Abstract
Odontogenic myxoma (OM) was diagnosed in an 8-year-old Labrador Retriever dog with an ulcerohemorrhagic mass located on the caudal area of the right maxillary gingiva. The neoplasm was characterized by a low mitotic index and moderate numbers of spindle, stellate, and round cells that were sparsely distributed in an alcian blue reactive myxomatous matrix. Individual neoplastic cells were characterized by small amounts of faintly eosinophilic staining cytoplasm, prominent nucleoli, and stippled amphophilic staining chromatin that was immunoreactive for vimentin but negative for cytokeratin and actin. To the authors' knowledge, this is the first reported case of canine OM from North America, and it shares histomorphologic and histochemical features with 3 other cases reported in dogs elsewhere. Whereas, a literature review suggests untreated canine OM is insidious and locally aggressive, the prognosis in the present dog remains unknown. These findings support previous recommendations for inclusion of canine OM on the World Health Organization list of odontogenic tumors.
Odontogenic tumors (OT) are neoplasms of the maxillary and mandibular bone system that may invade the surrounding periodontal soft tissues. These tumors originate from epithelial and mesenchymal components of the dental papilla 6,13 ,16 and manifest as a progressive but slow-growing enlargement of the gingiva and surrounding tissues. Although OTs are generally rare in domestic animals, 1 the neoplasms occasionally occur in small animals 6,14 and human beings. 3 In human beings, most OTs are benign but can also be malignant. 3 Odontogenic tumor phenotypes such as acanthomatous and fibromatous epulides do not usually present any diagnostic challenge, 14 but some variants may be difficult to diagnose either because of poor differentiation or because of a lack of published literature on their histomorphologic characteristics. Accurate diagnosis of these tumors, therefore, is of paramount importance because these tumors frequently show aggressive behavior, despite a deceptively slow growth pattern. 5,10
Odontogenic myxoma (OM) is a benign but locally invasive OT variant of mesenchymal origin that arises from the dental papilla, dental follicle, and periodontal ligament. 13,15 Also called odontogenic fibromyxoma, OM has been reported at a very low frequency in human beings 4,8,9,11 and is extremely rare in domestic animals. 1 In human beings, OM has been diagnosed in middle-aged individuals 3,7 but is also reported in young children. 12 Human medical data indicate that OM may run a locally destructive course3 and that it appears to have a predilection for the mandible. 7,8,12 Of the few cases documented in domestic animals, 1 report was in a 1.5-year-old mixed breed filly, 1 another in a 12-year-old female Doberman Pincher, 5 and the most recent cases were in a 14-year-old male Dachshund 2 and a 3-year-old Jack Russell terrier. 10 The current report compares the gross, microscopic, and prognostic attributes seen in the present tumor with those described in the other 3 canine OM cases reported elsewhere (Table 1). 2,5,10
A formalin-fixed biopsy from an 8-year-old Labrador Retriever was received for histologic evaluation at the North Dakota State University, Veterinary Diagnostic Laboratory (Fargo, North Dakota). The case history indicated that the biopsy was taken from an ulcerohemorrhagic mass located on the caudal area of the right maxillary gingiva. The formalin-fixed biopsy was routinely processed and paraffin embedded. Five-micron sections were deparaffinized and stained with hematoxylin and eosin (HE). Other sections were histochemically stained with alcian blue (pH 2.5) for the presence of glycosaminoglycans, and immunohistochemically tested by the horseradish peroxidase a system for the tumor markers cytokeratin, vimentin, and actin. For immunohistochemistry (IHC), archived positive control tissue samples routinely applied for tumor IHC at the North Dakota State University, Veterinary Diagnostic Laboratory were used in the characterization of the present tumor. These archived samples included the following blocks of paraffin-embedded samples: 1) vaginal tissue from a 13-year-old female Shih Tzu with a vaginal fibroma (vimentin), 2) cutaneous biopsy from an 11-year-old female spayed mixed-breed dog with a perianal adenoma along with an intact epidermis (cytokeratin), and 3) a cutaneous biopsy with subdermal skeletal muscle fibers from a dog of unspecified breed and age (actin).
