Abstract
Syringomyelia is a common clinical problem in the Cavalier King Charles Spaniel dog population. The underlying pathophysiology of the development and progression of syringes is currently unknown. The primary aim of the current study was to determine whether development of syringomyelia is accompanied by alterations in cerebrospinal fluid composition. Patient records of Cavalier King Charles Spaniels with Chiari-like malformations, with or without concurrent syringomyelia, that had magnetic resonance imaging and cerebrospinal fluid analysis between December 2004 and December 2009, were retrospectively reviewed. Total nucleated cell count per microliter, with differential count, red blood cell count per microliter, and protein concentration (g/l), were reviewed. Data were analyzed with the Mann–Whitney U-test. Spearman rank test was used to test association between cerebrospinal fluid composition and syrinx size, and the Fisher exact test was used to compare the presence of macrophages, reactive lymphocytes, and myelin. A P value of ≤0.05 was considered significant for statistical tests. Dogs with syringomyelia had a higher total nucleated cell count (syringomyelia: 4/μl [range, 0–15/μl] vs. without syringomyelia: 2/μl [0–8/μl]; P = 0.0047), higher protein concentration (syringomyelia: 0.26 g/l [0.07–0.42 g/l] vs. without syringomyelia: 0.2 g/l [0.12–0.39 g/l]; P = 0.039), and an increased neutrophil percentage (syringomyelia: 0% [0–37%] vs. without syringomyelia: 0% [0–21%]; P = 0.0203) than those with the Chiari-like malformation alone. There was a positive correlation between total nucleated cell count and syrinx size (r = 0.51; P = 0.0068). Development of syringomyelia is accompanied by alterations in cerebrospinal fluid composition. The pathogenesis of syringomyelia has not been fully elucidated; therefore, the exact origin of cerebrospinal fluid changes remains unclear.
Introduction
Chiari-like malformation (CM) is estimated to be almost omnipresent in the Cavalier King Charles Spaniel (CKCS) dog population. 3 The disease is recognized on magnetic resonance (MR) imaging as relatively reduced caudal fossa volume, with subsequent foramen magnum herniation of the cerebellum, 11,18,23 and has been associated with the development of fluid-filled cavities within the spinal cord parenchyma (syringomyelia [SM]). 11,17,21 CM/SM is a debilitating and painful disease complex likely to be multifactorial in origin, secondary to altered cerebrospinal fluid (CSF) dynamics and damage to the spinal cord parenchyma; clinical signs frequently exhibited by affected dogs include phantom scratching and vocalization, cervical scoliosis, thoracic and pelvic limb ataxia, and thoracic limb paresis. 21-23
Despite an abundance of literature having been published describing the clinical and imaging findings in dogs with CM and CM/SM and, additionally, the use of CSF analysis being a routine diagnostic procedure in dogs presenting with neurologic signs, there remains a paucity of information on CSF composition in CKCS with CM or CM/SM. It is currently unclear whether the development of SM is accompanied by CSF changes. The CSF composition from CKCS with CM/SM has been described in single case reports, and findings included normal CSF findings, a mixed pleocytosis with a mild elevation in protein concentration, and mononuclear pleocytoses with protein concentrations within reference range, according to reference intervals set by the laboratory. 7,23 In human medicine, it is generally accepted that an increase in CSF protein concentration can occur secondary to syringomyelia. 15 However, to the authors' knowledge, there has been no published study documenting that.
Syringes are thought to arise following damage to the spinal cord parenchyma, as a consequence of altered CSF dynamics, 5,17,21 and therefore, it can be hypothesized that the frequency of CSF changes is higher in CKCS with CM/SM than it is in dogs with CM alone. The current study was a retrospective study into the composition of CSF in CKCS with CM and CM/SM. The aim of the study was to determine whether dogs with CM/SM have CSF changes associated with syrinx formation, by documenting the composition of CSF in CKCS with CM and CM/SM and by elucidating the differences between the 2 groups. The data have implications for the clinical management of CKCS with CM and CM/SM.
