Abstract
In dogs, Staphylococcus pseudintermedius is a common colonizer and is associated with pyoderma, otitis externa, and urinary tract infections. In the current study, nasal, pharyngeal, and rectal swabs were taken from 175 healthy dogs and cultured for S. pseudintermedius. The organism was found in 153 dogs (87.4%), including individuals in which it exclusively colonized in the nares (n = 1), pharynx (n = 16), and rectum (n = 17). Antimicrobial susceptibility testing revealed that a remarkably susceptible population, 46.4% of isolates, was susceptible to all drugs tested, and resistance to penicillin (39.9%) and tetracycline (23.5%) were most common. No methicillin (oxacillin)-resistant isolates were identified. Although 3.3% of isolates were erythromycin resistant, no inducible clindamycin resistance was found. The data provide a baseline for future resistance surveillance and indicate that multiple body sites, including at least the pharynx and rectum, should be tested.
Staphylococcus pseudintermedius, first described as distinct from Staphylococcus intermedius in 2005, is a coagulase-positive staphylococcal species frequently associated with pyoderma, otitis externa, urinary tract infections (UTIs), and opportunistically infected sites in dogs. 2,6,18,19 Colonization with S. pseudintermedius is common, and it is reported in up to 68% of healthy dogs. 8 Although the nares, mouth, rectum, groin, and forehead have been sampled in previous studies, the prevalence of single-site colonization has not been evaluated. 8,9 In human beings and dogs, multiple body sites, including the nares, pharynx, and gastrointestinal tract, play a role in Staphylococcus aureus colonization, although some individuals are single-site colonized. 3,7,10,11,14 Knowledge of sites of colonization is essential for conducting surveillance studies; failing to sample relevant sites may result in false-negatives and an underestimation of its prevalence. Conversely, the increased expense and time required for excessive sampling are undesirable.
Antimicrobial resistance, including methicillin-resistant S. pseudintermedius (MRSP), is increasing among S. pseudintermedius. 18 Although clinical S. pseudintermedius isolates from the Saskatoon region of Canada have remained largely susceptible, the recent identification of a multidrug-resistant MRSP from a canine UTI suggests that the locally prevalent resistance patterns are changing. 2,13,15 The purpose of the current study was to determine the relative prevalence of nasal, pharyngeal, and rectal colonization of healthy dogs with S. pseudintermedius, and to characterize the antimicrobial susceptibility profiles of such isolates to an extensive panel of drugs.
Between May and November 2008, 175 dogs presenting to the Western College of Veterinary Medicine (Saskatoon, Saskatchewan, Canada) for routine health checks and vaccination were sampled. One dog per household was included in the study. Using individual, commercially prepared culturettes, a nasal, pharyngeal, and rectal samples were taken. Of the 175 dogs sampled, it was not possible to collect all 3 specimens from 8 fractious animals; therefore, only 167 dogs were completely sampled.
All samples were processed within 5 hr of collection. Swabs were streaked on chromogenic S. aureus medium b and mannitol salt agar c both with and without 4 μg/ml oxacillin d and trypticase soy agar with 5% sheep's blood. c Swabs were then put into a tube containing 2 ml of enrichment broth with 75 g/l sodium chloride. 17 After overnight incubation at 35°C, broth cultures were inoculated onto the same 5 solid media to detect low bacterial numbers. Plates were evaluated at 24, 48, and 96 hr. Identification of S. pseudintermedius was based on colony morphology on blood agar; Gram staining characteristics; the production of catalase, DNase, and coagulase; and lack of hyaluronidase or acetoin production. 6,20
The susceptibility of 153 S. pseudintermedius isolates (1 per dog) to 33 antimicrobials was tested using a commercially available system. e The minimum inhibitory concentrations (MICs) of the following antimicrobials were determined: β-lactams: ampicillin (AMP), ceftiofur (CEF), penicillin (PEN), and oxacillin (OXA); macrolide/lincosamide/ketolides: clindamycin (CLI), erythromycin (ERY), telithromycin (TEL), tilmicosin (TIL), tulathromycin (TUL), and tylosin (TYL); fluoroquinolones: ciprofloxacin (CIP), danofloxacin (DAN), enrofloxacin (ENR), gemifloxacin (GEM), and moxifloxacin (MOX); tetracyclines: chlortetracycline (CLO), oxytetracycline (OXY), and tetracycline (TET); aminoglycosides: gentamicin (GMS), neomycin (NEO), and spectinomycin (SPT); and other drug classes: chloramphenicol (CHL), daptomycin (DAP), florfenicol (FFC), linezolid (LZD), nitrofurantoin (NIT), quinupristin/dalfopristin (QDA), rifampin (RIF), trimethoprim/sulfamethoxazole (SXT), tiamulin (TIA), and vancomycin (VAN). Clinical and Laboratory Standards Institute (CLSI) testing protocols and interpretive criteria were used for all drugs except oxacillin, where the newly implemented resistance breakpoint (0.5 μg/ml) was used (Papich MG: 2010, Proposed changes to Clinical Laboratory Standards Institute interpretive criteria for methicillin-resistant Staphylococcus pseudintermedius isolated from dogs. J Vet Diagn Invest 22:160. Letter to the Editor). 4,5 For drugs without CLSI resistance breakpoints, MICs were not categorically analyzed. Mupirocin (MUP) and fusidic acid (FUS) susceptibility was determined using the disk diffusion method f according to British Society for Antimicrobial Chemotherapy guidelines. 1 Isolates resistant to ERY and susceptible to CLI were tested for inducible CLI resistance according to CLSI guidelines. 5 For quality control, S. aureus American Type Culture Collection (ATCC) 29213, S. aureus ATCC 25923, Enterococcus faecalis ATCC 29212, and Escherichia coli ATCC 25922 were used.