Gross, microscopic, and prognostic characteristics of canine odontogenic myxoma (OM) in a Labrador Retriever compared with previous reports of the same neoplasm in 3 other dogs.
For IHC, embedded tissue blocks were deparaffinized and hydrated in distilled water, after which they were rinsed with TBS (1× Tris buffer saline solution, with Tween 20, pH 7.6). a The dual endogenous enzyme block a was applied for 5 min followed by a 5 min wash in TBS. Except for the vimentin IHC, all the other sections were then permeabilized in proteinase K a for 5 min followed by a quick rinsing step in TBS. To localize all the target tumor markers, test and control sections were individually incubated with normal mouse serum a for 15 min followed by a rinsing step in TBS. The following “ready-to-use” monoclonal antibodies (mAb) were used as primary antibody: 1) mouse anti-human cytokeratin mAb, clone MNF116 a for localization of cytokeratin, 2) mouse anti-human muscle mAb (clone HHF35) a for localization of actin, and 3) mouse anti-swine vimentin mAb (clone V9) a for localization of vimentin. For negative control sections, the primary antibody was substituted with phosphate buffer saline solution. After incubation with the primary antibody, the slides were washed in TBS, after which they were incubated with the horseradish peroxidase–labeled anti-mouse mAb a for 18 min. This was followed by a rinsing step in TBS, after which the slides were treated twice with romulin red b for 5 min, and rinsed in deionized water followed by TBS. The slides were then stained with hematoxylin a for 2 min, followed by a rinsing step in TBS, air-dried, and dehydrated in ethanol and cleared in xylene. The sections were secured with a coverslip, and the slides were examined by light microscopy. To demonstrate sulfated glycosaminoglycans, replicate sections were stained with alcian blue (pH 2.5). Briefly, tissue blocks were deparaffinized and hydrated in distilled water after which the slides were placed in 3% acetic acid solution for 3 min and 1% alcian blue solution was then applied for 30 min. After this step, the slides were washed in