Abstract
Outbreaks of eastern equine encephalitis observed from May 2008 to August 2009 in the Brazilian states of Pernambuco, Ceará, and Paraíba are reported. The disease occurred in 93 farms affecting 229 equids with a case fatality rate of 72.92%. Main clinical signs were circling, depression or hyperexcitability, ataxia, and progressive paralysis with a clinical manifestation period of 3–15 days. Main histologic lesions were a diffuse lymphocytic encephalomyelitis with neuronal death, satellitosis, neuronophagia, and hemorrhages being more severe in the cerebral gray matter of the telencephalon, diencephalon, and mesencephalon. Some animals also had areas of malacia in the telencephalon, thalamus, and basal nuclei. From 1 case, the virus was isolated by mice inoculation, and in other 13 cases was identified as Eastern equine encephalitis virus by semi-nested reverse transcription polymerase chain reaction. After DNA sequencing, all samples were identified as eastern equine encephalitis through the BLASTn analysis, but samples from the Ceará and Paraíba states corresponded to the same cluster, while the sample from the state of Pernambuco corresponded to a different cluster.
Keywords
Eastern equine encephalitis (EEE) is a zoonotic disease of the family Togaviridae, genus Alphavirus. Viruses of this genus are mainly associated with disease of the central nervous system (CNS) of horses and human beings, both of which are the terminal hosts, while infected birds usually act as reservoirs and do not develop disease. 8 However, domestic and wild birds including pheasants, emus, and broilers have often been affected, and there are records of the disease in other species, including sheep, 2 dogs, 4 and deer. 14
In South America, there have only been 2 cases of fatal EEE reported in human beings, 1 in Brazil and another in Trinidad, while in North America, over 100 fatal cases have been reported. This difference can be attributed to the distinct pathogenic characteristics of the agents and habits of the vectors involved. It is suggested that the U.S. variants are more virulent for human beings and horses than the South America types. 17 Human fatality rate with a U.S. variant ranged from 50% to 75%. 18
In Brazil, there are few reports on the isolation of EEE virus from horses. 11 Most studies are based on antibodies detected in horses, human beings, and wild vertebrates. 1 , 5–7 ,15, 16 The objective of the current study was to describe the epidemiology, clinical signs, and pathology of outbreaks of EEE that occurred in 2008 and 2009 in the semiarid region of the states of Pernambuco, Paraíba, and Ceará, in northeastern Brazil.
The farms visited were those on which the farmers reported the occurrence of nervous signs in equidae. In the state of Pernambuco, from April to August 2008, 11 farms were visited in the municipalities of Parnamirim and Exú. Of the 100 equidae affected, most were horses; morbidity reached 61% (61/100), and the case fatality rate was 98.36% (60/61). From March to July 2009, 40 farms in the state of Ceará and 42 in the state of Paraíba were visited. Out of 960 equidae, mainly horses, 165 horses, 1 pony, 1 donkey, and 1 mule were affected (morbidity of 17.5%); 105 horses, the pony, and the donkey died (case fatality rate of 64.28%). Horses ranging from 6 months to 16 years old, both females and males, were affected. The spatial distribution of cases is shown in Figure. 1.
