Abstract
The aim of the current study was to determine the sensitivity and specificity of serum canine pancreatic elastase-1 (cPE-1) for the diagnosis of pancreatitis in dogs. The study was prospective, assessing dogs presenting with clinical signs similar to pancreatitis. Sixty-one dogs were recruited (49 with pancreatic disease and 12 with non-pancreatic disease). There was no significant difference in serum cPE-1 between dogs with pancreatic disease and non-pancreatic disease. However, there was a significant difference in serum cPE-1 between severe acute pancreatitis and non-pancreatic disease. A cut-off value for serum cPE-1 > 17.24 ng/ml resulted in sensitivity of 61.4% and specificity of 91.7% for diagnosis of all types of pancreatic disease. The sensitivity rose to 65.85% and 78.26% for the diagnosis of all types of acute pancreatitis and severe acute pancreatitis, respectively. Serum cPE-1 is more sensitive at diagnosing severe acute pancreatitis than chronic or mild acute pancreatitis, and has a high positive likelihood ratio. Dogs with chronic pancreatitis tended to have lower serum cPE-1 concentration, suggesting decreased exocrine function.
Introduction
Pancreatitis in dogs continues to provide a diagnostic challenge for veterinarians. The difficulty in diagnosis is due to the lack of reliable laboratory testing for all types of pancreatitis. Acute pancreatitis is defined as inflammation and/or necrosis in the pancreatic or peri-pancreatic tissues, with an absence of permanent histologic changes such as fibrosis and acinar atrophy. 20 Histologic findings alone cannot determine the severity of acute pancreatitis, therefore clinical criteria are also used. 11 Chronic pancreatitis is defined as a continuing inflammatory disease of the pancreas, with the presence of irreversible fibrosis and atrophy. When there is concurrent necrosis and evidence of irreversible histologic change then this form of the disease could be termed recurrent or “acute-on-chronic” pancreatitis. It is possible that recurrent pancreatitis is indeed very common in dogs, and this may compound the difficulties in diagnosis due to a lack of functional pancreatic mass able to produce circulating enzymes during times of inflammation. Serum amylase, lipase, and trypsin-like immunoreactivity have been shown to have poor sensitivities and specificities even in severe pancreatitis in dogs. 4,12
Pancreatic elastase-1 (PE-1) is synthesized in pancreatic acinar cells, and during active pancreatic inflammation, it is released into the blood stream at the same time or immediately after trypsin. 3 Pancreatic elastase-1 contributes to the ongoing inflammatory state during acute pancreatitis by enhancing neutrophil-mediated tissue injury, and has been shown experimentally to induce pancreatic microcirculatory failure in the presence of sera co-factors. 7 There have been inconclusive assessments about the clinical utility of serum PE-1 in human beings for diagnosing acute pancreatitis. 1,10,19 In chronic pancreatitis, the exocrine function of the pancreas may become diminished, and in human patients with chronic pancreatitis, serum and fecal PE-1 is often reduced, reflecting subclinical exocrine pancreatic insufficiency. 9 Pancreatic elastase-1 has been measured in the serum of dogs undergoing endoscopic retrograde pancreatography, 15 in healthy dogs, dogs with renal failure, and dogs with pancreatitis (Spillmann et al, J Vet Intern Med abstract 2002; 16:635). In the latter study, there was a significant difference in the median values of dogs with severe pancreatitis compared to healthy dogs or dogs with renal failure. Measurement of serum canine PE-1 (cPE-1) is not affected by lipemia according to the assay manufacturers, and is less affected by impaired renal function than most other pancreatic enzymes in human beings. 10,14 The current study was designed as a prospective project to assess the clinical utility of serum cPE-1 in the diagnosis and differentiation of pancreatic disease in dogs.
Materials and methods
Dogs presenting to 3 university veterinary referral centers (2 in Europe and 1 in Australia) with clinical signs that could be attributed to pancreatic inflammation, including both vomiting and abdominal pain, were recruited into the study. All centers operate under National Health and Medicine Research Council equivalent Animal Use codes, and the study was approved by the relevant institutional ethics committee.