Materials and methods
Electronic patient records a from a referral hospital were retrospectively reviewed. Cavalier King Charles Spaniels that were diagnosed with CM or CM/SM by MR imaging and that had CSF analysis between December 2004 and December 2009 were included. Chiari-like malformation was defined as an impingement of the cerebellum and subsequent indentation by the occipital bone or as cerebellar herniation through the foramen magnum. 18 Syringomyelia was defined as hyperintense lesions on the transverse and/or sagittal T2-weighted images with maximal dorsoventral dimension greater than 2 ml. 11,20 All images were subsequently reviewed using image viewer software, b and syrinx size was measured by a board-certified neurologist. Patient medical records were reviewed; the site and date of CSF collection, the age (in months), gender, and additional historical data were recorded. Cavalier King Charles Spaniels with historical evidence of prior steroid treatment were excluded. Dogs that required subsequent steroid treatment were also excluded because a diagnosis of inflammatory central nervous system (CNS) disease could not be definitively ruled out in these patients. Dogs diagnosed with significant concurrent neurologic disease, such as intervertebral disc disease, were also excluded because neurologic disease can lead to alterations in CSF composition. 28
Clinical pathology records of the CSF from the diagnostic laboratory at the Royal Veterinary College were reviewed, including the following parameters from the samples: total nucleated cell count (TNCC) per microliter, protein concentration (g/l), and the number of red blood cells (RBCs) per microliter. Cerebrospinal fluid samples with >500 RBCs per microliter were considered contaminated blood samples and were excluded because of findings from a recent study, 10 where blood contamination greater than this was associated with altered CSF composition, and because of previous reports 1 that have suggested that there is an increase in 1 nucleated cell per 500 RBCs per microliter.
Cerebrospinal fluid samples had all been routinely analyzed using a standard operating procedure for the laboratory. The TNCC per microliter and the RBC count per microliter were conducted using a modified hemocytometer c within 30 min of cisternal or lumbar sampling (TNCC < 5 cells per microliter were considered to be within reference intervals for the laboratory). Differential cell counts and morphologic evaluation were conducted following centrifugation of the 200-μl aliquots of CSF at 145 × g relative centrifugal force for 5 min with a cytocentrifuge d and subsequent modified Wright staining. The protein concentration of samples was also measured from the supernatant, using pyrogallol red protein assay e,f (protein concentrations of ≤0.25 g/l for cisternal samples and ≤0.45 g/l for lumbar samples were considered to be within reference intervals for the laboratory). A clinical pathologist reviewed the differential count of a cohort of samples that met the inclusion criteria to determine whether there were any significant interobserver differences. Samples with differential counts from a minimum of 100 cells from cisternal or lumbar samples were included, and the following percentages were recorded from clinical pathologist reports: neutrophils, small mononuclear cells, large mononuclear cells, and eosinophils.
The presence of activated macrophages, reactive lymphocytes, and myelin-like material were recorded if described in the clinical pathologist report. Activated macrophages were recorded as present if the cytology report described macrophages, phagocytic large mononuclear cells, or cells with a foamy cytoplasm, and reactive lymphocytes were recorded when reactive, activated lymphocytes or plasma cells were reported. Myelin is recognized in CSF as an amorphous, pink material; myelin was recorded as present when myelin-like material was described in the cytology report. Those samples in which eosinophilic material was reported that was suggestive of myelin or erythrocyte ghosts were reviewed by a clinical pathologist. Those that were unclear were excluded because there was insufficient sample volume to do further stains with Luxol fast blue, which specifically identifies myelin, to confirm or refute its presence.
Statistical analysis was performed with a commercially available software program. g All statistical analysis was conducted using nonparametric tests; median values were calculated for variables, and statistical significance was calculated using the Mann–Whitney U-test. The data are presented as medians with ranges. The Fisher exact test was used to compare the presence of activated macrophages, reactive lymphocytes, and myelin between CM and CM/SM and also to compare cisternal and lumbar samples from patients with SM. The association between syrinx size and TNCC per microliter, protein concentration (g/l), neutrophils (%), small mononuclear cells (%), large mononuclear cells (%), and eosinophils (%) was evaluated using Spearman rank correlation coefficient. A P value of ≤0.05 was considered significant for all statistical tests.
