Abstract
Newcastle disease (ND) remains a constant threat to the poultry industry and is a limiting disease for poultry producers worldwide. The variety of clinical presentations and the emergence and spread of new genetic variants make recognition and diagnosis challenging. The current review details the pertinent features of the clinicopathologic disease in the main susceptible species, including chicken, turkey, duck, goose, pigeon, and other birds such as cormorants, psittacines, and canaries. Furthermore, the available and emerging laboratory diagnostic methodologies for the detection and typing of the virus are reviewed, including traditional techniques such as virus isolation and immunohistochemistry as well as rapid procedures based on molecular tools, such as real-time polymerase chain reaction, gene sequencing, and microarrays. The relevant genetic variability of ND viruses probably represents the major limitation in the validation and application of the current, advanced diagnostic molecular techniques. This underscores the importance of a multidisciplinary and comprehensive diagnostic approach, which should include, next to the new generation assays of the genomic era, the more traditional techniques such as histopathology, immunohistochemistry, and virus isolation.
Keywords
Introduction
Newcastle disease (ND) is a viral disease of poultry caused by a single-strand, nonsegmented, negative-sense RNA virus known as Avian paramyxovirus 1 (APMV-1). The disease is present worldwide and affects many species of birds causing severe losses in the poultry sector. In developing countries, where the majority of chickens are reared under “backyard” subsistence conditions, ND can drastically limit the amount of dietary protein as well as damage the microeconomy due to loss of ability to sell off extra chickens or eggs. Where chickens are raised commercially, either in developing or developed countries, outbreaks have occurred in many locations, causing massive economic damage through control efforts and trade losses. For instance, during the last major outbreak in the United States, in California in 2002–2003, more than 2,500 premises were depopulated (4 million birds) at a cost of US$162 million.
According to the World Organization for Animal Health (OIE), 125 ND is an OIE notifiable disease when it meets certain criteria of virulence. These high consequence strains can cause enormous economic impact, and so prompt recognition and confirmation is of paramount importance. However, the lack of characteristic clinical signs in many bird species, the variations in virulence for APMV-1 isolates and their genetic variability pose a serious challenge for the rapid identification and diagnosis of this infection. The current review aims to present relevant information concerning recognition and diagnosis of Newcastle disease virus (NDV), including field presentation of the disease and traditional bench diagnostics, as well as some of the newer methods used in the laboratory.
The virus
All avian paramyxoviruses (APMV) are part of the genus Avulavirus, subfamily Paramyxovirinae, family Paramyxoviridae, order Mononegavirales. 71 There are 9 serotypes of APMV, but all isolates of Newcastle disease virus (NDV) belong to serotype 1 (APMV-1), therefore NDV is synonymous with APMV-1. The APMV-1 viral genome of approximately 15 kb is composed of 6 genes encoding 6 structural proteins (fusion [F], nucleoprotein [NP], matrix [M], phosphoprotein [P], RNA polymerase [L], and hemagglutinin-neuraminidase [HN]). Two additional proteins are encoded by RNA editing of the P protein, namely proteins V and W. The cleavability of protein F is the main determinant for viral virulence, but other proteins such as HN and V are also believed to influence pathogenicity. 32,50,82
Although all NDV isolates belong to a single serotype (APMV-1), there is great genetic variability among different strains. Based upon phylogenetic reconstruction, NDV can be divided into 2 classes (I and II), each of those respectively subdivided into 9 and 10 genotypes (Fig. 1). 83 In the last few years, there has been an increase of newly discovered genotypes, and some of the genotypes have been associated with increased virulence or expanded host range. 74,121,127 In addition, this genetic variability has raised concerns as to whether the commonly used commercial vaccines can provide protection against the very distant genotypes, not just in preventing clinical signs, but also in limiting shedding of the challenge viruses. 83,84
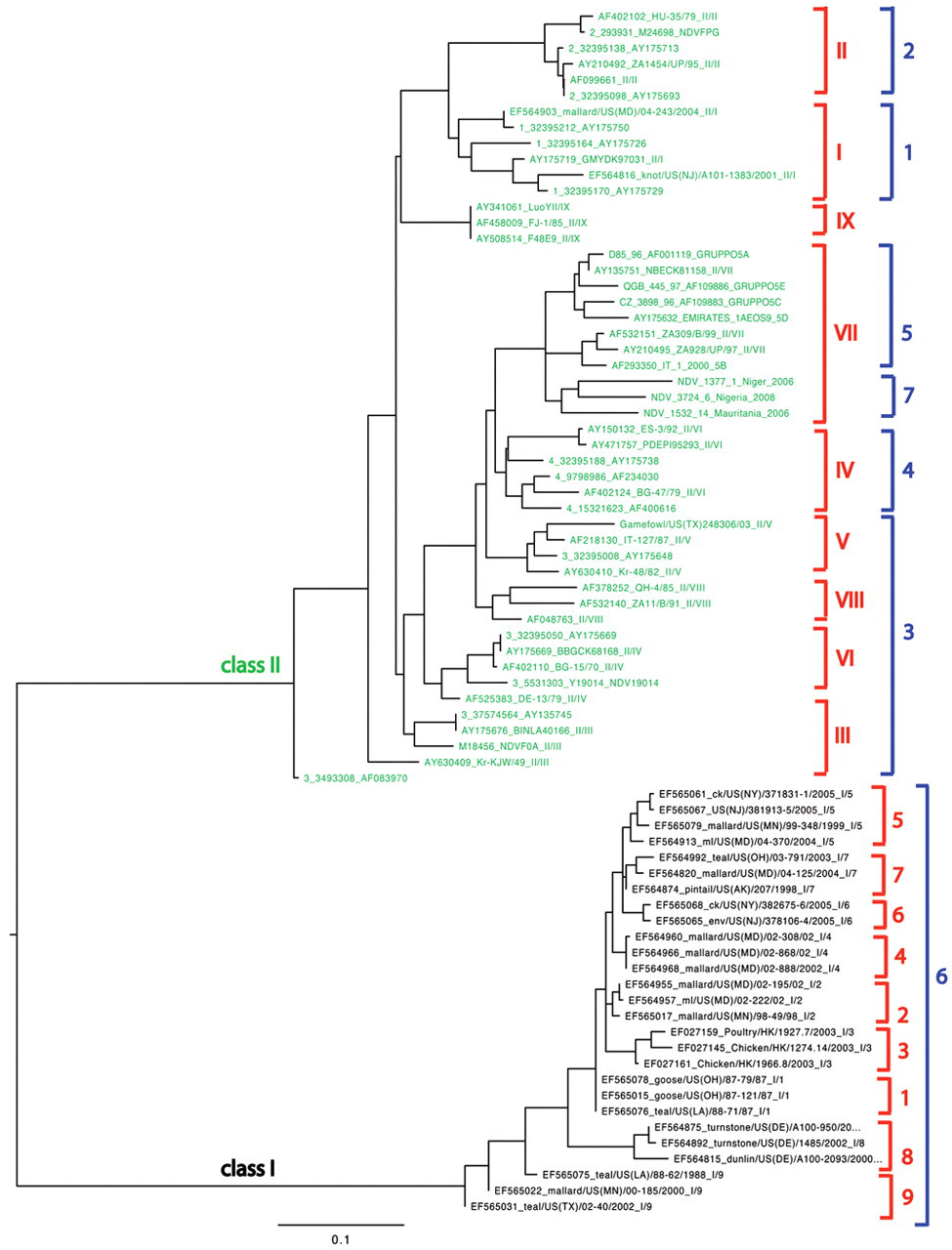
Phylogenetic tree for Avian paramyxovirus-1, demonstrating 2 distinct classes (I and II) with each having several genotypes. The nomenclatures of the 2 main genotyping systems are indicated in red (references 44, 83) and in blue (reference 5). Genotype X strains are not included due to the different location of the fusion (F) gene segment sequenced and deposited in the public database (GenBank accession nos. AY372135-37, AY372163).
