Abstract
Forty-seven canine cutaneous epithelial tumors and cysts were examined to determine coordinate expression of cytokeratins 7 (CK7) and 14 (CK14), vimentin, and Bcl-2 using commercially available antibodies. Within non-affected normal skin adjacent to tumors or cysts, CK7 expression was observed in luminal cells in apocrine glands; CK14 expression was observed in the stratum basale, stratum spinosum, stratum granulosum, basal layer of outer root sheath, sebaceous glands, and myoepithelial cells of apocrine glands; vimentin expression was observed in dermal papilla and scattered non-epithelial cells within the epidermis; and Bcl-2 expression was observed in scattered non-epithelial cells in the epidermis and some apocrine glands. The pattern of expression of CK7 and CK14 in cases of adenocarcinoma of the apocrine gland of the anal sac (CK7+/CK14–) and hepatoid gland tumors (CK7–/CK14+) may prove useful for diagnostic purposes. Loss of expression of CK14 and vimentin, identifying myoepithelial cells, was observed in apocrine and ceruminous adenocarcinomas. Differences in patterns of expression of Bcl-2 were observed between infundibular keratinizing acanthomas compared to trichoepitheliomas.
Introduction
Intermediate filaments provide structural integrity for cells and tissues.3,8 There are 6 types of intermediate filaments: type I and II are acidic and basic keratins, respectively, both of which contain cytokeratins (CKs) numbered in sequential order based on their size; type III includes vimentin, desmin, glial fibrillary acidic protein, and peripherin; type IV includes NF-L (neurofilament protein light), NF-M (medium), NF-H (heavy), α-internexin, and nestin; and type V are the nuclear laminins.3,8 Immunohistochemistry (IHC) is often used as a diagnostic tool to detect expression of intermediate filaments when characterizing tumors and can be helpful for making a diagnosis when a small tissue specimen is examined, when the location of origin of the tumor is unknown, or if the tumor is from a metastatic lesion.
Cytokeratin 7 (CK7) is a 54-kD type II keratin that is expressed in apocrine glands of normal canine skin, but not expressed in normal canine epidermis.3,9,13,27–29 CK7 expression utilizing either IHC or immunocytochemistry has been observed in apocrine gland tumors, squamous cell carcinomas, and a hepatoid gland carcinoma in dogs.1,12,27 Comparatively, in humans, CK7 expression was observed in 26% (7/27) of squamous cell carcinomas. 14
Cytokeratin 14 (CK14) is a 50-kD type I keratin that is expressed in the basal layer of the canine epidermis, inner root sheath, outer root sheath, sebaceous glands, and myoepithelial cells of the apocrine glands of canine skin.27–29 In 1 report, there was a lack of expression of CK14 in the epidermis of canine skin while other studies did not identify CK14 expression in root sheaths and myoepithelial cells of the apocrine glands.13,27,29 CK14 expression has been reported in canine cutaneous epithelial neoplasms, including trichoblastomas, papillomas, intracutaneous cornifying epithelioma (currently known as infundibular keratinizing acanthoma [IKA]), basal cell carcinomas, trichoepitheliomas, sebaceous gland tumors, hepatoid gland tumors, anal gland tumors, and squamous cell carcinomas.13,26,27,29 In humans, CK14 expression has been identified in squamous cell carcinomas and basal cell carcinomas.7,21
Vimentin is a 57-kD protein of the intermediate filament family. Vimentin is not expressed by keratinocytes in the canine epidermis. 4 Melanocytes, Langerhans cells, dermal papilla, and dermal fibroblasts express vimentin.4,15,20 In cutaneous squamous cell carcinomas, upregulation of vimentin has been reported. 2 This upregulation is observed in squamous cell carcinomas undergoing epithelial to mesenchymal transition (EMT).2,4 Upregulation of vimentin is considered to be a hallmark of EMT. 4