Animal selection
Dogs were designated to have primary pancreatic disease on the basis of abdominal ultrasound and/or histologic findings. The dogs were further subdivided into the following groupings:
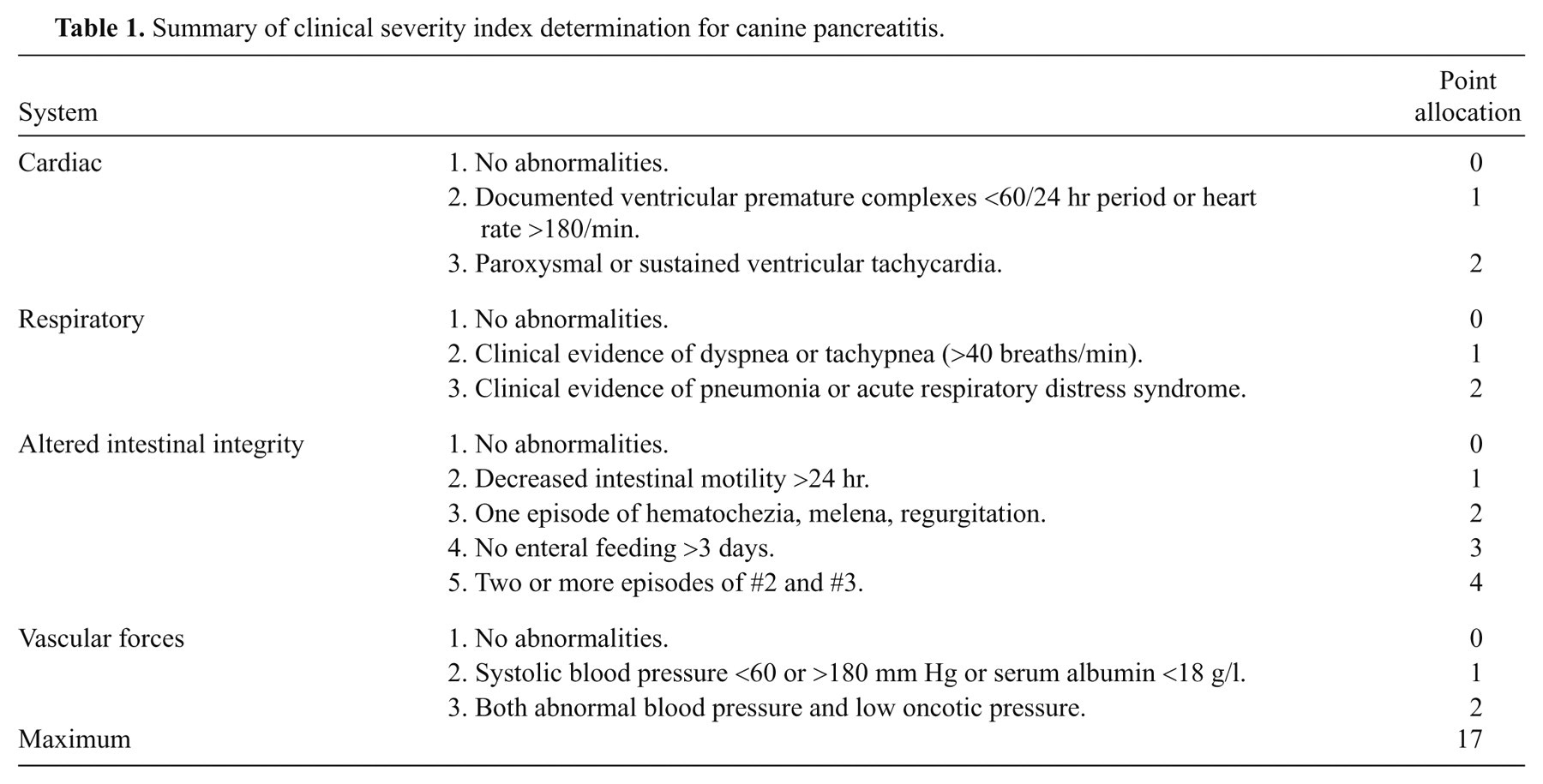
Severe acute pancreatitis: Dogs had marked pancreatic and peri-pancreatic fat necrosis evident histologically (obtained at exploratory laparotomy or postmortem), or marked peri-pancreatic hyperechogenicity combined with abnormal pancreatic echogenicity as determined by ultrasonography, 4,8,16 in addition to either a severe clinical severity index >2 11 (Table 1), or the need for intensive management.
Mild acute pancreatitis: Dogs had pancreatic necrosis on histologic evaluation (obtained at exploratory laparotomy or postmortem), but did not satisfy the clinical or ultrasonographic criteria for having severe disease.
Acute-on-chronic pancreatitis: Dogs had histologic evidence of both acute (necrosis) and chronic (fibrosis or atrophy) pancreatitis; and blood and pancreatic samples were taken during a time of active clinical signs. Clinical severity was not determined for dogs in this group.
Chronic pancreatitis: Histologic evaluation of the pancreas obtained at postmortem or exploratory laparotomy showed evidence of chronicity (fibrosis and atrophy), with no active inflammation or necrosis.
Pancreatic neoplasia: Primary pancreatic carcinoma was diagnosed via histologic evaluation of biopsies obtained at exploratory laparotomy or postmortem, with no concurrent inflammation
Summary of clinical severity index determination for canine pancreatitis.