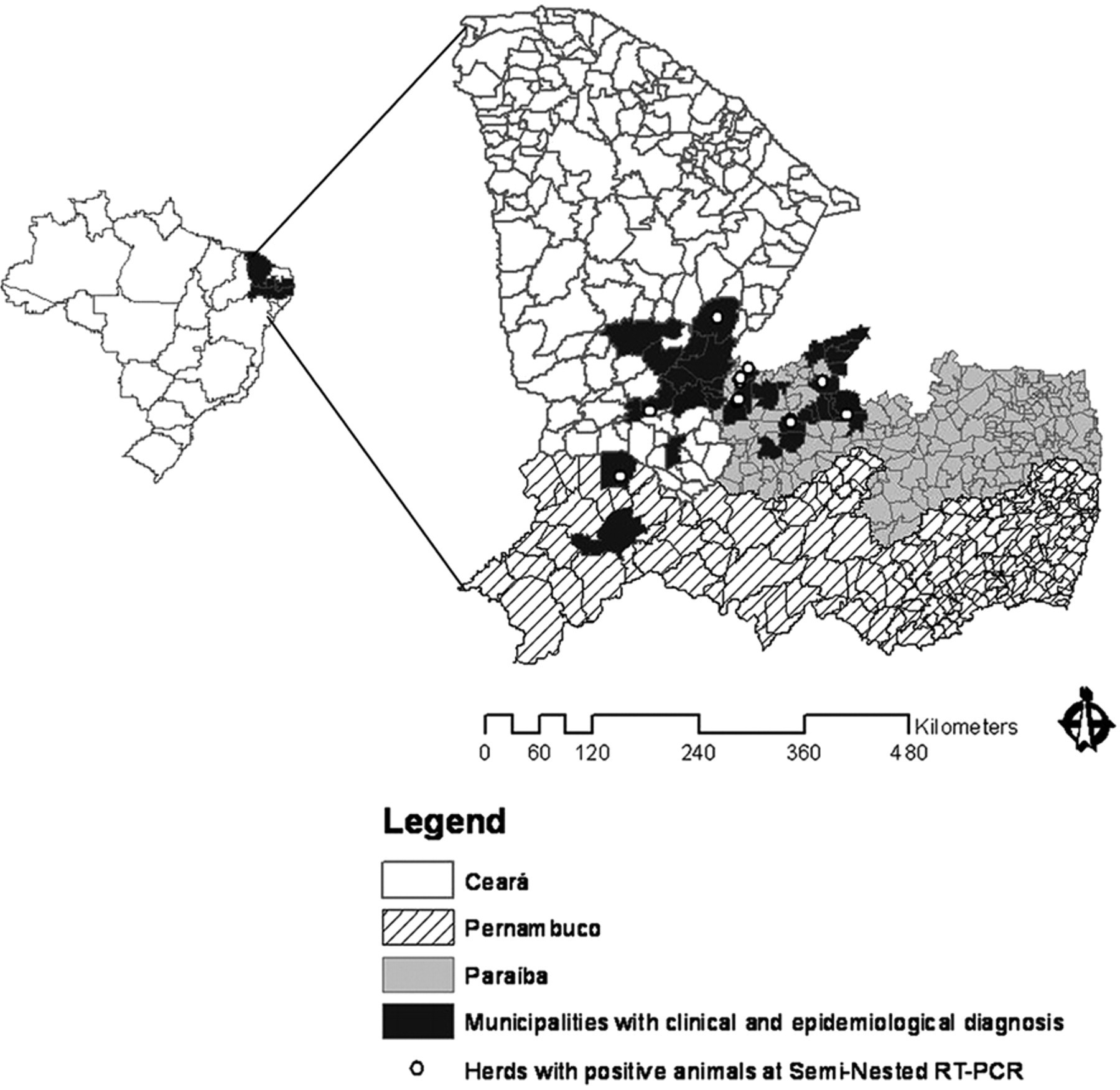
Map of the Brazilian states of Paraíba, Ceará, and Pernambuco showing the municipalities where eastern equine encephalitis occurred.
During visits, affected animals were examined clinically. The main clinical signs were ataxia, circling, blindness, head pressing, depression or hyperexcitability, lip movements, or other involuntary movements, tongue paralysis, ataxia, paresis progressing to paralysis, recumbence, paddling, and coma. Fever, lip or mandible paralysis, wide base stance, hypermetric gait, stringhalt, falls, salivation, and nasal and eye discharge were less frequently observed. The clinical manifestation period was 3–15 days.
Fourteen necropsies were performed. Traumatic lesions, including corneal opacity, subcutaneous periocular edema, and alopecia and erosions of the skin in different parts of the body were observed in 9 horses. Five animals showed congestion of the meningeal vessels. After fixation, red or brown areas were observed in the temporal and occipital cortex of 1 horse (Fig. 2).