B-cell lymphoma 2 (Bcl-2) is an antiapoptotic polypeptide that functions as a repressor of programmed cell death. 19 Bcl-2 is expressed in canine mast cell tumors and canine hemangiosarcomas.17,25 Overexpression of Bcl-2 has been linked to neoplastic transformation in canine mast cell tumors. 25 There are no studies evaluating expression of Bcl-2 in normal canine skin, to the authors’ knowledge. In humans, Bcl-2 expression has been demonstrated in a number of cutaneous neoplasms and has been used for diagnostic purposes to differentiate trichoepitheliomas from basal cell carcinomas and squamous cell carcinomas from basal cell carcinomas based on staining patterns.22,23
The purpose of our study was to determine distribution of antibodies to CK7, CK14, vimentin, and Bcl-2 expressed in canine cutaneous epithelial tumors and cysts. CK7, CK14, vimentin, and Bcl-2 were chosen due to their ability to aid in differentiation between various cutaneous neoplasms in humans including basal cell carcinomas and squamous cell carcinomas.14,20,22
Materials and methods
Samples
A total of 47 canine cutaneous tumors and cysts were retrieved from the archives of the Veterinary Diagnostic Laboratory, University of Illinois at Urbana–Champaign. The masses were submitted as surgical biopsies, and samples were submitted in 10% buffered formalin with fixation times of ~24–72 hr. Samples were embedded in paraffin, and sections were cut at 3 µm and stained with hematoxylin and eosin (HE). Diagnoses were verified by review of the HE-stained slides by 2 pathologists (AW Stern, SM LeClerc) according to the World Health Organization classification of tumors.
Antibodies and immunohistochemical staining
Immunohistochemistry for CK7, CK14, vimentin, and Bcl-2 was performed on formalin-fixed, paraffin-embedded tissues sectioned at 4 µm using standard techniques on 2 automated stainers (for CK7, CK14, and vimentin a and for Bcl-2 b ). The slides were placed in a 55–72°C oven for 1 hr and then deparaffinized. For antigen retrieval, slides were placed in 0.1% protease at 37–40°C for 20 min for CK7 and CK14; in antigen retrieval solution c using a decloaking chamber for vimentin; and in heat-induced epitope retrieval with ethylenediamine tetra-acetic acid buffer (pH 8.0) at 97°C for 20 min for Bcl-2 slides. To reduce nonspecific binding, all slides, except for Bcl-2, were incubated using blocking reagents for 5 min d and 10 min, e respectively. For Bcl-2, slides were incubated with peroxidase blocking reagent. f The slides were incubated for 30 min to 1 hr with the primary monoclonal antibodies CK7 g (1:25), CK14 h (1:20), vimentin i (predilute), and Bcl-2 j (1:500). A mouse-on-canine horse radish peroxidase (HRP) polymer detection method k was used for the CK7, CK14, and vimentin antibodies. A polymer HRP method l was used for the Bcl-2 antibody. Diaminobenzidine (DAB) as chromogen for 10 min was used for the CK7, CK14, and vimentin. m Bcl-2 had a DAB+ detection kit n applied. Finally, slides were counterstained in hematoxylin. Primary antibodies (CK7, CK14, and vimentin) were replaced with a polymer negative control serum o for negative controls. Primary antibody Bcl-2 was replaced with a universal negative mouse control. p Positive IHC controls consisted of urinary bladder (CK7), squamous cell carcinoma (CK14), small intestine (vimentin), and canine lymph node (Bcl-2).
Multiple fields of nonaffected normal skin and neoplastic cells from each slide were evaluated, and the results were expressed as positive or negative staining. Cytoplasmic immunoreactivity was assessed. Only those cases exhibiting >5% neoplastic cell positivity were regarded as positive.
