Abstract
Endocarditis lesions from 117 slaughter pigs were examined pathologically and etiologically in addition to 90 control hearts with cardiac valves. Lesions were located on the valves; however, the lesions had extended to the walls in 21 cases (18%). Lesions predominated on the mitral valve (59%). A total of 28 cases, from which no growth was obtained or a contamination flora was grown, were screened by fluorescence in situ hybridization (FISH) for bacteria (general bacterial probe) and probes specific for Streptococcus suis and Erysipelothrix rhusiopathiae, respectively. Using FISH, an additional 10 cases of endocarditis due to S. suis and E. rhusiopathiae were disclosed. Within lesions, streptococci predominated (53%) followed by E. rhusiopathiae (30%). Distinct features of both the lesions and the shape and localization of bacterial colonies were related to streptococci and E. rhusiopathiae. The propensity for streptococci to be localized on more than 1 valve in single hearts may be because S. suis–infected pigs tend to have been infected for a longer period compared with E. rhusiopathiae. Mineralization of endocarditis lesions was significantly associated with infection by streptococci, and was seen in 71% of the cases, whereas it was present in only 28% of lesions caused by E. rhusiopathiae. In addition, areas with mineralization were significantly correlated to the presence of a granulomatous reaction. Granulomatous endocarditis is likely a result of a foreign body reaction due to dystrophic mineralization. Local proliferation of valvular endothelial cells, found in 9 hearts in the current study, may increase the risk of developing thrombosing endocarditis in pigs.
Introduction
Porcine endocarditis is a well-known disease entity, and the lesions are usually well recognized at gross inspection. 18,24 Endocarditis occurs in all age groups of pigs (from just a few weeks old to adults), and in Denmark, where it is obligatory to open the heart for examination during meat inspection, approximately 0.009% of pigs slaughtered annually are diagnosed with endocarditis (Ministry of Food, Agriculture and Fisheries, Danish Veterinary and Food Administration, unpublished data, 2008).
Porcine endocarditis has been the focus of several studies. 6,8,10–14,16 However, the pathology in combination with the cause of endocarditis in slaughter pigs has only been examined sporadically. In the majority of studies focusing on spontaneous porcine endocarditis, the evaluation of lesions has been based solely on gross inspection usually combined with microbiological examinations. 16,18,20 If histopathology has been applied, only a few cases were examined or the age group in focus has been dominated by recently weaned piglets and/or sows, 8,11 or the lesions were selected to contain only a single bacterium, such as Streptococcus suis. 22,25 From such studies, it is well established that S. suis and Erysipelothrix rhusiopathiae are the dominant agents of porcine endocarditis. However, in single cases, other bacteria such as Arcanobacterium pyogenes, Staphylococcus aureus, other staphylococci, streptococci other than S. suis, Escherichia coli, Pasteurella multocida, Listeria monocytogenes, and Actinobacillus equuli have also been isolated from spontaneous cases of porcine endocarditis. 8,16,19,20
In general, information on the relationship between individual bacterial causes and the accompanying pathology of porcine endocarditis is mostly textbook based, in which some general views are presented. 18,24 Two studies concluded that no morphological difference between the endocarditis lesions caused by streptococci and those due to E. rhusiopathiae was found. 8,11 Contrasting the sparse information on the histopathology in spontaneous cases of porcine endocarditis, the initial and progressive inflammatory reactions have been elucidated in a number of porcine endocarditis models. 9,10,13,14
Recently, a hazard analysis for a contingent devolution to visual meat inspection at Danish slaughterhouses was carried out (Alban L, Vilstrup C, Steenberg B, et al.: 2009, Risk assessment for modernisation of meat inspection of Danish finisher pigs. In: Proceedings of the 12th International Symposium on Veterinary Epidemiology and Economics, p. 38. Durban, South Africa). During the decision-making process for that analysis it was decided that the lesions and causes of endocarditis in Danish slaughter pigs should be examined extensively. Therefore, in the present study, the relation between etiology, as determined microbiologically and by fluorescence in situ hybridization (FISH), and the characterization of lesions by gross and histologic examination of spontaneous endocarditis in slaughter pigs is described and analyzed.
