Abstract
Seven cases of hydranencephaly and cerebellar hypoplasia in a water buffalo herd are described. The herd of 133 females was divided for mating into 4 groups and mated with different bulls. The disease was observed in the offspring of 5 cows from only 1 group over a period of 6 years. These cows had all mated with the same bull during that period. All affected calves were unable to suckle, showed intention tremors, involuntary movements of the ears, depression, and blindness. Some calves were recumbent and others showed wide-based stance and inability to walk. At necropsy, all affected calves had similar lesions consisting of hydranencephaly and cerebellar hypoplasia. Histologically, the cavities were surrounded by normal-looking nervous tissue. The occurrence of the disease in the offspring of only 1 out of 6 bulls in different years, the similar lesions in all 7 calves affected, and the negative serology and immunohistochemistry for Bovine virus diarrhea virus and Bluetongue virus highly suggest that the disease is hereditary.
Congenital malformations can be hereditary or acquired in utero. The latter may be caused by viral infections, such as Bovine viral diarrhea virus (BVDV), 11,25 Bluetongue virus (BTV), and Akabane virus, 23,24 by drugs and toxic plants, such as Astragalus spp., Veratrum californicum, Lupinus spp., Conium sp., Nicotiana sp., 15 and Mimosa tenuiflora, 16 and by nutritional deficiencies. 9 Malformations of unknown etiology are also occasionally observed. 10
Congenital diseases are very frequent in the Brazilian water buffalo herd, with more than 3,000,000 head, most of them belonging to the offspring of approximately 200 animals imported from India in the 1960s. 13 This inbreeding has contributed to the occurrence of many hereditary diseases diagnosed in buffalo in Brazil, including arthrogryposis, 20 mechanobullous genodermatoses, 17 albinism and megaesophagus, 10 and myotonia due to a chloride channel 1, skeletal muscle (CLCN1) mutation (Borges AS, Mota LL, Barbosa D, et al: 2006, Nova desordem miotônica em búfalos: descrição clínica e eletromiográfica [Novel myotonic disorder in buffaloes: clinical and electromyographic description]. Proceedings of the 6th meeting of the Brazilian Society of Neurologic Investigation, May 12–13. Ribeirão Preto, São Paulo, Brazil. In Portuguese).
Recently, a previously unknown congenital neurological syndrome was recognized in water buffalo in southern Brazil. A thorough diagnostic work-up was carried out to try to determine the cause of the problem. The main objective of the current study was to describe the epidemiology, clinical signs, and pathological changes of the previously unknown syndrome and to discuss possible etiologies.
The farm where the disease was diagnosed is located in the municipality of Capão do Leão, state of Rio Grande do Sul, in southern Brazil. The herd was raised together with a herd of Charolais cattle, on native pastures and occasionally on rice stubbles. On the same farm, a herd of Jersey cattle was maintained in confinement. The whole buffalo herd was regularly treated with ivermectin at 3 months of age, and the females were treated again with the same product at 18 months of age. All animals were vaccinated against clostridial diseases, leptospirosis, and foot and mouth disease, and the young females were vaccinated against brucellosis, with a strain 19 vaccine.
The herd consisted of 133 females, which during mating season, were divided into 4 groups that were kept separately in 4 adjacent paddocks of approximately 50 hectares each. These paddocks were occasionally subjected to flooding, and they were covered with native pastures and brush. The whole area had been stocked with buffalo that were first introduced to the farm in 1980; cattle were also occasionally kept in these paddocks.
In group 1 (36 animals), the cows were inseminated with imported semen, after which they were subjected to natural service with 2 local buffalo bulls. Cows in groups 2–4 (35, 35, and 27 cows, respectively) were naturally mated with a different bull for each group.
Out of 158 buffalo calves born in a period of 6 years to cows from group 4, 7 calves were affected. After the birth of the second abnormal calf in group 4, the bull that fathered these calves was mated again to the cows of this group and with a few of his own daughters to investigate the possibility of the disease being hereditary. Cow VII-15, daughter of bull 4 (VI-9), (Fig. 1) delivered 2 affected calves, IX-18 and X-21 (Fig. 1); cow VI-12 delivered 2 affected calves, VIII-17 and IX-19 (Fig. 1). No affected calves were observed in any of the other groups.
All affected calves were submitted to the Veterinary Diagnostic Laboratory, Federal University of Pelotas (Rio Grande do Sul, Brazil) for a complete clinical and pathological diagnostic work up. Clinical signs in all affected animals were evident upon birth; they were similar in all animals and they varied only in intensity. Five calves were recumbent (Fig. 2) and could not stand up even when helped. The other 2 calves were able to stand, but showed wide-based stance. When forced to move, these calves were unable to walk and showed severe incoordination. All affected calves were unable to suckle, and they showed intention tremors, involuntary continuous movements of the ears, depression, and blindness. Intention tremors were severe in most of the buffalo, except for calf 7, which was very depressed and had only mild intention tremors. Four affected buffalo had a slightly dome-shaped skull (Fig. 2).
All affected calves were euthanized between 1 and 3 days of life. At necropsy, all buffalo had similar lesions in the nervous system (Fig. 3). The cerebral surface was flattened with almost total absence of gyri, sulci, and fissures. Attenuated gyri were observed on the occipital cortex of all animals (Fig. 3). The cerebral lateral ventriculi were severely dilated (hydrocephalus ex vacuo) and full of clear liquid. The cerebral hemispheres appeared as membranous sacs (approximately 0.5–2 mm thick; Figs. 3–5). Remnants of neural tissue were also observed as delicate membranes or thin cords crossing these cavities (Figs. 4, 5). The columna fornicis was flattened and rounded, the septum pellucidum was elongated, and the corpus callosum had variable degrees of atrophy (Figs. 4, 5). In the external capsule, small, bilateral, and symmetrical cavities (porencephaly) were observed (Figs. 4, 5). The lesions extended from the frontal to the occipital cortex. The thalamus in all affected buffalo was preserved but the hippocampus had different degrees of atrophy. In the remaining temporal and parietal cortex, there were small cystic cavities (porencephaly). The cerebellum presented varying degrees of hypoplasia (Fig. 3). No other significant gross abnormalities were observed in any of the carcasses except for a diffuse yellowish discoloration of the liver in 3 calves.