Classification of viral pathogenicity
Newcastle disease affects a wide range of domestic and wild avian species; however, the severity of the disease varies greatly, spanning from peracute disease with almost 100% mortality to subclinical disease with no lesions. Such variability makes it impossible to pinpoint ND as a single clinicopathologic entity. Based on severity of clinical disease, the strains of NDV were originally classified into 4 pathotypes, known as Doyle, Beach, Beaudette, and Hitchner forms. 7 At present, pathotypes are more commonly classified based on pathogenicity from least most pathogenic: “asymptomatic enteric,” “lentogen” (formerly Hitchner), “mesogen” (formerly Beaudette), and “velogen.” The velogens have been further divided into “viscerotropic” (formerly Doyle; velogenic viscerotropic NDV [VVNDV]) or “neurotropic” (formerly Beach; velogenic neurotropic NDV [VNNDV]) according to their ability to cause primarily visceral or nervous signs. 9 Additionally, some laboratory testing in embryos or chickens using standard pathogenicity parameters can be done, including MDT (mean death time), IVPI (intravenous pathogenicity index), and ICPI (intracerebral pathogenicity index). All involve the use of numeric criteria. The MDT is the time to death, measured in hours, after inoculation of embryonated eggs (if the embryos die in less than 60 hr, it is classified as a velogen; if the embryos survive for more than 90 hr, it is classified as a lentogen; anything in between is a mesogen). 9,125 The IVPI test involves scoring illness (0 = normal; 1 = sick; 2 = paralyzed or nervous signs; 3 = death) after intravenous inoculation of 6-week-old chickens. The IVPI scores are computed similarly to ICPI (see below) and range from 0 to 3. According to some authors, 7 velogenic NDV have IVPI scores between 2 and 3, mesogenic between 0.0 and 0.5, while lentogens have 0; however, to the author’s knowledge, there are no IVPI cut-off values to define notifiability to the international community. The IVPI test is not in widespread use today. At present, according to international standards, 125 the definitive in vivo assessment of virus virulence is based on the ICPI test, which is regarded as the most sensitive and widely used test for measuring virulence. The ICPI test is based on scoring sick or dead birds (0 = normal; 1 = sick; 2 = dead) every day for 8 days after inoculation of virus intracerebrally into ten 1-day-old chicks. 113,125 The score of the ICPI test is calculated using the mean score per bird, per observation, over the 8-day period. Scores range from 0 to 2, and any strain with an ICPI ≥0.7 is considered virulent or “notifiable” to the OIE. 125 The correspondence between these standard tests and pathotypes are reported in Tables 1 and 2. 7 Additionally, the OIE recognizes specific sequences of the F protein as a qualifier for virulence: “notifiable” are those strains that have, with respect to the amino acid sequence of the F protein, one pair of basic amino acids at residues 116 and 115 plus a phenylalanine at residue 117 and a basic amino acid (R) at residue 113. 125
Pathotype designation of Newcastle disease virus strains based on standard pathogenicity tests.*
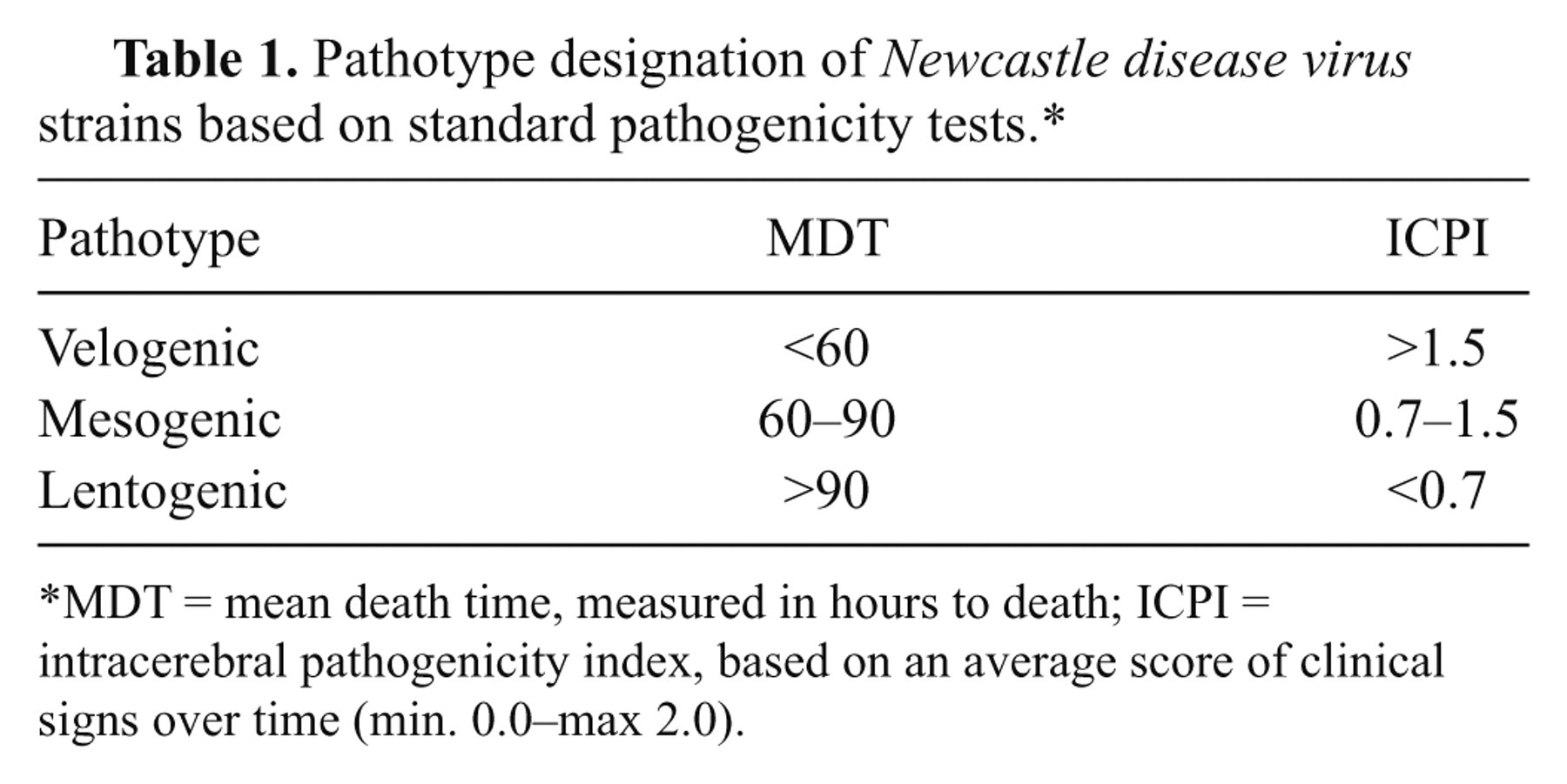
MDT = mean death time, measured in hours to death; ICPI = intracerebral pathogenicity index, based on an average score of clinical signs over time (min. 0.0–max 2.0).
Biological proprieties of selected well-characterized strains of Newcastle disease virus.*
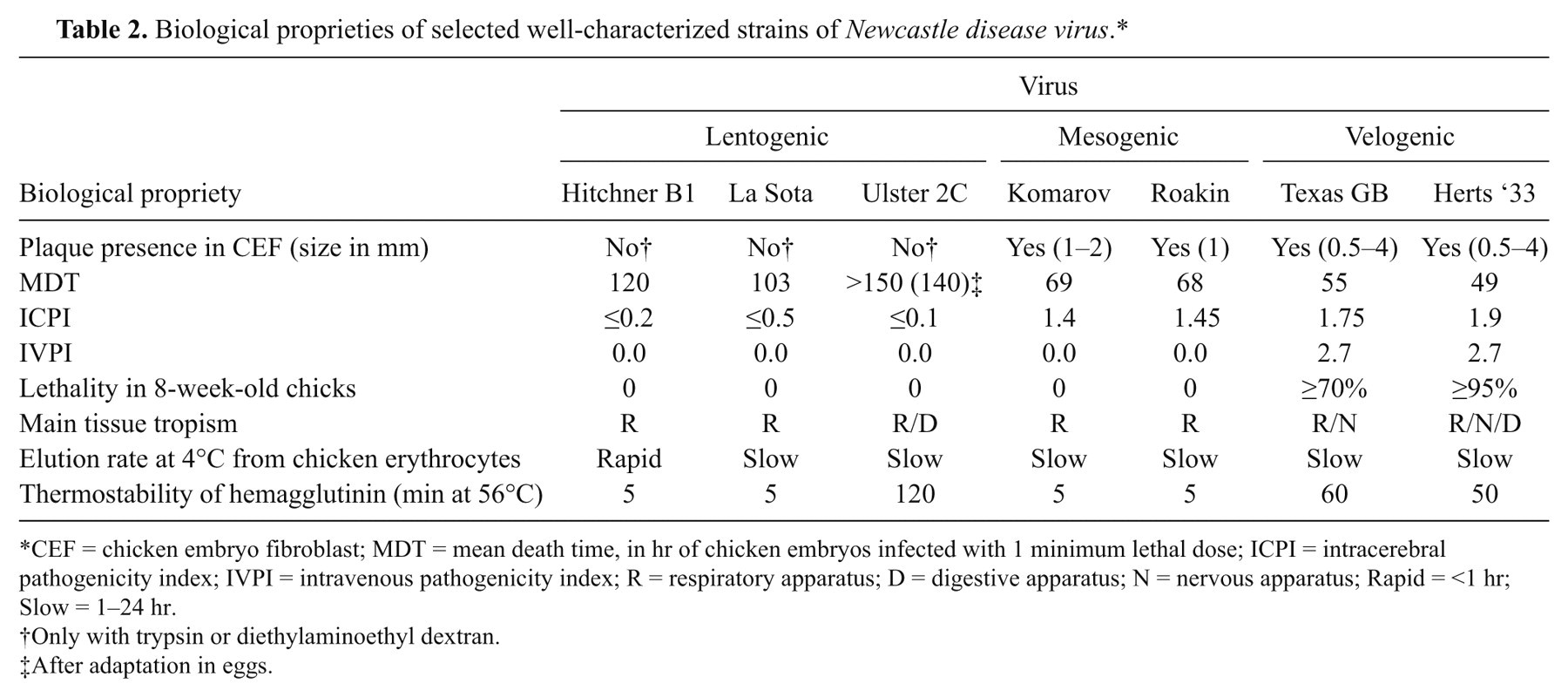
CEF = chicken embryo fibroblast; MDT = mean death time, in hr of chicken embryos infected with 1 minimum lethal dose; ICPI = intracerebral pathogenicity index; IVPI = intravenous pathogenicity index; R = respiratory apparatus; D = digestive apparatus; N = nervous apparatus; Rapid = <1 hr; Slow = 1–24 hr.
Only with trypsin or diethylaminoethyl dextran.
After adaptation in eggs.
Some drawbacks to these tests exist in the interpretation of pathotype results. For example, a previous study 96 reported 10 NDV isolates from pigeons to have ICPI values between 1.2 and 1.45 and a range of IVPI values from 0 to 1.3, suggesting the viruses were virulent; however, the lowest MDT recorded was 98 hr, a characteristic of lentogenic viruses. In fact, not all virulent strains have an MDT <60 hr (Table 2). The in vivo tests on strains isolated from species other than chickens can present some problems and may not produce accurate readings until passaged in chickens or embryonated chicken eggs. 11 The authors’ personal experiences show that a more accurate indication of the real pathogenicity of ND viruses for a susceptible species could come from experimental infection of a statistically significant number (≥10) of young and adult birds with a viral standard dose (e.g., 10 5 50% egg infectious doses [EID50]) administrated via natural routes (e.g., oronasal route or eye-drop).