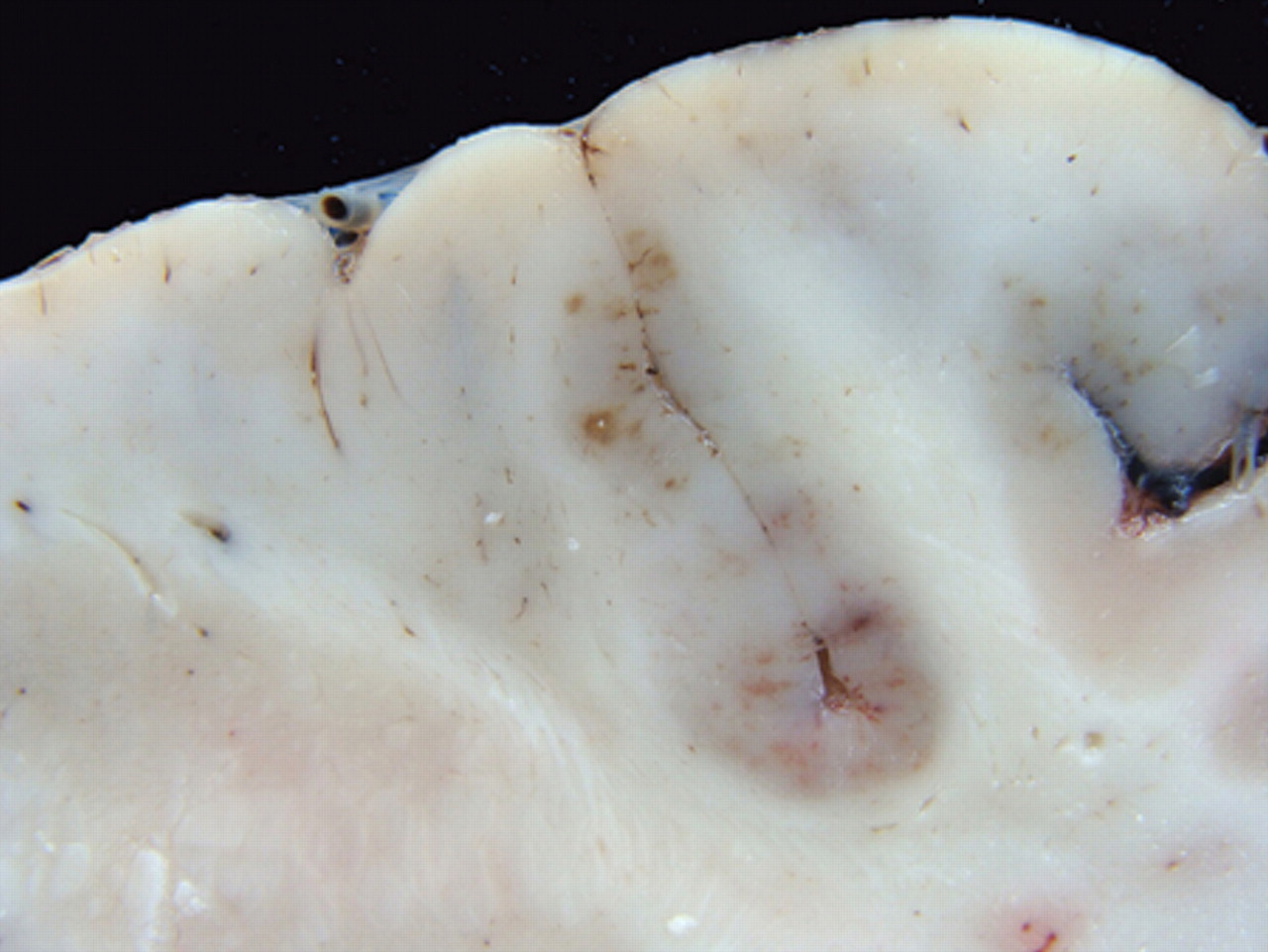
Cerebrum. Horse with equine encephalomyelitis from the Brazilian municipality of Jaguaribe, state of Ceará, showing red or brown areas of malacia in the occipital cortex.
Samples of the organs of the thoracic and abdominal cavities, and the CNS were collected for histologic examination. The samples were fixed in 10% buffered formalin solution. After fixation, the brain and spinal cord specimens were cut transversely into 3–5 mm thick sections, and samples of frontal, temporal, parietal, and occipital lobes, hippocampus, basal nuclei, thalamus, rostral and caudal colliculi, pons, cerebellum, medulla oblongata, and cervical, thoracic, and lumbar spinal cord were embedded in paraffin, cut into 4–5-µm sections, and then stained with hematoxylin and eosin.
