Abstract
The current study describes the recovery of Helcococcus ovis from an adult female Verata breed goat that was euthanized because of respiratory problems and emaciation. At necropsy, the affected animal showed a purulent bronchopneumonia with scattered pulmonary abscesses and fibrous pleural adhesions. A Gram-positive, catalase-negative, coccus-shaped bacterium was isolated from the lung tissue and confirmed as H. ovis by 16S ribosomal RNA gene sequencing.
Helcococcus ovis is a catalase-negative, facultatively anaerobic, Gram-positive coccus belonging to the family Peptostreptococcaceae that was first described 1 in 2 sheep as mixed infections with other known ovine pathogens. Since then, H. ovis has attracted an emerging interest in veterinary medicine where it has been reported as the primary pathogen in association with valvular endocarditis in cattle, 4 with a pulmonary abscess in a horse, 5 and in a sheep with pleuritis and bronchopneumonia. 6
In the current case, the only affected animal belonged to a Verata breed herd located on an estate near the city of Badajoz (Extremadura) in southwest Spain where the goats have been reared under a semi-extensive management system. The goats are grazed during the day and are confined and given feed supplements in the goat house at night. There are also cattle, pigs, and sheep on the premises, but they are raised in different paddocks without contact with the goats.
The affected goat was euthanized because of chronic ill health. It had suffered from dry cough, fever, lethargy, and progressive emaciation for weeks. The animal had been treated unsuccessfully with tetracycline.
At necropsy, the animal appeared thin and dehydrated. The main gross pathological findings were in the respiratory tract. The lungs had multiple variably sized encapsulated abscesses containing yellow–green purulent material, and there was a purulent bronchopneumonia. Both lungs were firmly adhered to the parietal pleura by dense, fibrinous adhesions (Fig. 1). No gross lesions were observed in any other tissues.
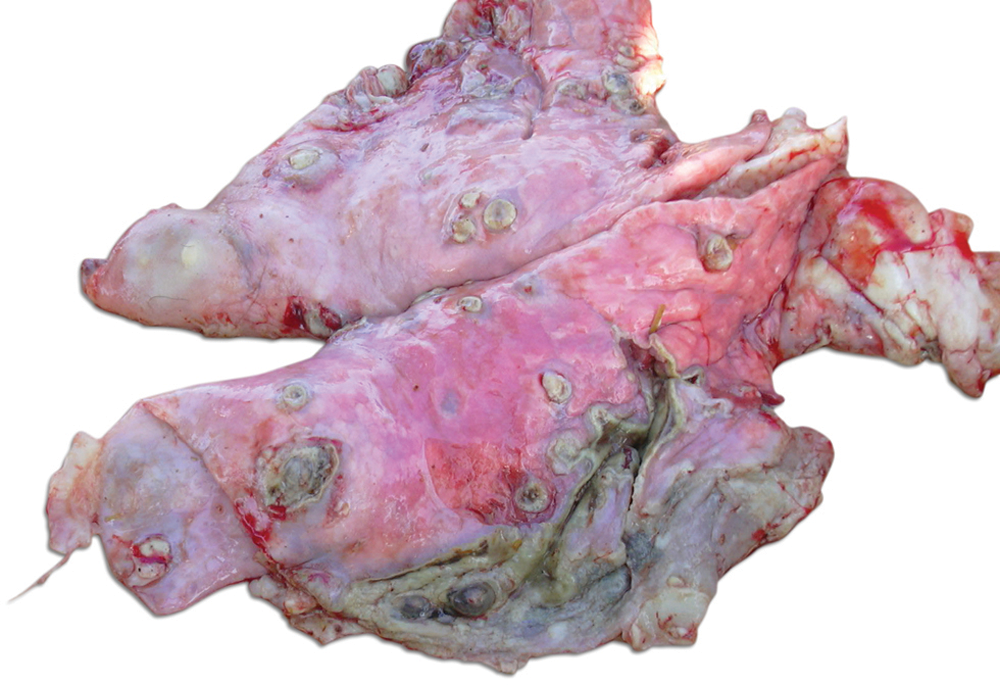
Lungs; goat. Multiple encapsulated abscesses scattered throughout all lung parenchyma containing green–yellow purulent to caseous material and numerous pleural adhesions.
Fresh and formalin-fixed lung tissues were submitted to the School of Veterinary Sciences at the University of Extremadura (Cáceres, Spain) for further diagnostic tests. Smears were prepared from fresh lung tissue samples and stained by Gram and Ziehl–Neelsen stains. Direct microscopic examination showed Gram-positive cocci in pairs or in short chains; the smears contained no acid-fast bacilli. The presence of Gram-positive cocci and absence of acid-fast bacteria helped to rule out tuberculosis.
