Abstract
The aim of this study was to determine the value of C-reactive protein (CRP) in the diagnosis of patients with neonatal sepsis by a meta-analysis. Potential relevant studies were searched through the PubMed, Embase, and Cochrane Library databases before February 2016. We combined estimates of sensitivity, specificity, positive likelihood ratio (PLR), negative likelihood ratio (NLR), and diagnostic odds ratio (DOR) with their 95% confidence intervals (CIs) of CRP for neonatal sepsis diagnosis. Summary receiver operating characteristic (SROC) curve was applied to evaluate the diagnostic value of CRP. The meta-regression and subgroup analysis were performed when heterogeneity was significant. In total, 31 studies were included in our meta-analysis with 5698 participants. The overall estimates for CRP in the diagnosis of neonatal sepsis were: sensitivity 0.69 (95% CI, 0.66–0.71), specificity 0.77 (95% CI, 0.76–0.78), PLR 3.83 (95% CI, 3.03–4.84), NLR 0.38 (95% CI, 0.31–0.45), and DOR 12.65 (95% CI, 8.91–17.94). The area under the curve (AUC) and Q* index were 0.8458 and 0.7773. Meta-regression analysis showed that heterogeneity was irrelevant to test time, cutoff value, assay method of CRP, neonates, and sepsis type. Heterogeneity still existed but decreased after subgroup analysis. CRP might be a valuable approach for the diagnosis of neonatal sepsis.
Introduction
Neonatal sepsis is a common severe bacterial infection of the bloodstream, presenting in a newborn baby, 1 with a high mortality. 2 It has been noted that neonatal sepsis causes over 520,000 deaths of newborns annually.3,4 Neonatal sepsis is often not diagnosed until serious because the early signs are neither specific nor sensitive. 1 It is divided into two categories: early-onset sepsis (<72 h) and late-onset sepsis (>72 h). 5 Nowadays, early and reliable diagnosis of neonatal sepsis has become a topic in the medical field.
The gold standard test for neonatal sepsis is the blood culture. However, it is limited by the disadvantages of time-consuming and false negative results. 6 Recently, several biochemical markers, such as C-reactive protein (CRP), procalcitonin (PCT), and tumor necrosis factor alpha (TNF-α), have been proposed as potential markers for diagnosis of neonatal sepsis. 7 CRP, a pentraxin protein, plays an important role in inflammatory and/or infectious stimuli, thus being regarded as an acute-phase protein in neonatal sepsis. 8 Previous studies have declared that CRP is a useful diagnostic marker for the early stages of neonatal sepsis.9,10 Another study has found that CRP is the best single marker of neonatal sepsis between 24 and 48 h of onset, with high sensitivity and specificity. 11 However, Benitz et al. thought that the positive predictive value of elevated CRP levels is low, especially for culture-proven early-onset infections. 12 What’s more, CRP has also been found just in the laboratory test the best method for the diagnosis of neonatal sepsis. 13 Therefore, the diagnostic value of CRP is still controversial when used for the diagnosis of neonatal sepsis.
In the present study, we aimed to adequately evaluate the accuracy of CRP in the diagnosis of neonatal sepsis by a systematical review and meta-analysis of the relevant studies. The diagnostic accuracy variables of sensitivity, specificity, positive likelihood ratio (PLR), negative likelihood ratio (NLR), and diagnostic odds ratio (DOR) with their 95% confidence intervals (CIs) were pooled to evaluate the diagnosis value of CRP. In addition, meta-regression and subgroup analysis were performed to explore the sources of between-study heterogeneity.
Materials and methods
Data sources and search strategy
A computerized search was conducted using the electronic databases of PubMed, Embase, and the Cochrane Library before February 2016. The search strategy was based on the following index terms variably combined: C-reactive protein (CRP), neonatal (newborn, infant) sepsis, neonatal septicemia, diagnosis. Moreover, reference lists of relevant studies were searched manually for additional publications. There was no language restriction in our study.