Main histologic lesions were a diffuse multifocal non-suppurative lymphoplasmacytic encephalomyelitis with perivascular infiltration by lymphocytes, plasma cells, macrophages, and some neutrophils, vasculitis with endothelial cells, swelling, neuronal death, satellitosis, neuronophagia, hemorrhages, edema, and multifocal lymphocytic infiltration of the neuropil (Fig. 3). These lesions were more severe in the gray matter of the telencephalon, diencephalon, and mesencephalon. Similar but mild lesions were observed in the cerebellum and spinal cord, mainly in the gray matter. The distribution and severity of the inflammatory lesions in 14 horses are presented in Table 1. In 5 cases, rare eosinophils were observed within the lymphoplasmacytic exudates. Nine cases showed lymphoplasmacytic meningitis with perivascular cuffing in the cerebral meninges, and in 4 of these horses, the meningitis also affected the cerebellum. Focal areas of malacia, with vacuolation of the neuropil and presence of macrophages, neutrophils, cellular debris, and occasional gitter cells were observed mainly in the telencephalon, thalamus, and basal nuclei. Axonal spheroids were occasionally observed. No significant lesions were observed in other organs examined.
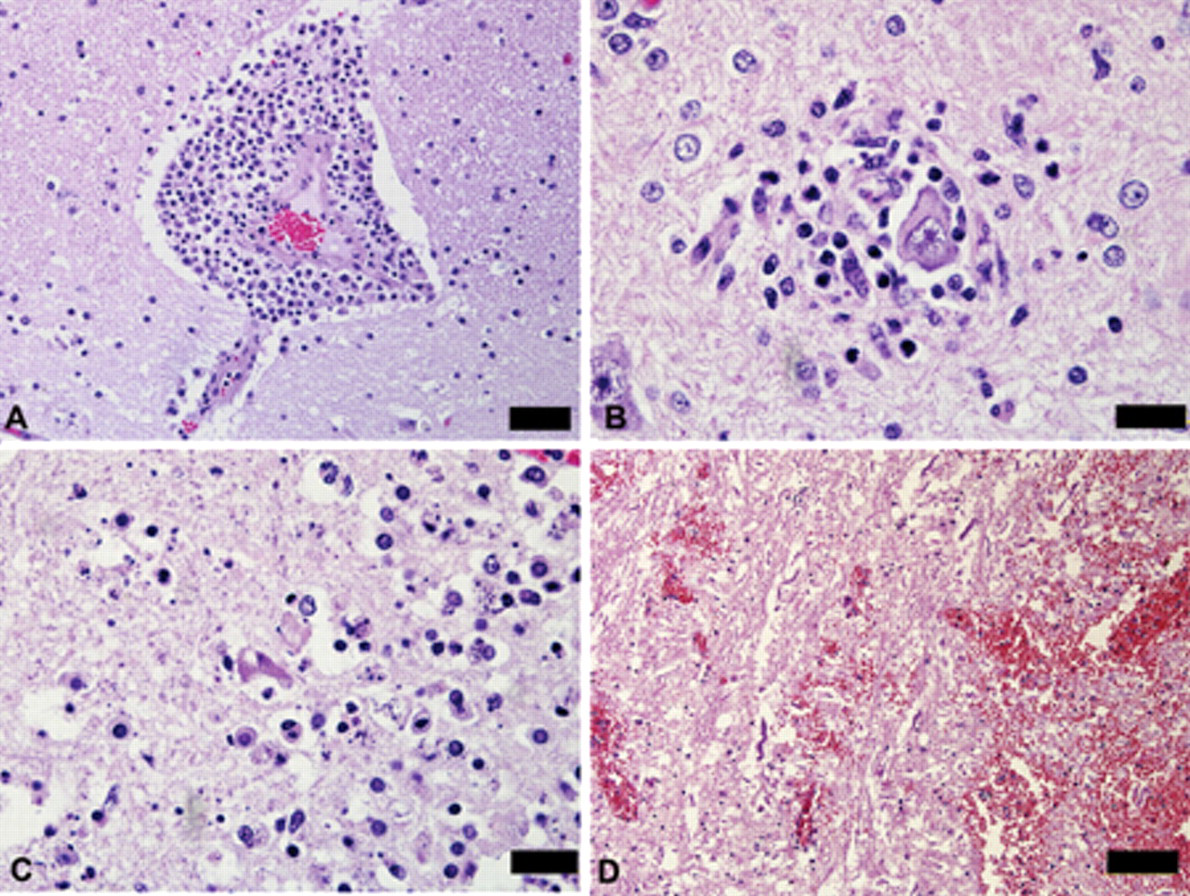
Brain. Horse with equine encephalomyelitis from the Brazilian municipality of Exú, state of Pernambuco.