The formalin-fixed lung samples were processed routinely and embedded in paraffin wax. Sections (5 µm) were mounted on glass slides and stained with hematoxylin–eosin and Brown and Brenn Gram stain. Significant microscopic changes observed included a severe, diffuse, purulent bronchopneumonia characterized by the presence of exudates and infiltration of inflammatory cells in the bronchiolar wall as well as in the lumen of bronchiole, the alveoli, and the inter-alveolar septa. In addition, there were multiple pulmonary abscesses scattered throughout the parenchyma, containing a central zone with necrotic cell debris surrounded by numerous colonies of Gram-positive cocci and large number of neutrophils, many of the latter degenerate, encapsulated by fibrous connective tissue (Fig. 2).
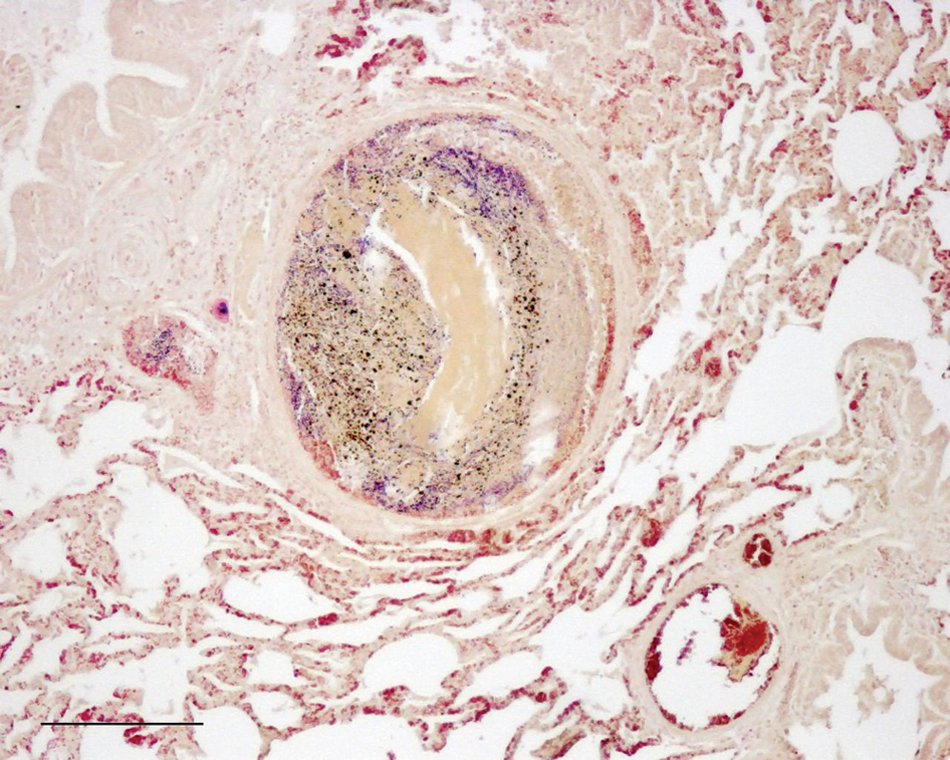
Lung; goat. A section of lung showing congestion, edema, and interstitial infiltrates of inflammatory cells. Focal lesion characterized by a central zone of necrotic cell debris and bacterial colonies surrounded by a fibrous capsule and presence of degenerated neutrophils in the periphery. Brown and Brenn Gram stain. Bar = 200 µm.
Lung tissue was also inoculated onto 5% sheep blood agar, Mycoplasma agar, and Sabouraud dextrose agar. The plates were incubated at 37ºC in both aerobic and in a 5% CO2-enriched atmosphere. After 3 days of incubation, a heavy growth of tiny, grayish, nonhemolytic satellite colonies were observed around a small number of contaminating staphylococcal colonies on the blood agar plate (Fig. 3). However, after subculturing, the isolate grew readily on blood agar, and pure cultures of Gram-positive, catalase-negative, coccus-shaped organisms were obtained after 48–72 hr of incubation in the presence of 5% CO2. No Mycobacterium or virus isolations were attempted.