Samples of remnants of frontal, parietal, temporal, and occipital cortex, corpus striatum, thalamus, rostral colliculi, cerebellar peduncles, cerebellum, pons, obex, spinal cord (cervical, thoracic, and lumbar segments), liver, heart, lungs, kidney, lymph nodes, and muscle were collected and fixed in 10% buffered formalin (pH 7) for 3–5 days, except tissues from calf 1, which were in formalin for more than 6 months. The tissues were embedded in paraffin, processed routinely for histology, sectioned at 5 µm, and stained with hematoxylin and eosin. Selected sections of the central nervous system (CNS) were also stained with Luxol fast blue. The CNS of a buffalo from group 1 that had died shortly after birth from non-CNS disease was used as control. Upon histologic examination, the remnants of the cerebral cortex surrounding the lateral ventriculi were composed of mostly histologically normal nervous tissue (Fig. 6). The thin cords remaining within the cavities were also formed by normal-looking nervous tissue (Fig. 6). In some areas, including the temporal and occipital cortex, the gyri were still present, but the white matter was markedly rarefied, sometimes with small cavities in neuropil, occasionally containing gitter cells (Fig. 7). Also, in some of these areas, the perivascular spaces were severely dilated, and axonal spheroids were also observed (Fig. 8). Diffuse astrocytosis was observed in some areas of the white matter. In calf 6, an area of hemorrhage, perivascular cuffs of mononuclear cells, and focal astrocytosis was observed in the cortex. In the corpus striatum of calf 7, multifocal areas of mild spongiosis of white matter and dilatation of perivascular spaces were observed. The cerebellum of all affected buffalo had different degrees of hypoplasia (Fig. 1), the severity of which varied in different regions of the cerebellum of the same calf. The more affected folia, found mainly in the vermis, showed marked reduced thickness of all gray matter layers. The molecular and granular layers were irregularly thin, the granular layer had a lower cell density, and there was decreased number of Purkinje cells that were replaced by Bergmann glia. The white matter was markedly rarefied and hypomyelinogenesis was observed in Luxol fast