running tap water for 10 min, rinsed in distilled water, and counterstained in nuclear fast red solution for 5 min. After washing the slides in distilled water for another 1 min, they were dehydrated in 2 changes each of 95% alcohol and absolute alcohol, cleared in xylene, and then mounted on slides with a synthetic resin. Publications of the 3 canine OM cases reported in Spain, India, and South Africa were reviewed, and the findings were tabulated against those of the present case for comparison.
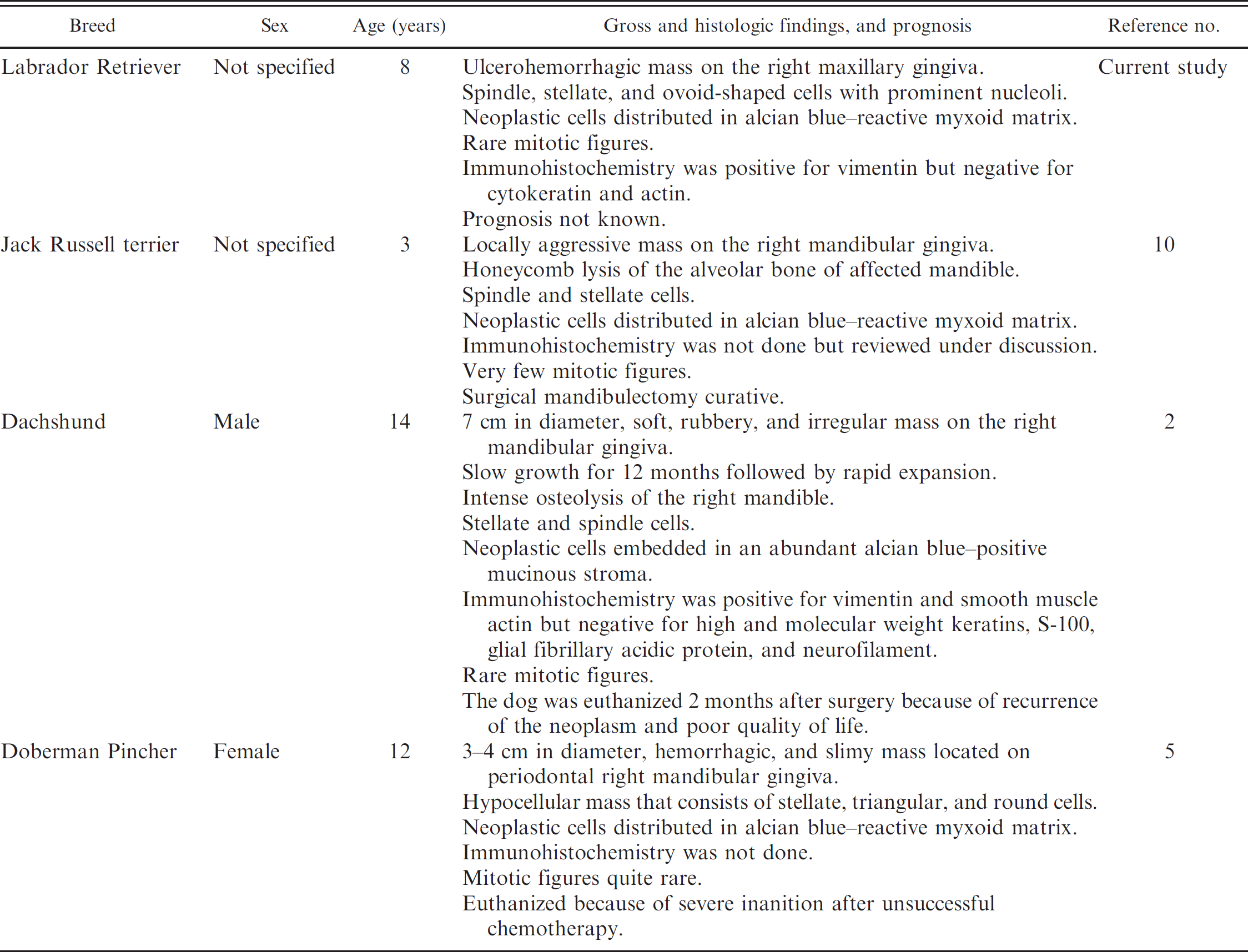
Gingival mass from the right maxilla of an 8-year-old Labrador Retriever dog. The neoplasm was characterized by low to moderate numbers of spindle and stellate–shaped cells embedded in a loose myxomatous matrix. Note the prominent nucleoli and stippled amphophilic staining to vesicular nuclei. Hematoxylin and eosin. Bar = 2000 μm.
Except for the maxillary location in the present case, OM was present on the mandibular gingiva in all the other canine cases reported in the veterinary literature. Multiple HE-stained sections were characterized by intense mixed cell inflammation, moderate amounts of subepithelial granulation tissue, focally extensive hemorrhages, and islands of necrotic tissue debris in some areas. The viable neoplastic tissue was composed of numerous spindle, stellate, and round-to-plump–shaped cells that were sparsely distributed in a myxomatous matrix (Fig. 1) that was strongly reactive with alcian blue stain (data not shown). There was a very low mitotic rate, and individual neoplastic cells were characterized by small amounts of faintly eosinophilic staining cytoplasm, prominent nucleoli, and stippled amphophilic staining chromatin. In addition, neoplastic cells were immunoreactive for vimentin (Fig. 2) but negative for actin (Fig. 3) and cytokeratin (Fig. 4). A large area of the overlying gingival stratified squamous epithelium was partially ulcerated and transmigrated by numerous degenerative neutrophils and contained low numbers of lymphocytes, plasma cells, and macrophages. Based on the above findings, a morphologic diagnosis of gingival OM was made.