Inclusion and exclusion criteria
Studies were eligible for this meta-analysis when they were accord with the following inclusion criteria: (1) the study was designed as a clinical trial; (2) CRP was involved in detection of neonatal sepsis; (3) the participants were neonates who were diagnosed with sepsis by golden standards; (4) the golden standards for diagnosis of sepsis were definite in the study; and (5) the numbers of true positive (TP), false positive (FP), false negative (FN), and true negative (TN) were provided in the original study or could be obtained by calculation.
The major exclusion criteria were: (1) studies with incomplete data; (2) reviews or reports or comments; and (3) reduplicates or meeting records or letters.
Data extraction and quality assessment
The included studies were reviewed and data were extracted by Liyun Xu and Qiubo Li independently. The data items from the eligible studies included: (1) general information, such as the first author’s surname, year of publication, country of origin, study period, study population characteristics (number of cases, age and weight), assay method, golden standard, test time, and cutoff value; and (2) information of diagnostic parameter, such as TP, FP, FN, and TN. Any disagreement during the data extraction was solved by discussing with PY to reach a consensus.
The quality of the included studies was assessed with the Quality Assessment of Diagnostic Accuracy Studies-2 (QUADAS-2) [14]. There were four domains involved in QUADAS-2, consisting of patient selection, index test, reference standard, and flow and timing. All the domains were involved in the risk of bias assessment, and three domains of patient selection, index test, and reference standard were evaluated for applicability concerns. The methodological quality assessment figure of the included studies was drawn by using the software of Review Manager version 5.2 (RevMan 5.2; The Cochrane Collaboration, Oxford, UK).
Statistical analysis
Sensitivity, specificity, PLR, NLR, and DOR with their 95% CIs were chosen as the diagnostic accuracy variables. Heterogeneity across the included studies was tested according to Cochran Q test and I2 statistic. The random effects model was used to pool the data when significant heterogeneity was observed (P <0.05, I2 > 50%), and oppositely, the fixed effect model was performed. In addition, a summary receiver operating characteristic (SROC) curve was applied. The area under the curve (AUC) and an index Q* were computed to evaluate the diagnostic value of CRP for neonatal sepsis. Threshold effects were assessed graphically by visual inspection of the SROC curve and statistically by calculating the Spearman correlation coefficient. The meta-regression and subgroup analyses were performed when heterogeneity was significant. Those statistical analyses were performed using Meta-DiSc v.1.4 (http://www.hrc.es/investigacion/metadisc_en.htm). α=0.05 was used as the significance level.
Results
Search results
The flow chart of study selection was illustrated in Figure 1. The literature search yielded 851 potentially relevant studies in total. There were 503 studies remained after removing non-clinical trials and duplicated studies. By abstract evaluation, 457 studies were excluded for lack of CRP-related diagnostic data. Full-text assessment was done with the left 46 studies, and 15 studies were excluded (10 studies without appropriate CRP diagnostic data; three studies with other diseases infected by bacteria; two studies in which analytical data unmatched with the cases). Ultimately, 31 studies7–13,15–38 were included in this meta-analysis.