Results
Ninety-two CKCS had MR imaging and CSF analysis between December 2004 and December 2009. A total of 31 cases were excluded: 22 because of blood contamination (>500 RBCs per microliter); 4 because of a diagnosis of meningoencephalomyelitis of unknown etiology; 2 because of previous treatment with steroids; 2 because of the small CSF sample size, which precluded analysis; and 1 because of a possible history of trauma. Of the remaining 61 dogs, 27 were diagnosed with CM alone (44%), and of these, 24 were cisternal samples, and 3 were lumbar samples. Thirty-four dogs were diagnosed with CM/SM (56%), and of these, 25 were cisternal samples, and 9 were lumbar samples. The median age of CKCS with CM was 43 months (11–124 months), and of those, there were 6 intact males, 9 neutered males, 6 intact females, and 6 neutered females. The median age of CKCS with CM/SM was 40 months (11–132 months), and of those, there were 7 intact males, 11 neutered males, 4 intact females, and 12 neutered females. There was no significant difference between the age of dogs with CM and CM/SM.
Cisternal CSF analysis showed abnormal results in 9 out of 24 samples (38%) in the CM group and in 17 out of 25 samples (68%) in the CM/SM group. There was a change in CSF composition due to an increase in TNCC (CKCS with CM: 2/24 [8%]; CKCS with CM/SM: 10/25 [40%]) or protein content (CKCS with CM: 7/24 [29%]; CKCS with CM/SM: 14/25 [56%]). There was an elevation in either TNCC or protein concentration in samples from CKCS with CM; however, 7 out of 25 of the samples (28%) from dogs with CM/SM had an increase in both of these parameters. Cerebrospinal fluid analyses of lumbar samples from CKCS with CM were within reference intervals for the laboratory; however, only 3 samples were included in the study. Cerebrospinal fluid analyses of lumbar samples from CKCS with CM/SM had abnormal findings in 2 out of 9 of the cases (22%), and the change in CSF in both samples was attributable to an increase in TNCC.
A differential cell count was performed on 6 out of 27 of the cisternal and lumbar CSF samples (22%) from CKCS with CM and on 21 out of 34 with CM/SM (62%). There was no difference in the proportion of small mononuclear or large mononuclear cells from cisternal samples between dogs with CM and dogs with CM/SM (Table 1). Dogs with CM/SM had a higher neutrophil percentage than did the CM group (P = 0.0203; Fig. 3). In contrast, very few samples contained eosinophils, and the proportion present in cisternal samples did not differ between the 2 groups (Table 1). Furthermore, the proportion of cisternal samples in which activated macrophages (CKCS with CM: 11/24 [46%] vs. CKCS with CM/SM: 10/25 [40%]), reactive lymphocytes (CKCS with CM: 1/24 [4%] vs. CKCS with CM/SM: 1/25 [4%]), and myelin (CKCS with CM: 2/22 [9%] vs. CKCS with CM/SM: 3/25 [12%]) were present did not differ between the 2 groups.
Differential count of cisternal samples from Cavalier King Charles Spaniel dogs with Chiari-like malformation (CM) and those with Chiari-like malformation/syringomyelia (CM/SM).*
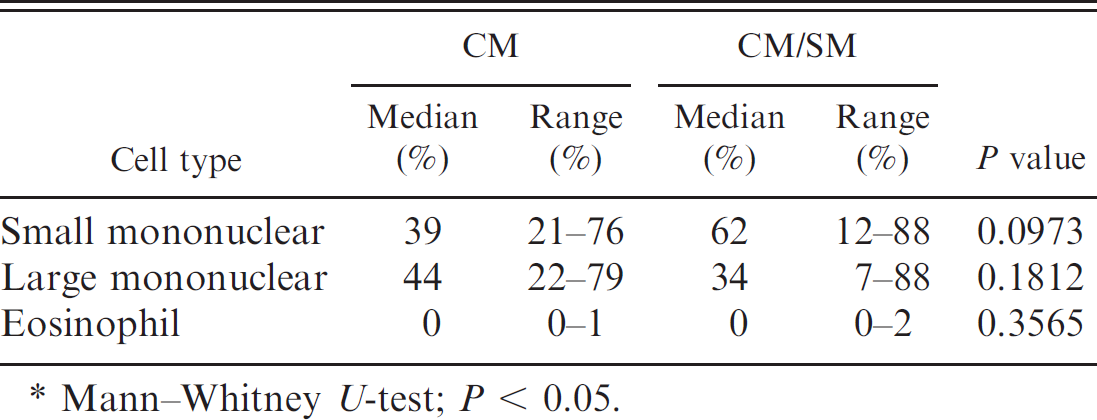
Mann–Whitney U-test; P < 0.05.