blue stained sections. Despite these alterations the organization of the layers of the cerebellum was still normal. Frequently folia with different degrees of hypoplasia were observed in the same region (Fig. 9). No significant histological abnormalities were observed in the brainstem or cervical, thoracic, and lumbar regions of the spinal cord.
Selected sections from the CNS of all affected calves were processed by an avidin–biotin complex (ABC) immunoperoxidase test using primary antibodies against BVDV and BTV, according to the Standard Operational Protocols of the California Animal Health and Food Safety Laboratory, University of California, Davis. Immunohistochemistry for these 2 diseases was negative on all samples tested.
Blood samples of all cows from group 4 were collected for serological examination. For BVDV antibodies, serum samples were submitted to a standard virus neutralization (VN) assay, testing two-fold dilution of sera against a fixed virus dose (100 mean tissue culture infectious doses [TCID50]). Madin–Darby bovine kidney cells were used as indicators of virus replication, and readings were performed at 96 hr of incubation. All sera were tested in duplicate against 2 BVDV strains (BVDV-1 Singer; BVDV-2 VS-253). All tested sera were negative for BVDV antibodies (VN titers < 2), regardless of the strain used in the test. Antibody detection against BTV was performed by enzyme-linked immunosorbent assay with commercial kits. a The results of serological tests were all negative for BTV.
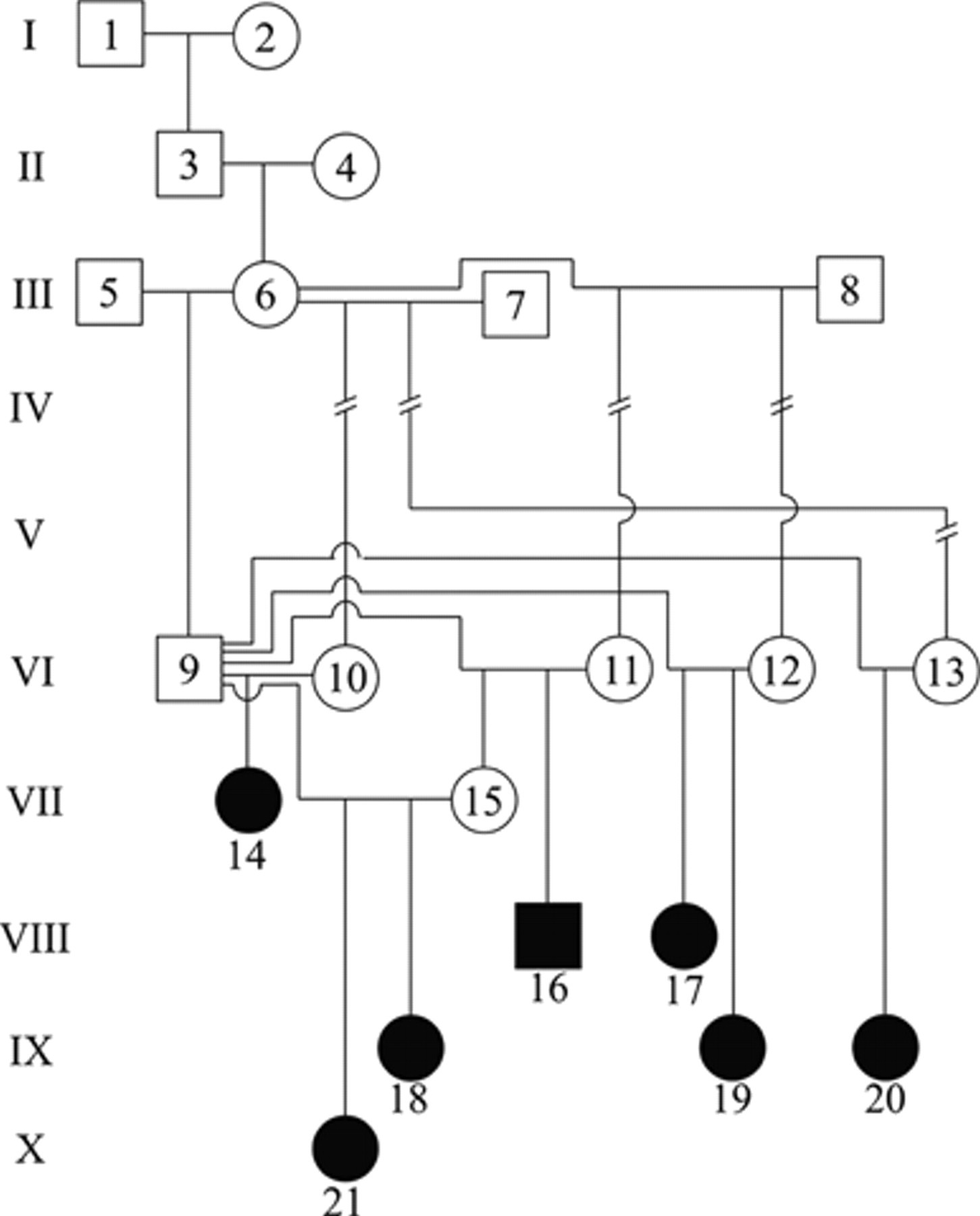
Genetic analysis of bull 4 (VI-9) and cows from the affected group. Solid black circle indicates affected female; solid black square indicates affected male.

Calf 4 affected by congenital hydranencephaly showing recumbence, depression, and a slightly dome-shaped skull.
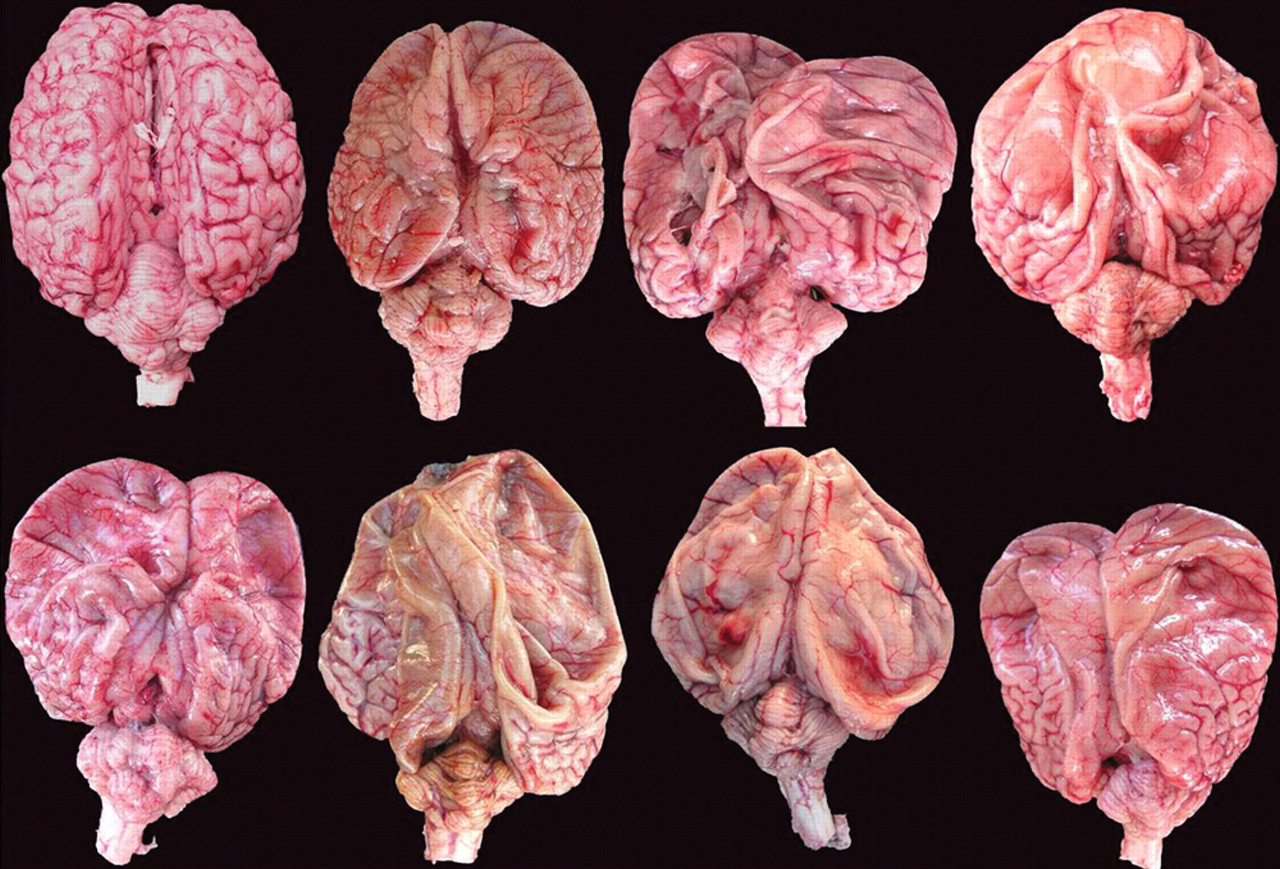
Congenital hydrencephaly, buffalo calves, brains. Brains of the 7 affected buffalo calves have similar lesions including absence of gyri mainly in the frontal and parietal cortex. Some gyri remain mainly in the occipital cortex. The cerebellum show different degrees of hypoplasia. The brain at left on the top is from the control calf followed by brains from calves 1–3 at top and 4–7 at bottom.