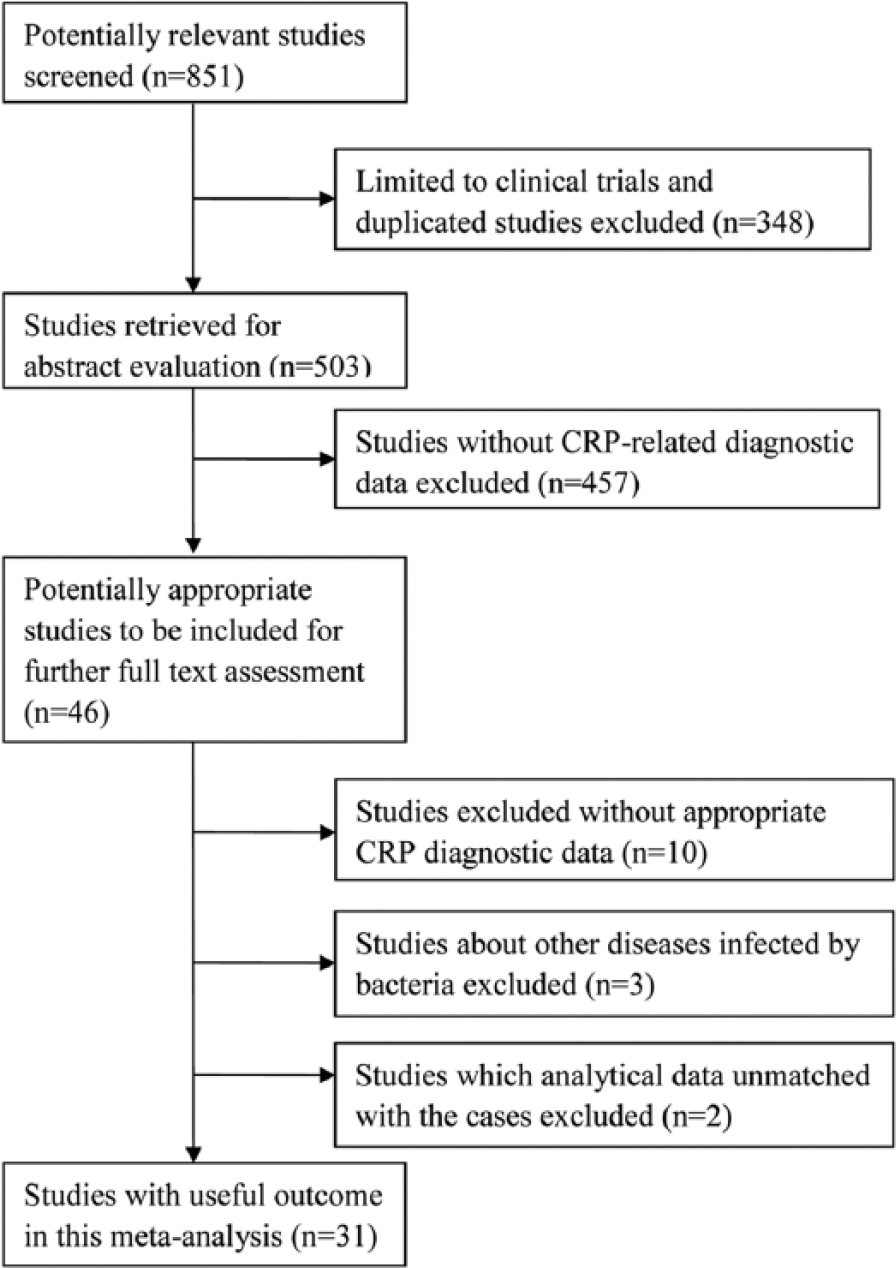
The flowchart of literature search and study selection. CRP, C-reactive protein.
Characteristics of eligible studies
The characteristics of the included studies were summed up in Table 1. There were 5698 participants involved in our meta-analysis. The 31 included studies were published between 1993 and 2015. Countries of origin distributed worldwide, such as Iran, Canada, Finland, Turkey, Italy, and America. The study period ranged from 3 to 33 months. The participants were patients in the neonatal intensive care unit with various ages (within 40 h). Assay methods of CRP in those included studies mainly included: immunonephelometry, nephelometry, an enzyme multiplied immunoassay technique, and ELISA. Cutoff values were different among the included studies (mostly 10 mg/L). All the patients were confirmed neonatal sepsis using clinical or/and blood culture. The quality assessment was shown in Figure 2. Twenty-five studies were high-quality and six studies were low-quality.
The characteristics of the included studies in this meta-analysis.