The comparison of cisternal and lumbar samples of the CM/SM group revealed that myelin was detected more frequently in lumbar (4/7 [57%]) than in cisternal (3/25 [12%]; P = 0.0258) samples. There was no difference between cisternal and lumbar CSF analysis in the occurrence of activated macrophages (cisternal: 10/25 [40%] vs. lumbar: 4/9 [44%]) or reactive lymphocytes (cisternal: 1/25 [4%] vs. lumbar: 1/9 [11%]). Additionally, there was no difference in the median differential count between the cisternal and lumbar CSF analysis: small mononuclear cells (median: 62%; range: 12–88% vs. 55%; 24–69%), large mononuclear cells (median: 34%; range: 7–88% vs. 32%; 5–48%), and neutrophils (median: 0%; range: 0–37% vs. 0%; 0–67%).
Interestingly, CKCS with CM/SM had a higher TNCC (median: 4/μl; range: 0–15/μl) than did the CKCS with CM (median: 2/μl; range: 0–8/μl; Fig. 1).
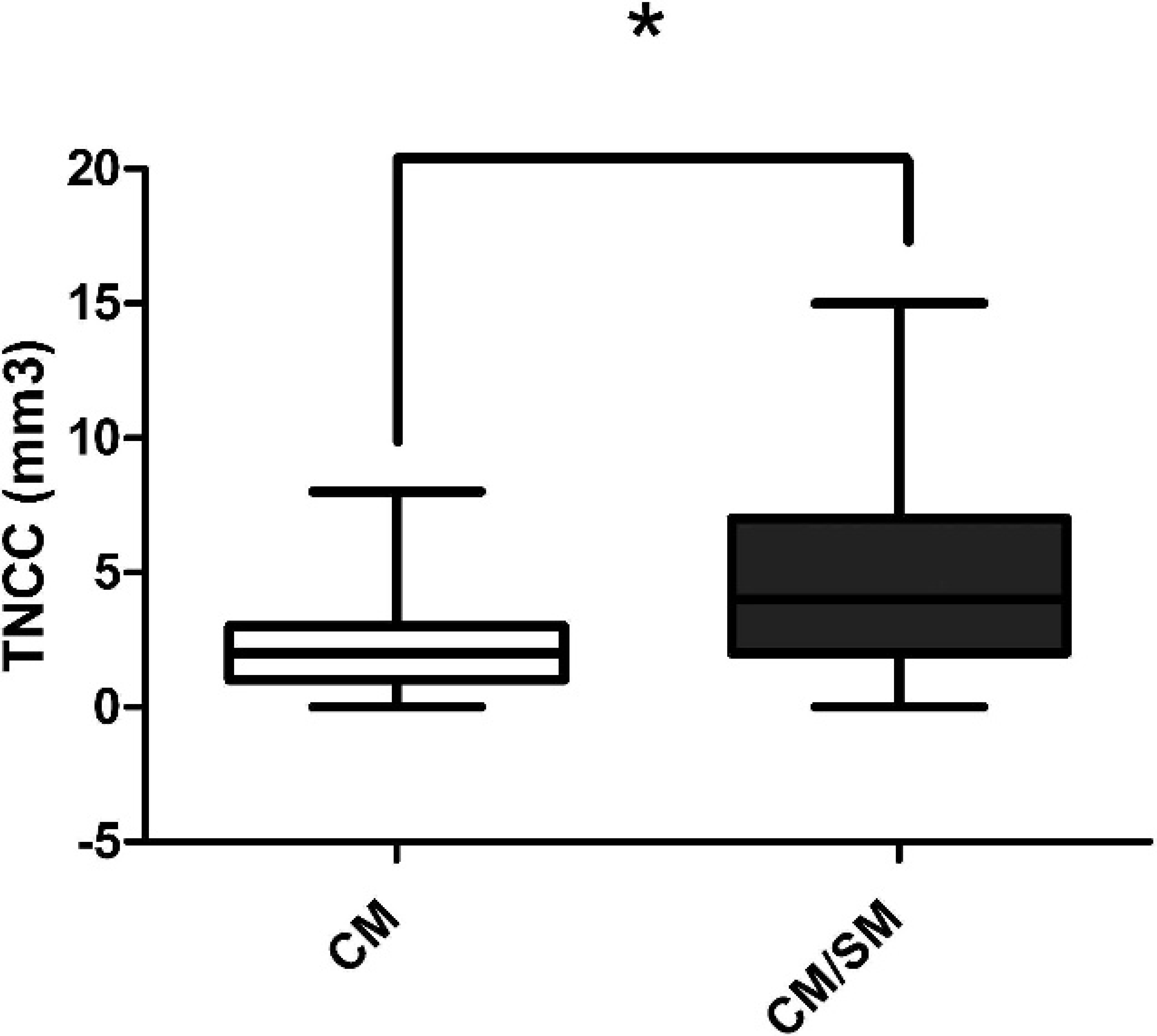
Box-and-whisker plot of the distribution of total nucleated cell count (TNCC) from cisternal cerebrospinal fluid samples of Cavalier King Charles Spaniel dogs with Chiari-like malformation (CM) and those with Chiari-like malformation/syringomyelia (CM/SM). Dogs with CM/SM had a higher TNCC than did those with CM alone (P = 0.0047). Solid horizontal line represents the median. Mann–Whitney U-test, * P < 0.05.