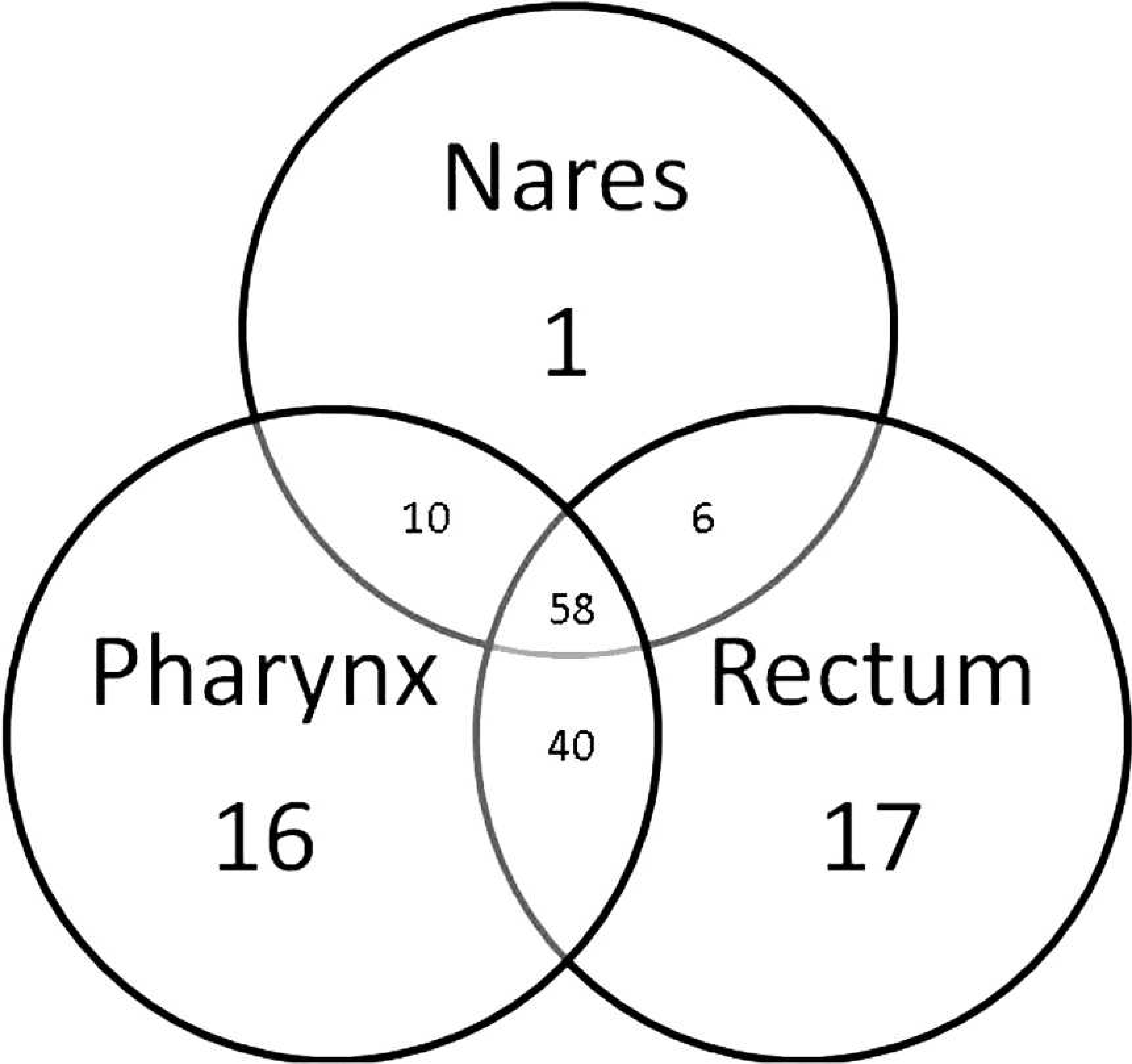
Venn diagram of sites of colonization.