Bird studies
Although the standard pathogenicity indices can often offer a good idea of the virulence, they do not always correlate exactly with what is observed in animal experiments, especially when the virus is administered to adult birds via a possible natural route of inoculation. 109,119 In other words, the MDT and ICPI do not always correlate with the clinicopathologic syndrome, and the “disease-inducing ability” of each strain. Furthermore, it should be noted that the status of “notifiability,” as indicated by the OIE (ICPI ≥0.7 and/or virulent F protein cleavage sequence) covers a broad span of pathogenic potential, encompassing viruses that are capable of causing very severe lesions or none. 125 Therefore, animal experiments done in conditions similar to those in the field (e.g., 4-week-old chickens inoculated via eye-drop instillation) are useful, as they help to completely characterize newly isolated strains. Such information can augment researchers’ understanding of the strains and can improve diagnostic ability by describing more accurately the clinical and pathologic presentations associated with a particular isolate.
In the next sections, the clinical signs and pathologic findings will be presented based upon species infected. Data and findings are drawn from numerous sources, but mainly from original research papers, case reports, and textbook chapters. In addition, much is based on observations in one of the authors’ laboratories (South East Poultry Research Laboratory [SEPRL], University of Georgia, Athens, Georgia), where a large number of strains have been studied in an identical and systematic way. In this system, a standard target dose (105 EID50) is given via a natural route (eye-drop inoculation) to uniform age (4-week-old) and source (SEPRL source flock) birds, with consistent observation, collection, and examination methods.
Clinical signs and pathologic findings with NDV infection
Chickens
The vast majority of references on NDV in poultry are related to chickens, as this species is the most seriously impacted by NDV. 9 There are such widely varying disease forms that clinical findings for this species are further divided according to pathotypes. However, the severity of clinical signs does not vary only accordingly to the inherent virulence of the virus, but also according to some host-related factors. These factors are mainly age, route of infection, immune status, and concomitant environmental stress. For example, younger animals tend to have more severe and acute disease than older animals, intravenous inoculation is more likely to elicit neurologic signs, and aerosolization of high viral doses tends to impact the upper respiratory tract preferentially. 1,6,9,12,68
Velogenic viscerotropic Newcastle disease
With VVND, mortality can easily reach 100%, and in experimental conditions, the course of disease is rapid, usually 2–4 days. Clinical signs are first recognizable starting at 2 days postinfection (dpi). 23,64,66,109,117 The main signs are conjunctival swelling and reddening centered over the lymphoid patch located in the lower eyelid (Fig. 2A), anorexia, ruffled plumage, prostration, weakness, tremors, and diarrhea; labored breathing is variably reported. 9,23,64,66,109,117 In numerous animal experiments conducted with the same technique used in the current authors’ laboratory (infection via eye-drop instillation in 4-week-old chickens), respiratory signs were observed very rarely and were limited to open-mouth breathing in a few animals. 109 In the absence of respiratory lesions (as reported in the same studies), the open-mouth breathing was interpreted as polypnea and a consequence of a generalized febrile state.
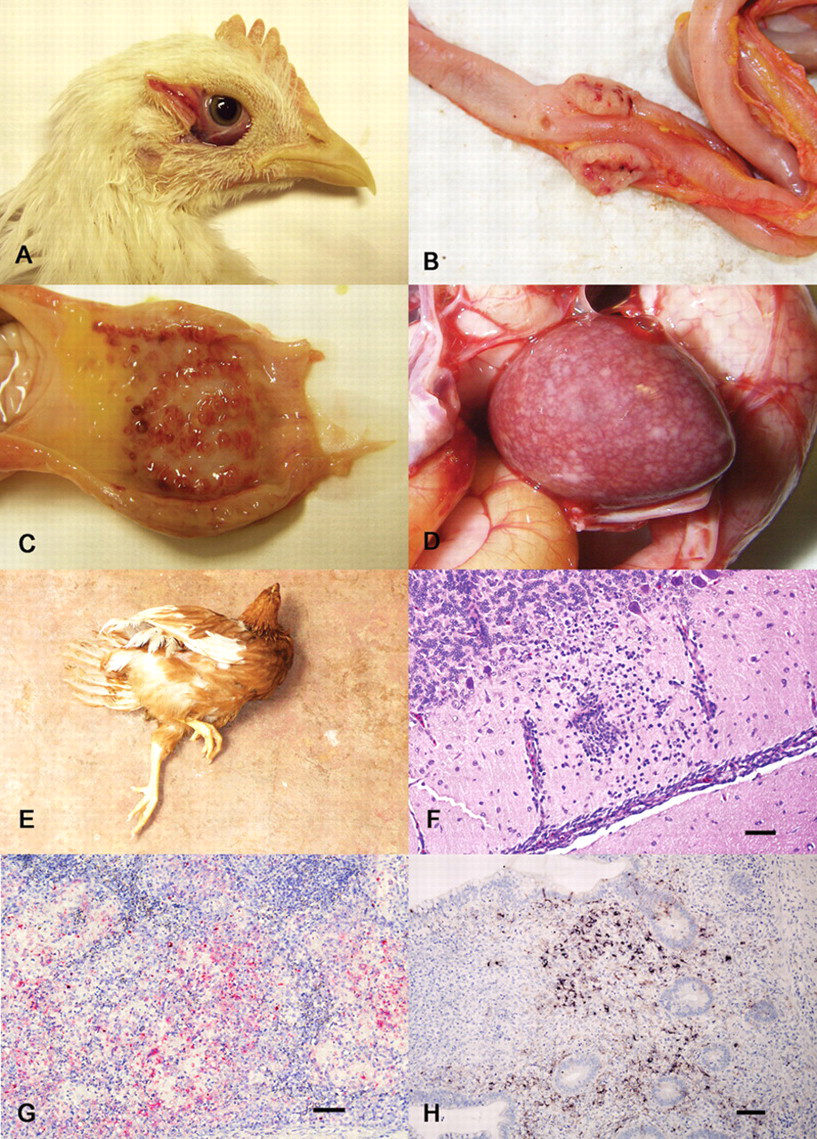
Clinical and pathologic features of Newcastle disease virus (NDV). Velogenic viscerotropic (VVNDV):
The presence of multifocal hemorrhages seen through the serosal surface of the intestines, multifocal areas of necrosis and/or ulceration of the gut-associated lymphoid tissue, and disseminated foci of necrosis in the spleen are highly suggestive of VVNDV infection. 8, 9, 12, 23, 64, 66, 109, 117 The cecal tonsils, which are especially prominent gut lymphoid aggregates located in the proximal portion of the ceca, are often regarded as the “old faithful” lesion for VVND, as they most consistently display hemorrhage and necrosis grossly (Fig. 2B). Other common intestinal lesions are multifocal hemorrhages and ulceration in the junction between proventriculus and gizzard, which is a site of lymphoid aggregate development (Fig. 2C). Spleens are enlarged and severely mottled, showing multiple foci of white to yellow discoloration (necrosis) in the most severe cases (Fig. 2D). 23,64,66,109,117 Perithymic hemorrhages are occasionally observed, 23 and as the disease progresses, there is severe atrophy of thymus and bursa. 66,109 Tracheal hemorrhages have been rarely described, but were notable features in many chickens infected with the CA02 isolate, especially in the cranial portion of the trachea, and were the consequence of necrosis in the laryngeal tonsils. 117 Comb and/or wattle edema are variably present. 81 Eyelid edema and hemorrhage are consistent findings in animals inoculated via the conjunctival route. 23,87
The most unifying histologic feature is severe necrosis of the lymphoid tissues scattered throughout the body, most especially prominent in spleen and gut-associated lymphoid tissue, which corresponds to the foci hemorrhage and ulceration noted grossly. 9,23,64,67,110,119 In the less severe, or initial stages, there is lymphoid depletion and hyperplasia of macrophages with large vacuolated cytoplasm (commonly referred to as the “starry-sky” effect). In later stages, there is accumulation of cellular and karyorrhectic debris, pyknosis, and numerous macrophages with vacuolated cytoplasm that contain nuclear debris (tingible body macrophages). 23,64,66,109,117 In the thymus, very early in the infection, there is almost complete necrosis of the cortex. The medulla usually has less severe lymphoid depletion. In the bursa of Fabricius, there is severe loss of lymphocytes both in the cortex and medulla of numerous follicles. The intrafollicular epithelial cells become prominent, and there is accumulation of numerous macrophages. Occasionally, there is formation of epithelium-lined cysts within the lymphoid-depleted lobules. Although numerous follicles are affected at the same times, it is not unusual to observe severely affected lobules adjacent to less affected or normal ones.
Microscopic changes in the brain are minimal with VVND, even in birds dying with neurologic signs. Perivascular cuffing is occasionally described. 9,23,64,66,109,117 One report of a field outbreak caused by a VVNDV strain describes multifocal necrotizing encephalitis characterized by multifocal extensive areas of malacia. However, when the same isolate was inoculated into specific pathogen–free (SPF) chickens, no encephalitic lesions were observed, suggesting a co-participation of other factors in the pathogenesis of the field lesions, rather than just pathogenic potential of the virus alone. 88
Occasionally, vascular changes such as hydropic degeneration of the media, hyalinization, and development of hyaline thrombosis are reported in VVNDV infections. 9,87 However, in numerous experiments involving SPF chickens infected via eye-drop instillation, 22, 23, 64–66, 109, 117, 120 vasculitis has never been a prominent feature. Most likely, it is possible that vessels adjacent to intense inflammatory foci might undergo nonspecific inflammatory changes that alter the vessel’s morphologic appearance. In the experience of the authors of the current study, vessels adjacent to primary areas of infection can show some degree of hyalinosis, most likely caused by intense exudation of proteins.
With respect to other body organs, VVND infection has been reported to cause multifocal areas of necrosis in the pancreas, liver, and gall bladder. 9,64,66,109,117 A 2008 NDV outbreak in a poultry facility in Japan 88 was characterized, among other lesions, by hepatic necrosis. When the same strain was inoculated in SPF chickens, fibrin thrombi occurred in the liver.