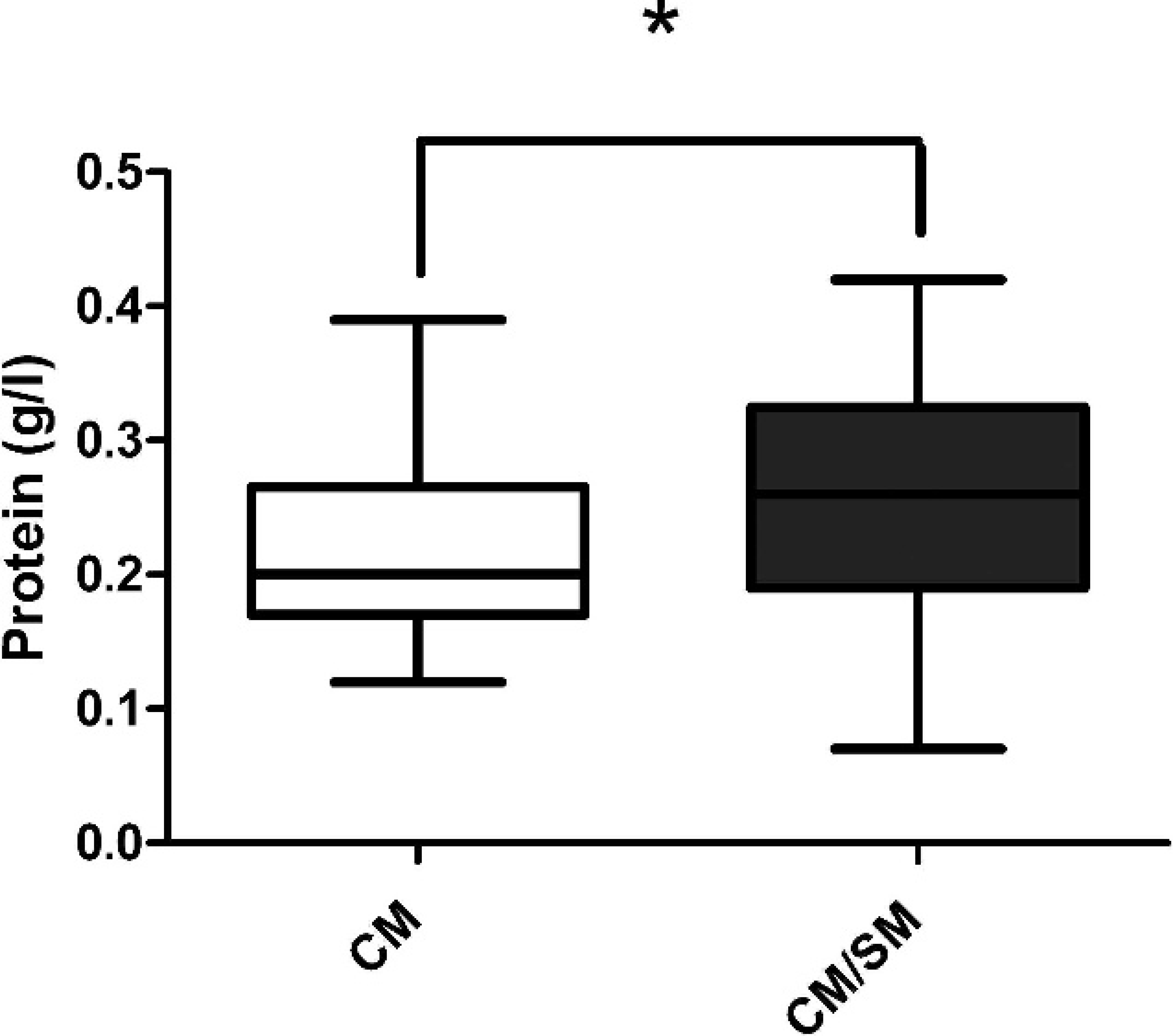
Box-and-whisker plot illustrating the distribution of protein concentration (g/l) from cisternal cerebrospinal fluid samples of Cavalier King Charles Spaniel dogs with Chiari-like malformation (CM) and those with Chiari-like malformation/syringomyelia (CM/SM). Dogs with CM/SM had a higher protein concentration than did dogs with CM alone (P = 0.039). Solid horizontal line represents the median. Mann–Whitney U-test, * P < 0.05.
