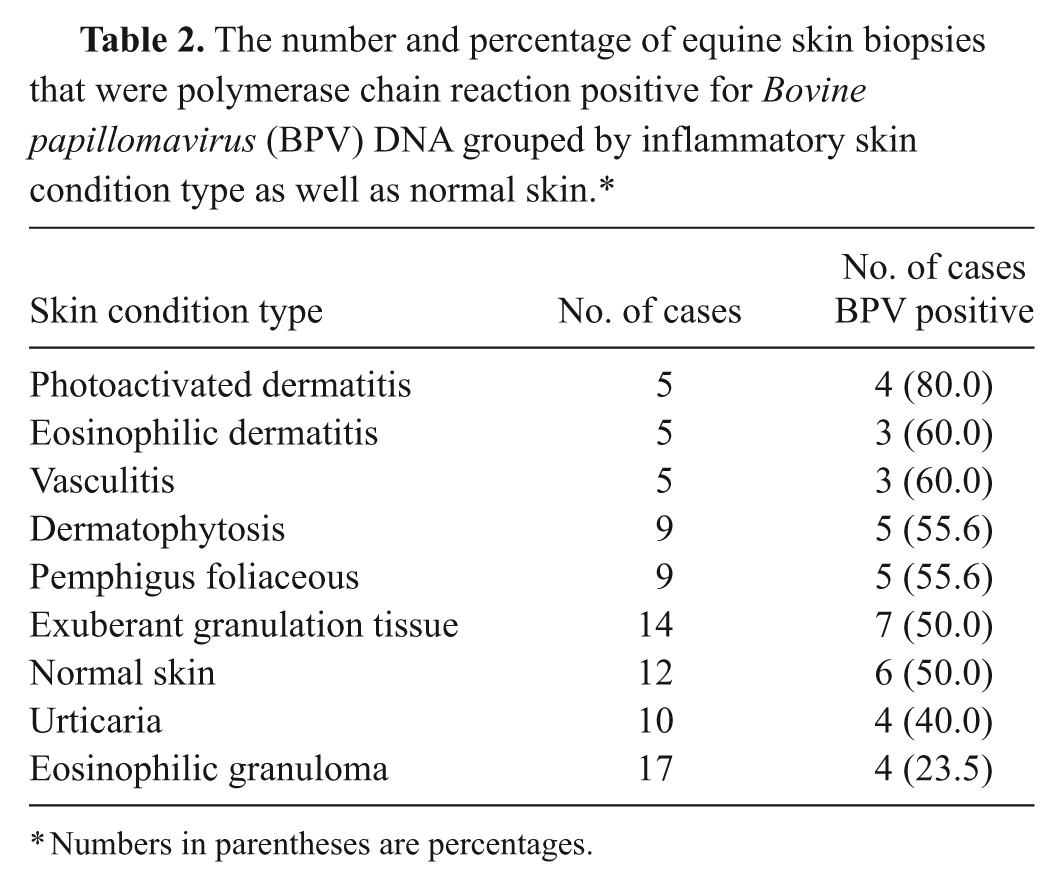
Numbers in parentheses are percentages.
Polymerase chain reaction and isothermal loop-mediated amplification DNA amplification primers used in formalin-fixed, paraffin-embedded equine sarcoid and nonsarcoid skin biopsies.
Laser microdissection, polymerase chain reaction, and isothermal loop-mediated amplification of Bovine papillomavirus–positive equine sarcoids and nonsarcoid equine skin conditions
The 41 tissue biopsies from part A that were positive by PCR for BPV DNA and all BPV-positive equine sarcoid biopsies with sufficient remaining tissue (70 samples) collected for a previous study 28 were used for the current study. Sections 5-µm thick were cut from the FFPE blocks, affixed to specialized membrane-covered glass slides designed for use with LM, a and stained with hematoxylin and eosin. No coverslips were affixed.
Using a laser microscope dissection system, b each slide was examined under 200× magnification by a Diplomate of the American College of Veterinary Pathologists (B.K. Wobeser). When present on the slide, portions consisting of various tissue compartments (Table 4) were excised with the laser and catapulted into individual 500-µl microcapillary tubes. c A minimum of 5 × 106 µm3 of tissue was collected into each tube and followed by DNA extraction, as previously described. 25 The extracted and precipitated DNA was resuspended in 10 µl of Tris–ethylenediamine tetra-acetic acid buffer.
