Abstract
Bovine viral diarrhea virus 1 (BVDV-1) subtype b was isolated from premature Holstein calves from a dairy herd that experienced an outbreak of premature births, late-term abortions, brachygnathism, growth retardation, malformations of the brain and cranium, and rare extracranial skeletal malformations in calves born to first-calf heifers. Experimental inoculation of 3 colostrum-deprived calves aged 2–4 months old with this BVDV isolate resulted in thrombocytopenia, lymphopenia, and leukopenia. Outbreaks of brachygnathism are rarely associated with BVDV, and thrombocytopenia is rarely associated with BVDV-1 strains.
Infection with Bovine viral diarrhea virus 1 and 2 (BVDV-1 and BVDV-2, respectively; family Flaviviridae, genus Pestivirus) results in a wide variety of clinical presentations in cattle, including abortion; congenital defects; fetal mummification; persistent infection; diarrhea; sudden death; necrosis of lymphoid tissues and squamous epithelium in the oral cavity, esophagus, rumen, omasum, and skin; crypt epithelial necrosis in the intestinal tract; vasculitis; and immunosuppression. 2,3 A hemorrhagic syndrome associated with thrombocytopenia has previously only been found with BVDV-2 strains. 3 The current study documents the occurrence of an outbreak of congenital deformities, including brachygnathism, late-term abortions, and premature births in Holstein heifers from which a BVDV-1b strain was isolated. Infection of calves with this virus resulted in thrombocytopenia.
In May 2004, first-calf heifers in a dairy with 650 milking cow Holsteins experienced an outbreak of late third-trimester abortions and premature live births with skeletal and neurologic deformities. Over a 4-week period, 19 of 37 heifers in their last month of gestation aborted or gave birth to calves approximately 2–4 weeks early. In the first week of May, 7 of 7 premature stillborn and live calves had notably shortened mandibles and appeared undersized for their age and development. One of these, a live 2-day-old calf (calf A) that had been tube-fed colostrum because it was unable to stand, was submitted to the California Animal Health and Food Safety Laboratory (Tulare, CA). Another live calf (calf B) was euthanized by the referring veterinarian. The euthanized calf and 2 stillborn calves (calves C and D) were necropsied in the field by the referring veterinarian, and their tissues (calf B) or heads (calves C and D) were submitted to the laboratory.
Upon presentation to the laboratory, calf A was in lateral recumbency and unable to obtain a sternal position, and it exhibited ventral strabismus, mild opisthotonus, mandibular brachygnathism, and occasional random paddling. After euthanasia, necropsy revealed that one foreleg was swollen due to marked hemorrhage around a fractured third metacarpal bone; carpal and hock joint fluid were red tinged and of normal consistency. The brain was grossly and histologically unremarkable. Luxol fast blue staining for myelin of medulla and cerebellum was within normal limits. The spinal cord was not examined. Calf B had exhibited neurologic signs, and necropsy by the referring veterinarian revealed ascites and mandibular brachygnathism; the parenchymal tissues were grossly and histologically unremarkable. The brain and spinal cord were not examined. The calvarium of calf C was markedly domed with no bone covering the dorsal central 9 cm of the brain; in addition, the maxilla and mandible were shortened (brachycephaly; Fig. 1), and there was a complete cleft palate. The referring veterinarian reported that there were only short stubs of fleshy tissue where the forelimbs should have been. Calf C had hydranencephaly with red-tinged fluid filling the lateral ventricles and mild cerebellar atrophy. Histologically, the medulla, midbrain, and thin remnants of cerebrum were hypercellular. The cerebral cortex had very thin, poorly organized gray matter and little or no white matter. There was multifocal thinning and mild dysplasia of the granular layer of the cerebellum. Luxol fast blue staining of the medulla and cerebellum revealed decreased myelin throughout. The head of calf D was slightly domed, and there was mandibular brachygnathism, moderate to marked hydrocephalus, and mild cerebellar atrophy. Histologically, the cerebrum had attenuated, normally organized gray matter, decreased white matter, and increased reserve cells around the distended ventricles. The thalamus, midbrain, and medulla had multiple hypercellular areas. The cerebellum exhibited multifocal thinning and dysplasia of the granular layer. Luxol fast blue staining of medulla and cerebellum revealed multifocal hypomyelination in the cerebellum, particularly in the tips of the folia.