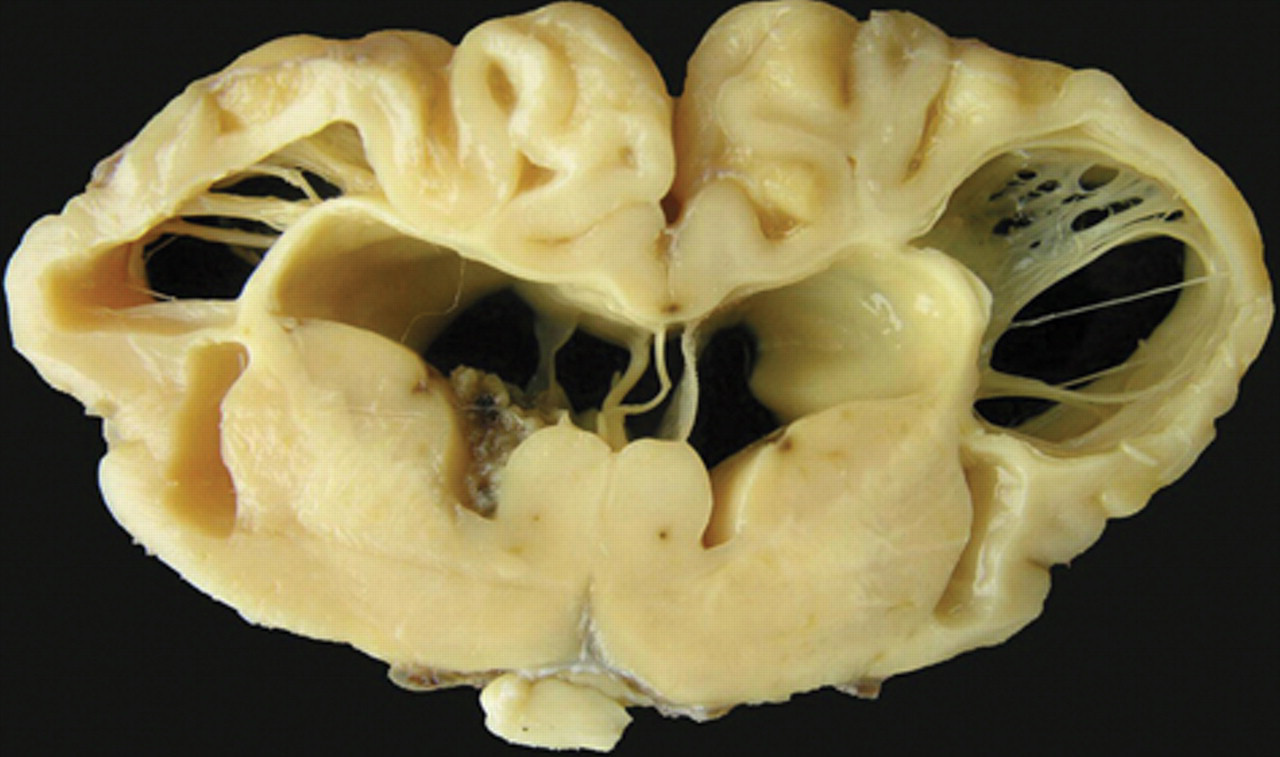
Congenital hydrencephaly, buffalo, brain; calf 1. Transversal section showing large bilateral cavities crossed by remnants of nervous tissue. Smaller bilateral cavities (porencephaly) are also observed in the external capsule. The columna fornicis is flattened and rounded, the corpus callosum is extremely atrophic, and the septum pelucidum is elongated. Ex vacuum hydrocephaly is also observed. The cingulate gyrus and some gyri of the parietal cortex are still present.