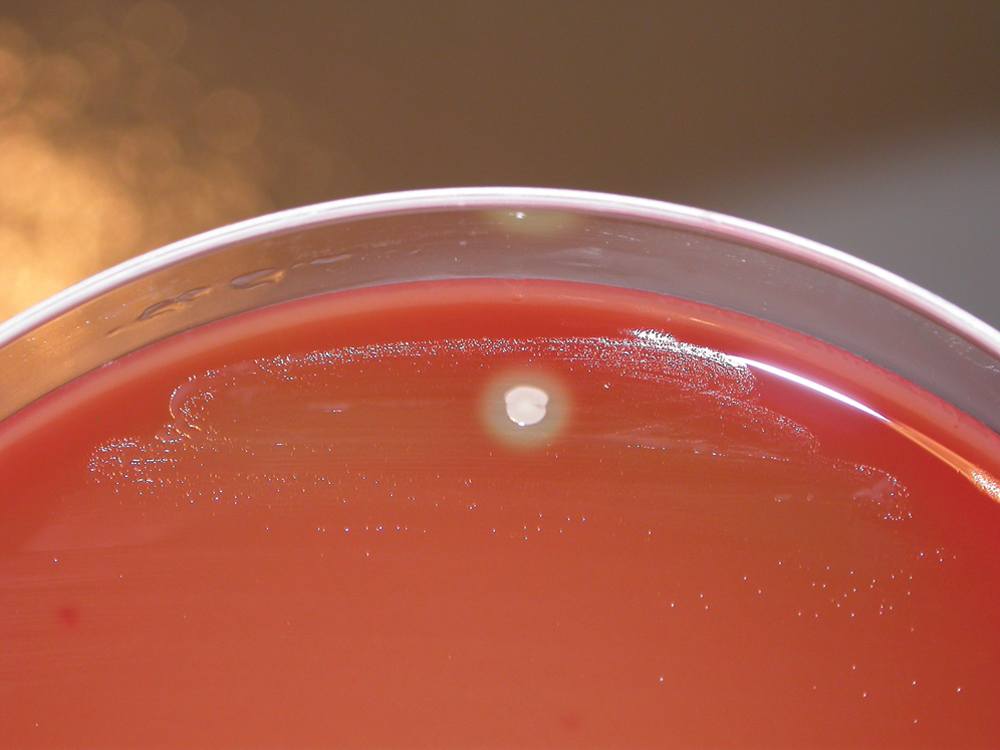
Blood agar plate. Growth of tiny, gray, nonhemolytic colonies was observed on the 5% sheep blood agar plate showing satellitism around a Staphylococcus spp. colony after 72 hr of incubation at 37°C in 5% CO2.
Biochemical identification was attempted with a commercial system. a The isolate showed a biochemical profile that did not match any of the species identified by this system. The isolate reacted positively in the following tests: L-phenylalanine-7-amido-4-methyl-coumarin (L-phenylalanine-AMC), hydrolysis of 4-methylumbelliferyl (MU)-phosphate, L-tryptophan AMC, L-arginine-AMC, 4MU-β-D-glucuronide, p-nitrophenylphosphate, p-nitrophenyl β-D-glucoside, p-nitrophenyl β-D-cellobioside, p-nitrophenyl α-D-galactoside, p-nitrophenyl α-D-maltoside, and proline and leucine p-nitroanilide and N-acetyl-β-D-glucosaminide. Acid was produced from lactose, fructose, and trehalose, but not from sucrose, arabinose, mannitol, maltotriose, and glycerol. Moreover, neither esculin nor urea was hydrolyzed.
To obtain a definitive identification of the isolate, a 1,400-bp fragment of the 16S ribosomal RNA (rRNA) gene was amplified by polymerase chain reaction, using the primers (LPW57 5’-AGTTTGATCCTGGCTCAG-3’ and LPW205 5’-CTTGTTAC GACTTCACCC-3’) previously described, 3 and the resultant product was sequenced. A BLAST homology search 7 (www.ncbi.nlm.nih.gov/BLAST) revealed that the nucleotide sequence of the amplified product matched (96% identity 974/1018 bp) the 16S rRNA gene (partial sequence) of H. ovis strain H41-Yamagata-080523 (GenBank accession no. AB542087.1).
Antimicrobial susceptibility testing using a modified Kirby–Bauer procedure determined that the isolate was sensitive to ampicillin, amoxicillin, cephalothin, ceftazidime, and vancomycin, but it showed intermediate susceptibility to tetracycline in vitro. This circumstance, together with the inability of the drug to penetrate the abscesses and the delay of its administration, may explain the lack of response to treatment in the affected goat.
The fact that H. ovis was the dominant organism isolated from the lungs of a goat with pulmonary abscesses and purulent bronchopneumonia suggests an etiologic role for the organism. Helcococcus ovis should be included in the list of possible etiological agents of animal respiratory disease because this bacterium has been previously reported as the primary pathogen associated with respiratory disease from other animal species.5,6
Based solely on gross morphology, it is quite difficult to differentiate between abscesses caused by H. ovis and other species of the genera Corynebacterium spp., Staphylococcus spp., Streptococcus spp., and Rhodococcus spp. Even with a Gram stain, the morphology of these Gram-positive bacteria is quite similar. In addition, the carbon dioxide requirement and satellite growth on initial isolation as well as the slow growth of the organism may have resulted in the agent being overlooked or misidentified. Veterinarians must be alert to the threat posed by this uncommon organism and the apparent pathogenic capacity of H. ovis for goats. Although the identification of H. ovis and other newly described organisms can be achieved by phenotypic tests, difficulties can often occur, and the use of molecular tools such as 16S rRNA gene sequencing has been shown to be extremely useful for the identification of unusual animal pathogens.1,2
Footnotes
Notes
The authors declared that they had no conflict of interests with respect to their authorship or the publication of this article.
Alfredo García acknowledges the INIA-CCAA program for his research contract (ref. DR07-0027).