In general, there is a paucity of evidence that VVND affects the lung. In multiple experiments using various VVND strains in the current authors’ laboratory, there has been no indication of pulmonary parenchymal involvement. 23,64,66,109,117 One report of a VVNDV field outbreak in Japan describes a pneumonia characterized by histiocytic proliferation and tracheitis. 88 However, when the same isolate was inoculated into SPF chickens, no pulmonary lesions were observed.
Velogenic neurotropic Newcastle disease
Morbidity with VNND often reaches 100%, and mortality is usually 50% (but can rise to 100% in young chickens). The most prominent clinical signs are neurologic and consist of head twitch, tremors, opisthotonus, and paralysis (Fig. 2E). 9,23,113 Despite the fact that the neurologic involvement can be dramatic, the animals are characteristically bright and alert, and if able to reach food, will eat. The course of the disease is longer than with VVND, and the neurological signs are most prominent between 5 and 10 dpi, which is beyond the point of survival with most VVND strains, where animals often die at 4 or 5 dpi. 23
While, according to some key reviews, 9,113 respiratory signs are considered a prominent feature of infection with velogenic neurotropic strains, there is an absence of original reports (at least in the recent literature) that describe respiratory clinical signs or respiratory lesions in animals experimentally infected with VNNDV. When 4-week-old chickens were infected via eye-drop instillation with 4 neurotropic strains (Turkey ND, Texas GB, Cor-MI, Cor-MN) respiratory distress was not observed, and the neurologic signs predominated. 23,62 To the authors’ knowledge, only one original report, published in 1976, of experimental infection with VNNDV, described severe respiratory signs (i.e., mouth breathing and gasping by 4 dpi) followed by nervous signs at 11–12 dpi. 108
Gross lesions are often absent, and the involvement of the visceral organs appears to be minimal, although animals euthanized in the early stages of disease may have splenic or proventricular congestion. 23 Despite the neurotropism of these strains, gross lesions in the central nervous tissue are not present. 9,23 In comparison to VVND, there are no characteristic gross lesions for VNND. In fact, in most cases, gross lesions are completely absent.
Histopathologic changes in chickens infected with VNND strains are largely restricted to the central nervous system. There is multifocal mononuclear perivascular cuffing, associated with hypertrophy/hyperplasia of vascular endothelium, moderate gliosis, and multifocal necrosis of the Purkinje cells (Fig. 2F). 23,123 The lesions are more prominent in the cerebellum, especially within the molecular layer, where they first appear around 5 dpi. 123 Nervous lesions are most prominent 5–10 dpi. Other reported histologic lesions with VNNDV are lymphoid depletion, and myocarditis. 23 No reports of documented pneumonia with VNNDV were found in the literature.
Mesogenic Newcastle disease
As reported previously, 9 mesogenic viruses in field conditions cause mild clinical signs, mainly respiratory. Field outbreaks with mesogenic strains also have been associated with a drop in egg production and misshapen eggs. 9 Concurrent viral and secondary bacterial infections are thought to be common complications of mesogenic NDV that result in more severe morbidity. 9,19,33,89,113
In contrast to what is observed in the field with mesogenic strains, experimental inoculation of SPF chickens with most mesogenic strains causes very minimal clinical signs (mostly slight depression), but not any signs specifically related to the respiratory system. In numerous animal experiments conducted with similar methodology, mesogenic strain infection will in rare cases result in neurologic signs, similar to those observed with VNND, but much milder, and with lower mortality rates. 64,109,110
Gross lesions with mesogenic strains are minimal. As shown previously, 23 SPF chickens infected with mesogenic strains had mild splenomegaly and some degree of conjunctivitis when inoculated via eye-drop instillation. In the field, infection with mesogenic strains is often associated with secondary bacterial infections, which have their own set of morphologic correlates. 6,8,9
Histologically, there is a range of changes seen with mesogenic strains. The more virulent strains, those that cause a notable degree of clinical disease, consist mainly of nonsuppurative encephalitis that has many similarities to the cases caused by the VNND strains (i.e., perivascular cuffing and gliosis). Some birds may also have myocarditis, especially within 5–10 dpi. 23,64,109 In addition, splenic and pancreatic necrosis 126 can be observed.
When pigeon-isolated strains that were considered to be mesogenic after passage in chicken 65 were inoculated in 4-week-old chickens via eye-drop instillation, nervous lesions were the most severe, and were characterized by perivascular cuffing, gliosis, chromatolysis, and neuronal necrosis, all of which were most prominent in the cerebellum and medulla oblongata between 5–10 dpi. 63,66 In the same studies, multifocal myocardial necrosis and mild splenic necrosis were observed.
Lentogenic Newcastle disease
It is generally accepted that lentogenic viruses do not cause disease in adult chickens. Although some textbooks refer to La Sota as causing severe respiratory disease in very young animals, no peer-reviewed references could be found in the scientific literature. 9 When the lentogens B1 and QV4 were experimentally inoculated into 4-week-old chickens, 23 or when QV4 was inoculated into 7-week-old chickens, 43 in both cases via eye-drop instillation, no clinical signs were observed.
Some lentogenic isolates in Australia have been associated with respiratory disease in commercial broilers in the field (“late respiratory syndrome”) with very low mortality, detectable gross lesions (reddening of the trachea), and chronic nonsuppurative tracheitis histologically. 48 However, Escherichia coli was consistently isolated from the tracheas of the diseased birds, indicating that the clinical disease may well have been multifactorial in nature. 47
In another report, there were mild clinical signs consisting of rales, coughing, anorexia, and depression observed between 2–12 dpi when a lentogenic strain (Ishii) was aerosolized at high concentration into 40-day-old SPF chickens. 68 However, the high dose delivered directly to the respiratory system may have affected the clinicopathologic syndrome.
Lentogenic strains produce minimal, if any, gross lesions. 9 In one report, mild pulmonary hemorrhages and splenomegaly were described with the QV4 strain, when inoculated via eye-drop instillation. 43 In another experiment, inoculation of B1 and QV4 via eye-drop (using the current authors’ laboratory standard methodology and dosing) caused no gross lesions. 23 In a 1999 report, in which fields outbreaks are described in farmed broilers in Australia, lentogenic strains of NDV had been isolated together with E. coli, and gross lesions consisted mainly of tracheal hemorrhages 48 ; when the same NDV isolate was experimentally inoculated into SPF chickens, no gross lesions were detected. 47
Histologic changes seen in lentogen-infected chickens are minimal. When the lentogenic strains B1 and QV4 are inoculated via eye-drop into 4-week-old chickens, hyperplasia of the lymphoid follicles in spleen and air sacs were present. 23 In a similar experiment with QV4, but with slightly older birds (7 weeks), there was lymphoid follicle proliferation mainly in the lamina propria of the trachea. 43 Some lentogenic isolates in Australia caused nonsuppurative tracheitis in association with E. coli in field outbreaks, or, when experimentally inoculated in SPF chickens, induced mild changes, including lymphocytic infiltration, loss of cilia, and squamous metaplasia in the proximal trachea. 47,48 Aerosol delivery of lentogenic virus in an experimental setting commonly results in tracheal changes (i.e., deciliation, congestion, goblet cells hyperplasia, edema, and multifocal submucosal infiltration of scattered heterophils, lymphocytes, and plasma cells). 68,80 In one report of B1 infection via aerosol and air sac instillation, the main lesion consisted of lymphoid follicle proliferation in the lung and in the air sacs. 42
Newcastle disease virus in other avian species
Turkeys
Turkeys are susceptible to NDV, and clinical signs are similar to those present in chickens but are less severe. 10,20 As shown previously, 117 3-week-old SPF and 6-week-old commercial turkeys infected with CA02, a VVNDV isolated from the 2002 outbreak in California, all became sick, with clinical signs first appearing around 2 dpi, and all were dead or humanely euthanized by 5 dpi. Clinical signs consisted mainly of depression, nasal discharge, blood-tinged diarrhea, and incoordination. In the same study, commercial turkeys appeared more resistant than SPF turkeys. In another experiment, 100 6-week-old SPF turkeys infected with the velogenic viscerotropic strain CA1083 had body tremors, dyspnea, and incoordination, and all died by 7 dpi. Those infected with the Turkey ND and Iowa 1519 (velogenic neurotropic) strains were recumbent and uncoordinated, with leg and wing paralysis, and head twitching by 5 dpi, with all animals infected with Iowa 1519 strains dying by 7 dpi. In the same experiments, 4-week-old commercial turkeys had less severe clinical signs, and longer disease progression.
Turkeys infected with VVND strains developed splenic necrosis and/or splenomegaly, conjunctivitis, multifocal areas of hemorrhage and ulceration mainly in the small and to a lesser extent large intestine, multifocal hemorrhages in the upper third of the trachea that are associated with necrosis of the laryngeal tonsils, just caudal to the epiglottis, cloudy air sacs, and multifocal pancreatic necrosis. Histologically, the most prominent lesion was lymphoid depletion and necrosis of the organs, and ulceration of the intestine overlying the affected lymphoid patches. 100,117 Turkeys infected with VNNDV (Turkey ND and Iowa 1519) had multifocal gliosis and perivascular cuffing in the brain, necrosis of Purkinje cells, multifocal necrotizing myocarditis, and mild lymphocytic infiltration within the airsacs. 100 Turkeys infected via eye-drop with Roakin strain had conjunctivitis, splenomegaly, multifocal myocardial necrosis, lymphocytic infiltration in tracheal mucosa and airsacs, and by 10 dpi, multifocal areas of pancreatic necrosis. Infection with La Sota (a lentogen) via eye-drop did not cause any lesions. 100 In turkeys aerosolized with a very high dose of lentogenic NDV (La Sota, B1, ET, 2024), the most prominent lesion was mild to moderate fibrinonecrotizing tracheitis. 1
Ducks
Ducks can become infected and spread the virus; however, no clinical signs are usually reported when animals are experimentally infected, even with velogenic strains. 91,94 Only one paper describes neurological signs in ducklings that were experimentally infected with a mesogenic strain that was responsible for a previous field outbreak in ducks. 46
Geese
Geese are considered susceptible to infection, but the development of clinical disease is variable. There have been numerous reports 50,53,74,121,127 of clinical disease in geese caused by NDV strains in China. As reported previously, 121 the same field isolates were able to experimentally reproduce similar clinical disease in SPF geese. The involved strains belonged to genotypes VIId (isolated more frequently), VI, and IX. Clinical signs started to appear at 3 dpi and were characterized by moderate to severe depression, anorexia, diarrhea, ocular and nasal discharges, and swelling of the eyelids. Deaths occurred between 3–12 dpi. 121
Both natural and experimental infections of geese with some novel strains circulating in China 121 were characterized by multifocal areas of ulceration and hemorrhages in the esophagus, gizzard, and multifocal necrosis of the intestinal mucosa. Histologically, there was ulceration and fibrin deposition in the intestinal mucosa and over the cecal tonsils, severe atrophy of lymphoid organs, and lymphoid depletion in some animals, multifocal areas of necrosis in the pancreas and, less frequently, in the liver. In a few cases, brain was affected, with neuronal degeneration present.