Calf C. The maxilla and mandible are shortened (brachycephaly). Marked doming of the head is associated with hydranencephaly.
Bovine viral diarrhea virus infection was confirmed in all 4 calves by various methods. Direct BVDV fluorescent antibody testing on frozen tissue sections using a 1:50 dilution of fluorescein-conjugated, porcine-origin, anti-BVDV polyclonal antibody was performed according to the manufacturer's recommendations a and was positive on kidney and lung of calf A and kidney of calf B. Immunohistochemistry 1 on formalin-fixed, paraffin-embedded tissue with the use of monoclonal antibody 15c5 b at a 1:1,000 dilution using the avidin–biotin–immunoper-oxidase technique c was positive on skin from calves A, C, and D. The real-time reverse transcription polymerase chain reaction (RT-PCR) modified from previously published primers and probes 5 confirmed the presence of BVDV-1 in the kidney and whole blood of calf A, the kidney of calf B, and the brain of calf D. The BVDV realtime RT-PCR was negative on lung and spleen from calf B and brain from calf C.
Sera were submitted from 6 heifers that had affected calves, and BVDV serum neutralization titers ranged from 1:32 to 1:2,048 for BVDV-1 and 1:128 to 1:1,024 for BVDV-2. Of the 6 heifers, 4 had 2-fold higher BVDV-1 than BVDV-2 titers, and one heifer had an 8-fold higher BVDV-1 titer. Only the dam of calf B had a higher BVDV-2 titer (1:128) than BVDV-1 titer (1:32). Calf A received colostrum at birth and had a 1:128 BVDV-1 titer and 1:256 BVDV-2 titer.
The heifers from this dairy had been vaccinated with a single dose of modified live vaccine containing BVDV-1a at 1 week of age. At 5 months of age, heifers were sent to a heifer ranch until 12 months of age when they were moved to a second heifer ranch. At the first heifer ranch, no vaccinations were given. At the second heifer ranch, animals were vaccinated on arrival with a killed vaccine containing cytopathic BVDV-1a and noncytopathic BVDV-1. The home dairy vaccinated lactating cows once a year with a killed BVDV vaccine. The second heifer ranch received animals from 3 other dairies. The heifers returned to the home dairy when they were approximately 6–7 months pregnant and were vaccinated with a killed vaccine containing BVDV-1a and BVDV-2. The pregnant heifers were housed with the dry cows on their return. Approximately 8 weeks before the onset of abortions in the heifers, a number of postpartum cows experienced high fevers (over 40°C) and some had diarrhea. Paired serology on 3 affected cows taken 3 weeks apart revealed a rising BVDV-1 seroconversion in 1 cow, falling seroconversion (acute titer >1:8,196) in a second, and a static titer of 1:2,048 in the third cow. Based on the presumptive diagnosis of BVDV infection, the late lactation cows and dry cows, but not the heifers, were vaccinated with a modified live vaccine containing BVDV-1a and BVDV-2. In July 2003, 9 months earlier, the herd began using a modified live vaccine containing BVDV-1a and BVDV-2 at the postpartum examination, which was the reason they only vaccinated late lactation and dry cows in response to the fever and diarrhea problem.
Tissues from calf A (live born) were submitted to the National Animal Disease Center (Ames, IA) for further characterization. A BVDV was isolated and identified as a BVDV-1b strain based on phylogenetic analysis of the 5' untranslated region. 7 The BVDV phylogenetic tree in Figure 2 shows the relationship of this strain to other pestivirus strains. Four colostrum-deprived mixed-breed calves, aged 2–4 months, were tested as free of BVDV in buffy coat samples by virus isolation and free of BVDV antibodies in serum. Three of these calves were inoculated with the BVDV-1b isolate, and the fourth served as a nonexposed control. Calves were housed individually in climate-controlled pens. There was no animal to animal contact between pens, but there was shared airflow.