Overall, S. pseudintermedius was identified in 153 (87.4%) of 175 dogs and in 148 (88.6%) of 167 completely sampled dogs, higher than previously reported. 8 Nasal, pharyngeal, and rectal colonization was identified in 75, 124, and 121 dogs, respectively (Fig. 1). A total of 1 (0.7%), 16 (10.5%), and 17 (11.1%) dogs were exclusively nasally, pharyngeally, and rectally colonized, respectively. To maximize the sensitivity of S. pseudintermedius detection, future studies should include at least the pharynx and rectum, which together accounted for 99.3% of carriers.
Susceptibility to all drugs was the most common phenotype identified, including 71 isolates (46.4%; Table 1). No resistance to OXA (MRSP), CEF, GMS, MOX, CIP, ENR, CHL, SXT, RIF, NIT, VAN, LZD, QDA, DAP, or FUS was found (Fig. 2). Although no MRSPs were identified, the isolates were not screened for mecA, the gene conferring methicillin resistance; however, susceptibility to OXA is reported to be 97% sensitive in detecting methicillin resistance and was considered sufficient for the purposes of the current study. 16 Resistance to PEN and TET was found most commonly, occurring in 39.9% and 23.5% of isolates, respectively. However, nearly half of PEN-resistant (30 of 61) and half of TET-resistant (18 of 36) isolates were resistant to only 1 drug. Five isolates (3.6%) were macrolide resistant, including a single isolate only resistant to ERY. Two of four CLI-resistant isolates were also PEN resistant but remained susceptible to TET. No inducible CLI resistance was found. A single isolate resistant to MUP was identified, which was also resistant to PEN, AMP, and TET, making it the only multidrug-resistant isolate (resistant to 3 or more antimicrobial classes). High rates of MUP resistance (>50%) have been reported among human MRSA patients in Saskatchewan, but little is known about the susceptibility of canine staphylococci to this drug. 12 No isolates were simultaneously resistant to β-lactams, tetracyclines, and macrolides, affirming the utility of these therapeutic options. A previous study including clinical canine S. pseudintermedius from Saskatoon reported lower levels of PEN resistance, but a higher prevalence of TET, ERY, CLI, and SXT resistance. 13 The lack of resistance to the fluoroquinolones GMS, CHL, and RIF is consistent with earlier studies from this area. 2,13
Antimicrobial susceptibility profiles of Staphylococcus pseudintermedius isolated from 153 healthy dogs.*
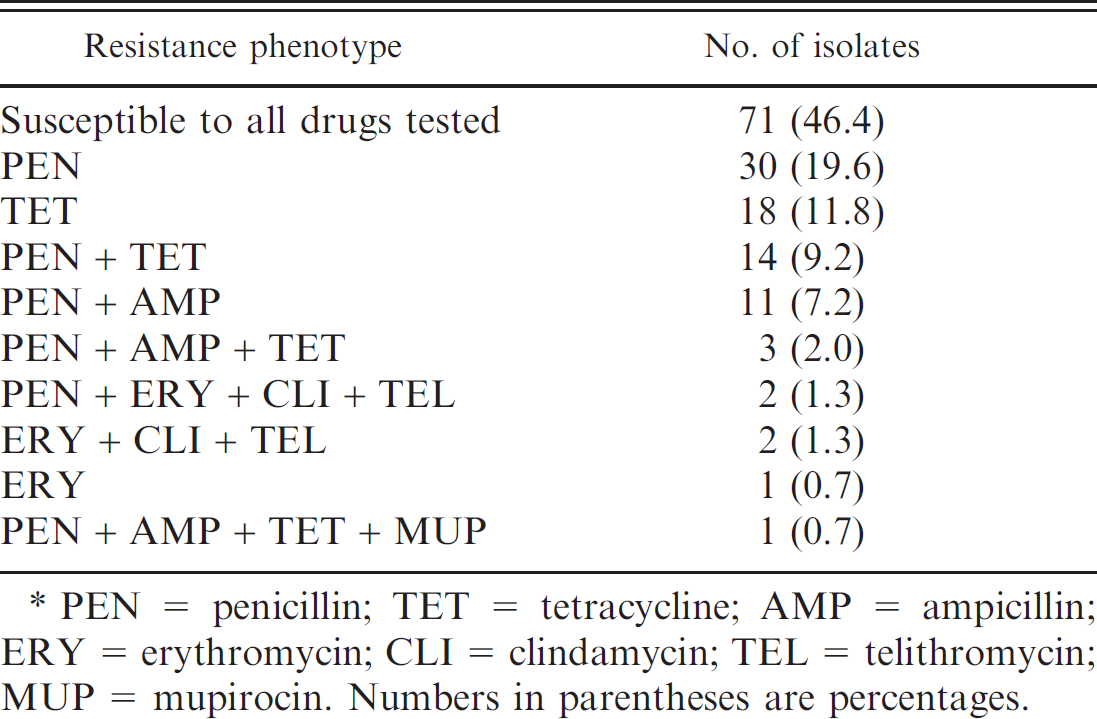
PEN = penicillin; TET = tetracycline; AMP = ampicillin; ERY = erythromycin; CLI = clindamycin; TEL = telithromycin; MUP = mupirocin. Numbers in parentheses are percentages.