Dogs that were classified as having non-pancreatic disease had a confirmed primary diagnosis with normal to minimal pancreatic changes observed histologically. Additionally, dogs did not have exogenous glucocorticoids within 3 days of admission.
Assay
Blood was taken from all dogs recruited into the study within 12 hr of admission to the university referral center. Separated serum was stored at −20°C, then shipped frozen to Giessen, Germany for analysis by operators blinded to the disease classification of the samples. A species-specific sandwich enzyme immunoassay based on 2 monoclonal antibodies highly specific for cPE-1 was performed. a This assay has been shown to have an intra-assay coefficient of variation (CV) ranging from 2.1% to11.6% and an interassay CV ranging from 3.4% to 10.8% in dogs. 15 An earlier abstract of 16 healthy dogs had a minimum value of 32.1 ng/ml, and a maximum value of 659.3 ng/ml, with a median concentration of 55.8 ng/ml (Spillmann et al, J Vet Intern Med abstract 2002; 16:635). Unfortunately, the mean values were not reported in that study.
Statistical analysis
Statistical software package b was used for all calculations. The sensitivity and specificity of serum cPE-1 was calculated using receiver operating characteristic (ROC) curves to determine the optimal cut-off value for diagnosis, and 95% confidence intervals (CIs), positive, and negative likelihood ratios were also determined. Statistical difference between groups was determined using Mann–Whitney t-test at 95% CI, with significance being a P value < 0.05.
Results
Animal information
A total of 107 dogs were initially recruited into the study, but there was only sufficient information obtained in 61 dogs to be included. Forty-nine had pancreatic disease (severe acute pancreatitis, n = 23; mild acute pancreatitis, n = 11; acute-on-chronic pancreatitis, n = 7; chronic pancreatitis, n = 3; and