Animals were inoculated via the nasal route with 5 ml of freeze–thaw lysate prepared from infected Madin–Darby bovine kidney cells 5 days after inoculation that contained approximately 1 × 106 tissue culture infectious doses of virus per milliliter. Temperatures were taken daily, and blood samples were collected before inoculation and on days 3, 6, 9, 11, and 13 after inoculation. Lymphocyte and platelet counts were determined using a cytometer per the manufacturer's directions. d Two of three inoculated calves and the control had a mild increase of body temperature to a maximum of 0.44°C, and one challenged calf had an increase of 1.22°C (40.2°C for 4 days). Baseline values for white blood cell, lymphocyte, neutrophil, and platelet counts were established on a single blood sample taken from each calf before inoculation.
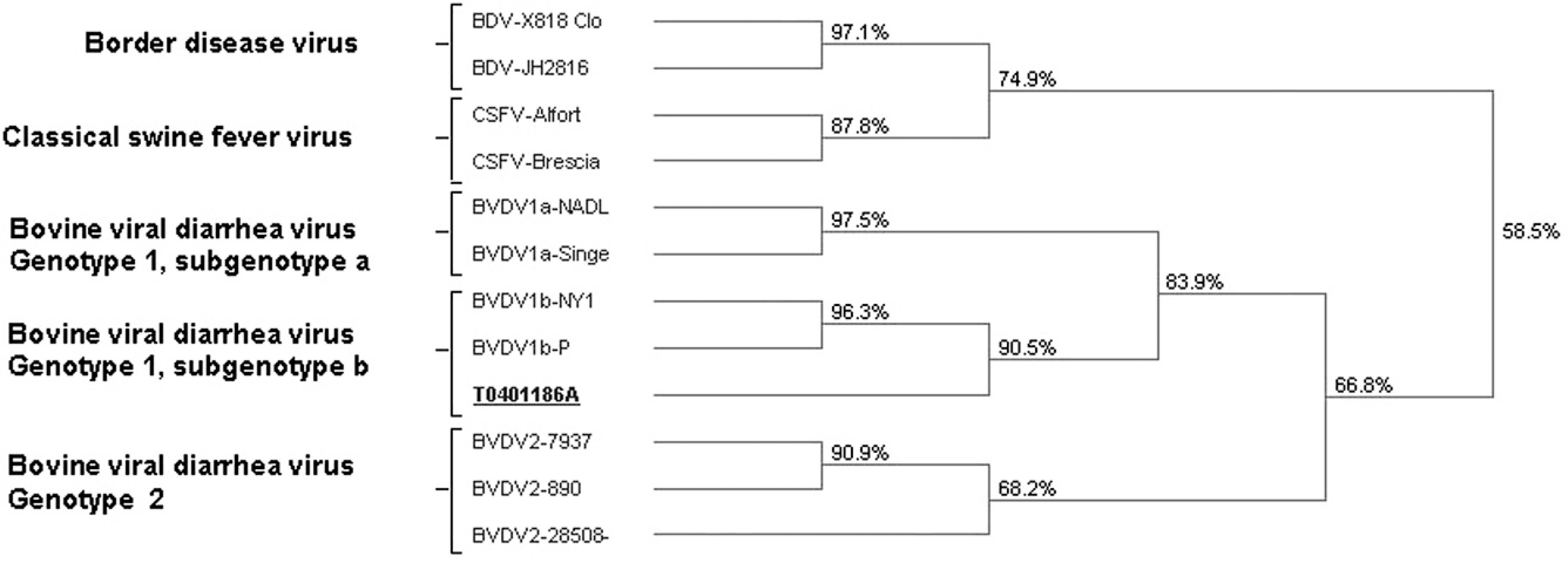
Bovine viral diarrhea virus (BVDV) phylogenetic tree demonstrating relationship of virus T0401186A, BVDV genotype 1 subgenotype b, compared to other pestiviruses.
Lymphocyte, total white blood cell, and platelet counts declined in all inoculated calves and rose in the control. Peak declines in lymphocyte counts below baseline occurred on day 3 in 1 calf (33.4% decline) and day 6 in the other 2 calves (20.7 and 44.4%, respectively). All 3 calves had maximal platelet count decreases on day 9 after inoculation. The decreases ranged from 35.5% to 46.8% below baseline with absolute counts of 136,000–199,000 platelets per microliter of blood in BVDV-challenged calves compared with 416,000 for the control calf. On gross necropsy, patchy areas of discolored and collapsed lung were seen in the challenged calves but not the control calf. One challenged calf had petechial hemorrhages in the ileocecal valve and linear hemorrhages in the rectum. Lung tissue from 2 challenged calves was examined histologically. One calf had mild bronchitis and atelectasis and the other had multifocal hemorrhage and areas of alveolar edema and fibrin exudation.