Distribution and intensity of inflammatory histologic lesions observed in 14 cases of eastern equine encephalitis in northeastern Brazil.*
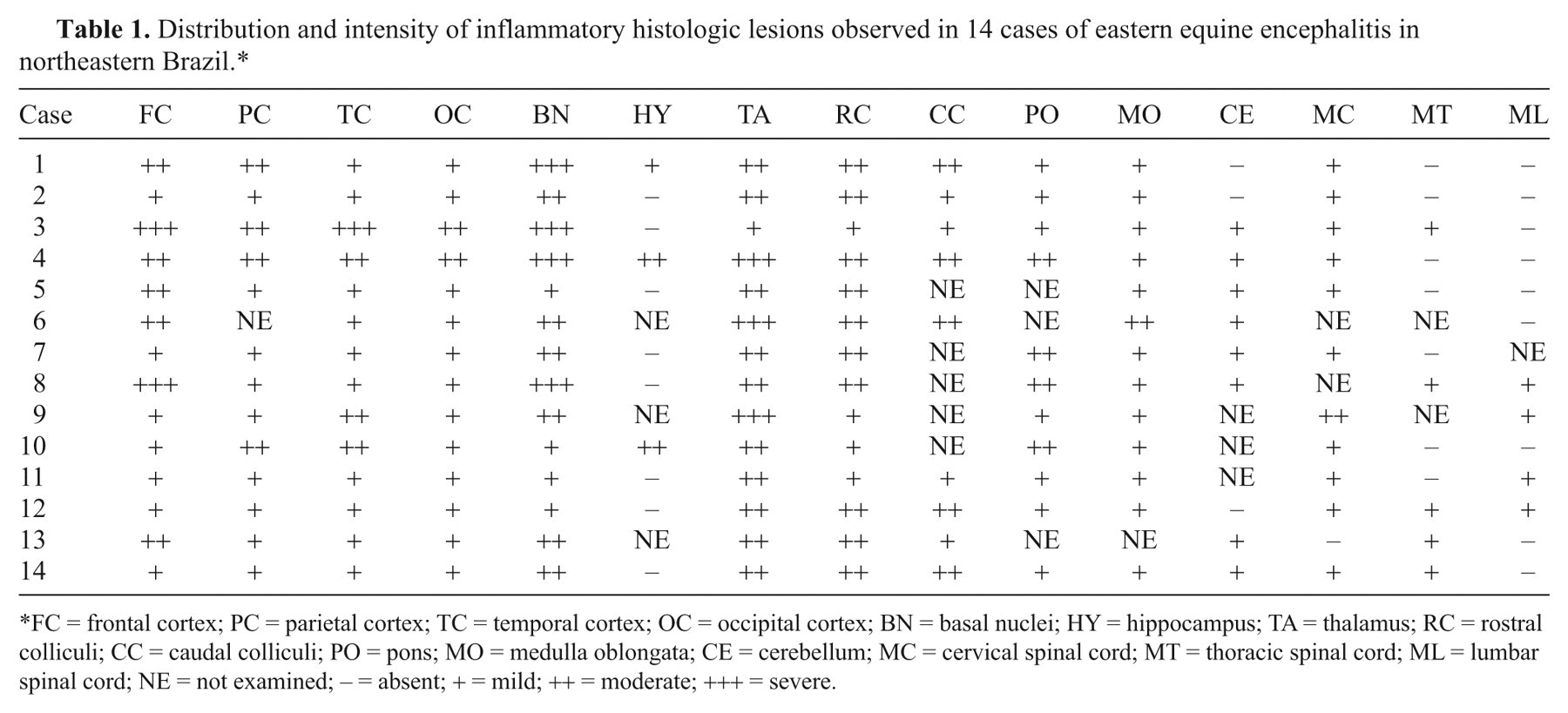
FC = frontal cortex; PC = parietal cortex; TC = temporal cortex; OC = occipital cortex; BN = basal nuclei; HY = hippocampus; TA = thalamus; RC = rostral colliculi; CC = caudal colliculi; PO = pons; MO = medulla oblongata; CE = cerebellum; MC = cervical spinal cord; MT = thoracic spinal cord; ML = lumbar spinal cord; NE = not examined; − = absent; + = mild; ++ = moderate; +++ = severe.