Furthermore, the protein concentration in the CM/SM group (median: 0.26 g/l; range: 0.07–0.42 g/l) was higher than was the concentration in the CM group (median: 0.2 g/l; range: 0.12–0.39 g/l; Fig. 2). Further analysis of the CKCS with CM/SM revealed a positive correlation between syrinx size and TNCC (r = 0.51; P = 0.0068).
Discussion
To the authors' knowledge, there have been no previous studies in canine or human medicine that have described the CSF composition in patients with CM and CM/SM. Cavalier King Charles Spaniels with CM/SM had a higher TNCC, protein concentration, and neutrophil percentage than did those CKCS with CM alone, although considerable overlap existed, and these were not discriminatory between groups. There was also a positive association between syrinx size and TNCC in CKCS with CM/SM.
Differences in CSF cellular and protein composition between the 2 groups may arise as a consequence of syrinx formation or be due to as yet unidentified factors that predispose dogs toward the development of CM/SM and that are absent in dogs with CM alone. Elevated protein concentration in cases of SM has been reported in human medicine textbooks, 15 but there remains a lack of data on CSF composition both in CKCS with CM/SM and in human beings with the Chiari malformation and associated SM. In the current study, elevated protein concentration was the most prevalent abnormal finding in the CSF of CKCS because high concentrations were detected in 29% of CKCS with CM and 56% with CM/SM.
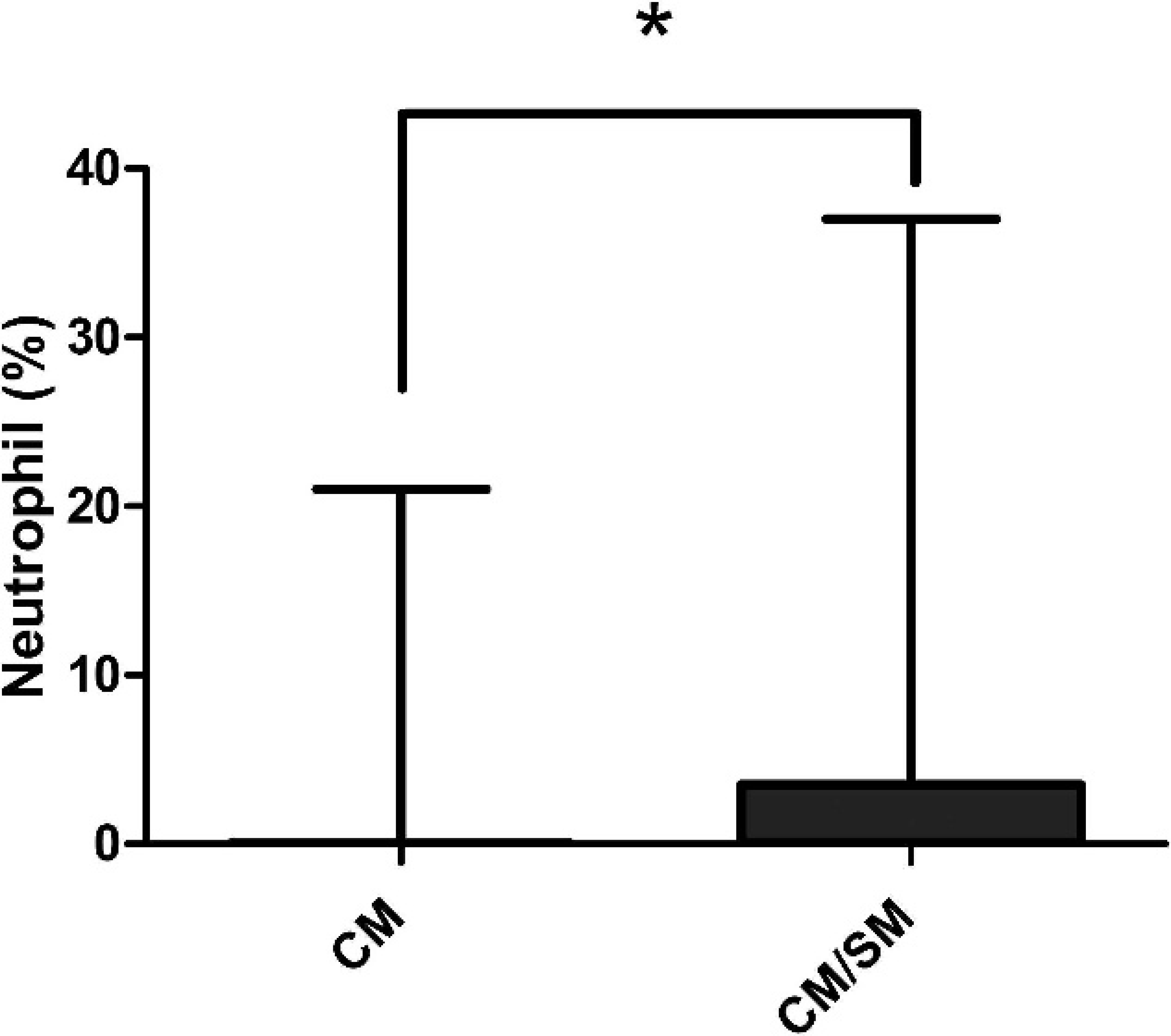
Box-and-whisker plot of the neutrophil cell percentages from cisternal cerebrospinal fluid samples of Cavalier King Charles Spaniel dogs with Chiari-like malformation (CM) and those with Chiari-like malformation/syringomyelia (CM/SM). Dogs with CM/SM had a higher neutrophil percentage than did those in the CM group (P = 0.0203). Solid horizontal line represents the median. Mann–Whitney U-test, * P < 0.05.
Healthy CSF contains very low protein concentrations, 9 and elevations can be seen with a large variety of neurologic disease. 6 Reported mechanisms of protein elevation include disruption and increased permeability of the blood–brain or spinal cord barrier; globulin synthesis, most commonly a consequence of inflammatory disease processes; local necrosis; and alterations in CSF flow and absorption. 6 Recent theories 12,17 provide a potential explanation for elevated protein concentrations in SM. Before the intramedullary and vascular theory, 12,17 syringes were thought to contain CSF; however, it is now accepted that they are a consequence of an accumulation of extracellular fluid, secondary to repeated mechanical stress and high pressure in the microcirculation of the spinal cord. Therefore, the most likely mechanism of increased protein concentration seen in the CSF of the dogs in the present study is disruption of the blood–spinal cord barrier.