Materials and methods
Tissue samples
During a 10-month period (March–December 2008), hearts with gross signs of endocarditis lesions were collected at 11 slaughterhouses from different regions in Denmark. The material was selected as a part of a hazard analysis in the process of a contingent devolution to visual meat inspection at Danish slaughterhouses (Alban L, et al.: 2009, Risk assessment for modernisation of meat inspection of Danish finisher pigs). The affected hearts were obtained during routine meat inspection. Together with the diseased hearts, a control heart (i.e., the heart from the pig after the diseased heart in the slaughter line) was sampled as a control. At arrival, it was ensured that each heart chamber was opened exposing the atrioventricular (AV) valves, the pulmonary valve, and the aortic valve. 11 Lesions were characterized by gross inspection. From the diseased cusps and the corresponding cusps from the control hearts, material was sampled for bacteriologic and histologic examination.
Bacteriology
Lesions on the heart valves were sampled with a sterile loop and subsequently plated on blood agar, prepared from Oxoid blood agar base a supplemented with 5% sterile bovine blood and incubated at 37°C for 2 days. Colonies were subcultured and identified biochemically using standard phenotypical methods. 4 Streptococcal isolates were further analyzed by means of partial 16S ribosomal RNA gene sequencing. Gene sequencing was accomplished using universal eubacterial primers 37F (5′-GGCTCAGRWYGAACGC-3′) and 519R (5′-GTRTTACCGCGGCTGCTG-3′). b The polymerase chain reaction (PCR) products were purified using a PCR extraction kit c and then sequenced. d
Histology
Sampled tissues from all hearts were fixed in 10% neutral buffered formalin, processed through graded alcohols and xylene, and embedded in paraffin wax. Tissue sections were cut at 3–4 μm and stained with hematoxylin and eosin. Selected sections were stained by von Kossa for mineralization with calcium deposition, and Masson trichrome and van Gieson stain for collagen in granulation tissue. 3 For immunohistochemistry and FISH, sections were mounted onto positively charged glass slides. e
Immunohistochemistry
An indirect immunostaining technique was applied for the demonstration of endothelial cells by the use of antibodies toward von Willebrand factor (A0082) f as the primary reagents. Briefly, after deparaffinization, antigen retrieval was performed in 0.072% protease g solution for 5 min. This was followed by blocking of endogenous peroxidase by 0.6% H2O2 for 15 min, and blocking of unspecific binding sites by Ultra V Block. h After incubation with the primary reagents, a primary antibody enhancer was applied. h The horseradish peroxidase polymer was applied, and the reaction was developed with 3-amino-9-ethylcarbazole as described by the manufacturer. h Throughout the immunostaining protocol, with the exception of the step between blocking of unspecific binding and the application of the primary reagent, the slides were washed in Tris-buffered saline. After immunostaining, the sections were counterstained with Mayer hematoxylin. Negative controls for the immunostaining were run on parallel sections without the primary reagent and with a nonsense polyclonal antibody of the same concentration as the primary reagent.
Fluorescence in situ hybridization and statistical analysis
In etiologically undetermined cases, FISH was applied with the following oligonucleotide probes: 1) a general bacterial probe, S-D-Bact-0338-a-A-18 (5′-GCTGCCTCCCGTAGGAGT-3′), i targeting nearly all members of the bacterial domain 1 ; 2) a probe specific for S. suis, S-S-S.suis-0183-a-A-18 (5′-CCATGCGGTAAATACTGT-3′) i , 5 ; and 3) a specific probe for E. rhusiopathiae, S-S-E. rhusiopathiae-449-a-A-18 (5′-TCCCTCTTCCTATCGT TC-3′) i designed by the use of the software ARB 15 and the database Greengenes (http://greengenes.lbl.gov/cgi-bin/nph-index.cgi). j Specificity of the new probe was tested in silico by BLASTn (http://www.ncbi.nlm.nih.gov/), k and affinity was confirmed on formalin-fixed lung tissue injected with a pure culture of E. rhusiopathiae strain S31A5. The oligonucleotide probes i were 5′ labeled with the isothiocyanate derivative cyanine (Cy)3. The slides were deparaffinized in xylene and dehydrated in 100% alcohol for 30 min before hybridization. The hybridization was carried out at 45°C with 40 μl of hybridization buffer (100 mM Tris [pH 7.2], 0.9 M NaCl, 0.1% sodium dodecyl sulfate) and 200 ng of each probe for 16 hr in a slide rack. l The samples were then washed 3 times in prewarmed (45°C) hybridization buffer for 15 min and subsequently 3 times in prewarmed (45°C) washing solution (100 mM Tris [pH 7.2], 0.9 M NaCl). The samples were rinsed in water, air dried, and mounted in a fluorescence mounting medium m for epifluorescence microscopy. An epifluorescence microscope n equipped for epifluorescence with a 100-W lamp n and filter sets 23 or 43 were used to visualize Cy3. Images were obtained using a camera and the software AxioVision version 4.5. n Entire tissue sections of in situ hybridization were scanned. o The significance of observations was evaluated with the chi-squared test with a significance level of P < 0.05.