Results
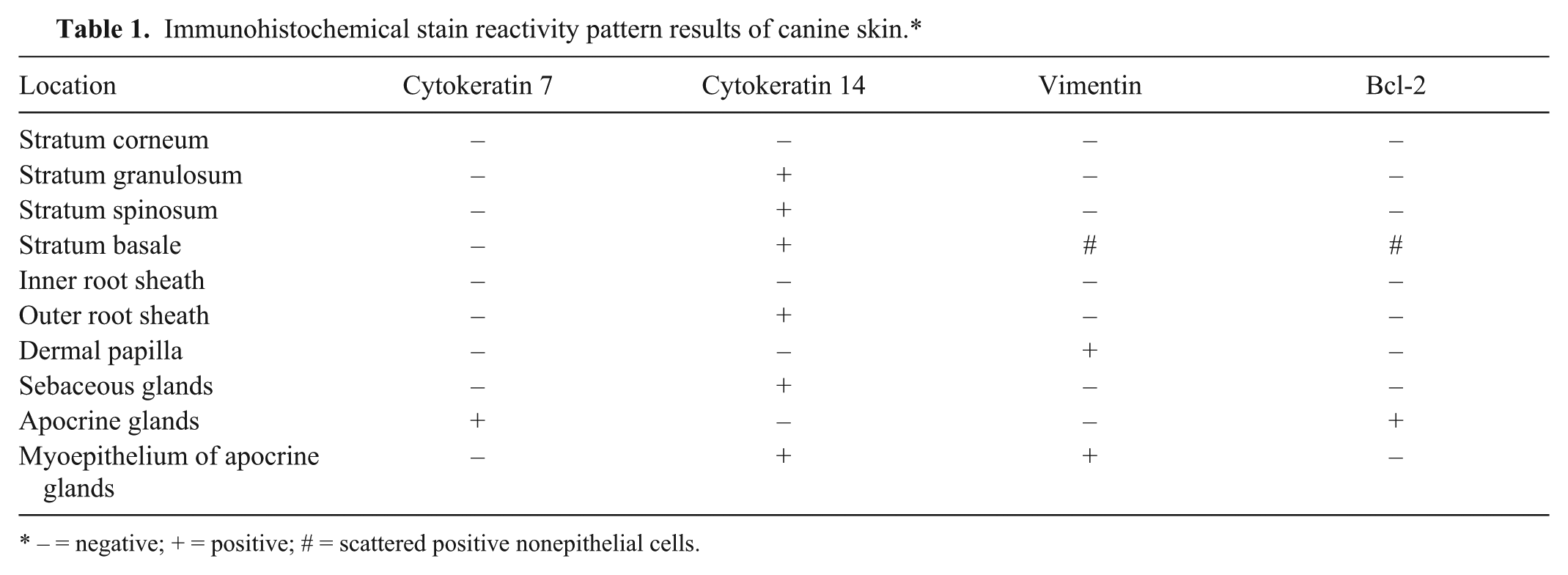
Nonaffected normal canine skin samples adjacent to tumors or cysts are summarized in Table 1. CK7 IHC staining was observed in the luminal cells in apocrine glands, and no staining was observed in the epidermis (Fig. 1). CK14 expression was observed in the stratum basale, stratum spinosum, stratum granulosum, basal layer of outer root sheath, sebaceous glands, and myoepithelium of apocrine glands (Fig. 1). Dermal papilla, myoepithelial cells of apocrine glands, and scattered individual cells within the epidermis, consistent with Langerhans cells, and melanocytes expressed vimentin (Fig. 1). Bcl-2 was expressed by solitary cells in the epidermis (consistent with melanocytes) and in some apocrine glands (Fig. 1).
Immunohistochemical stain reactivity pattern results of canine skin.*
– = negative; + = positive; # = scattered positive nonepithelial cells.