Serum samples, 1 taken during the course of the clinical disease and 1 taken 15 days later, were collected from 2 horses that were not vaccinated against equine encephalitis, and sent to the Biological Institute of São Paulo (Brazil) to determine the antibody titers for equine eastern and western encephalitis by means of neutralization test in cell culture. The antibody titers against EEE virus were 1:10 and 1:10 in the first sample, increasing to 1:320 and 1:160 in the second.
Direct fluorescent antibody test 3 and mouse inoculation test 10 were negative for Rabies virus. A litter of 5-day-old mice inoculated with CNS suspension collected from 1 horse immediately after euthanasia began to show signs 24 hr after inoculation. The inoculated mice were isolated from the other littermates, failed to nurse, and had difficulty breathing and paresis progressing to flaccid paralysis. Death occurred within 6 hr after the onset of first clinical signs. Forty-eight hours after inoculation of the newborn mice, the mother presented nervous signs including circling movement, head pressing on the box, and paresis of the hind limbs progressing to flaccid paralysis, and died 72 hr after inoculation. The virus isolated from mice was confirmed as EEE by the semi-nested reverse transcription polymerase chain reaction (RT-PCR).
Identification of the virus was performed by semi-nested RT-PCR on 13 horse specimens collected from the municipality of Exú, in the state of Pernambuco (1); from the municipalities of Jaguaribe (1) and Várzea Alegre (2), both in the state of Ceará; and from the municipalities of São João do Rio do Peixe (2), Coremas (2), Poço de José de Moura (2), Uiraúna (1), Paulista (1), and Patos (1), all in the state of Paraíba. RNA extraction was carried out using TRIzol a according to the guidelines of the manufacturer, from the original specimens of horses. In 10 samples, the extraction was performed in a pool of brain stem and cerebellum, and in 3 samples, in a pool of cervical cord, hippocampus, and frontal cortex. As a positive control, a sample of fixed virus of Western equine encephalitis virus (WEEV), assigned by the Instituto Evandro Chagas (IEC), state of Pará, and maintained in Swiss albino mice was used. As a negative control, ultra pure water free of DNase and RNase was used. The positive and negative controls, together with the 13 CNS samples, were submitted to the semi-nested RT-PCR technique directed toward amplification of a fragment of 310 base pairs (nucleotides 283–592) of the nonstructural protein 1 of the genus Alphavirus, according to the protocol described previously. 12
In the semi-nested PCR, a negative control was used for every 3 samples tested, to obtain greater control over possible contamination of the reaction. The primers used in the semi-nested RT-PCR are described in Table 2. The fragment of 310 bp resulting from the semi-nested RT-PCR was purified from 1% agarose gel with a gel extraction kit b according to the manufacturer’s instructions and quantified with low DNA mass ladder c 2% agarose gel, according to the manufacturer’s instructions. For each reaction of DNA sequencing, between 2.5 ng and 10 ng of DNA were used.
Description of the primers used in the current study for semi-nested reverse transcription polymerase chain reaction (RT-PCR).

The DNA fragments were subjected to bidirectional DNA sequencing, with primers A2 and A3 in separate reactions using a cycle sequencing kit. d The purification of the sequencing reaction was performed using cross-linked dextran gel e on plates with a multi-screen filter in 96-well plates. After purification, the sequences were generated in automatic genetic analyzer. f The confirmation of the sequencing and determination of viral species was performed using the application BLASTn (http://www.ncbi.nlm.nih.gov/BLAST).