Tissue compartment descriptions for use in laser microdissection for the detection of Bovine papillomavirus in sarcoid and nonsarcoid skin samples.

To confirm that amplifiable DNA was present in each sample following extraction, PCR using primers that amplify a 247-bp sequence of the equine actin gene was utilized (Table 3). The reaction mixture was as follows: 5 mM potassium chloride and 1 mM Tris–hydrogen chloride, 3 mM magnesium chloride, 200 µM of each deoxyribonucleotide triphosphate (dNTP), 1 µM of each primer, 1.25 units of Taq DNA polymerase, 3 µl of template DNA, and sufficient water for a final reaction volume of 50 µl. Amplification conditions were 94ºC for 3 min, followed by 40 cycles of 94ºC for 30 sec, 59ºC for 60 sec, and 72ºC for 60 sec, followed by a final extension period of 72ºC for 10 min. Confirmation that the PCR product was equine actin was conducted by nucleotide sequencing.
After confirmation that amplifiable DNA was present in the sample, PCR for BPV DNA using the same method used in part A was performed on each sample with the exception that the number of cycles was increased from 40 to 45 to account for the smaller sample volume acquired by LM. In addition to PCR, LAMP was also performed on each sample. Positive and negative controls for both PCR and LAMP included sequenced, BPV-positive bovine papilloma, tissue-free sections of slide membrane to act as negative controls, and standard no-template controls. The LAMP primer set that was used amplifies a portion of the same sequence of the E2 and E5 genes used for PCR (Table 3). Using Primer Explorer V4 d and the 244-bp sequence of E2 and E5 that the BPV PCR amplifies, 4 sets of primers were designed. All were optimized using a cloned BPV E2 and E5 amplicon originally derived from an equine sarcoid. The primer set used in the current experiment was chosen from the 4 designed sets based on consistent and strong amplification. The LAMP reaction solution was composed of 2 mM Tris–hydrogen chloride, 1 mM ammonium sulfate, 1 mM potassium chloride, 0.2 mM magnesium sulfate, 0.1% Triton X-100 (pH 8.8), 2 mM magnesium chloride, 200 µM of each dNTP, 1.6 µM of FIP and BIP primers, 0.2 µM of F3 and B3 primers, 32 U of Bst DNA polymerase large fragment, 3 µl of template DNA, and sufficient water for a final reaction volume of 50 µl. The reaction was heated at 65ºC for 60 min. Following isothermal amplification, positive products were identified using electrophoresis on a 2% agarose gel stained with ethidium bromide and visualized under ultraviolet light.
Presence of Bovine papillomavirus DNA in the keratinocytes of different clinical types of sarcoids
The clinical types of sarcoids used in part B of the current study had been previously identified. 28 Sarcoids from 4 different clinical types (occult, verrucous, nodular, and fibroblastic) were considered in this portion of the study. A fifth clinical type of sarcoid (mixed) used in part B was not utilized for this portion of the study as mixed sarcoids exhibit features from 1 or more of the other 4 sarcoid types and are, therefore, not a homogenous clinical type. The BPV DNA positivity in the epidermal compartments from each of the 4 different clinical types of sarcoids was compared.
Statistical analysis
Comparisons of the recovery rates of BPV DNA between sarcoid and nonsarcoid groups were made using Fisher exact tests. Comparisons of recovery rates of BPV DNA between matched samples of sarcoid or nonsarcoid groups were made using McNemar test. Relative risk of BPV positivity in the epidermis among different clinical types of sarcoids was made based on comparison with the clinical types with the lowest risk of BPV positivity. The comparison of agreement between LAMP and PCR was made using an unweighted kappa test. P-values < 0.05 were considered significant.
Results
Presence of Bovine papillomavirus in nonsarcoid conditions of equine skin
A total of 86 cases of nonsarcoid, non-neoplastic skin conditions including histologically normal skin from horses with no history of sarcoids were examined. Results are summarized in Table 2. Positive BPV DNA samples were found in all conditions examined including normal skin. Overall, 41 out of 86 (47.6%) samples were positive for BPV DNA. Such a finding is significantly lower than the proportion of sarcoid samples found to contain BPV DNA by the same methodology (P = 0.0001). No significant difference in the proportion of positive samples was present among any skin conditions or normal skin. All negative controls including blank paraffin blocks cut during the microtome cutting procedure were negative.