pancreatic carcinoma, n = 5). A further 12 dogs with non-pancreatic inflammatory disease were evaluated: 4 dogs with inflammatory bowel disease, 2 dogs with small intestinal foreign bodies, 2 dogs with renal failure, and 1 each of immune-mediated thrombocytopenia, gastric carcinoma, multi-centric lymphoma, and insulinoma.
Median and intervals
The median, mean, and intervals for serum cPE-1 in all groups are presented in Table 2, and graphically in Figure 1. Overall, the median and mean for all dogs with pancreatic disease was 19.3 ng/ml and 47.46 ng/ml, respectively. For dogs with acute (mild, severe, and acute-on-chronic) pancreatitis, the median and mean was 20.85 ng/ml and 55.04 ng/ml, respectively. Dogs with non-pancreatic disease had a median and mean of 5.81 ng/ml and 8.19 ng/ml, respectively. There was a significant difference in serum cPE-1 concentrations between all pancreatic disease groups and the non-pancreatic disease group (P = 0.005) as well between the acute pancreatitis group (including mild, severe, and acute-on-chronic) versus the non-pancreatic disease group (P = 0.002), but not between the acute pancreatitis groups versus the pancreatic neoplasia group (P = 0.08). The difference between severe and mild acute pancreatitis was also significant (P = 0.04), and there was a strongly significant difference between severe acute pancreatitis when compared to non-pancreatic disease (P = 0.002).
Minimum, maximum, median, and mean concentrations of serum canine pancreatic elastase-1 (cPE-1) in the nominated disease categories.
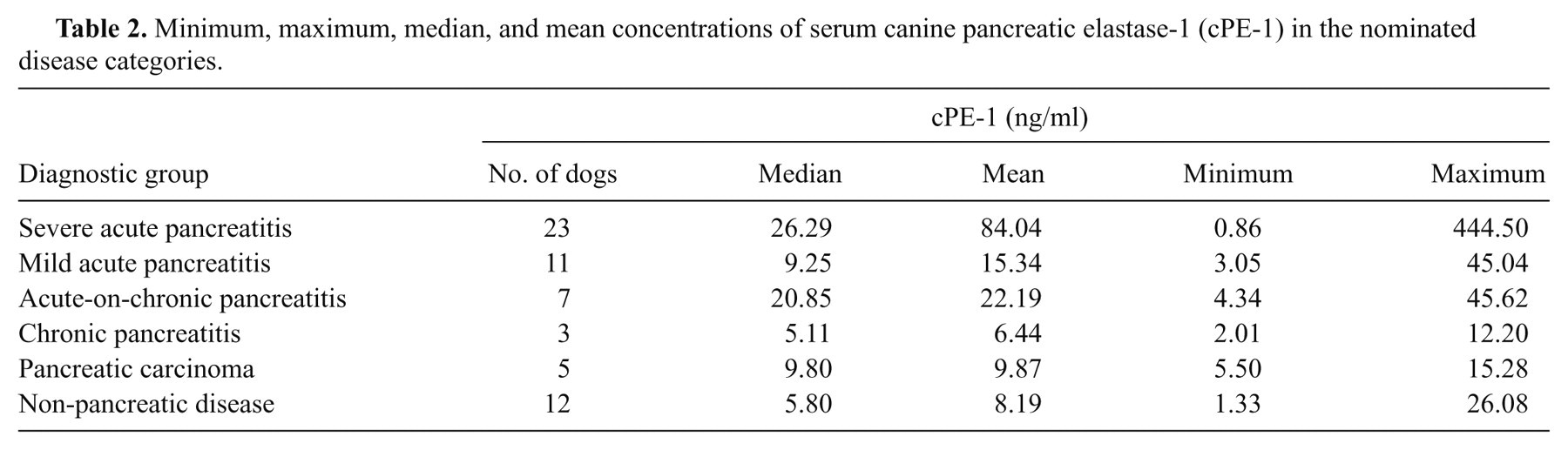
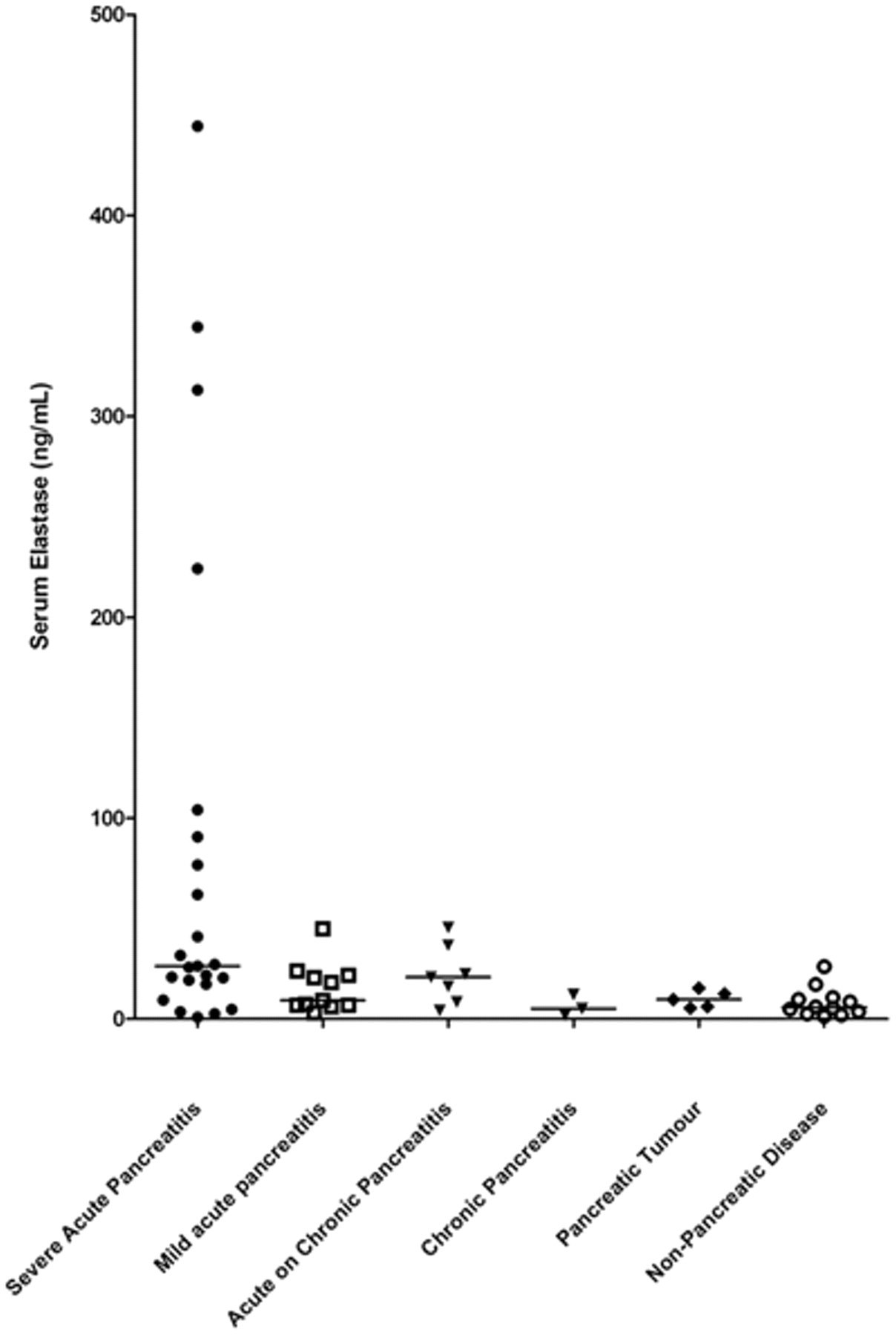
Serum canine pancreatic elastase-1 concentrations for all disease groups. There is a significant difference between the means of all groups except when comparing all dogs with acute pancreatitis (mild, severe, and acute-on-chronic) to dogs with pancreatic carcinoma.