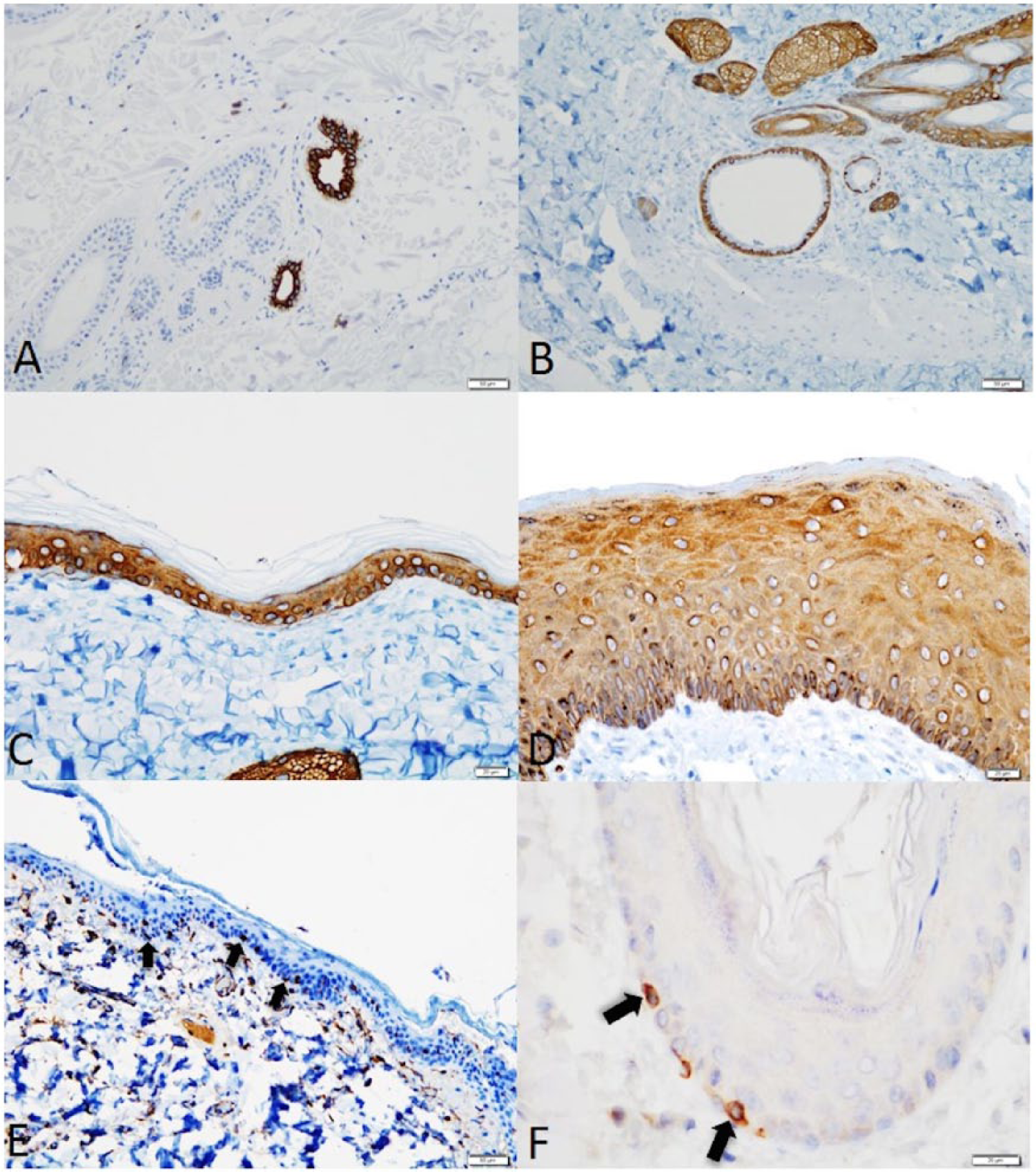
Normal and hyperplastic canine skin.
CK7+/CK14– was observed in all of the adenocarcinomas of the apocrine gland of the anal sac (Fig. 2; Table 2). None of the other tumors examined had this pattern of staining. CK7–/CK14+ was observed in the hepatoid gland carcinoma (1/1), hepatoid gland adenomas (3/3), squamous cell carcinomas (5/5), follicular cysts (2/2), trichoblastomas (3/3), sebaceous adenoma (1/1), sebaceous gland carcinomas (3/3), sebaceous epitheliomas (2/2), basal cell carcinomas (2/2), basosquamous cell carcinoma (1/1), infundibular keratinizing acanthomas (2/2), trichoepitheliomas (2/2), malignant trichoepitheliomas (2/2), and pilomatricomas (2/2). CK7+/CK14+ was observed in the apocrine cyst (1/1), apocrine ductal adenoma (1/1), apocrine adenocarcinomas (3/3), and ceruminous gland carcinomas (3/3). No tumors were CK7–/CK14–.