Because the current study is intended to serve as a baseline for S. pseudintermedius resistance surveillance, MICs of an extensive panel of drugs were determined. Monitoring antimicrobial MICs allows changes in susceptibility below the resistance breakpoint to be detected compared with categorical analyses, which are relatively insensitive. Nearly uniform susceptibility to RIF, TIA, NIT, LZD, DAP, VAN, CIP, GEM, MOX, GMS, CEF, OXA, and QDA was found, with all isolates inhibited by the lowest 2 concentrations tested, and below the resistance breakpoint, where available. As old drug classes are “rediscovered” to treat resistant infections, such as pleuromutilin (retapamulin) and its derivatives, which are now used for treating MRSA in human beings, historical susceptibility data to related compounds (tiamulin) will be invaluable in detecting the emergence of resistance. 21
The inclusion of S. pseudintermedius isolated from healthy dogs may be more indicative of isolates involved in first-time community-associated infections than of diagnostic samples, addressing an important gap in the veterinary literature. The recognition of highly susceptible S. pseudintermedius is important in balancing the publication bias toward resistant organisms. The emphasis on resistance may encourage the empiric use of broad-spectrum antimicrobials by both veterinarians and human health professionals. Although empiric therapy is often unavoidable in clinical practice, it should be stressed that culture and susceptibility testing are cornerstones of prudent use.
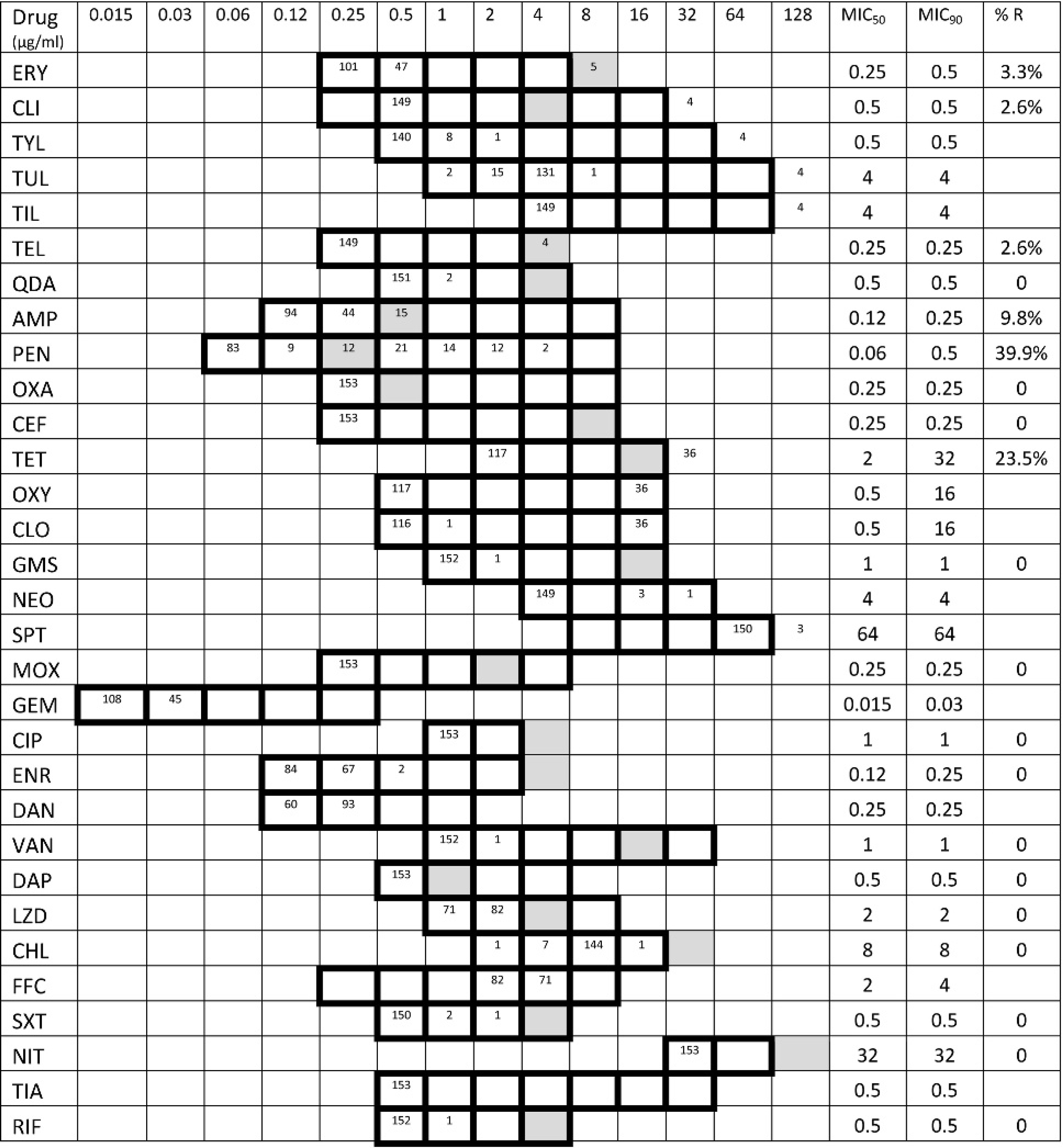
Antimicrobial minimum inhibitory concentration (MIC) distribution of Staphylococcus pseudintermedius (n = 153). Cells corresponding to concentrations tested are outlined in bold. The number of isolates inhibited at each concentration are noted in each cell; isolates inhibited at the lowest concentration tested should be considered to have an MIC less than or equal to