Sensitivity and specificity
Using ROC analysis, an optimal diagnostic cut-off value of 17.24 ng/ml for serum cPE-1 was calculated to maximize the area under the curve, when comparing all groups. The 95% CIs for the sensitivity, specificity, positive likelihood ratio, and negative likelihood ratio were calculated with the cut-off value of 17.24 ng/ml, as well as with the previously reported median concentration of 5.5 ng/ml for healthy dogs (Table 3). 15 There was a high specificity for the diagnosis of severe acute pancreatitis (91.7%) using a cut-off value of 17.24 ng/ml, resulting in a high positive likelihood ratio, but a lower sensitivity (78.3%) with wide confidence intervals. The sensitivity also decreased when assessing all types of pancreatic disease (61.4%), and all cases of acute pancreatitis (65.9%), but the specificity remained the same. When the median value 5.5 ng/ml was used, the specificity greatly reduced (50%) with an increased sensitivity for severe acute pancreatitis (82.61%), all types of acute pancreatitis (85.37%), and all pancreatic disease (81.62%; Fig. 2). There was overall a low negative likelihood ratio, which was lower when assessing acute pancreatitis.
The sensitivity, specificity, positive likelihood ratio (PLR), and negative likelihood ratio (NLR), with 95% confidence intervals (CIs) were calculated for diagnosis of severe acute pancreatitis (as compared to non-pancreatic disease [NPD]), diagnosis of all forms of pancreatitis (severe acute, mild acute, acute-on-chronic, and chronic), and diagnosis of dogs with all forms of acute pancreatitis (severe, mild, and acute-on-chronic).*
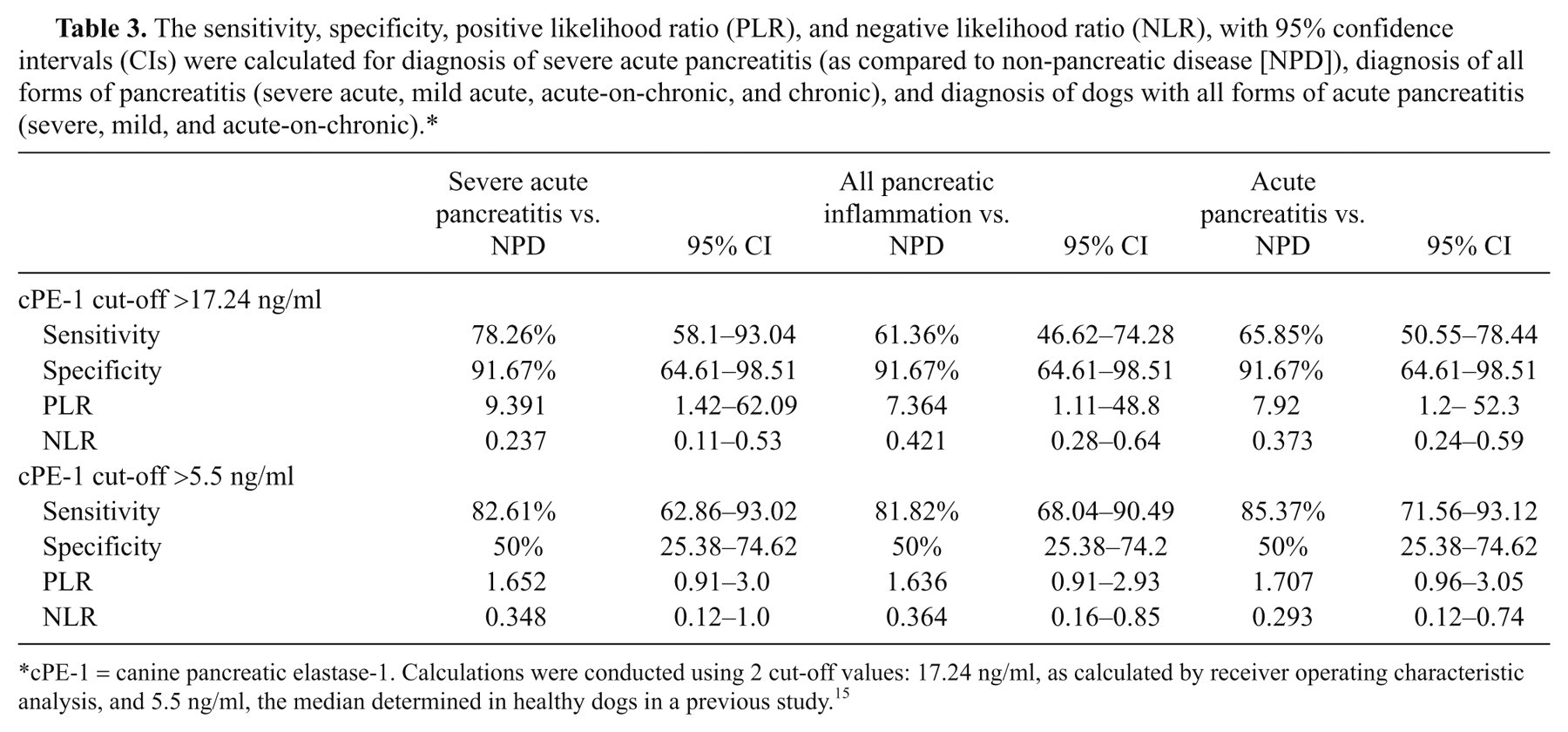
cPE-1 = canine pancreatic elastase-1. Calculations were conducted using 2 cut-off values: 17.24 ng/ml, as calculated by receiver operating characteristic analysis, and 5.5 ng/ml, the median determined in healthy dogs in a previous study. 15
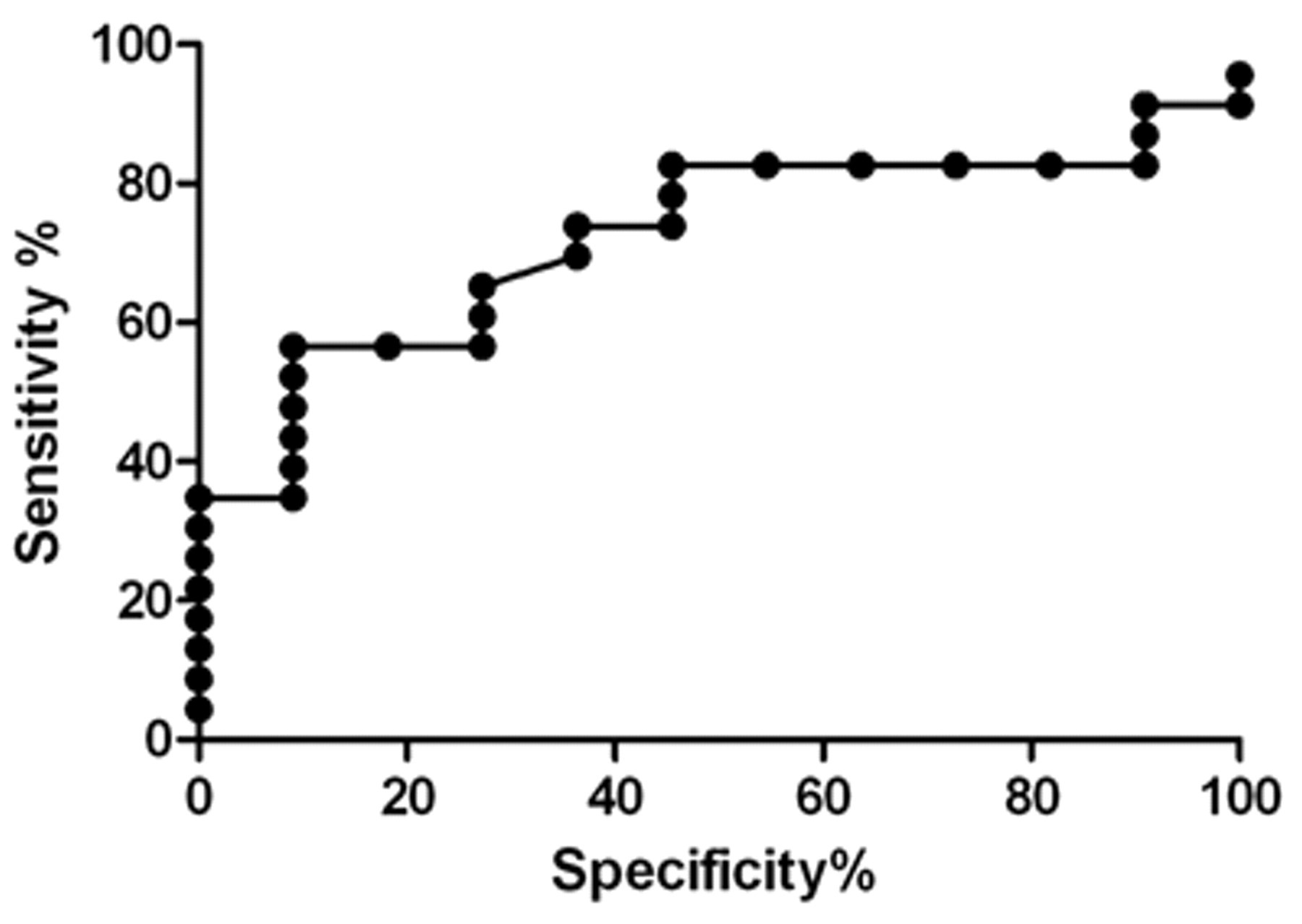
An area under the curve of 0.8261 is determined using receiver operating characteristic analysis comparing severe acute pancreatitis to non-pancreatic disease in measurement of serum pancreatic elastase-1, with a calculated cut-off value of 17.24 ng/ml.