The protein concentration in the CSF of dogs with CM and of dogs with CM/SM, although higher than that of healthy CSF, is still much lower than that in extracellular fluid. The vascular theory 17 provides an explanation for this because it hypothesizes that disruption of the blood–spinal cord barrier leads to ultrafiltration of the plasma, whereby there is passage of crystalloids in the absence of macromolecules. As stated previously, an increase in protein concentration was also present in dogs with CM in the absence of SM. It is possible that these dogs may develop SM in the future but did not have detectable syringes at the time of their MR imaging. This is highly likely, given that SM is a developmental process and has a higher incidence in older dogs. 4
Despite there being a number of proposed theories, the exact mechanism of SM formation and progression currently remains unclear. 17,21 A recent study 5 in CKCS outlined one difference between the 2 groups in providing evidence of an association between altered CSF flow dynamics, involving turbulence and velocity and the presence of SM. One possible explanation of the increased TNCC and neutrophilia in dogs with SM is that alterations in CSF flow dynamics in the syrinx may cause further microtrauma in the spinal cord parenchyma, in addition to the trauma caused by processes described in the aforementioned vascular and intramedullary theories. Altered CSF flow dynamics may incite an inflammatory response that leads to changes in CSF composition. This also provides an explanation for the findings in the present study of increasing TNCC with increased syrinx size because there was an association between turbulent flow and the severity of SM. 5
Another explanation for the neutrophilia might be a local arachnoiditis. Arachnoiditis is a chronic inflammatory process that can occur as a consequence of a variety of processes, including trauma to the spinal cord, and is frequently associated with the development of SM. 16 Although not previously reported, recurrent trauma, secondary to the caudal displacement of cerebellum that occurs in CM, 19 may lead to arachnoiditis. The MR images of the CKCS in the current study did not have any findings compatible with inflammatory CNS disease at this site. Further imaging sequences in these cases would, however, be necessary because arachnoiditis may not be detected on standard MR imaging sequences. 17 This may provide a further explanation for the increased TNCC and higher proportions of neutrophils detected in dogs with CM/SM in the present study.