Pigeons
Birds in the Columbiformes order, which includes pigeons and doves, can be infected with NDV. 34,117 But most ND in pigeons is due to pigeon-specific viruses, which are known as Pigeon paramyxovirus-1 (PPMV-1) to distinguish them from the rest of the APMV-1 viruses. The first PPMV-1 outbreak was reported in the Middle East during the 1970s, then spread to Europe during the 1980s, 73 and at present is considered to be endemic worldwide. 116 Clinical signs in pigeons vary mainly according to age. In young animals, mortality can reach 100%, whereas in adults, mortality is minimal, and morbidity is approximately 10%. The incubation period is 10–14 days, and viral shedding can be observed beginning at 2 dpi. 13 Clinical signs consist mainly of nervous signs (most prominent in young birds) and diarrhea. 9,116
Unlike PPMV-1, NDV strains isolated from chickens, even when highly virulent, cause minimal or no clinical disease in pigeons. In a previous study, 117 eye-drop instillation of CA02, a velogenic viscerotropic strain, caused observable clinical disease (mild tremors) in only 1 out of 10 inoculated birds. In other studies when pigeons were inoculated via eye-drop with CA1083 (also a VVND), clinical disease was observed in 9 out of 21 juvenile birds and in 5 out of 10 adult birds. Clinical signs consisted of head tremors, wry neck, opisthotonus, wing droop, and leg paralysis. In the juvenile pigeons, 7 of 21 died, and in the adults, only 1 in 10 died. 34
Lesions associated with NDV in pigeons vary according to the virulence of the strains. In general, isolates from chickens cause minimal pathological changes, whereas pigeon variant of NDV (PPMV-1) can cause a series of lesions that vary based upon the age and the inoculation route. 61 Gross lesions in pigeons infected with PPMV-1 from natural outbreaks consist of pancreatic necrosis, enteritis, and proventricular hemorrhages. Histologically, lesions consist of nonsuppurative encephalitis, multifocal necrosis in spleen, bursa, liver, larynx, and pancreas, and multifocal accumulation of lymphocytes in several organs. 79,96,126 Grossly, pigeons infected with viscerotropic velogenic CA02 showed only moderate spleen enlargement at inspection, while histological lesions consisted mainly of perivascular cuffing and gliosis in the cerebellum and brainstem by 14 dpi. 117
Upland game birds
Partridges and pheasants are considered to be extremely susceptible to NDV. 3,14 Clinical signs have been considered similar to those observed in chickens, and can span from acute onset with rapid death, severe nervous signs, to inapparent infection. 9,30,60 Also, lesions are similar to those observed in other poultry. 9
Cormorants
There are several reports describing NDV outbreaks in double crested cormorant (Phalacrocorax auritus) populations. Reports from the field describe mainly neurologic signs 15 ; however, experimental infection of cormorants infected with strains isolated from recent outbreaks did not cause mortality in 16-week-old cormorants. Clinical signs including tremors and ataxia were recorded in a small number of inoculated birds. 70
In field reports, 15,17 the most prominent gross lesions were enlarged and mottled spleen associated with bursal atrophy and multifocal hemorrhagic foci in the meninges. Histologically, lesions were multifocal nonsuppurative encephalitis with areas of gliosis, which appeared more prominent in the cerebellar white matter, interstitial nephritis, and multifocal myocarditis. In experimentally infected 16-week-old cormorants, no lesions were observed at necropsy. 70
Pet birds
In psittacine birds, clinical signs vary from inapparent to severe neurologic disease. The incubation period is usually short (2–3 days) but can be up to 14 days. 113 In a survey including 7 species of wild birds in a zoological collection, 3 species of parrots (macaw parrot, white cockatiel, red breasted parakeet) shed VVND without showing clinical signs. 102 In 1991, cases were reported 95 of ND in psittacine birds from 6 states of the United States, and in 4 of which the disease assumed outbreak proportions. Clinical signs included tremors, lateral recumbency, respiratory distress, greenish diarrhea, ruffled plumage, and head drawn back between the shoulders, and often death. The isolated viruses were categorized as VVNDV. Birds affected were yellow-headed Amazon parrots (Amazona ochrocephala oratrix), yellow-naped Amazon parrots (Amazona ochrocephala auropalliata), cockatiels (Nymphicus hollandicus), and conures (Aratinga spp.).
In another study, 35 aerosol exposure of budgerigars (Melopsittacus undulates), Amazon parrots, and conures with a VVNDV isolated from an Amazon parrot, caused mainly neurological signs consisting of tremors, ataxia, wing droop, and uni- or bilateral leg paralysis that culminated about 2 weeks post infection. Animals with bilateral leg paralysis died, while those with single leg paralysis adapted to the condition or recovered. Budgerigars showed the most severe signs, followed by Amazon parrots and conures. In the same study, budgerigars, Amazon parrots, and conures were able to spread the virus and infect cage-mates.
Psittacine birds also have been implicated in the maintenance of NDV infections and in transmission of the disease to poultry species. The California outbreak of 1971 has been traced back to a psittacine isolate. 115 This appears even more important since excretion of VVNDV has been shown to last for more than 1 year after exposure in Amazon parrots and for more than 80 days in budgerigars. 35
Gross and histological lesions in psittacines after NDV exposure are not well documented. In one report, 35 following aerosol exposure to VVNDV, conures, Amazon parrots, and budgerigars had hemorrhages and necrosis of the intestinal mucosa, hemorrhages on the skullcap and around the orbit, fibrinous peritonitis, hepatosplenomegaly, focal hepatic necrosis, airsacculitis, and hemorrhagic tracheitis.
Canaries (Serinus canarius) show variable clinical disease. In one study, 35 aerosolization of a VVNDV strain into canaries caused viral shedding and cumulative mortality of 25%; however, no characteristic clinical findings were observed before death. In another study, canaries showed very low mortality upon infection with a VVNDV strain and, when present, clinical signs included severe depression prior to death and neurological deficit (Terregino C: 2004, Evaluation of vaccination against Newcastle disease in canary birds. In: Proceedings of the 53rd Western Poultry Disease Conference, March 7–9, Sacramento, CA).
Immunohistochemistry and in situ hybridization as tools for understanding the pathogenesis of NDV
Immunohistochemistry (IHC) and in situ hybridization (ISH) have been used extensively to study the distribution of NDV protein in the tissues of infected birds. 23, 49, 64–66, 76, 93, 109, 110, 117, 120, 123 Studies have allowed a more thorough understanding of the tropism and distribution of the virus. As such, they help to supply information about pathogenesis and so can clarify for the diagnostician the pattern of lesions seen grossly and histologically. 21 Immunohistochemistry and ISH are not in routine use as diagnostic assays but the information from the experimental studies can supply a roadmap in understanding the clinicopathologic picture presented with the various pathotypes. The pathotypes (VVNDV, NVNDV, mesogens, and lentogens) all appear to have different tropism and viral distribution in the body tissues.
Velogenic viscerotropic Newcastle disease
Animal experiments in chickens conducted with similar methodologies 23,64-66,109,117 showed that the VVNDV strains, in comparison to the other pathotypes, have the most intense and widespread distribution of virus in various tissues. Virus was constantly found as early as 2–3 dpi in the lymphoid tissues throughout the body, including thymus and bursa (primary lymphoid organs), spleen, and cecal tonsils. Within the spleen, both by IHC (Fig. 2G) and ISH, signal was mainly observed in the macrophages surrounding the penicilliary arteries, whereas in the other lymphoid organs, virus was detected in the center of the lymphoid follicles, in cells morphologically compatible with macrophages and lymphocytes. In the cecal tonsil, NDV first appears in cells morphologically consistent with macrophages within the lamina propria (Fig. 2H), and this is followed rapidly by depletion and ulceration. Other sites where NDV immunohistochemical signal has been observed are the conjunctiva, nasal turbinates, multifocally and rarely in the esophageal mucosa, crop, bone marrow (in the lymphoid dependent areas and within the osteoclasts), in the epithelium of the comb, interstitium, and proximal tubules of kidney, in Kupffer cells in the liver, in cardiac myocytes, pancreas, and in epicardial and pericardial lining cells. The VVND viruses have been detected by both IHC and ISH within scattered neurons in the cerebrum and cerebellum, and in the submucosal and myenteric plexuses within the intestine. 88,109 In the respiratory tract, moderate signal has been detected in laryngeal tonsils by IHC, alveolar septa by ISH, and occasionally in the epithelial cells lining both air capillaries and atria and within the epithelium of the air sacs, by IHC. 23,109,117 So it appears that after initial replication in lymphoid tissue, VVNDV can have a very extensive dissemination to multiple body systems.