Discussion
Serum cPE-1 had less than optimal sensitivities when assessing all dogs with pancreatic disease (61.4%) in the current study. However, when only dogs with severe acute pancreatitis were considered, the sensitivity increased to 78.3%, and there was a high positive likelihood ratio. Of significant interest was the finding of a specificity of 91.7%. This combined with a high positive likelihood ratio and low negative likelihood ratio would suggest that a high cPE-1 is likely to be due to pancreatitis. This benefit was greatest when using a cut-off value of 17.24 ng/ml to assess severe acute pancreatitis.
The current study did not assess healthy dogs, as the aim was to determine the clinical utility by comparing groups of dogs that could be considered to have pancreatitis on presentation. As such, the cut-off value determined in the present study by comparing dogs with similar clinical presentations is more clinically relevant than comparison to healthy dogs. Additionally, the authors did not compare this test modality to clinical suspicion (based on clinical signs at presentation), as 46 dogs were excluded due to an unknown final diagnosis.
Serum cPE-1 appears to be less sensitive than current modalities such as abdominal ultrasound for the overall diagnosis of pancreatic disease in dogs (61.4% vs. 66%). This finding is contrary to initial studies in human beings where good correlation with disease was demonstrated, although overlap between groups was observed. 1,9,10 Later studies did not show such a convincing correlation. 13,18,19 The major reason for this discrepancy is that the earlier studies used a radioimmunoassay (RIA), which detects polyclonal elastase (bound to 1-α-anti-trypsin complex), with a half-life of 2.2 days. As a result, the RIA detects greater amounts for a longer period. The enzyme-linked immunosorbent assay, as used in the current study, is a monoclonal assay that detects free or unbound elastase with a half-life of 0.4 days, 13 thus decreasing the sensitivity of the test after a short period of time has elapsed since onset of clinical signs. Conversely, this makes a positive result more likely to be a true positive if there is persistently an increased value.
Most human studies suggest that serum cPE-1 is most clinically useful for diagnosis of severe acute pancreatitis. This is also supported by the current findings, as well as previous reports in dogs. One abstract from 2002 describes a reference interval of elastase in 16 healthy dogs of 32.1– 659.3 ng/ml (median: 55.8); 14 dogs with pancreatitis had a range of 24–1720 (median: 160), and 6 dogs with renal disease had a range of 5–182 (median: 43.3; Spillmann et al, J Vet Intern Med abstract 2002; 16:635). When the dogs with pancreatitis were classified into severe (n = 9) or mild (n = 6) forms, there was a significant difference in serum cPE-1 between healthy dogs and dogs with renal failure compared to severe pancreatitis.
It is uncertain why so many of the healthy dogs in the aforementioned study had such high serum cPE-1 results compared to those measured in the current study. However, a later publication from the same authors also reported a wide-ranging baseline cPE-1 concentration in 7 healthy dogs (0.1–411.6 ng/ml) but with a median value of 5.5 ng/ml, similar to the dogs with non-pancreatic disease in the present study. 15 This initially wide reference interval raises the question of whether sample handling or hemolysis may have contributed to the variability of measured values. The manufacturers of the assay report that hemolysis or storage above 8°C for more than 72 hr can artifactually increase serum cPE-1 measurements. Hemolysis was recorded in only 8 of the 61 samples measured in the present study. The results of these 8 samples were consistent with diagnosis, and statistical analysis was unaffected by inclusion or exclusion of the dogs, so to mimic conditions expected in clinical practice the results were retained. In this light, more weight is lent to the premise that serum cPE-1 is highly specific.
It is also possible that the high results observed in the study of healthy animals may be outliers, have subclinical pancreatic disease, or that serum cPE-1 excretion may decrease with age, as non-renal excretory mechanisms may diminish. 14 It is not possible from the available data of the previous study 14 to evaluate these possible reasons.
Each of the 3 dogs in the current study that had severe renal failure and no pancreatic inflammation (urea: 70.4 mmol/l, reference <12; creatinine: 804 µmol/l, reference <120) had a serum cPE-1 below the calculated value. There is a strong suggestion that serum elastase is not affected by renal clearance, as compared to many other pancreatic enzymes. In a previous study of 24 healthy people and 47 people with various degrees of renal insufficiency (but no known pancreatic disease), the measurements of elastase, lipase, amylase, pancreas-specific amylase, phospholipase-A2, and trypsin were compared. 14 Elastase was less affected than the other enzymes, only increasing to a significant amount in the serum when the creatinine clearance was reduced below 10 ml/min. All other enzymes were increased when creatinine clearance fell below 40 ml/min, although lipase tended to be less so. One of the dogs with renal failure and no pancreatic inflammation in the present study had a canine pancreatic-specific lipase result markedly above the reference interval, but a serum cPE-1 concentration less than 5.5 ng/ml.