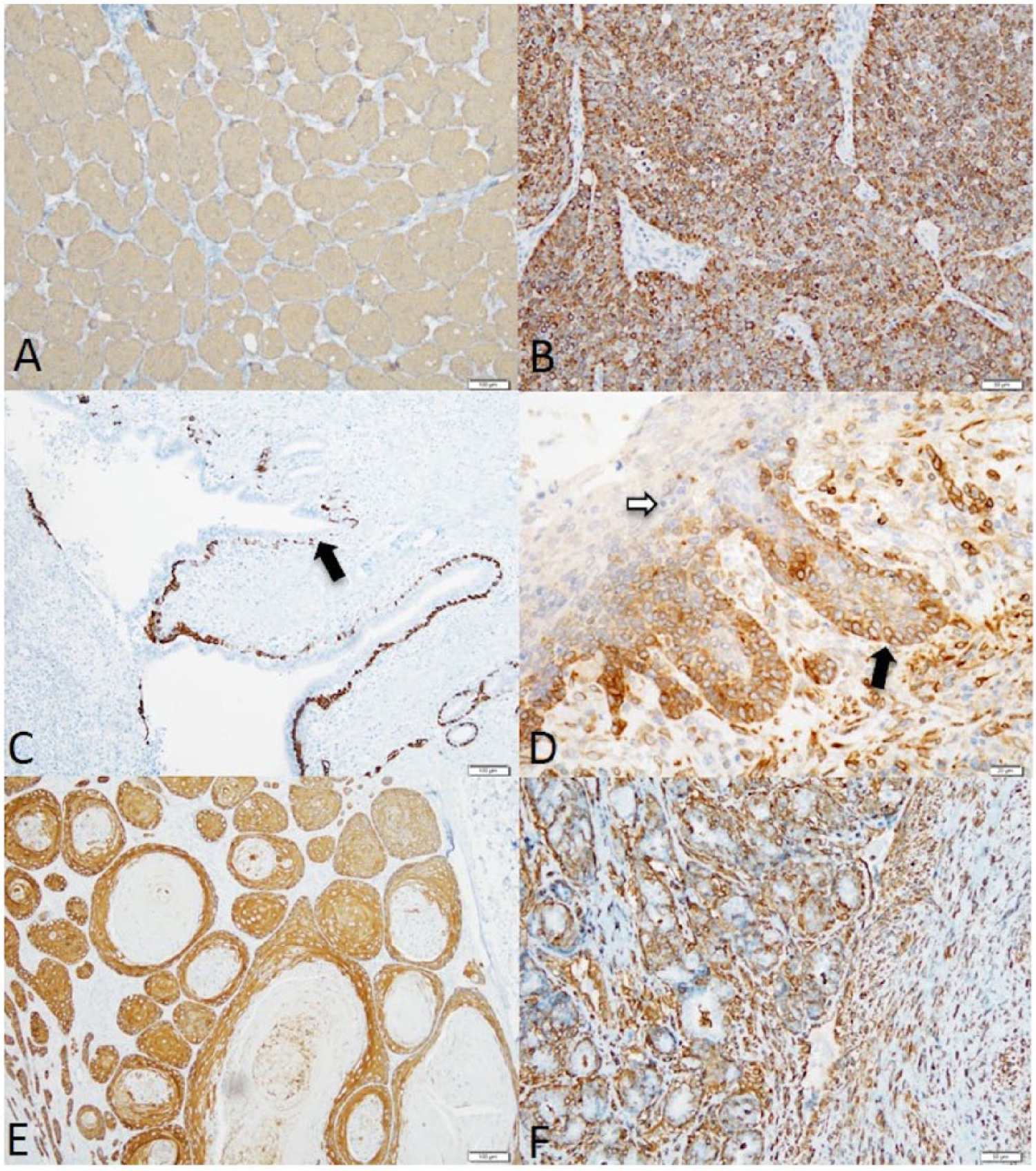
Canine cutaneous tumors.