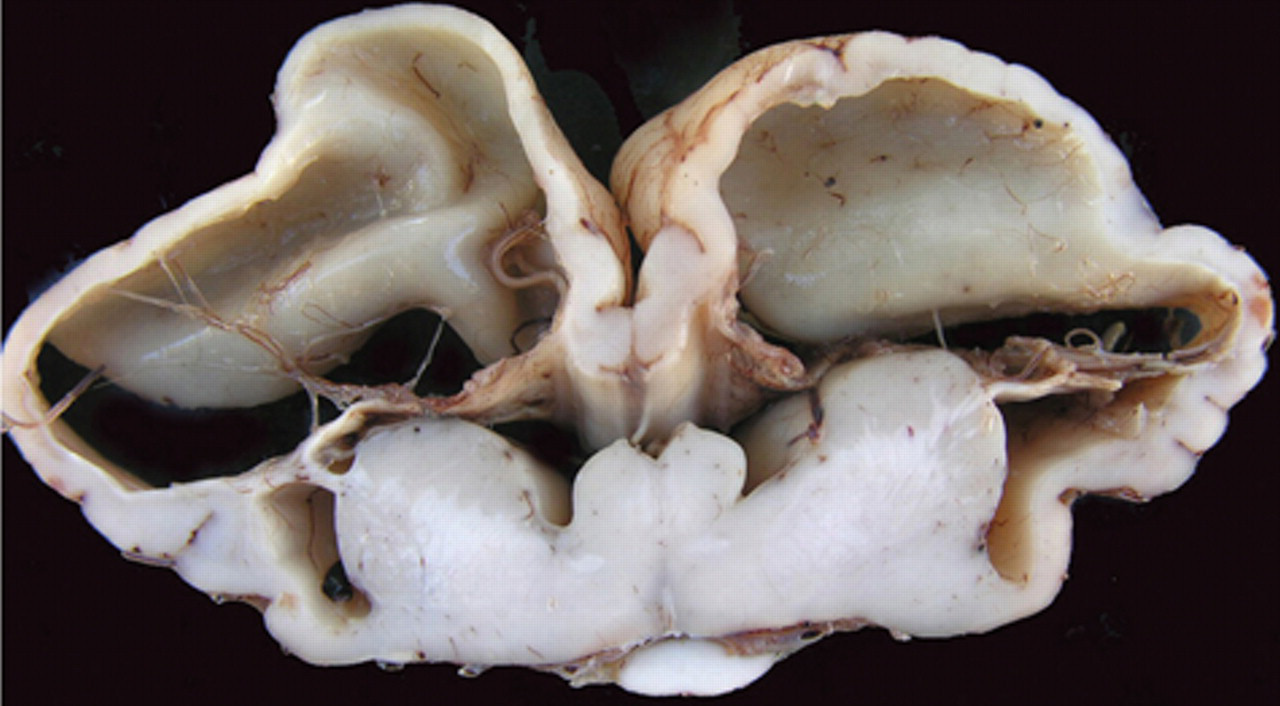
Congenital hydrencephaly, buffalo, brain; calf 4. Transversal section showing very similar, but more severe lesions than calf 1 in Figure 4. The cavities are larger and covered by thinner cortex with absence of gyri. Flattened and rounded columna fornicis, extremely atrophic corpus calosum, elongated septum pelucidum, and hydrocephaly ex vacuum are also observed.
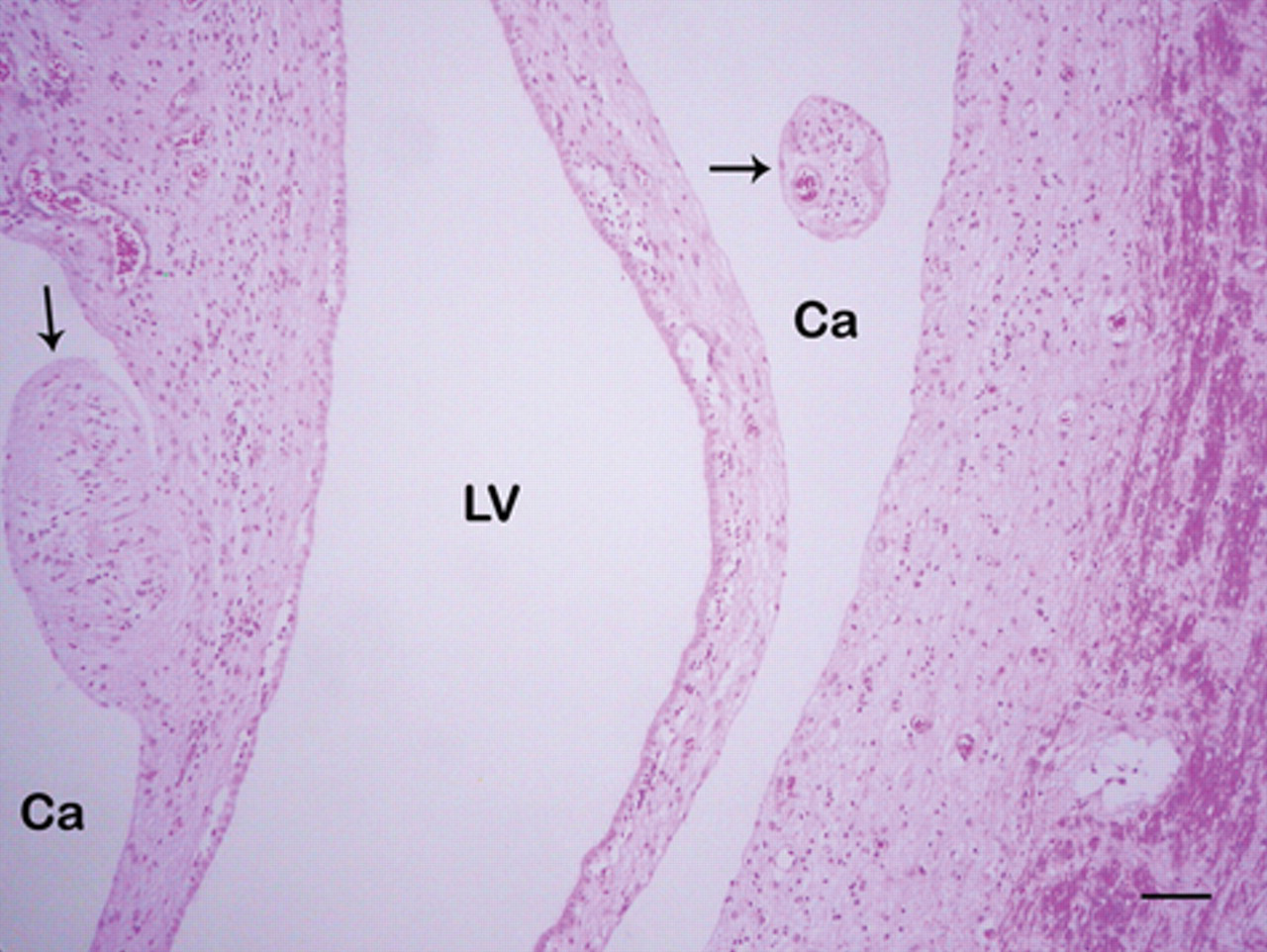
Congenital hydrencephaly, buffalo, brain, frontal cortex; calf 1. Cavities (Ca) crossed by nervous tissue remnants (arrows) are observed. The lateral ventricles (LV) are dilated and covered by flattened ependymal cells. Hematoxylin and eosin. Bar = 120 µm.