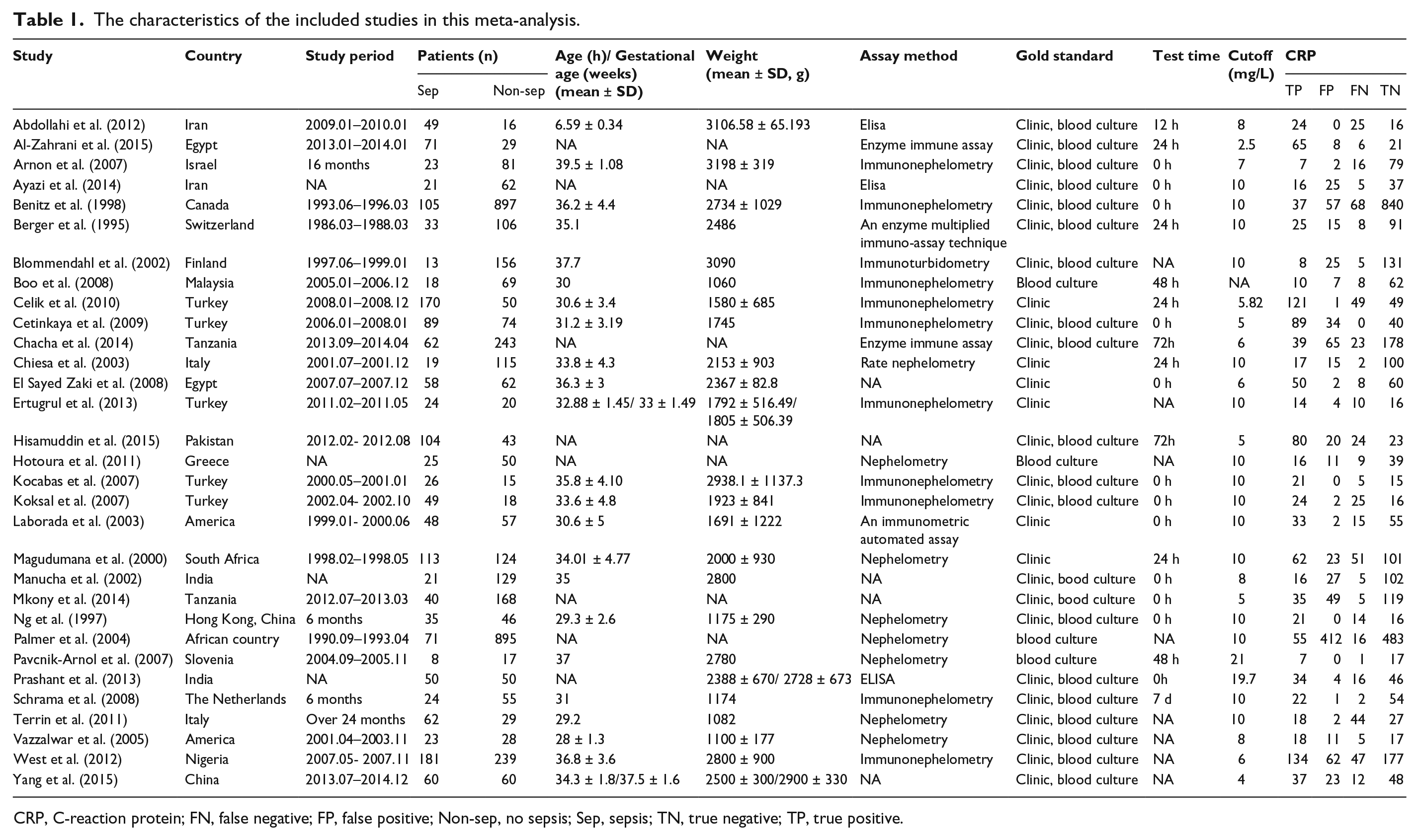
CRP, C-reaction protein; FN, false negative; FP, false positive; Non-sep, no sepsis; Sep, sepsis; TN, true negative; TP, true positive.