Immunohistochemical stain reactivity pattern results of canine skin tumors and cysts.*
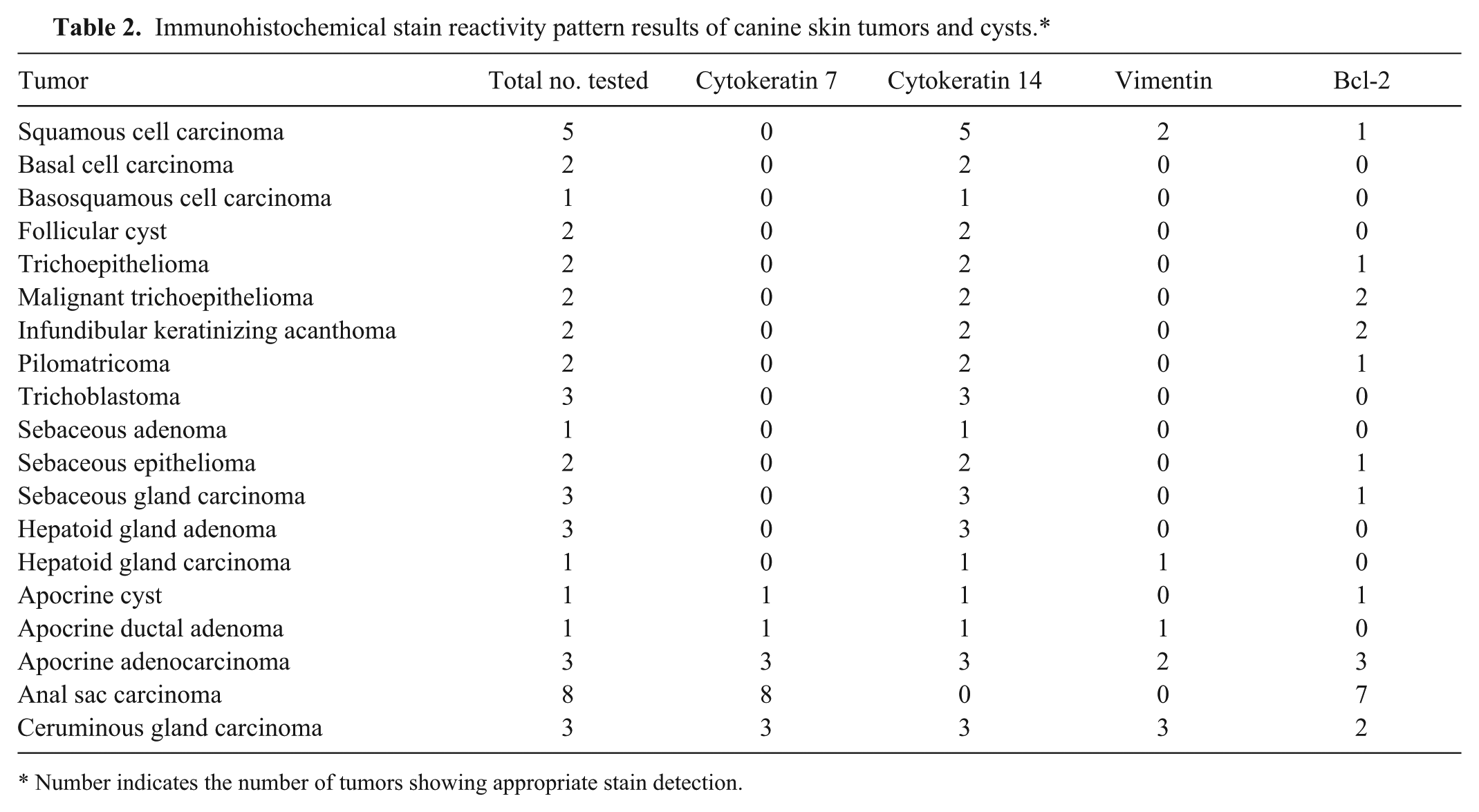
Number indicates the number of tumors showing appropriate stain detection.
Vimentin expression was observed in the hepatoid gland carcinoma (1/1), myoepithelial cells of apocrine ductal adenoma (1/1), myoepithelial cells of apocrine adenocarcinoma (2/3), myoepithelial cells of ceruminous gland carcinoma (3/3), and squamous cell carcinomas (2/5) (Table 2).