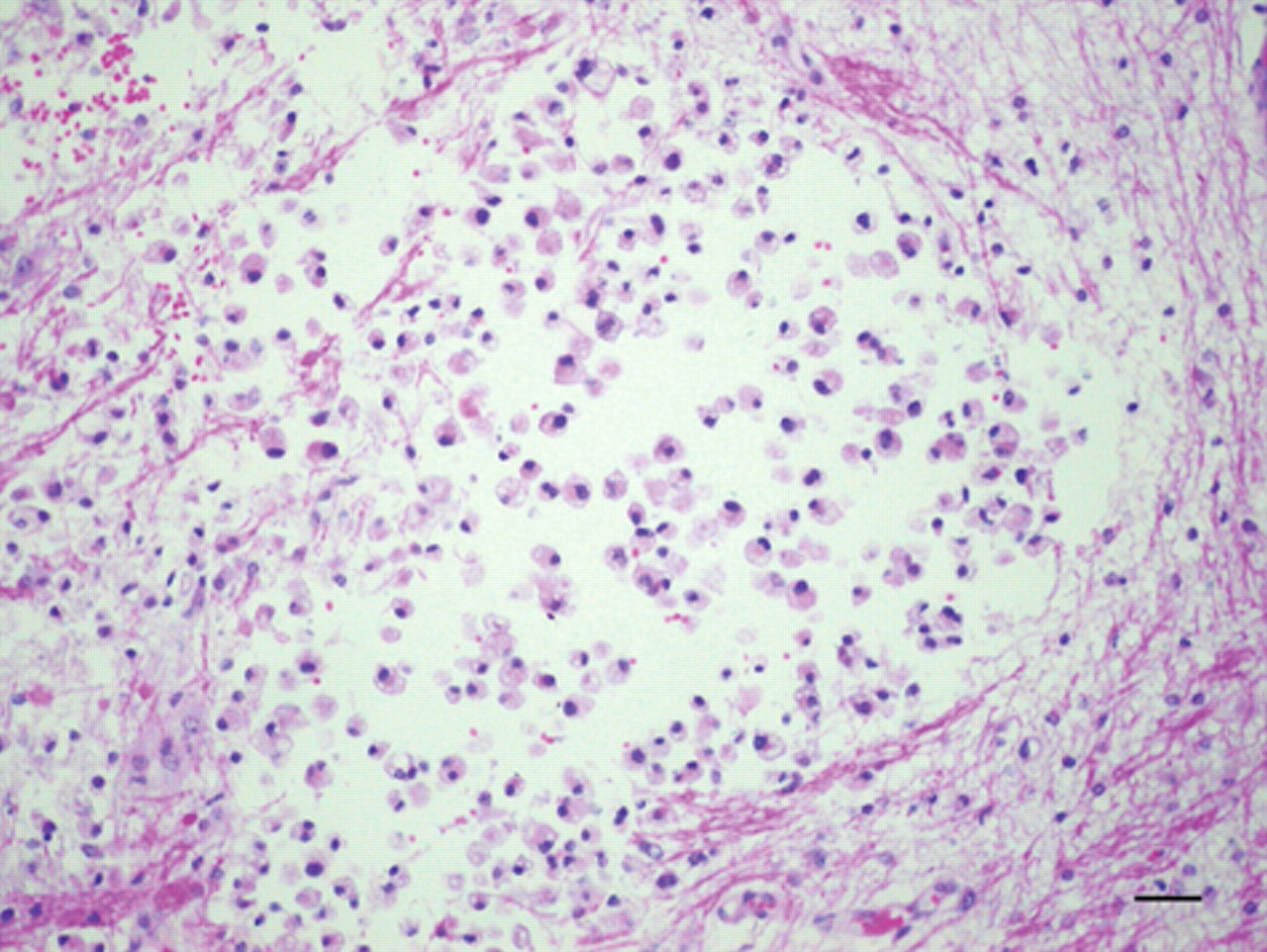
Congenital hydrencephaly, buffalo, brain, temporal cortex; calf 2. Rarefied white matter with a small cavity containing gitter cells is observed. Hematoxylin and eosin. Bar = 60 µm.