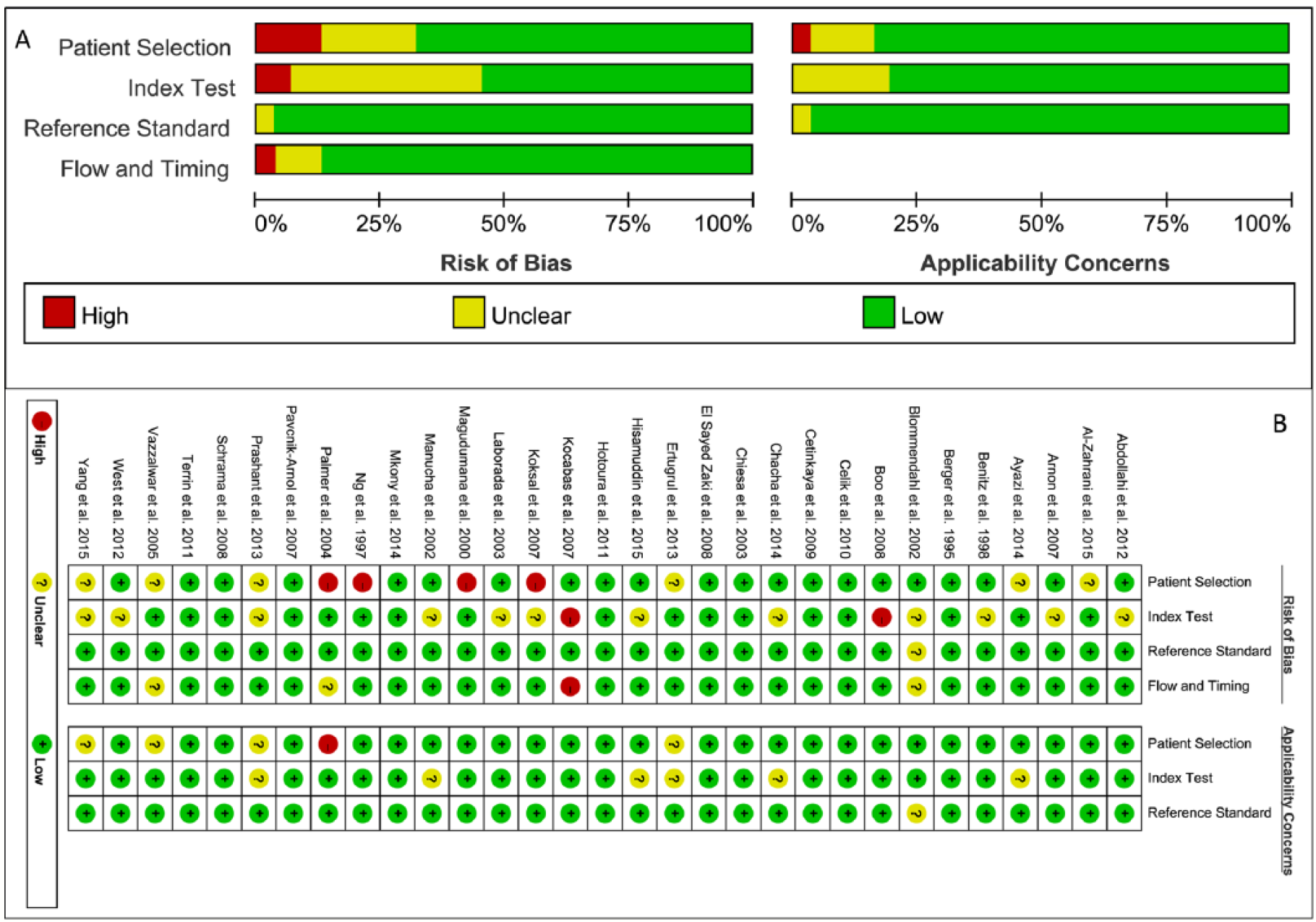
(a) The results of quality assessment of the included articles based on the risk of bias and applicability concerns. (b) The results of quality assessment of the included articles.
Meta-analysis
No threshold effect was detected according to the shape of the SROC curve (Figure 3) and Spearman correlation coefficient (r = 0.143, P = 0.526). The AUC was 0.8458 and Q* was 0.7773. The results of the meta-analysis were summarized in Table 2. Significant heterogeneity of the included studies was shown on the estimates of sensitivity (P <0.01, I2 = 88.9%), specificity (P <0.01, I2 = 95.3%), PLR (P <0.01, I2 = 84.9%), NLR (P <0.01, I2 = 84.1%), and DOR (P <0.01, I2 = 68.3%). Therefore, we chose the random effects model to pool the data. The overall sensitivity, specificity, PLR, NLR, and DOR were 0.69 (95% CI, 0.66–0.71), 0.77 (95% CI, 0.76–0.78), 3.83 (95% CI, 3.03–4.84), 0.38 (95% CI, 0.31–0.45), and 12.65 (95% CI, 8.91–17.94), respectively.