Another interesting finding was that serum cPE-1 was generally lower in the chronic pancreatitis group than the other pancreatic disease groups, as well as being below the reference interval in many dogs with acute pancreatitis. This would suggest that these dogs may have reduced exocrine pancreatic function.
No information is available regarding the time interval between onset of clinical signs and the blood sample collection for the patients included in the present study, although all samples were collected within 12 hr of admission to the veterinary referral centers. It may well be possible that cPE-1 is more useful when measured in the first 24–36 hr after the onset of clinical signs, but this strategy may not be a practical suggestion for clinical veterinary practice, and the current study was designed to assess the clinical utility of the assay. In general, animals with severe pancreatitis will likely be presented much sooner after the onset of clinical signs than those with chronic disease.
No attempt was made to characterize the clinical severity of the acute-on chronic pancreatitis group, as this was a histological-based classification. It is possible that the sensitivity of serum cPE-1 for the diagnosis of severe disease could be altered if animals in this group were re-classified. The fact that all of the dogs in the non-pancreatic disease group had pancreatic histopathology performed increases the clinical significance of findings in the present study. Although not all dogs in the severe acute pancreatitis group had histopathology performed, the combination of clinical and ultrasonographic changes were strongly supportive of a diagnosis of pancreatitis, although the presence of necrosis could not be confirmed. It is possible that some of these dogs had “acute-on-chronic” disease, and if they were excluded from the analysis then the sensitivity of the assay may be even higher. However, in veterinary practice, pancreatic biopsies are seldom performed in dogs that have severe clinical signs of disease and concurrent abnormalities on abdominal imaging.
In the human literature, it has been proposed that increased serum cPE-1 may be specific for pancreatic carcinoma, 5 but this specificity is not supported in other studies. 1 Similarly, in the current study, none of the dogs with pancreatic carcinoma had serum cPE-1 concentrations greater than 17.24 ng/ml (the determined cut-off value), which may be related to the short serum half-life, reduced functional pancreatic mass, or the lack of concurrent pancreatic inflammation. Conversely, all dogs in the severe acute pancreatitis group had follow-up of sufficient duration to rule out the presence of concurrent pancreatic neoplasia.
Abdominal imaging is often relied upon for confirmation of the diagnosis of acute pancreatitis. This modality has a reported sensitivity of 66–68%. 4,12,17 Although the sensitivity of ultrasound is reported as being low, these studies are not recent, and are assessing animals with chronic and mild disease as well as severe disease, decreasing the sensitivity of ultrasound as a result. 8 Additionally, the specificity of ultrasound is actually quite high, meaning it is unlikely that any false positives were included in the patient group with severe disease in the current study. In a 2008 study, all dogs with severe pancreatitis had a positive ultrasound diagnosis. 17 The presence of hyperechogenicity surrounding the pancreas on ultrasound reflects peri-pancreatic fat necrosis, and necrosis is considered the most significant indicator of severe disease in human beings. 2,6
Serum cPE-1 and canine pancreatic-specific lipase appear to have similar sensitivities in the diagnosis of severe acute pancreatitis (78% and 82%, respectively). 17 The use of blood tests alone to diagnose pancreatitis should not be relied upon in clinical practice, and combination with abdominal ultrasound may prove to have the highest sensitivity and specificity for diagnosis of severe acute pancreatitis in dogs. Additionally, abdominal imaging is very important to rule out non-pancreatic causes of “acute abdomen” that would require immediate surgical management, such as intestinal foreign bodies.
Serum cPE-1 is useful for the diagnosis of severe acute pancreatitis, but less so for milder forms of the disease. The diagnosis of milder or more chronic forms of the disease remains dependent on histologic confirmation for definitive diagnosis. The usefulness of serum cPE-1 in assessing residual exocrine pancreatic function following acute pancreatitis should be further evaluated.
Footnotes
Acknowledgements
The authors are grateful for the assistance of ScheBo Biotech in the United Kingdom and Germany for their logistical assistance and performing the serum canine pancreatic elastase measurements; and also to the staff of their university clinical departments and clinical pathology laboratories, in particular Amanda Lawlor and Joel Eluk.
a.
ScheBo®.Elastase 1-Canine, ScheBo Biotech AG, Giessen, Germany.
b.
GraphPad® Prism 5, GraphPad Software Inc., La Jolla, CA.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
This study was supported by the European College of Veterinary Internal Medicine Clinical Studies Fund.