Of the 13 CNS samples submitted to semi-nested RT-PCR, all were positive for the genus Alphavirus. After DNA sequencing, all positive samples were identified as EEEV through the BLASTn analysis. Sequencing showed that the samples from the states of Ceará and Paraíba corresponded to the same cluster, while the sample from the state of Pernambuco corresponded to a different cluster. Detailed results of the sequencing of these samples will be published elsewhere.
After diagnosis of the disease, nearly all equidae from the affected regions were vaccinated and new outbreaks were not observed during 2010. In the 2008 and 2009 outbreaks, the presumptive diagnosis of EEE was made by the epidemiological, clinical, and pathological features of the disease, and confirmed by virus isolation, semi-nested RT-PCR, and the increased antibody titer in 2 horses that had recovered from the disease. The results of the sequencing of the isolates proved that the outbreak in 2008 in Pernambuco was caused by a distinct EEEV variant different from the virus variant causing the outbreak in Ceará and Paraíba in 2009. The diagnosis of 2 outbreaks of EEE in the semiarid regions of 3 northeastern states represents the first full description of the disease in Brazil, including epidemiology, clinical signs, pathology, and virus identification.
During the visits to the farms, some experienced owners and veterinarians mentioned that this disease is known as roda (circling) and occurs periodically in the region. The occurrence of both outbreaks of EEE at the end of the rainy season suggests that the disease is seasonal and epidemic in the Brazilian semiarid region.
The semiarid region is characterized by a warm climate with an average temperature of 26°C and rainfall of approximately 800 mm per year, and in some regions of only 500–600 mm of rain per year. The rains are irregular, with insignificant to low rainfall occurring for some years. The rainy season is short, from January-February to April-May. The humidity is low, ranging from 60% to 75%, and the vegetation is typical of caatinga (“white forest” in the Tupi-Guarani language), an exclusive Brazilian biome, occupying almost 11% of the country. This vegetation is characteristic of the arid conditions (xerophytic) with a strong presence of bushes with twisted branches and deep roots, and the presence of cacti and bromeliads. Bushes lose almost all of their leaves in times of drought.