A total of 17 of the 41 positive samples were sequenced and compared to sequences in GenBank using the Basic Local Alignment Search Tool (BLAST). 1 Of the 17 samples sequenced, 8 were identical to BPV-1 obtained from equine sarcoids in western Canada (GenBank accession no. FJ895875) and 9 of the 17 had 100% identity to BPV-2 found in sarcoids in western Canada (GenBank FJ895874).
Laser microdissection, polymerase chain reaction, and isothermal loop-mediated amplification of Bovine papillomavirus–positive equine sarcoids and nonsarcoid equine skin conditions
Equine actin PCR was positive in all 334 tissue compartments, which indicates that amplifiable DNA was present in all samples. The PCR, LAMP, or both assays detected BPV in 157 out of 334 tissue compartments (47%). Results are summarized in Table 5. The PCR assay detected BPV DNA in more samples than did LAMP, detecting BPV DNA in 155 compartments, including 19 not detected by LAMP. Meanwhile, the LAMP assay detected BPV DNA in 138 compartments including 2 not detected by PCR. Overall, results from PCR and LAMP agreed on 313 out of 334 samples (93.7%), which corresponds to an unweighted kappa score of 0.87 (95% confidence interval: 0.76–0.98) and is considered “almost perfect” agreement. 15 Given this finding, tissue compartments from samples that were positive by either PCR or LAMP were considered positive for BPV DNA for the other parts of the current study.
Comparison of number of samples positive and negative for the presence of Bovine papillomavirus (BPV) DNA using 2 different amplification techniques, polymerase chain reaction (PCR) and isothermal loop-mediated amplification (LAMP), in 334 laser microdissected tissue compartments of equine skin.

Bovine papillomavirus DNA was found in 47% of tissue compartments in both sarcoid and nonsarcoid samples (Table 6). Not all tissue compartments were present in any given sample. For example, in normal skin, areas of inflamed epidermis or dermis did not exist. Similarly, some samples of sarcoid no longer contained areas of morphologically neoplastic fibroblasts. Such areas were likely contained within sections of the FFPE block that were consumed during other testing. In sarcoid samples, BPV DNA was most commonly detected within areas of morphologically neoplastic fibroblasts in the dermis. The BPV DNA was detected within 57 of the 65 samples where such tissue compartments existed. Other areas of the superficial and deep dermis that were morphologically normal also contained BPV DNA (4/14 and 10/21, respectively; Table 7). Overall, in sarcoid samples, BPV DNA was significantly more likely to be recovered from morphologically neoplastic fibroblasts than from other tissues on the same histological section (P = 0.0005; Table 8).
Sarcoids and nonsarcoid equine skin samples dissected into tissue compartments using laser microdissection and examined for the presence of Bovine papillomavirus (BPV) DNA using polymerase chain reaction and isothermal loop-mediated amplification.

Numbers in parentheses are percentages.
Number and percentage of sarcoid and nonsarcoid equine skin samples dissected into tissue compartments using laser microdissection and examined for the presence of Bovine papillomavirus (BPV) DNA using polymerase chain reaction and isothermal loop-mediated amplification.