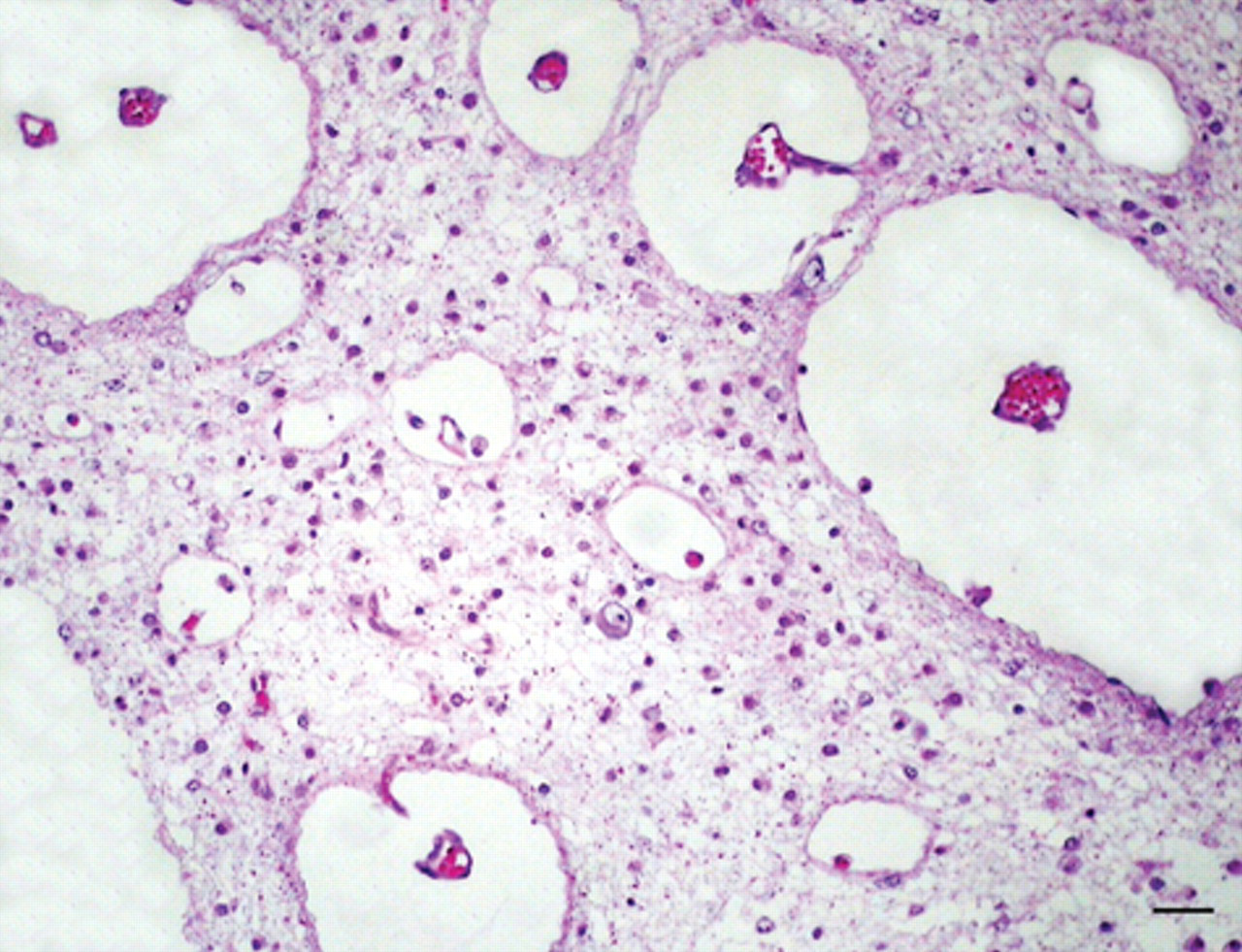
Congenital hydrencephaly, buffalo, brain, occipital cortex; calf 1. Very enlarged perivascular spaces are observed in the white matter, which is rarefied and infiltrated by gitter cells. Hematoxylin and eosin. Bar = 60 µm.
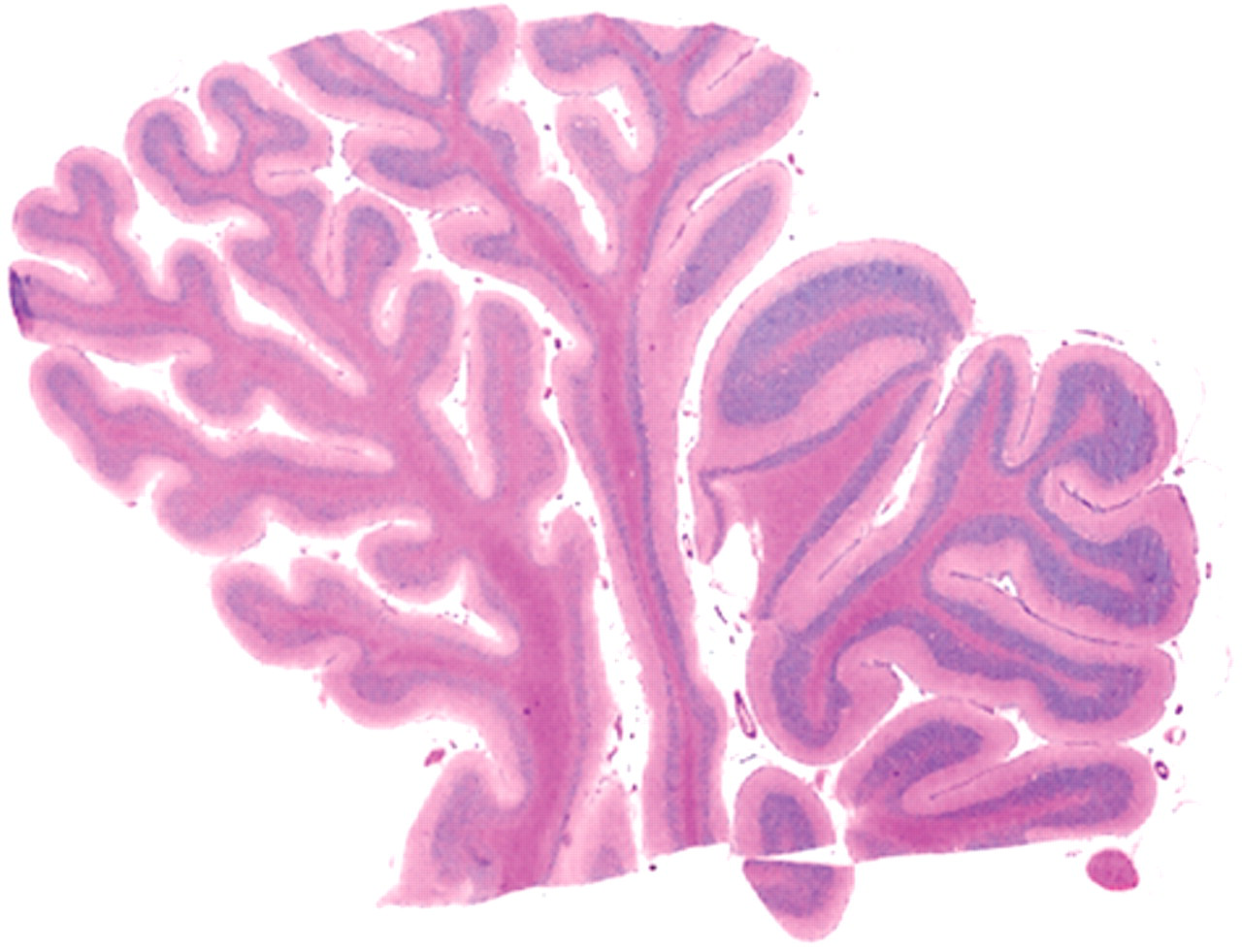
Cerebellar hypoplasia with congenital hydrencephaly, buffalo, cerebellum; calf 5. On submacroscopic view, there is different degree of hypoplasia in different folia. The folia at left had thinner granular and molecular layers than the folia at right. Hematoxylin and eosin.