The summary receiver operating characteristic curve of C-reactive protein. Each red circle represents each study in the meta-analysis. The size of the red circle indicates the size of each study. AUC, area under the curve; SROC, summary receiver operating characteristic.
Summary of the outcomes of the overall meta-analysis and subgroup analysis.

Meta-regression analysis
We conducted a meta-regression analysis to explore the source of heterogeneity due to no threshold effects. DOR was used for the accuracy measure, as it is a unitary measure of diagnostic performance, encompassing both sensitivity and specificity or both PLR and NLR. 39 The variables included cutoff value (10 mg/L vs. others), test time (0 h vs. others), and assay method of CRP (immunoturbidimetry vs. others), as well as neonates (premature infants vs. term infant vs. others) and sepsis type (early-onset sepsis vs. late-onset sepsis vs. others). Meta-regression analysis was performed by seriatim excluding each concomitant variable. The results showed these variables did not contribute to the between-study heterogeneity (Table 3).
The results of meta-regression analysis.

CI, confidence intervals; Coeff, coefficient; RDOR, ratio of diagnostic odds ratio; Std. Err., standard error; Var, variable.
Subgroup analysis
The subgroup analysis was performed with six studies11,12,21–23,31 which used the standard of cutoff value = 10 mg/L and test time = 0 h. The results (Table 2) revealed that heterogeneities still existed for sensitivity (P <0.01, I2 = 84.8%), specificity (P <0.01, I2 = 91.6%), PLR (P <0.01, I2 = 85.7%), NLR (P <0.01, I2 = 79.6%), and DOR (P = 0.0273, I2 = 60.3%), with slight decrease. Thus, we used the random effects model to pool the data. The pooled sensitivity, specificity, PLR, NLR, and DOR were 0.54 (95% CI, 0.48–0.59), 0.92 (95% CI, 0.90–0.93), 6.30 (95% CI, 2.49–15.94), 0.44 (95% CI, 0.31–0.62), and 13.99 (95% CI, 5.62–34.80). The AUC was 0.8037 and Q* was 0.7391.
Discussion
Neonatal sepsis is a common and catastrophic illness. 40 It is indispensable to find an early and accurate diagnosis to decrease morbidity of neonatal sepsis. In the present study, we provided an insight understanding of the diagnostic value of CRP in neonatal sepsis. This meta-analysis included 31 studies, involving 1695 cases and 4003 controls. The overall results suggested that CPR had a moderate specificity (77%) and high diagnostic value (AUC 84.6%) and diagnostic accuracy (with high DOR 12.65) for neonatal sepsis, while the sensitivity (69%), PLR (3.83), and NLR (0.38) were moderate.
Several biomarkers have been used for diagnosis of neonatal sepsis, such as PCT. 41 Kocabas et al. 21 compared the diagnostic effects of PCT, CRP, IL-6, IL-8, and TNF-α, and recommended PCT (cutoff 0.34 ng/mL) and TNF-α (cutoff 7.5 pg/mL) as the best markers (sensitivity 100% and 100%, specificity 96.5% and 96.6%) and CRP (cutoff 10 mg/L) had a sensitivity of 80.7% and specificity of 100%. Besides, Celik et al. 19 have proposed that the combination of CRP and IL-6 is useful for the early diagnosis of neonatal sepsis, however, Kocabas et al. 21 indicated that the diagnostic effect mainly attributes to CRP, not IL-6. CRP has a similar diagnostic accuracy with PCT. A recent meta-analysis reported a AUC of 0.87 of PCT for neonatal sepsis 42 which is consistent with our study for CRP (AUC = 0.85), whereas in our study, the sensitivity (69%) and the specificity (77%) were lower than PCT (sensitivity 81%, specificity 79%). Enguix et al. also suggest a similar result that CRP (cutoff 23.0 mg/L) has a lower accuracy (sensitivity 95.8%, specificity 83.6%) than PCT (cutoff 6.1 ng/mL; sensitivity 98.6%, specificity 88.9%). 43 As for TNF-α, Lv et al. 44 conducted a meta-analysis which indicated a similar diagnostic efficiency of TNF-α for diagnosis of both early-onset (sensitivity 66%, specificity 76%) and late-onset neonatal sepsis (sensitivity 68%, specificity 89%) to the efficiency of CRP in our study. A direct comparison of CRP with PCT or TNF-α by a meta-analysis is critical for selection of biomarkers of neonatal sepsis. Besides, another study has found that it is safe and accurate to use CRP combined with serum amyloid A in diagnosis and follow-up of neonatal sepsis. 8 Therefore, we conjecture the specificity of CRP may be elevated when used in combination with other diagnostic markers for the diagnosis of neonatal sepsis.