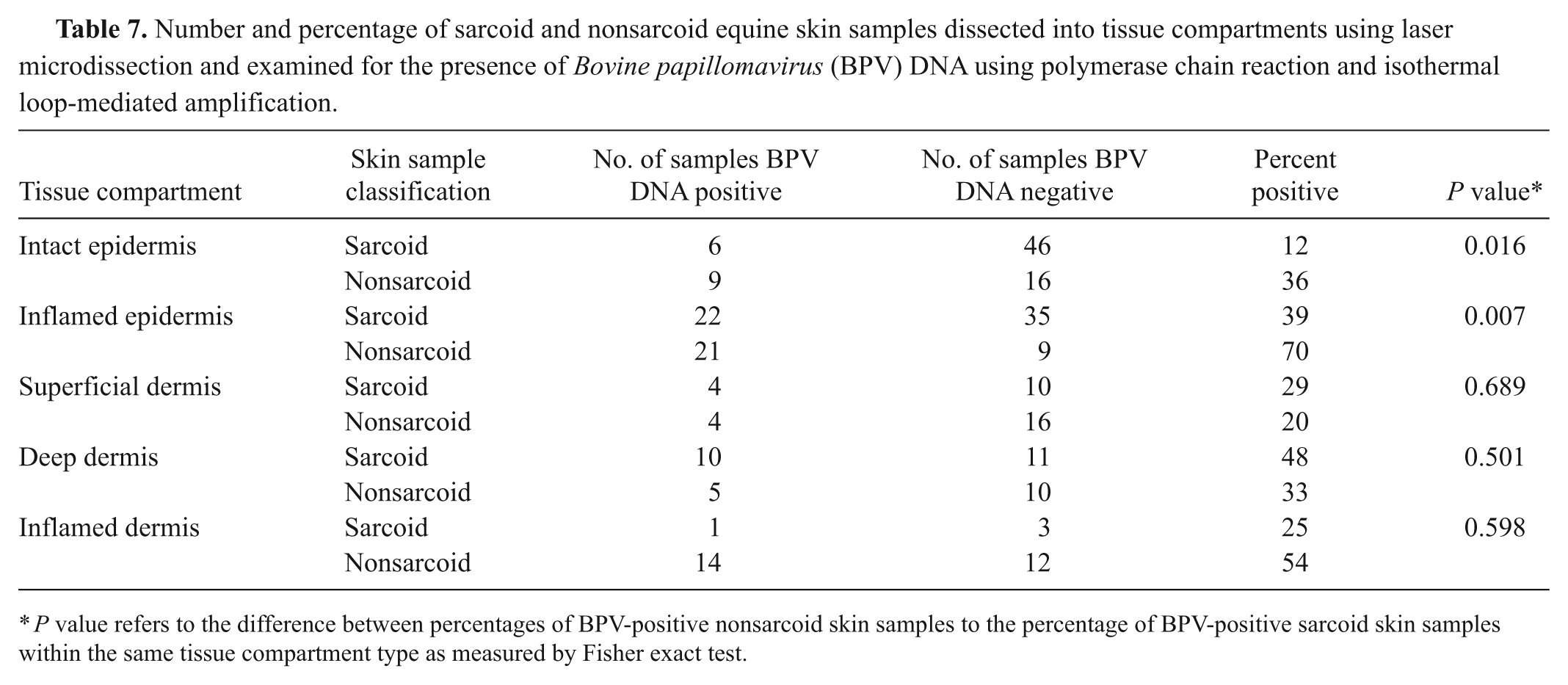
P value refers to the difference between percentages of BPV-positive nonsarcoid skin samples to the percentage of BPV-positive sarcoid skin samples within the same tissue compartment type as measured by Fisher exact test.
Comparison of Bovine papillomavirus (BPV) DNA detection rates from laser microdissected equine skin samples where both compared tissue compartments were present within the same histologic sample.

P value refers to the association of presence of BPV DNA from matched equine skin samples within the compared compartment types as measured by McNemar test.
Nonsarcoid samples were more likely to have BPV DNA in intact or inflamed epithelium than were sarcoid samples (P = 0.016 and P = 0.007 respectively; Table 7). Sarcoid samples were more likely to have BPV DNA in noninflamed dermis than nonsarcoid samples, but the differences were not significant. Nonsarcoid samples were more likely to contain BPV in inflamed dermis and epidermis as compared to normal dermis and epidermis (P = 0.008 and P = 0.009, respectively; Table 8).
Presence of Bovine papillomavirus DNA within the epidermis of different clinical types of sarcoids
A total of 49 sarcoids of 4 clinical types contained sufficient epidermis for analysis. When subdivided based on clinical type into occult, verrucous, nodular, and fibroblastic, BPV DNA was present within the epidermis of all sarcoid types (Table 9). There was a significantly higher chance of BPV DNA being present in the epidermis of occult sarcoids compared to fibroblastic or nodular sarcoids. There were no significant differences between the detection rate of BPV between verrucous sarcoids and occult ones.
Presence of Bovine papillomavirus (BPV) within the epidermis of laser microdissected equine sarcoids of different clinical types and the relative risk of BPV positivity based on comparison to BPV positivity in occult sarcoids.