Turkeys had viral distribution very similar to that observed with chickens, with the main target of viral replication being the lymphoid organs. 100,117 Pigeons infected with CA02 had minimal positive signal only by ISH in bursa, spleen, and thymus. 117
Velogenic neurotropic Newcastle disease
In chickens, the VNNDV strains Texas GB and Turkey ND were detected only in the brain (few scattered neurons), myocardium, and air sacs by ISH or IHC. 23 In general, the detection signals increase with time, starting at 5 dpi and increasing until the animals become severely clinically ill. 23 In turkeys infected with VNNDV, distribution of immunohistochemical labeling was similar, and mainly detected in cerebellum, pancreas, and heart. 100
Mesogenic Newcastle disease
Numerous IHC and ISH studies done on tissues of chickens infected with mesogenic strains demonstrate that localization of the virus is mainly limited to the site of inoculum (mainly conjunctiva), heart, and in those strains that cause neurologic disease, the brain. 63,64,66,109 In these neurologic cases, immunolabeling or hybridization demonstrate that the virus is preferentially located in the clustered neurons within the cortex, medulla oblongata, scattered Purkinje cells, and within multifocal areas of the molecular layer in the cerebellum. With Roakin, Anhinga, Pigeon TX, Pigeon GA, Pigeon 84, and 84-44407 strains (all mesogens), viral messenger RNA or NP was constantly detected in clusters of cardiac myocytes (usually associated with areas of inflammation) by 5 dpi. 23,63,64,66 With ISH, Roakin and Anhinga strains were also detected in the air sac epithelium. 23 Rare positive cells by ISH were observed in the spleen of animals infected with the Anhinga strain. 23 Turkeys infected with the Roakin strain showed minimal immunolabeling by IHC, only in few cells in the heart and crop. 100
Lentogenic Newcastle disease
Systemic detection of lentogens is challenging. In an experiment conducted with 4-week-old chickens inoculated via eye-drop instillation, hybridization with the B1 strain occurred only in very small amounts in air sac epithelium and in the myocardium; the QV4 strain was detected only in the heart. 23 Immunolabeling for NP was detected only minimally in few epithelial cells of the trachea in commercial turkeys infected with the La Sota strain. No systemic spread was observed. 100
Newcastle disease infection and respiratory pathology
Newcastle disease virus is often grouped with the respiratory pathogens, and there seems to be a common assumption that NDV is primarily a respiratory disease. Based on a review of the literature as well as the current authors’ own experimental findings, it seems advisable to caution against labeling NDV as such.
The velogens have specific tropisms for lymphoid tissue (VVND) or central nervous system (VNND). Although respiratory tissues may show evidence of infection, as detected by IHC or ISH, the involvement of this system seems relatively minor compared to the massive damage to other body systems. The mesogenic strains have a particularly wide array of clinicopathologic presentations, with experimental infection resulting in anything from mild depression to severe neurologic impairment. Field outbreaks associated with mesogenic strains are often reported to be respiratory in nature, although these most commonly have additional secondary bacterial pathogens. In experimental studies with mesogens, there is a lack of respiratory involvement.
Although some lentogenic NDV strains have been shown experimentally to cause moderate lesions in the respiratory system, these changes were obtained only through aerosolization or use of very high viral titers, 1,43,68 direct air sac instillation of the virus, 42 or when very young chickens (1-day-old) were used. 80 In the current authors’ ISH and IHC studies, viral RNA or protein within cells throughout the respiratory tract have been identified in varying degrees, especially with the VVND viruses. However, the progression to lesion development is not remarkable.
It seems that NDV is truly a kaleidoscope of disease presentations, with the most virulent viruses targeting lymphoid tissues and central nervous system, and minor presence of virus with or without pathologic changes in multiple other body systems. In the field, where secondary respiratory pathogens are always lurking, minor pathologic changes in airways induced by the less virulent NDV strains might allow for these pathogens to become established and create a respiratory syndrome.
Conventional diagnosis of APMV-1 virus Isolation of virus
Historically, diagnosis of APMV-1 infection relies on the detection of the infectious agent by virus isolation in embryonated eggs or cell cultures. 2 At present, virus isolation is the prescribed test for international trade 125 and remains the method of choice for confirmatory diagnosis or as the “gold standard” method for the validation of other techniques. 9,12,113 Interestingly, antigen immunodetection of NDV has received little attention, as demonstrated by the absence of data published in available international scientific journals on this issue. Furthermore, to the authors’ knowledge, only 2 commercial products based on immune chromatographic assay exist on the market at present. This poor development might be due to the limited information this type of assay can provide particularly in a vaccinated population (i.e., positivity for APMV-1 without additional information on the pathotype and strain involved).
Virus isolation and typing in embryonated chicken eggs
Harmonized protocols for virus isolation in embryonated eggs follow OIE standards. 125 If commercial eggs are used, the presence of specific antibodies for NDV as a result of vaccination programs or natural exposure to the antigen of the parent flocks should be checked as they may reduce the overall ability of the virus to grow, 12 particularly if samples are inoculated into yolk sac where maternal antibodies are concentrated.
As described in the OIE Diagnostic Manual, at least five 9–11-day-old SPF embryonated chicken eggs for each sample should be inoculated into the allantoic cavity, then incubated at 35–37°C for 4–7 days and candled daily to check vitality. The mortality of inoculated eggs earlier than 24 hr post-inoculation is generally considered nonspecific, although some very virulent strains if present in high concentrations in the sample may cause embryo mortality as early as 24 hr post-inoculation. Specific embryo mortality more often occurs within 3–5 days post-infection and is influenced by the virus strain, age of embryo, and inoculum concentration. In general, embryonic death is hastened when younger embryos and higher inoculum concentrations are used. Death of the embryo is often very quick if the virus is inoculated in the yolk sac and amniotic sac, while it is slower if inoculation is via the allantoic cavity.
Generally speaking, more than 85% of ND isolations are made on the first passage, with less than 10% needing one blind passage. Isolation of ND viruses after 2 blind passages is considered very rare. 69 To accelerate the final isolation, it is possible to carry out 2 passages at a 3-day interval, obtaining results comparable to 2 passages at 4–7-day intervals. 12 The allantoic fluid containing dead embryos, or those chilled at the end of the fourth through seventh day, are tested for hemagglutinating (HA) activity, as hemagglutination is a key feature of ND viruses. However, avian influenza (AI) viruses and other avian paramyxoviruses will also cause hemagglutination, so distinction is essential. If HA activity is detected, the hemagglutinating agents should be identified by means of the hemagglutination inhibition (HI) test, which uses specific sera, or by molecular tests, which may provide information on the pathotype and genotype.
Some APMV-1 strains lose the hemagglutinating capacity when heated at 56°C for 5 min, but retain infectivity for chicken embryos even after 30 min at the same temperature. Influenza viruses, instead, always lose their infectivity before the loss of HA ability. On the basis of the response to heat-treatment, it may be also possible to distinguish between 2 types of lentogenic viruses. In fact, classical vaccine viruses (e.g., La Sota or B1 strain) can be heat-inactivated while other lentogenic viruses as well as mesogenic and velogenic strains remain infectious after the treatment. 77
In the HI test, some level of cross-reactivity may be observed among the various avian paramyxovirus serotypes. Cross-reactivity can be observed between APMV-1 and APMV-3 viruses (particularly with the psittacine variant of APMV-3, commonly isolated from pet or exotic birds) or APMV-7. The risk of mistyping an isolate can be greatly reduced by using a panel of reference sera or monoclonal antibodies (mAbs) specific for APMV-1, APMV-3, and APMV-7. The use of mAbs also permits characterization of antigenic differences within different strains of APMV-1 or even between subpopulations of the same strain. 104
Other more traditional laboratory techniques, such as agar gel immunodiffusion, fluorescent antibody test, hemolysis test (APMV-1 viruses cause hemolysis while influenza viruses do not), or the identification of virus particle morphology by electron microscopy can be applied but such methods allow only a generic identification of APMV without any information on pathotype.
Virus isolation in cell cultures
Suspension of homogenated organs, feces, or swabs prepared as for isolation in eggs may also be used for attempted isolation in cell cultures. The APMV-1 strains can replicate in a variety of cell cultures of avian and non-avian origin, among which the most widely used are: chicken embryo liver cells, chicken embryo kidney cells, chicken embryo fibroblasts, African green monkey kidney cells, avian myogenic, and chicken embryo–related cells. 113 Primary cell cultures of avian origin are the most receptive. Viral growth is usually accompanied by cytopathic effects typically represented by disruption of the monolayer and formation of syncytia. The virus also causes the formation of plaques, which according to the level of the cytopathic effect can appear clear, dull, or very dark and have a variable diameter from 0.5 to 4.0 mm. The majority of velogenic and mesogenic strains cause the formation of clear plaques. Effective replication and plaque formation in chick embryo cells for lentogenic viruses is conditioned on the presence in the culture of Mg2+ ions and diethylaminoethyl dextran or trypsin (0.01 mg/ml) in the culture medium.
Some strains of PPMV-1 and some strains of APMV-1 such as the nonpathogenic Ulster strain can be isolated in chicken liver or chicken kidney cells but not in embryonated eggs. 69 If possible, mainly when dealing with samples suspected of being infected with PPMV-1, virus isolation should be attempted using both substrates (embryonated eggs and primary chicken embryo cells). Since the viral titer obtained in cell culture is usually very low, additional replication steps in embryonated eggs should be performed prior to characterization of the isolate by HI or other phenotypic methods.
Application of molecular methods for APMV-1 detection and typing
Being able to identify APMV-1 using molecular techniques has become increasingly common. As for many other RNA viruses, genetic variability of APMV-1 is significant, and recent analyses have revealed the existence of 2 main distinct genetic clades, termed class I and II (Fig. 1). 31 Class I includes almost exclusively low virulence strains recovered from live bird markets in the United States or wild waterfowl worldwide, while class II is comprised of the vast majority of viruses of high and low virulence isolated from poultry and wild birds. 31,58 Within classes, relevant genetic variability exists, and several distinct genogroups have been identified in each class. 5,44,83 For example, a previous study described relevant genetic variability among class I viruses, and 9 novel genotypes within this class were identified. 58 Figure 1 schematically illustrates the genogrouping according to the 2 main distinct classifications.