A combination of CRP (1 mg/L) and PCT (1 μg/mL) results in a higher specificity (86%) but lower sensitivity (58%) which are similar with those of CRP alone (specificity 84%, sensitivity 58%). 18 The results of CRP plus blood immature to total neutrophil leucocyte ratio are also unsatisfactory. 18 The combination of CD64 (6136 antibody-PE molecules bound/cel) and CRP (10 mg/L) results in a sensitivity of 81%, and specificity of 82%. 45 Ng et al. suggests an ideal model for late-onset neonatal sepsis diagnosis that CRP (0 h, 12 mg/L) plus CD64 (24 h, 4000 PE-molecules bound/cell) produces a high accuracy (sensitivity 100%, specificity 90%, positive predictive value 80%, negative predictive value 100%). 46 Thus, further studies are needed on the cutoff of CRP and different combinations of these biomarkers.
It should be noted that although no threshold effect was detected, significant heterogeneity was observed across the included studies when used the estimation of diagnostic accuracy variables. Meta-regression analysis showed that the sources of between-study heterogeneity were irrelevant to test time, cutoff value, assay method of CRP, neonates, and sepsis type. However, subgroup analysis of studies with cutoff 10 mg/L and test time 0 h still showed significant heterogeneity. Thus, cutoff value (10 mg/L) and test time (0 h) may be potential for diagnosis of neonatal sepsis. Importantly, the specificity reached to 92% in the subgroup analysis.
A total of 31 studies were included in our meta-analysis with high quality. However, there were four included studies11,22,24,26 with the risk of patient selection bias. Studies by Boo et al. 10 and Kocabas et al. 21 were at the risk of index test bias. Kocabas et al. 21 also had a high risk of flow and timing. The index test remains unclear in 12 included studies,12,15,16,18,22,23,25,30,35–38 in which CRP was not described in detail (such as assay method and study period). These aspects may influence the analysis of the accuracy of CRP. Therefore, further study is required for more high-quality studies.
Nevertheless, some limitations in our meta-analysis should be aware. First, the considerable degree of between-study heterogeneity was observed in this meta-analysis. This issue was addressed by adoption of the more conservative random effects model to estimate each effect size. Besides, a subgroup analysis by excluding studies with cutoff value ≠ 10 mg/L and test time ≠ 0 h. In addition, meta-regression analysis of several potential factors (cutoff, test time, infants, and sepsis types) was performed to identify potential sources of heterogeneity; however, regrettably, we could not find the exact sources of the heterogeneity. Second, only CRP, as one of the diagnostic markers of neonatal sepsis, was considered in this meta-analysis without comparison between CRP and other markers for the diagnosis of neonatal sepsis. Third, sample sizes in some included studies were small and the study periods were short.
In conclusion, CRP may be a valuable approach for the diagnosis of neonatal sepsis (sensitivity 69%, specificity 77%). It may be feasible for CRP to elevate sensitivity by combining with other diagnostic markers. Further investigation is warranted with more high-quality studies, larger sample sizes, and standardization of the techniques via international collaboration. Comparisons between CRP and other biomarkers or combination of them are imperative to explore a better biomarker for neonatal sepsis diagnosis.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