Significantly higher relative risk of being BPV positive.
Discussion
Bovine papillomavirus DNA was present in all categories of skin lesions examined, as well as in normal skin (Table 2). The overall rate of BPV DNA–positive cases was surprisingly high. In a study by the current authors examining the epidemiology of equine sarcoids in western Canada, identical methods to determine the presence of BPV DNA in sarcoid biopsies was used. 28 In that study, 74 out of 96 sarcoid samples were positive for BPV DNA. While the recovery rate of BPV DNA in the current study was lower than that found in sarcoids (P = 0.0001), BPV DNA was found with considerable frequency.
Two types of BPV are commonly associated with equine sarcoids, BPV-1 and BPV-2. Sequencing of the BPV recovered from these nonsarcoid skin samples confirmed that one or the other of these types of BPV was present in the samples described herein and that their partial gene sequences were identical to those found in equine sarcoids from the same geographic region. A 2008 study on the presence of BPV DNA in sarcoids and the skin of nonsarcoid-bearing horses found that the proportion with BPV-2 positivity was higher in nonsarcoid-bearing horses than in sarcoid-bearing horses suggesting that BPV-2 may be a more easily cleared infection than BPV-1. 7 However, in the current study, BPV-1 and BPV-2 DNA were recovered in roughly equal proportions in nonsarcoid-bearing horses. Such result was the opposite of that found in the 2008 study 7 in that the level of BPV-1 recovery was actually higher than the recovery rate from sarcoid-bearing horses in western Canada. The observation suggests that there may be no difference in the duration of infection between BPV-1 and BPV-2.
To enhance the sensitivity of detection, researchers have used other sample collection methodologies in combination with PCR for DNA amplification, including skin swabbing and scraping techniques to determine the presence of BPV on the surface of sarcoid-bearing and nonsarcoid-bearing horses. In one such study, BPV DNA was detected in most of the sarcoid samples, but none of the nonsarcoid control samples, 18 which is in contrast to the frequency with which some studies, including the current one, were able to find BPV DNA in nonsarcoid equine skin samples. 4 In a 2005 study, 4 BPV DNA was detected on the skin surface of both nonsarcoid- and sarcoid-bearing horses. Perhaps, it is not simply the presence of BPV DNA alone that is important in sarcoid development; but also its location within the skin.
In a previous study, the use of swabbing, cleaning, and biopsy of equine sarcoids, followed by PCR on the collected samples, was attempted to determine where within the lesion BPV was located. 7 The results were somewhat difficult to interpret as the methodology lacks the ability to precisely determine what is being sampled. For example, in 1 horse, BPV DNA was not present on a swab of the uncleaned skin surface, but was present after cleaning the skin surface. To avoid this problem, LM was used in the current study. The advantages of LM are the ability to choose specific areas or individual cells from a histologic slide of a lesion, and the ability to excise and capture these target areas without contamination from surrounding areas. The LM scope uses an ultraviolet laser to excise the cells of interest and then uses a diffuse blast of laser light to propel the excised cells into a collection chamber so that DNA contamination from surrounding cells or instruments does not occur. Very recently, a study has used LM on equine sarcoids following the same reasoning. 6
Using this methodology, in the current study, it was found that there were significant differences in the anatomic location from which BPV can be recovered from nonsarcoid and sarcoid skin samples (Table 7). Nonsarcoid skin samples were more likely to have BPV DNA on or in the epidermis, than were sarcoid samples.
In addition, the presence of BPV DNA was significantly associated with the presence of inflammation. The BPV DNA was more likely to be found in inflamed epidermis or dermis than in noninflamed areas on the same slide (Table 8). Skin trauma has been suggested as an important mechanism to allow BPV access to the dermal fibroblasts and to provide an environment conducive to the proliferation of BPV transformed cells. 7 The current results suggest that macroscopic trauma is not required and that inflammation alone may be sufficient to allow BPV access to the dermal fibroblasts. This may explain sarcoid development in locations on horses with no previous history of injury at that site. In addition, inflamed areas may represent potential sites for transmission of BPV from one horse to another by either contact or potentially via fomites. In support of this theory is a report that the same BPV DNA found within equine sarcoids could also be found in face flies (Musca autumnalis) in the horses’ surroundings (Kemp-Symond JG: 2000, The detection and sequencing of bovine papillomavirus type 1 and 2 DNA from Musca autumnalis face flies infesting sarcoid-affected horses. MSc thesis, Royal Veterinary College, London, UK).