Finally, a low number of nondegenerate neutrophils have been reported in CSF of healthy dogs, which has been hypothesized to be a consequence of blood contamination. 6 Red blood cells are not present in healthy CSF; however, iatrogenic blood contamination is commonly encountered following analysis and is reported to confound interpretation. 6,8 A recent study 10 provided evidence that samples with a low TNCC and blood contamination (>500 RBCs per microliter) had a significantly higher protein concentration and neutrophil percentage compared with noncontaminated samples. Formulas used to correct the TNCC and protein concentration in samples with blood contamination are reported to be unreliable, and there are differing opinions as to the level of blood contamination that leads to alterations in CSF composition. 14,24,25,27 An increase in 1 nucleated cell per 500 RBCs per microliter has been previously reported; therefore, samples with >500 RBCs per microliter were excluded 1 in the present study. Thus, the findings in the current study are unlikely to be attributable to blood contamination. Mild elevations of neutrophil percentage have been also associated with a wide range of CNS diseases, such as degenerate disc disease and neoplasia. 6,28 Because CKCS with significant concurrent neurologic disease were excluded, the elevations in neutrophils occurred in the absence of other CNS pathology and may, therefore, be associated inflammatory process secondary to trauma as described.
Myelin-like material was detected in a greater proportion of lumbar than cisternal samples in CKCS with CM/SM, although its presence did not differ between CKCS with CM/SM and those with CM alone. Previously, detection of myelin in CSF samples was commonly associated with a disease process in which demyelination was a feature. 2,13,26 However, a recent study 29 provided evidence that myelin is also detected in common neurologic diseases, in which demyelination is not a feature, such as intervertebral disc disease, and provided further evidence of an association between the presence of myelin and the collection site. The findings in the present study are supportive of this hypothesis. However, it must be noted that a limitation of the study was that there was insufficient sample to perform specific stains to confirm or refute the presence of myelin and, therefore, it could not be verified in any case.
In conclusion, abnormal CSF findings are more prevalent in CKCS with CM/SM than in those with CM alone. In general, CSF changes in CNS diseases are nonspecific and have been associated with a vast variety of pathologic processes. Because the pathogenesis of syringomyelia has not been fully elucidated, the origin of CSF changes remains unclear; therefore, further studies are necessary to determine the underlying pathophysiology. Further analysis of lumbar samples would be beneficial in future studies because many clinicians prefer the lumbar sampling site in CKCS because of the potential risk of iatrogenic trauma of the cerebellum and the small sample size, which precluded statistical comparisons between the 2 sites in dogs with CM.
Acknowledgements
The authors are grateful to the Clinical Investigation Centre, Royal Veterinary College, University of London, for their role in collection of samples.
Footnotes
a.
Rx Works 3.8.1, Rx Works Inc., Newbury, Berkshire, United Kingdom.
b.
Image Viewer 3.7.03, Visbion, Chertsey, Surrey, United Kingdom.
c.
Fuchs–Rosenthal haemocytometer, Hawksley, Lancing, United Kingdom.
d.
Shandon Cytospin, Thermo Scientific Labmedics Ltd., Manchester, United Kingdom.
e.
UP1570/1571 Randox Laboratories Ltd., Crumlin, United Kingdom.
f.
Ilab 600, Instrumentation Laboratory Ltd., Warrington, United Kingdom.
g.
GraphPad Prism 5 version 5.0, GraphPad Software Inc., La Jolla, CA.