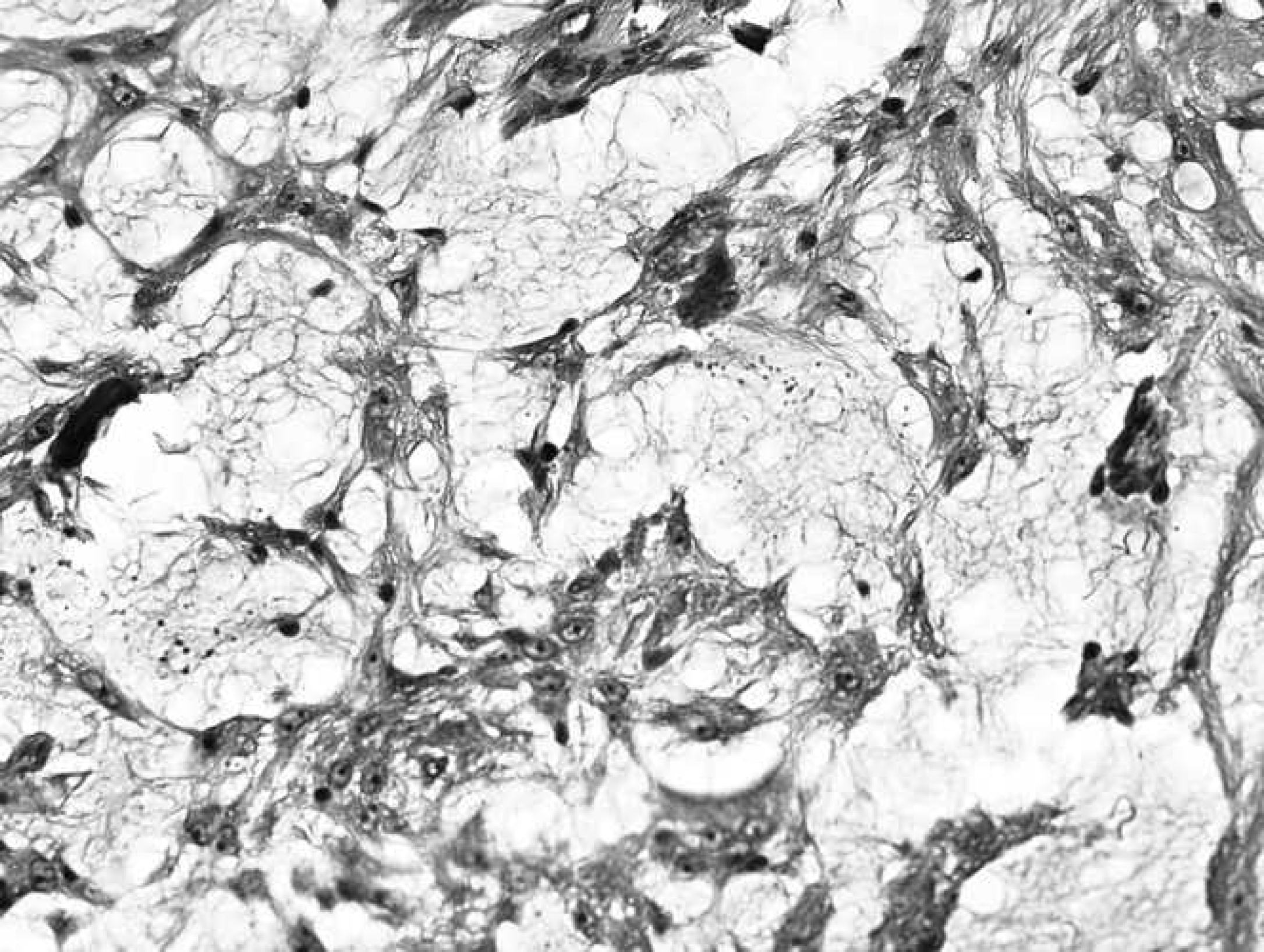
Gingival mass from the right maxilla of an 8-year-old Labrador Retriever dog. Neoplastic cells were characterized by strong cytoplasmic staining for vimentin. Immunohistochemistry. Bar = 1000 μm.