Bcl-2 was positive in anal sac carcinomas (7/8), apocrine adenocarcinomas (3/3), apocrine cyst (1/1), ceruminous gland carcinomas (2/3), squamous cell carcinomas (1/5), infundibular keratinizing acanthomas (2/2), trichoepitheliomas (1/2), malignant trichoepitheliomas (2/2), sebaceous gland carcinomas (1/3), pilomatricomas (1/2), and sebaceous epitheliomas (1/2) (Table 2).
Discussion
We observed expression of CK7 only in the luminal cells of apocrine glands and expression of CK14 in multiple layers of the epidermis (stratum basale, stratum spinosum, and stratum granulosum), outer root sheath, sebaceous glands, and myoepithelium of the apocrine glands. In our study, the CK7 findings are in agreement with previous studies examining CK7 expression in normal canine skin.9,29 Basal keratinocytes synthesize CK14, which is retained by keratinocytes in the stratum spinosum. 16 CK14 has previously been identified in the stratum spinosum in normal human skin. 5 In our study, we observed CK14 staining of canine basal keratinocytes and stratum spinosum in agreement with previous studies examining expression of CK14 in dogs, but we also observed additional expression of CK14 in the stratum granulosum. 29 CK14 expression has been noted in suprabasal layers with hyperproliferative disorders, which may be the reason for stratum granulosum expression in our study. 6 In this case series, cutaneous epithelial tumors and cysts that were positive for CK7 included tumors of apocrine gland origin (ductal adenoma, adenocarcinoma, and adenocarcinoma of the apocrine gland of the anal sac), apocrine cyst, and ceruminous gland tumors.
A complete history will provide valuable information regarding the primary location of the biopsied tissue; however, in some cases, incomplete histories are provided, and utilization of coordinate expression of CK7 and CK14 may prove useful. An additional utility of coordinated expression of CK7 and CK14 is differentiating between anaplastic tumors arising in the perianal area. Poorly differentiated hepatoid gland carcinomas and adenocarcinoma of the apocrine gland of the anal sac may be difficult to diagnose using conventional HE light microscopy. Coordinated expression of CK7 and CK14 may be useful to differentiate hepatoid gland tumors (CK7–/CK14+) from adenocarcinoma of the apocrine gland of the anal sac (CK7+/CK14–). This information would be of value for both treatment and prognostic purposes. Given the small number of adenocarcinomas of the apocrine gland of the anal sac and hepatoid gland tumors evaluated in our study, additional studies to further characterize the hypothesis for use of coordinate expression of CK7 and CK14 to allow for differentiation of these neoplasms should be performed.
Another potential utility of coordinate expression of CK7 and CK14 is differentiation between a benign and malignant apocrine neoplasm. Although all tumors of apocrine gland origin and ceruminous gland tumors in our study were CK7+/CK14+, we observed multifocal loss of CK14 and vimentin staining (myoepithelial cells) in the adenocarcinomas. In light of the fact that myoepithelial cells express both CK14 and vimentin, coordinate loss of both CK14 and vimentin would be expected where there is loss of myoepithelial cells of apocrine and ceruminous glands. 18 Complete loss or diminished number of myoepithelial cells has been reported in human breast neoplasms.11,24 This loss of myoepithelial cell layer is considered a crucial step in the progression toward invasive carcinoma. 24 However, this feature alone cannot be relied on to diagnose a malignant process independent of other features of malignancy.11,24
Bcl-2 is expressed in non-affected normal canine skin adjacent to tumors or cysts in scattered individual cells within the epidermis, consistent with melanocytes. Expression of Bcl-2 is also observed in apocrine glands but inconsistently between animals. An extensive study evaluating the expression of Bcl-2 in normal canine skin has not previously been performed for comparison purposes, to the authors’ knowledge. Human skin expresses Bcl-2 in the stratum basale, melanocytes, outer root sheath, dermal papillae, epithelial cells of the secretory coils of eccrine sweat glands, and in the excretory ducts of sebaceous glands. In our study, all the IKAs (2/2), as well as 1 pilomatricoma (1/2), were positive for Bcl-2. Bcl-2 expression was observed in a diffuse pattern only in the trabeculae of the IKAs. Epithelial cells lining the central cyst did not express Bcl-2. Basaloid cells resembling matrical cells within a trichoepithelioma (1/2) and malignant trichoepitheliomas (2/2) were observed to express Bcl-2. Differentiating IKAs from trichoepitheliomas can be difficult with histopathology. 10 The pattern of Bcl-2 expression in IKAs appears to differ from the pattern of expression of Bcl-2 in trichoepitheliomas in our limited sample size.