The seasonal variation of EEE, as well as other arboviral diseases, is associated with a particular temperature range, rainfall, and other environmental factors that determine the density of vectors. 9 The rainfall of Paraíba, in 2009 (1,345 mm), was among the highest in the last 15 years (mean of 864 mm), suggesting that the increased rainfall is an important factor for the occurrence of outbreaks, which occur preferentially at the end of the rainy season (Fig. 4) when there is an increase in vector population.
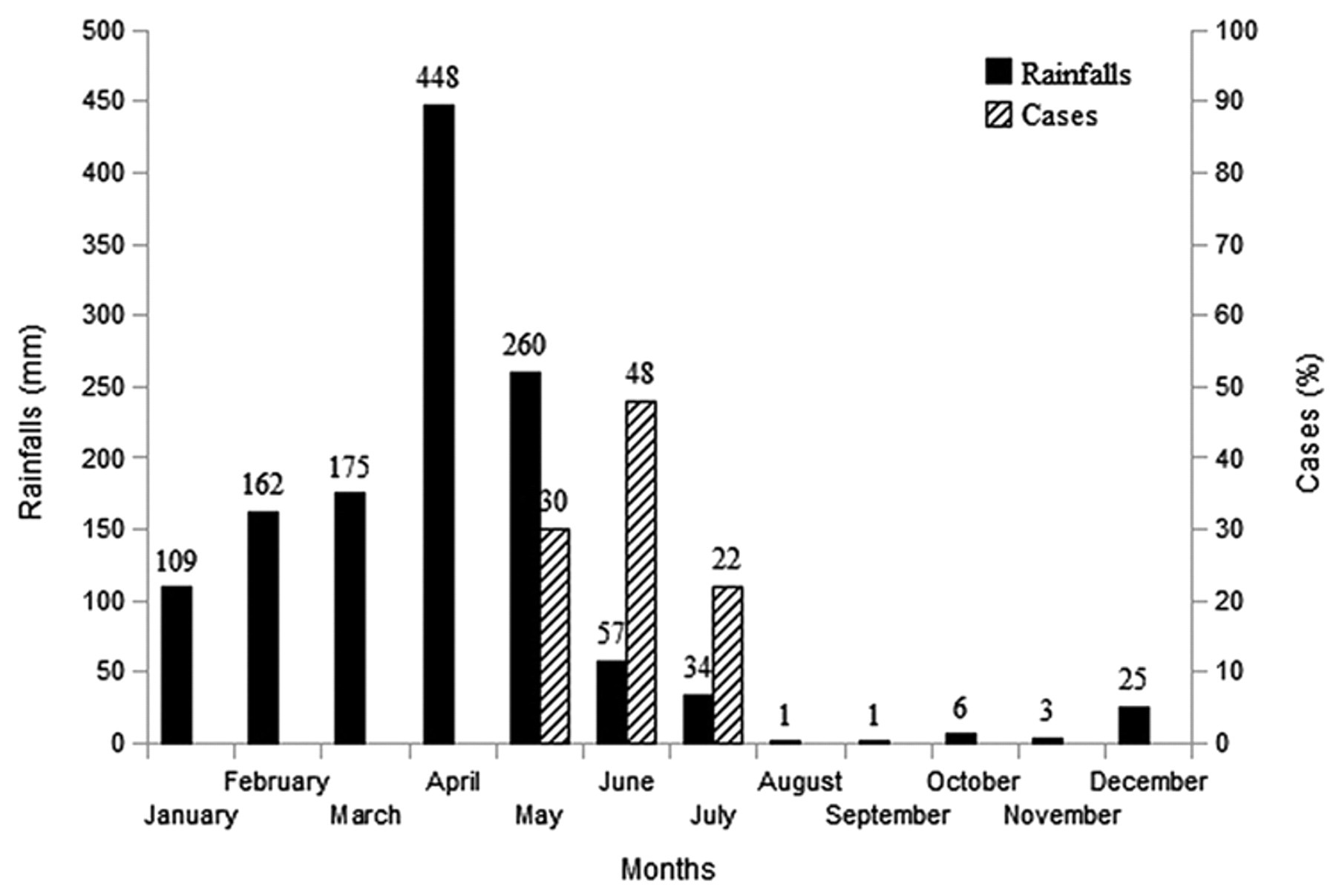
Mean monthly rainfall, from January to December 2009, in 16 Brazilian municipalities (Belém do Brejo do Cruz, Cajazeiras, Catolé do Rocha, Condado, Coremas, Itaporanga, Malta, Patos, Sousa, São Francisco, Vista Serrana, Poço de José de Moura, São Bentinho, São José do Brejo do Cruz, Uiraúna, São João do Rio do Peixe) in the state of Paraíba where eastern equine encephalitis occurred. The monthly distribution of cases observed in the State is also presented.
The morbidity and fatality rates suggest that the isolate from the first outbreak was more pathogenic than the isolate from the second outbreak. Eastern equine encephalitis virus is considered more pathogenic than WEEV and Venezuelan equine encephalomyelitis virus, although considerable variation in pathogenicity may occur. 8 Epidemiologic characteristics (epidemic and seasonal) as well as clinical signs suggesting mainly cerebral and brain stem involvement allowed differentiation of EEE from rabies, Crotalaria retusa poisoning, and leukoencephalomalacia, which are common diseases of horses in the Brazilian semiarid. 13
It is concluded that EEE is an important disease of horses in northeastern Brazil. Although the epidemiology, clinical signs, and pathology allow the presumptive diagnosis of the disease, it is necessary to submit specimens for laboratory diagnosis to determine the etiologic agent of the encephalitis. Such identification is very important for the surveillance of equine arboviruses because of their public health importance, in addition to any economic loss.
Footnotes
a.
TRIzol, Invitrogen Brasil Ltda., São Paulo, Brazil.
b.
QIAquick®, QIAGEN Biotecnologia Brasil Ltda., São Paulo, Brazil.
c.
Low DNA Mass™ Ladder, Invitrogen Brasil Ltda., São Paulo, Brazil.
d.
BigDye® Terminator v3.1, Applied Biosystems do Brasil, São Paulo, Brazil.
e.
Sephadex™ G-50, GE Healthcare do Brasil Ltda., São Paulo, Brazil.
f.
ABI 3130, Applied Biosystems do Brasil, São Paulo, Brazil.
The authors declared no potential conflicts of interests with respect to the authorship and/or publication of this article.
The authors received no financial support for the research and/or authorship of this article.