To the authors' knowledge, this is the fourth case of canine OM to be reported in peer-reviewed literature to date. 2,5,10 Similar to what has been observed in human beings, 7,8,12 data obtained from a comparison of the present case with 3 other canine OM cases, 3 of 4 (75%) were located on the mandible. Evaluation of the present case revealed histomorphologic similarities between the present case and the other canine OM cases (Table 1). Immunohistologic evaluation showed that this mass was positive for vimentin but negative for cytokeratin and actin. Despite the lack of a statistically significant sample size because of the very low numbers of canine OM cases, evaluation of this and the other cases in the literature suggest that untreated OM cases may be chronically progressive and locally destructive (Table 1). Given the fact that surgical veterinary pathology is a vibrant industry in Canada and the United States, detection of a canine OM case in North America at this time underscores the rarity of this tumor in dogs. The present findings show that histomorphologic features of the present OM case are remarkably similar with those reported in human beings 3,4,7 and in other canine OM cases reported in Spain, 2 India, 5 and South Africa. 10 It is important to note that the most recent North American report of OM among domestic animals was published 11 years ago and involved a 3.5-year-old filly tested at the University of Florida (Gainesville, Florida). 1 Immunohistochemical evaluation of the present case showed that the mass was positive for vimentin but negative for cytokeratin and actin. Vimentin immunoreactivity is consistent with a mesenchymal ontogeny as previously reported in human beings. 13,15 Although a review of the literature suggests that untreated OM is rather slow in onset and locally destructive (Table 1), the prognosis in the present dog remains unknown.
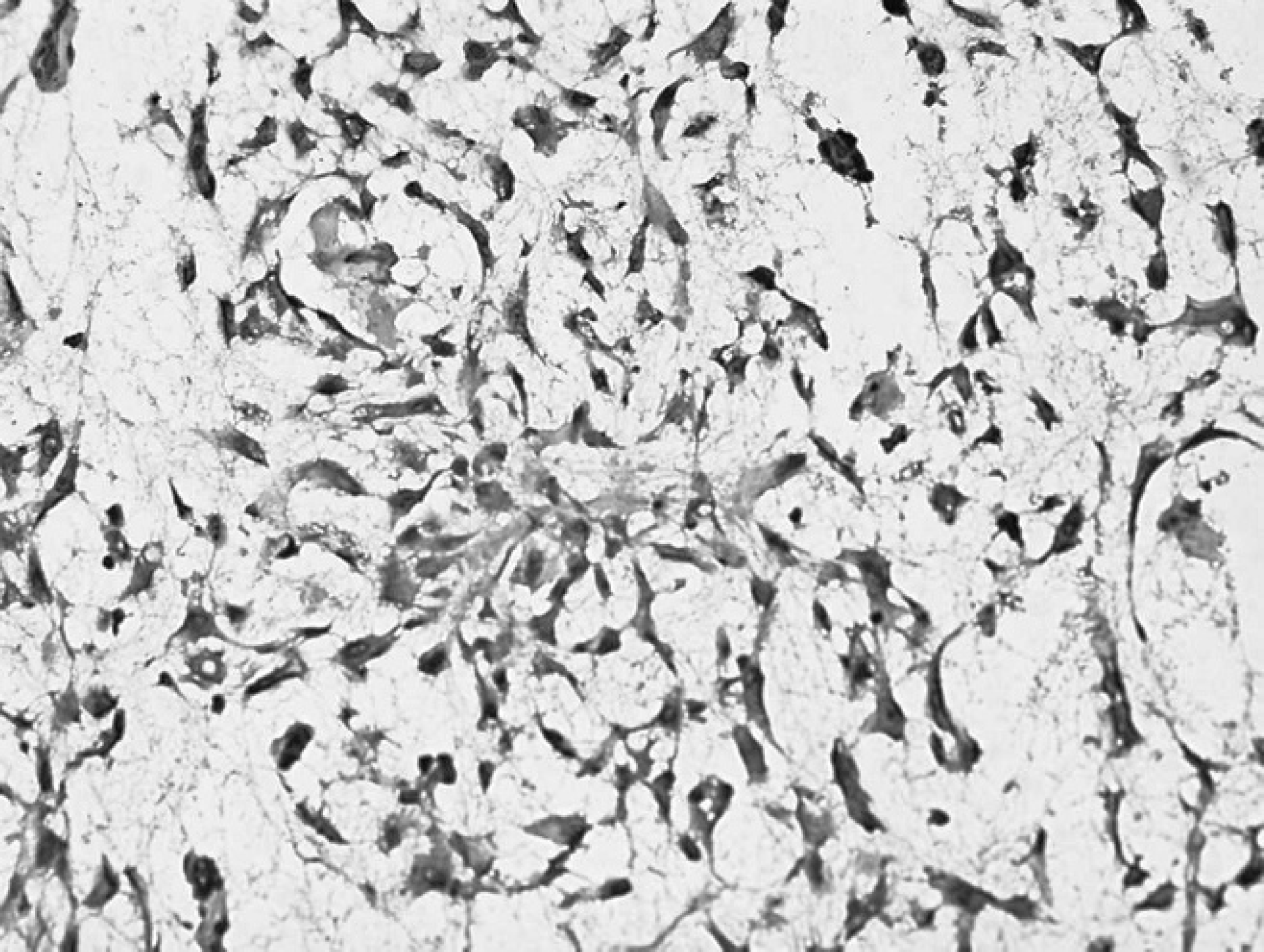
Gingival mass from the right maxilla of an 8-year-old Labrador Retriever dog. Neoplastic cells stained remarkably negative for actin. Note the spindle and stellate–shaped neoplastic cells with prominent nucleoli. Immunohistochemistry. Bar = 2000 μm.