A 2008 study demonstrated the presence of BPV DNA in circulating peripheral blood mononuclear cells of sarcoid-bearing horses. 8 This suggested a possible alternate explanation for the presence of BPV DNA in inflamed areas. Rather than the environment being the source of BPV DNA infection at these sites, circulating blood mononuclear cells within the horse are the sources and are concentrated at these sites of inflammation. However, although areas of inflammation were more likely to have BPV DNA within them, BPV DNA was also found in tissue compartments without inflammation. So a strictly internal source of infection seems unlikely.
A third possible explanation for the frequent presence of BPV in inflamed areas of the skin is that BPV infection itself may be a contributing cause of the inflammation. Epidermal changes associated with BPV infection have been described.5,6 Whether these changes predispose horses to the development of other inflammatory skin conditions in these areas is unclear. It may be that most horses are able to clear the inflammatory processes associated with these BPV infections, but others are unable to avert infection and progression to sarcoid development occurs.
Recently, BPV DNA has been recovered from keratinocytes within the epidermis of sarcoids. 6 In that study, BPV DNA was amplified from keratinocytes in occult sarcoids but not from “advanced” sarcoids. The clinical classification of the sarcoids described as “advanced” was not identified. The current study demonstrated that BPV DNA could be found within the epidermis of all examined clinical types of sarcoids. However, BPV DNA in the epidermis was significantly more likely to be identified in occult sarcoids than in fibroblastic or nodular ones (Table 9). Presuming that fibroblastic and nodular sarcoids are types of the “advanced” sarcoids described in the other study, 6 results from more recent studies would agree that occult sarcoids are more likely to contain BPV DNA in the epidermis than are fibroblastic or nodular ones. This may represent a shift in the location of BPV DNA as sarcoids change from less to more clinically aggressive.
Two methods of DNA amplification were utilized in the current study, PCR and LAMP. The PCR assay has been extensively used to detect BPV in sarcoids in FFPE tissue,3,8-10,16,22,25,28 while LAMP has not. The LAMP assay is a relatively new DNA amplification technique first reported in 2000. 20 Some studies have shown it to be more sensitive for detection of low copy numbers than PCR. 13 In the current study, agreement, as measured by unweighted kappa, between PCR and LAMP was almost perfect. The PCR assay was more sensitive than LAMP, detecting 19 positive samples that LAMP did not, while LAMP detected 2 positive samples that PCR did not. This level of agreement suggests that LAMP is a viable DNA amplification technique for use with LM.
In summary, BPV DNA was present in over 40% of all samples of nonsarcoid equine skin biopsies tested. It was found in a variety of inflammatory skin conditions as well as normal equine skin. The distribution of BPV within the skin was different in the nonsarcoid samples than in the equine sarcoids, as it was more likely to be found on or in the epidermis. The BPV was more likely to be found in microscopically inflamed areas than in microscopically normal areas of nonsarcoid samples. This inflammation may cause defects in the skin barrier, which allows BPV access through the skin surface to the underlying dermis of the skin where it may contribute to equine sarcoid formation. Alternately, the inflammation itself may be partly a result of BPV infection, and the inability of some horses to resolve the inflammation in these areas may lead to sarcoid formation. The results of the current study may provide new insight into the pathogenesis of sarcoid formation and also imply that diagnostic techniques for equine sarcoids, which rely upon detection of BPV DNA from skin swabs, scrapings, or biopsies, are unlikely to be adequately specific.
Footnotes
a.
MembraneSlide 1.0 PEN, Carl Zeiss Canada Ltd., Toronto, Ontario, Canada.
b.
PALM MicroBeam, Carl Zeiss Canada Ltd., Toronto, Ontario, Canada.
c.
AdhesiveCap 500, Carl Zeiss Canada Ltd., Toronto, Ontario, Canada.
d.
Eiken Chemical Co. Ltd., Tokyo, Japan.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The Equine Health Research Fund of the Western College of Veterinary Medicine and the Equine Foundation of Canada funded the present study.