Congenital malformations due to BVDV infection are associated with fetal infection at 75–150 days of gestation and are commonly reported to involve the brain and eye. Eye tissue was not examined in the current study except to note the absence of microphthalmia. Hydrocephalus, hydranencephaly, cerebellar hypoplasia, growth retardation, and mandibular brachygnathism have all been reported with BVDV infection. 2 Mandibular brachygnathism is rarely reported to be associated with BVDV but was found in all 7 calves examined by the referring veterinarian. In the veterinary literature, mandibular brachygnathism has been reported with ocular lesions (cataracts and/or retinal lesions) and cerebellar hypoplasia. 8,9 Natural and experimental reports suggest that BVDV infection between 79 and 90 days of gestation can result in brachygnathism, and this period is also associated with milder cerebellar hypoplasia. 8,9 Exposure to BVDV between 30 and 125 days of gestation can result in persistent infection, which appears to have occurred in the calves in the present study based on the presence of the virus in all 4 calves 6–7 months after the likely onset of fetal infection.
Most BVDV-induced congenital deformities result from infection at 100–175 days of gestation, when the calf is becoming immune competent, and therefore the virus is often no longer present at the time of birth. 2 Failure of limb development has not been reported previously and was found in 1 of the 7 calves that also had multiple craniofacial skeletal and brain deformities. Calves born alive or aborted before and after the visit by the veterinarian were not examined, so the total number of calves with congenital deformities is not known. Exposure to the BVDV-1b isolate at the second heifer ranch could have resulted in a few acutely infected, asymptomatic, shedding heifers returning to the dairy where they exposed the co-housed dry cows to the virus strain, thereby resulting in the fever and diarrhea reported in postpartum cows. These heifers probably gave birth to healthy, congenitally infected, and recovered calves as their exposure was later in gestation. The herd vaccination history of a single yearly killed BVDV vaccine could produce a susceptible population of cows if a heterologous virus strain were introduced. Because the home dairy had begun the use of a modified live BVDV vaccine at postpartum examination approximately 10 months earlier, only late lactation and dry cows had not been vaccinated with modified live virus and were more susceptible to a newly introduced virus strain. The herd owner chose not to pursue testing of calves born to heifers for detection of persistent BVDV infection, so it is unknown what percentage of the live calf crop was infected. The BVDV-1b strain from the outbreak in the current study was associated with premature live births, abortion, multiple congenital defects, persistent infection, postpartum diarrhea, and fevers and, after experimental inoculation, with thrombocytopenia.
The BVDV-2 strains that experimentally cause a hemorrhagic syndrome decrease platelet counts below 100,000 platelets per microliter of blood. 4,10 With the challenge dose used in the experimentally inoculated calves in the present study, the lowest recorded level was 136,000 platelets per microliter of blood. Other BVDV-1 strains have not been previously reported to cause thrombocytopenia. Calf A had very extensive hemorrhage associated with a metacarpal fracture, but because a peripheral blood platelet count was not determined, it is uncertain whether BVDV-1b–associated thrombocytopenia may have contributed to the severity of the hemorrhage. Lymphopenia is reported with all BVDV strains regardless of genotype. Thrombocytopenia has only been associated with a minority of BVDV, and those BVDV tend to be strains with increased virulence. 4 Furthermore, although congenital abnormalities have been previously reported with BVDV, the present report documents a relatively large number of animals with multiple fetal malformations and significant impact on lymphocytes and platelets in acute infection making this a potentially good challenge virus to prove vaccine efficacy for fetal protection. More extensive challenge studies using this virus strain have been published and substantiate the limited challenge study results reported in the current study. 6
Footnotes
a.
American BioResearch Laboratories, Pullman, WA.
b.
IDEXX MoAb 15.c.5 (anti-BVDV), IDEXX Laboratories Inc., Westbrook, ME.
c.
Vectastain Elite ABC Mouse IgG kit and Vector NovaRED substrate, Vector Laboratories Inc., Burlingame, CA.
d.
CDC Technologies Inc., Irvine, CA.