Techniques based on biomolecular methods have been developed not only for the detection, but also for the rapid genetic characterization of the virus, and specifically to determine its pathotype (i.e., identification of avirulent or virulent strain). Molecular detection of APMV-1 has progressed in parallel with the increased variety of modern biotechnologies becoming available.
Origins of detection and typing of APMV-1 by nucleic acid amplification-based methods
The first documented attempt to detect APMV-1 by reverse transcription polymerase chain reaction (RT-PCR) in allantoic fluids of embryonated fowl eggs dates back to 1991. 52 Since then, a variety of laboratory protocols ranging from gel-based RT-PCR to real-time RT-PCR, restriction enzymes–based procedures, and rapid sequencing have been developed and published. Initially, RT-PCR for the detection of APMV-1 was applied to confirm the presence of the virus in allantoic fluids of embryonated chicken eggs inoculated following the standard procedures. For proper diagnosis of ND, it is essential not only to reveal APMV-1 viral RNA, but also to deduce the pathogenicity of the virus involved. Since the nucleotide and consequently amino acid composition of the cleavage site of the F gene is regarded as a major determinant for pathogenicity, 40,114 the RT-PCR protocols aiming to pathotype the virus all target this gene. Procedures for identification of NDV in allantoic fluids or tissue homogenates, with some including discrimination between virulent and nonvirulent viruses, were published by numerous investigators (Jestin V, Arnauld C: 1994, Direct identification and characterization of APMV1 from suspicious organs by nested PCR and automated sequencing. In: Proceedings of the joint first annual meeting of the National Newcastle Disease and Avian Influenza laboratories of the European communities, ed. Alexander DJ, pp. 89–97. Brussels, Belgium). 41,51,52,56,92,105 The main drawbacks of these earlier procedures were low sensitivity and/or inability to detect all strains. Nested PCR was also developed and showed good promise but this technique is prone to sample contamination and production of false-positive results, therefore it is not ideal for diagnostic laboratories with high sample throughputs. 55
A 2000 study 90 first reported the application of a 2-step nested RT-PCR followed by restriction enzyme analysis directly on clinical specimens. The procedure had high specificity, and further modification allowed discrimination between lentogenic, mesogenic, and velogenic strains. 29,90 Furthermore, the test could potentially identify dual infections (i.e., lentogenic and meso- and/or velogenic strains in the same sample). 29 Alignment of more than 300 APMV-1 F gene sequences available in GenBank and subsequent in silico determination of the presence of the BglI restriction site indicated that this approach could correctly identify the vast majority of the circulating viruses sequenced to date, with only few exceptions accounting for approximately 10% of the viral sequences tested. In the in silico simulation, all exceptions were represented by lentogenic sequences, which could potentially be misidentified as meso- and /or velogenic by the BglI restriction profile (Cattoli and De Battisti, personal communication).
Real-time PCR era of detection and typing of APMV-1
The advent of real-time PCR using fluorogenic hydrolysis (TaqMan) probes provided highly sensitive and rapid testing procedures. Generally speaking, these types of procedures showed sensitivity limits in the same order of magnitude of the nested RT-PCR assays; increased specificity of the test was due to the application of labeled probes and reduced risks of contamination by avoiding post-amplification manipulations. A previous study 4 applied fluorogenic probe–based real-time RT-PCR targeting the F gene to several virulent and nonvirulent isolates. Six distinct probes were designed in order to be capable of hybridizing with a wide panel of virulent or avirulent APMV-1, and the results obtained correlated well with the in vivo and sequencing results in 43 out of 45 isolates. Two isolates provided false-negative results likely due to variations in the nucleotides composition of the region targeted by the probes. This procedure was not tested on clinical specimens.
The increased amount of genetic sequence data available particularly in the last decade has highlighted the high degree of genetic variability of APMV-1 and particularly of the F gene. This variability may well explain the occurrence of false-negative results provided by different probe-based real-time RT-PCR protocols developed for this target 57,59,103,124 and the development of alternative protocols. To reduce the need for several distinct and expensive fluorogenic probes, a previous study 28 developed a ligase chain reaction (LCR) targeting the F gene for APMV-1 detection and pathotyping. Since DNA polymerases are not employed for amplification during LCR, misincorporation of nucleotides and generation of amplification artifacts as primer-dimer formation is greatly reduced, 54 facilitating the inclusion of many different primers in the same reaction mix to balance the large nucleotide variation of the targeted gene. The LCR was based on the discrimination of the single nucleotide “T” or “C” at position 395 of the F gene, associated to virulent or nonvirulent pathotypes, respectively.
In an attempt to reduce the risk of false-negative results due to target and/or probe mismatches, probe-free real-time RT-PCR protocols were designed based on the use of DNA intercalant (SYBR Green) 98 or light-upon-extension (LUX) fluorogenic primers. 16 By applying SYBR Green real-time RT-PCR protocol, the differentiation of APMV-1 pathotype was based on melting curve analysis subsequent to a 2-step RT-PCR amplification using primers targeting the F gene. According to the authors, the melting temperature (Tm) of the PCR amplicons were 91.25 ± 0.14°C, 90.17 ± 0.35°C, and 89.23 ± 0.27°C for lentogenic, mesogenic, and velogenic strains, respectively. 98 The test was positive in 38 out of 40 (95%) experimentally infected chickens, and the test limit of detection was 900 plasmid copies. However, this protocol was tested on a limited number of strains (e.g., only 2 lentogenic strains, 9 mesogenic strains, and 26 velogenic strains mostly coming from the same country). Considering the genetic variability of the F gene, variations in the Tm should be expected, making the predictive value of the melting curve analysis for pathotyping questionable. A protocol based on one-step real-time RT-PCR using LUX primers indicated that this relatively novel technique can be applied for APMV-1 detection but not for its pathotyping. 16 The primers designed, one of which was degenerated, showed 100% homology in most of the alignments with sequences of various origin deposited in GenBank. 16 The test, conducted on 32 virus strains of various origins and on samples collected in experimentally infected chickens, exhibited analytical sensitivity equivalent to 101.2/0.2 ml and 102.2/0.2 ml for virus culture and chicken feces, respectively, and the limit of detection was equivalent to 20 plasmid copies.
It is apparent from the literature that the high nucleotide variability of the F gene is primarily responsible for mismatches between oligonucleotides (i.e., primers and/or probes) and cDNA templates, which lead to false negatives. As demonstrated by previous studies, 39,57 the number but also the positions of the nucleotide mismatches of the probes are extremely important for the efficiency of the assay. From the above-mentioned studies, it seems that primer mismatches are better tolerated than probe mismatches.
In order to detect strains responsible for false-negative results in the molecular assays, alternative and strain-specific RT-PCR or real-time RT-PCR were developed, particularly for the genetically divergent pigeon paramyxoviruses (PPMV-1) 18 and APMV-1 of goose-origin. 67 Modifications of existing F gene–targeting protocols by the insertion of degeneracy in the oligonucleotides employed in the assays were considered 57 as well as a completely novel F-gene probe applied to a pre-existing USDA-validated protocol. 103 Also, different approaches to the amplification of the F gene have been attempted, such as the development of real-time RT-PCR using combination of distinct, shorter minor groove binder (MGB) probes within the same reaction mix 37 or the insertion of nucleotide degeneracy and locked nucleotides (LNA) in the probes. 39 In both studies, the assays were designed to rapidly differentiate virulent and avirulent strains by the application of multiple set of primers/probe. The limit of detection was established between 10 1 and 103 EID50/0.1 ml. 37,39
Although not based on real-time RT-PCR protocol, RT loop-mediated isothermal amplification assays (RT-LAMP) targeting the F gene of APMV-1 were described. 72,99 The RT-LAMP consists of an isothermal reaction, thus no need for a thermal cycler, and the final amplification products can be visualized by color change in the reaction tube, thus no need for special visualization equipment. The RT-LAMP assay for NDV has been improved recently but tested on a limited amount of reference strains, and field and experimental samples. 72 The results were promising; however, more extensive validation is needed, particularly with reference to the capability of this assay to detect different viral genogroups.
Genetic variability of APMV-1 as the main constraint to the application of molecular tests
Taking into account that variant viruses may emerge and circulate at any time, molecular-based assays targeting the hypervariable F gene could not be considered completely reliable for detection, even if indispensable for pathotyping. Other genes supposed to be more conserved were considered for the development of APMV-1 molecular detection assays. Recently, a protocol aimed at detecting and differentiating velogenic and lentogenic strains using SYBR Green I chemistry and PCR primers targeting the NP gene have been developed. 111 The assay was designed based on the fact that nucleotide sequence alignment of NP genes from different APMV-1 pathotypes revealed that velogenic and lentogenic strains showed distinct nucleotide variations at specific positions in the NP gene. The protocol developed was a 2-step real-time RT-PCR employing the first set of primer in the RT phase to synthesize 750 bp of cDNA. Subsequently, the real-time PCR was applied using a common forward primer for both velogenic and lentogenic strains and 2 reverse primers, each designed to be specific for lentogenic or velogenic strains (in both cases, the expected PCR product is 231 bp). The sensitivity limit of the assay was 3 × 105 DNA plasmid copies per reaction, 111 therefore 1,000 times less sensitive than a previous protocol for APMV-1 detection based on SYBR Green chemistry. 98 The discrimination between velogenic and lentogenic strains was based on the melting curve analysis, with the Tm being 86.0 ± 0.28°C and 87.4 ± 0.21°C for velogenic and lentogenic strains, respectively. However, mesogenic and lentogenic could not be discriminated in the study, 111 and the assay was evaluated on a limited number of field strains, mainly collected in the same region. Therefore, caution in the interpretation of the melting curve results for discrimination is imperative due to potential genetic variability among strains also for this gene.