that concentration. Cells corresponding to Clinical and Laboratory Standards Institute resistance breakpoints are shaded; for daptomycin (DAP) and linezolid (LZD), susceptible breakpoints are shaded. Erythromycin (ERY), clindamycin (CLI), tylosin (TYL), tulathromycin (TUL), tilmicosin (TIL), telithromycin (TEL), quinupristin/dalfopristin (QDA), ampicillin (AMP), penicillin (PEN), oxacillin (OXA), ceftiofur (CEF), tetracycline (TET), oxytetracycline (OXY), chlortetracycline (CLO), gentamicin (GMS), neomycin (NEO), spectinomycin (SPT), moxifloxacin (MOX), gemifloxacin (GEM), ciprofloxacin (CIP), enrofloxacin (ENR), danofloxacin (DAN), vancomycin (VAN), DAP, LZD, chloramphenicol (CHL), florfenicol (FFC), trimethoprim/sulfamethoxazole (SXT), nitrofurantoin (NIT), tiamulin (TIA), and rifampin (RIF).
Continued surveillance of antimicrobial resistance among coagulase-positive staphylococci, including S. pseudintermedius, is required to monitor the emergence and dissemination of resistance. Relatively little is known about the epidemiology, frequency of zoonotic transmission, or antimicrobial resistance of S. pseudintermedius colonizing healthy dogs. Necessitated by both animal and public health concerns, more research is needed to address these important issues.
Acknowledgements
The authors would like to thank the staff, students, and administration of the Western College of Veterinary Medicine teaching hospital for their support in conducting this study, and the Companion Animal Health Fund for funding. The University of Saskatchewan Committee on Animal Care and Supply and the biomedical Research Ethics Board approved this study. The authors have no conflicts of interest to declare.
Footnotes
a.
BBL™ CultureSwab™, BD Diagnostic Systems, Sparks, MD.
b.
CHROMagar, Paris, France.
c.
BD Diagnostic Systems, Sparks, MD.
d.
Sigma-Aldrich, St. Louis, MO.
e.
Sensititre®, TREK Diagnostic Systems Inc., Cleveland, OH.
f.
Oxoid Ltd., Cambridge, United Kingdom.