In this study, basal cell carcinomas and squamous cell carcinomas have the same CK7–/CK14+ pattern. Basal cell carcinomas were negative for both vimentin and Bcl-2. One squamous cell carcinoma was positive for vimentin and 1 squamous cell carcinoma was positive for both vimentin and Bcl-2. There were no trends noted using these markers to differentiate basal cell carcinomas from squamous cell carcinomas in the dog, in contrast to humans where expression of Bcl-2 has been reported to be diagnostically useful to help differentiate basal cell carcinomas (Bcl-2+) from squamous cell carcinomas (Bcl-2–). 22 It is of interest that only a single dog with squamous cell carcinoma (aural location) was euthanized due to progression of the disease and that this neoplasm was the only Bcl-2–positive neoplasm and 1 of 2 that was positive for vimentin.
In our study, 2 squamous cell carcinomas had expression for vimentin. Previous studies have identified that some squamous cell carcinomas express vimentin, and this is believed to be due to EMT. 4 Most of the squamous cell carcinomas in previous studies with expression of vimentin in the neoplastic cells was indicative of EMT, which is consistent with one of our cases. 4
In conclusion, coordinate expression of CK7, CK14, vimentin, and Bcl-2 identified by IHC may be helpful in differentiating between some canine cutaneous epithelial tumors and cysts. In particular, the pattern of expression of CK7 and CK14 in cases of adenocarcinoma of the apocrine gland of the anal sac (CK7+/CK14–) and hepatoid gland neoplasms (CK7–/CK14+) may prove useful for diagnostic purposes. Loss of expression of CK14 and vimentin in apocrine and ceruminous adenocarcinomas may prove useful to differentiate a benign versus malignant process especially in small biopsy specimens. Differences in patterns of expression of Bcl-2 were observed between IKAs compared to trichoepitheliomas. There were no helpful differences observed in the IHC staining for differentiating canine basal cell carcinomas from canine squamous cell carcinomas.
Footnotes
Acknowledgements
We would like to thank Renee Walker (University of Illinois) and Brad Charles (Colorado State University) for technical help with IHC staining of tissues.
Authors’ contributions
JB Pieper, AW Stern, and SM LeClerc contributed to conception and design of the study. KL Campbell contributed to design of the study. AW Stern and SM LeClerc contributed to acquisition, analysis, and interpretation of data. JB Pieper contributed to analysis and interpretation of data, and drafted the manuscript. All authors critically revised the manuscript; gave final approval; and agree to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
IntelliPath, Biocare Medical, Concord, CA.
b.
Link48, Dako North America Inc., Carpinteria, CA.
c.
Diva antigen retrieval solution (DV2004), Biocare Medical, Concord, CA.
d.
Peroxidazed 1 (PX968), Biocare Medical, Concord, CA.
e.
Background Punisher (BP974), Biocare Medical, Concord, CA.
f.
EnVision FLEX peroxidase blocking reagent, Dako North America Inc., Carpinteria, CA.
g.
M7018, Dako North America Inc., Carpinteria, CA.
h.
AM146-5M, BioGenex Laboratories Inc., Fremont, CA.
i.
PM048, Biocare Medical, Concord, CA.
j.
BCL-2-486, Leica Microsystems, Buffalo Grove, IL.
k.
MC541, Biocare Medical, Concord, CA.
l.
EnVision FLEX polymer HRP method, Dako North America Inc., Carpinteria, CA.
m.
intelliPATH FLX DAB chromogen kit, Biocare Medical, Concord, CA.
n.
EnVision FLEX DAB+ detection kit, Dako North America Inc., Carpinteria, CA.
o.
Polymer negative control serum, Biocare Medical, Concord, CA.
p.
Universal negative mouse control, Dako North America Inc., Carpinteria, CA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding was provided by Dermapet Small Melman and Anderson Dermatology endowments.