The M gene was also found to have more conserved regions, particularly near the 5’ end of the gene. 106 These regions were considered for the development of a one-step TaqMan-based real-time RT-PCR intended to be used for screening the presence of a wide variety of APMV-1 genomes in clinical specimens collected in poultry. 124 The assay could detect approximately 103 genome copies and 10 EID50 and correlated well (>90%) with virus isolation, although not all the samples tested positive by virus isolation were also positive by this real-time RT-PCR protocol.
All of these protocols were subjected to extensive validation, and they were approved by the USDA. The protocols were applied and field evaluated in USDA-APHIS laboratories for the testing of approximately 35,000 specimens between 2004–2007 (Senne, personal communication; cited in Kim 59 ). Being considered more reliable and sensitive, the intended use of the assay based on the conserved M gene is for primary screening, with the test for F gene providing the confirmation for virulent or avirulent viruses. 57 The protocol targeting the M gene described previously 124 also has been applied to wild bird surveillance in Europe 24 and for screening of samples collected in Europe, Africa, and Asia. 25,26 The extended use of these protocols in the field has revealed the occurrence of false-negative results due to the genetic variability in the M gene. 27,57,58,124 It became evident that the highly genetically divergent APMV-1 viruses belonging to genotype 6 (class I) were not detected by the assays targeting the supposedly conserved M or NP genes in the vast majority of cases. 39,59 Therefore, novel assays more fitting of this lineage should be developed. 59 Recently, a real-time RT-PCR protocol targeting the L gene of APMV-1 has been developed with the aim of detecting class I and II viruses. In this protocol, a single primer set and 2 different hydrolysis probes were applied to broaden the applicability of the different genogroups, but a lack in sensitivity (limit of detection > 105 EID50) was revealed when the assay was tested on class I viruses. 38 It has been recently demonstrated that also within class II APMV-1 viruses, the M gene is not truly highly conserved and false negatives occurred in case of outbreak investigations or routine surveillance in poultry using the USDA-validated real-time RT-PCR assay targeting the M gene. 25,26 In this case, mismatches occurred in regions targeted by both the primers and the probe but mismatches in the primers were well tolerated, similar to what has been described for the F gene. 39,57 In contrast, the number and position of the probe’s mismatches were the cause of the false-negative results, therefore the probe was shortened and modified through the introduction of degeneracies and LNA. 25
Alternative applications of molecular assays for APMV-1
The capability of the PCR-based tests to reveal the presence of a pathogen regardless of its viability offered the opportunity to expand the applicability of protocols aiming to detect APMV-1 genome to samples where viral infectivity is supposed to be abolished, such as inactivated vaccines and formalin-fixed tissues. A previous study 107 described the development and the application of a RT-PCR protocol aiming to reveal contamination with ND viruses in inactivated poultry vaccines.
One of the main limiting factor of conducting PCR tests using inactivated vaccines as original material is the low amount and/or poor quality of target nucleic acid obtained during the phase of extraction. This is mainly due to the diluted antigen suspended in the vaccine formulation, the presence of mineral oil used as adjuvant, and the treatment with viral inactivating chemicals, which degrade viral RNA. Interestingly, in a 1995 study, 107 the pre-amplification phase consisted of a very simple treatment of the sample, and a true RNA extraction phase was not applied. The sensitivity of the RT-PCR method applied allowed the detection of 5 × 102 EID50 (in live vaccine preparations) and 105 EID50 or 0.056 HA units of NDV (in inactivated preparations). Considering the vaccine content of one vaccine dose, which is 106 and 108 EID50 in live and inactivated vaccines, respectively, the sensitivity of the method was considered to be sufficient by the authors. 107 In this study, beta-propiolactone concentrations between 0.025% and 0.1% commonly used in the manufacturing process of vaccines did not interfere with the RNA detection.
The treatment with formalin or other fixatives, use of paraffin, and the prolonged storage of tissue samples prepared for histopathological examination also negatively affect the quality of the RNA, thus the final result of PCR assays applied on this type of specimens. However, a semi-nested RT-PCR was developed targeting the NDV M gene and applied on 35 mg of deparaffinized tissue. 118 The performances of this method were compared to the results obtained by the application of IHC and ISH on samples collected in experimentally infected chickens. Although the successful and sensitive amplification in formalin-fixed, paraffin-embedded (FFPE) tissues was successfully demonstrated, the authors described some limitation of this technique applied on this type of specimen. Tissues should be fixed rapidly postmortem to reduce RNA degradation, and subsequent processing into paraffin should not be delayed because prolonged fixation in formalin can alter the targeted RNA. 118
A safe way to ship and store inactivated viruses could be represented by the Flinders Technology Associates (FTA) filter papers, which consist of chemically treated filter papers originally developed for the collection and storage at room temperature of biological samples expected to be processed for DNA analysis. An additional potential advantage of this technology is that the filters are supposed to inactivate pathogens, as demonstrated for mycoplasma and Infectious bronchitis virus (IBV). 85,86 A previous study 97 tested the use of FTA filter papers for the molecular detection of NDV isolates in allantoic fluids demonstrating the ability of this system to inactivate the lentogenic NDV strain tested (La Sota strain). Newcastle disease virus RNA was amplified even after 30 days at room temperature, but the sensitivity of the method was relatively low, detecting 105.8 EID50 /ml at the lowest limit of detection. Tissue imprints on FTA filter papers of organs, but not tracheal or cloacal swabs collected in experimentally infected chickens, provided positive results, indicating that the system could be suitable for the virus detection in cases where high viral load is expected.
An alternative and interesting application of the real-time RT-PCR detection of APMV-1 was applied for environmental air sampling during the ND outbreak in California in 2003. 45 The experiment was conducted in 2 flocks composed of 3,000 breeder chickens and 60,000 layers, respectively, using a commercially available air-sampler paired with real-time RT-PCR targeting the F gene and virus isolation. The birds were reported to show nonspecific clinical signs, but no increased mortality for the 2 days prior to sampling. Viral RNA was detected after 2 hr of air sampling in both flocks. Although the technique needs some refinement, it appears applicable for routine surveillance or targeted detection of viral pathogens, and could be used in sale yards, shipping containers, and other environments characterized by high animal density. 45
Molecular methods in the differential diagnosis of Newcastle disease
The development of multiplex assays
Differential diagnosis of ND is extremely important in case of acute diseases and sudden death. The prompt recognition of highly contagious OIE notifiable diseases such as ND or AI and differentiation from other viral infections of poultry causing similar clinical signs, such as Gumboro disease (infectious bursal disease [IBD]) is essential for the implementation of appropriate control measures and to limit economic losses 2,112,113 The RT-PCR–based assays were developed in order to test for the multiple presence of major poultry viral pathogens directly in clinical specimens, in principle limiting the cost of the analysis and reducing the turn-around time. The use of these tests could be particularly useful in diagnostic laboratories with small to medium throughputs, and with limited staff and budget but having the basic equipment and facilities to conduct PCR tests.
In multiplex RT-PCR, the detection of APMV-1 RNA is commonly associated with the detection of AI genomes. 27,36 In addition, gel-based RT-PCR protocols for the detection of poultry respiratory pathogens, such as IBV, AI, and avian pneumoviruses, or other pathogens, such as IBD virus (IBDV), have been published. 78,101
A microarray approach was also investigated for its applicability in the multiple detection of poultry pathogens. A protocol aimed at differentiating between NDV and AI was developed, with the simultaneous identification of APMV-1 pathotypes and H5 and H7 AI subtypes. 122 More recently, a protocol based on asymmetric RT-PCR in combination with oligonucleotide microarrays has been developed for the detection of 4 poultry virus pathogens, namely NDV, IBDV, IBV, and AIV with the differentiation of H5, H7, H9, and N1, N2 subtypes. 112 Despite promising results, these protocols are still restricted to research purposes, and their extensive use during field surveillance or outbreak investigation remains to be evaluated.
The genetic variability of APMV-1 pushed investigators to consider the design of multiplex assays to embrace the wide genetic variety of the APMV-1 strains. A multiplex real-time RT-PCR was developed to detect a broad range of class I and II APMV-1 viruses 59 targeting the polymerase (L) gene of class I strains coupled with an existing protocol targeting the M gene of the majority of class II strains. 124 This duplexed format had a moderate decrease in sensitivity when compared to the single format, 59 but information on its extensive use during surveillance is not yet available. More recently, a duplex one-step RT-PCR targeting the F gene of class I and II APMV-1 viruses has been published. 75 Using this assay, APMV-1 classes can be differentiated by the different size of the amplified products on agarose gel, and the pathotype can be determined by sequencing the RT-PCR product. As for the class I and II real-time RT-PCR protocol recently described, 38 this protocol has limited sensitivity (104 EID50/0.1 ml).
Conclusion
Newcastle disease is one of the most important animal diseases in the world, both for the number of animals affected every year and for the severe economic impact on the poultry industry. Rapid and reliable detection and confirmation of ND is important to help limit economic losses and contain the disease.
Because NDV can cause a wide variety of disease presentations, it is important to enhance the awareness of field personnel as well as utilizing the most efficient and accurate laboratory testing procedures. A thorough understanding of NDV pathology is important in order to recognize the disease in the field and to formulate a list of differential diagnoses. Laboratory testing is essential to confirm field suspicion, to characterize the virus, and to comply with international reporting requirements. In addition, the use of pathological investigation as a diagnostic tool coupled with the laboratory techniques of traditional virology and molecular biology can help to gather the “whole picture” and accurately characterize risk. It appears evident from the literature how the rapid detection, typing, and subtyping of NDV still remain laboratory challenges not only for the variety of disease presentations and lesions, but also for the genetic variability of the strains involved. This variability perhaps represents the major limitation in the validation and application of the current, advanced molecular techniques for NDV diagnosis. It also reminds diagnosticians of the importance of a multidisciplinary and comprehensive diagnostic approach, which should include, not only the new generation assays of the genomic era, but more traditional techniques such as histopathology, IHC, and virus isolation.
Footnotes
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