Based on the clinical, gross, and histological findings, a morphological diagnosis of congenital hydranencephaly and cerebellar hypoplasia was established. The occurrence of the disease in different years, manifested only in the offspring of 1 out of 6 bulls (group 4), the similar lesions observed in all 7 calves affected, and the negative results of serology and immunohistochemistry for BVDV and BTV is highly suggestive of a hereditary etiology. This hypothesis is also supported by the genetic analysis (Fig. 1), which shows high level of inbreeding in the herd. This is suggestive of an autosomal recessive trait. Due to the strong possibility of the disease being hereditary, the bull that fathered all affected calves was culled in 2008 and replaced by a new bull; no new cases of the disease have been observed since.
To the authors’ knowledge, hydranencephaly has not been reported previously in buffalo, but it is a common disease of cattle, generally associated with viral infections including BTV, 23,24 Akabane virus, 8 Rift Valley fever virus, 3 Aino virus, 4,22 Chuzan virus, 4,14 and BVDV. 25 Hydranencephaly associated with cerebellar hypoplasia was described in a Holstein–Friesian herd in Canada, for 2 consecutive years, with a prevalence of 12.6%, but the etiology was not determined, although the authors suggested the possibility of a viral cause. 7
In the current case, only the two viral etiologies mentioned above were investigated, because BVDV and BTV have been described in Brazil and it was plausible that they had infected the buffalo herd in the present study. Nevertheless, BTV has only been reported in sheep in Brazil, 1,2 while BVDV has been diagnosed in cattle. 5,19
The remaining viral diseases known to cause hydranencephaly in cattle, including Akabane, Rift Valley fever, Aino, and Chuzan, were not specifically investigated but they are considered foreign animal diseases in Brazil. Nevertheless, epidemiological evidence did not support the role of any of these viruses in the cases described. Generally, viral infections present with a variety of malformations, including cerebellar hypoplasia, microphthalmia, arthrogryposis and palatoschisis, and they may also cause abortion or embryonic death, which greatly depends on the time in which the fetuses are infected. 7 Those variations as well as other reproductive problems were not observed in the disease reported in the current case. Also, in viral infections, malformations occur with higher frequency than that observed in the buffalo herd described herein, and generally during only 1 calving season. 23,24 Furthermore, cattle and sheep raised together with the buffalo described in the present case on the same farm were not affected. Hydranencephaly is frequently considered a model for virus-induced malformation in animals, 6 and it should be noted that the same malformation can be produced by other causes such as genetic defects. 9
The possibility of the disease being caused by toxic plants or nutritional abnormalities was ruled out, because the affected and unaffected groups of buffalo were grazing in paddocks with similar vegetation. Furthermore, the disease was not observed prior to 2003, when bull 4 was introduced to the herd.
The clinical signs and lesions were similar in all calves. Depression, inability to suck, involuntary movements, and central blindness are clinical signs associated with cerebral cortex lesions; while intention tremors, wide-base stance, and loss of balance are associated with cerebellar lesions. 12,18 Dome-shaped skull, observed in 4 calves, is uncommonly seen in cases of hydranencephaly, although it may occasionally be observed in cattle with this lesion. 11 All affected animals were born alive despite the almost complete absence of cerebral hemisphere tissue. In previous reports of hydranencephaly, calves with severe lesions in the cerebral cortex survived from several days to 2–3 months, and they even walked. 7,8,23 This is thought to be due to the presence of the relatively unaffected brainstem. In cases of other severe malformations of the CNS, such as brain aplasia and prosencephalic hypoplasia, in which there is preservation of the brainstem, affected animals can survive for relatively long periods after birth, and they may even remain standing and walk. 21
The histological changes observed in the affected buffalo in the present study were characterized by loss of nervous tissue, leaving cavities surrounded by more or less normal nervous tissue. However, the presence of gitter cells in a few sections is suggestive of previous tissue necrosis. No inflammatory leukocyte infiltrate was observed in 6 out of 7 buffalo. However, this finding cannot be used to rule out a viral etiology because perivascular cuffs and gliosis in the neural parenchyma are only occasionally seen in cases of viral hydranencephaly. 8,23 Apparently, the presence of inflammatory infiltrate is not usually seen in cases of viral hydranencephaly because most of the intense inflammatory reaction that occurs initially during the infection, clears up before birth. 11 In conclusion, congenital hydranencephaly and cerebellar hypoplasia appears to be a new disease of buffalo probably due to an autosomal recessive gene.
Footnotes
a.
ID Vet, Montpelier, France.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The project to study this disease was financially supported by the National Council of Scientific and Technological Development (CNPq; grant no. 471718/2007-6).