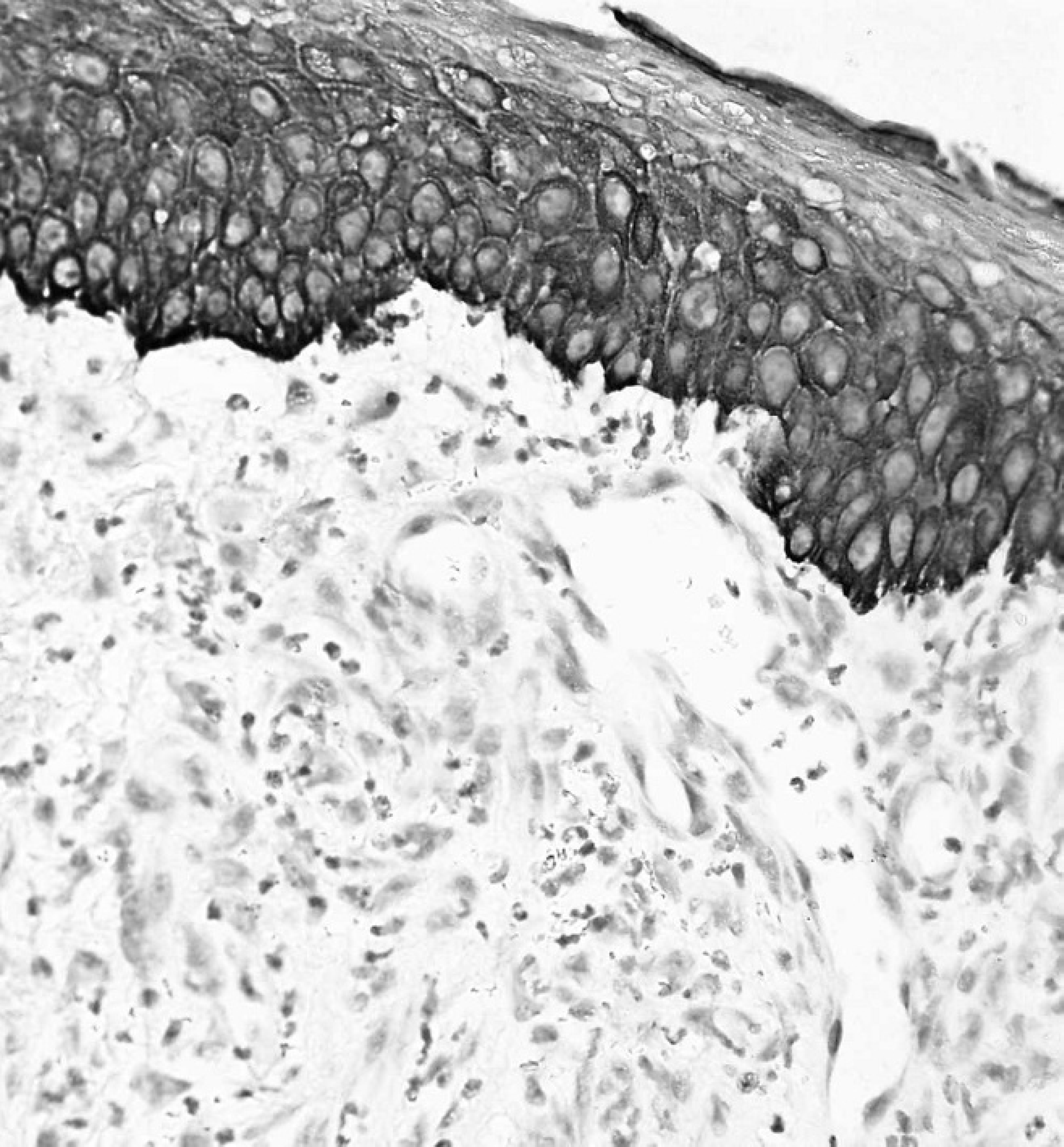
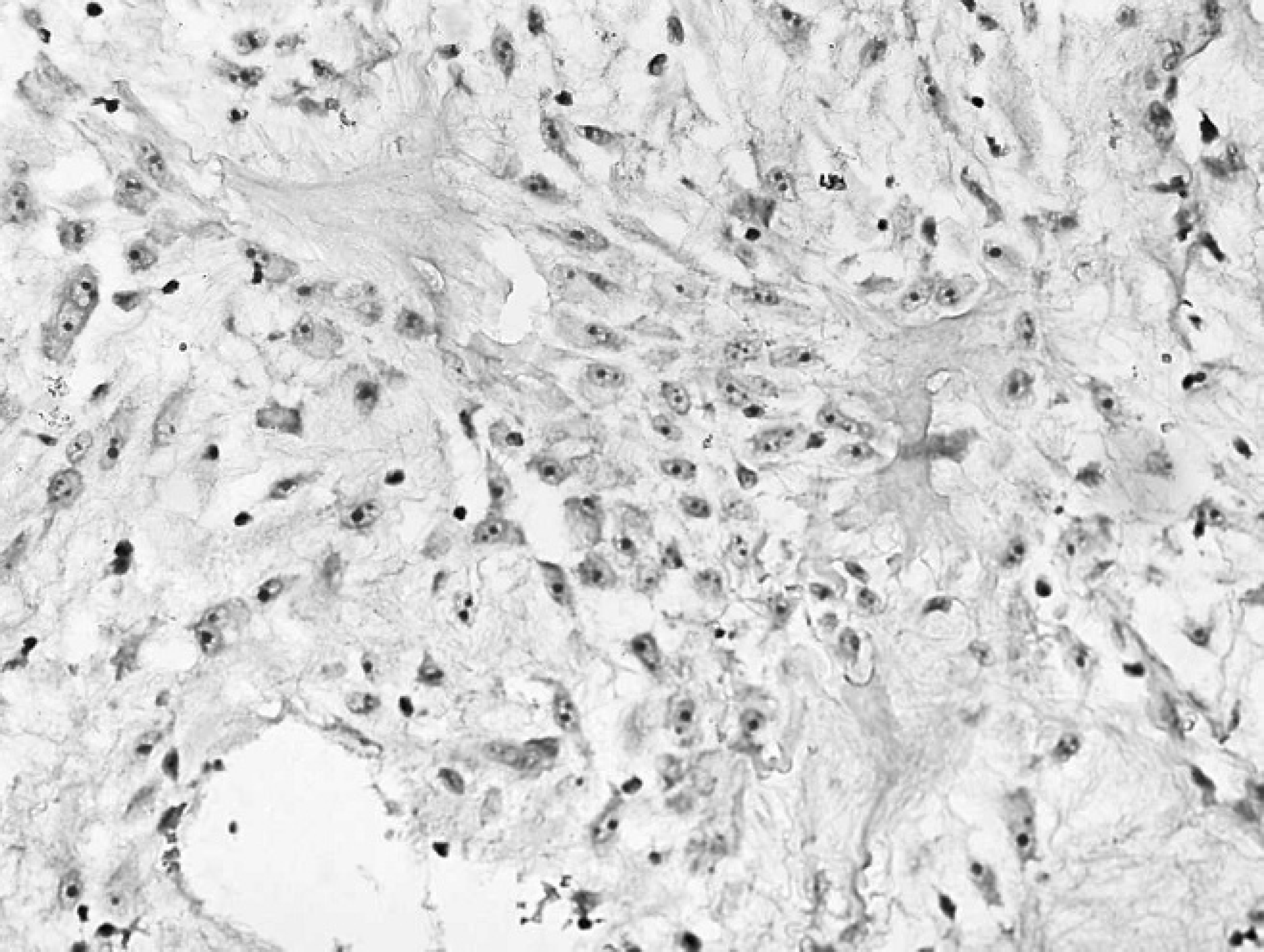
Gingival mass from the right maxilla of an 8-year-old Labrador Retriever dog. Neoplastic cells stained remarkably negative for cytokeratin. Note the intensely cytokeratin reactive stratified squamous epithelium overlying the area of neoplasia. A low-grade inflammatory response is clearly discernible. Immunohistochemistry. Bar = 2000 μm.
In conclusion, the current report corroborates evidence of the existence of OM in dogs and further verifies characteristic immunohistomorphologic and histochemical features of the mass that could help diagnosticians in arriving at a definitive diagnosis of the tumor in dogs. Similar to what has been observed in human beings but subject to corroboration by future investigations, canine OM appears to have a predilection for the mandible. The findings strongly suggest that, although canine OM may be rare, the tumor variant should be considered an important differential in dogs with OTs of unusual histomorphologic features that include a myxomatous matrix. In a previous report, 10 it was recommended that the World Health Organization and the Armed Forces Institute of Pathology should include canine OM on the classification list of OTs. Findings presented in this report further support and justify that recommendation.
Footnotes
a.
EnVision™, Dako Wash Buffer 10× (product no. S3006), Peroxidase and Alkaline Phosphatase Blocking Reagent (Dual Endogenous Enzyme-Blocking Reagent, product no. S2003), Proteinase K (product no. S3020), Mouse Serum (product no. N1698), Monoclonal Mouse Anti-Human: Cytokeratin (code no. M0821), Monoclonal Mouse Anti-Human: Actin (Muscle, code no. M0635) Monoclonal Mouse Anti-vimentin (code no. M0725; pretreatment using H.I.E.R. with Ready-to-Use Target Retrieval Solution, product no. S1700), EnVision+ System-HRP Labeled Polymer Anti-mouse (product no. K4001), Hematoxylin (product no. S3301); Dako North America Inc., Carpinteria, CA.
b.
Catalog no. RAEC810L, Biocare Medical, Concord, CA.