The localization and cause of valvular endocarditis in 117 slaughter pigs. *

AV = atrioventricular.
Identified by bacteriological isolation: 30; identified by in situ hybridization: 5.
Identified by bacteriological isolation: 40; identified by in situ hybridization: 5.
Records on the localization were missing.
Results
Gross pathology and etiology
A total of 117 hearts with macroscopically visible endocarditis lesions were received in addition to 90 control hearts free of endocarditis and with grossly normal valves (i.e., in 27 submissions a control heart was not included). The microbiological results together with the localization of lesions are shown in Table 1.
Erysipelothrix rhusiopathiae was identified as the causal agent in 35 cases (30%), whereas streptococci were the etiology in 62 of the diseased hearts (53%), with S. suis being the predominant species (73%). However, from Table 1, it is also apparent that 15 isolates of streptococci could not be characterized to the species level. Two cases with A. pyogenes were identified, and in 1 of the 2 cases, Fusobacterium necrophorum was also found. Pathogenic bacteria were not isolated from the valves of the control hearts.
All endocarditis lesions were situated on the cusps of valves. In 21 cases, the lesions also involved the adjacent wall. Regardless of etiology, the most common location of endocarditis was the left side of the heart (69 cases; 59%) with only the left AV valve affected in 49 cases, the left AV valve and the aortic valve in 11 cases, the left and right AV valves in 6 cases, the left and right AV and aortic valve in 1 case, and the left AV valve and the pulmonary valve in 2 cases (Table 1). Of the 49 cases with involvement of only the left AV valve, E. rhusiopathiae was identified in 19 (39%) and streptococci in 24 (49%). Of 37 cases involving only the right AV valve, E. rhusiopathiae was identified in 9 (24%) and streptococci in 18 (49%). None of the pigs with E. rhusiopathiae infection had lesions in both the left and right AV valves, whereas 5 (8%) of the hearts infected with streptococci had lesions on both valves. Overall, 2 hearts infected with E. rhusiopathiae (6%) and 17 hearts with streptococcal infection (27%) had lesions involving more than 1 valve.
Of 28 cases where no cause was identified by bacterial culture, FISH identified 5 cases caused by S. suis and E. rhusiopathiae, respectively. Eighteen cases remained etiologically undetermined. In 10 of these cases, single bacteria were detected by the general bacterial probe, whereas in the remaining 8 cases, which all contained heavily mineralized foci, bacteria were not disclosed by the general bacterial probe using the FISH technique.
Histopathology
All endocarditis lesions were histopathologically characterized as chronic vegetative endocarditis cases. Generally, all vegetations were present with a core of granulation tissue overlaid by fibrin similar to previous descriptions. 8,11,22 The histomorphology of the inflammatory reaction in endocardial vegetations caused by streptococci was characterized by a low-to-moderate infiltration with neutrophils. The cells were usually situated in small foci, embedded in the granulation tissue, and surrounded by fibroblasts and macrophages. Bacterial colonies, in which individual coccoidal bacterial cells were easily visualized, were typically situated in the outermost layer of fibrin and were distinctly separated from the fibrin by an enclosing capsule-like structure (Figs. 1, 2). Mineralization (of especially bacterial colonies as demonstrated by von Kossa) was significantly correlated with lesions containing streptococci and was present in 71% (44) of these cases (Fig. 2).
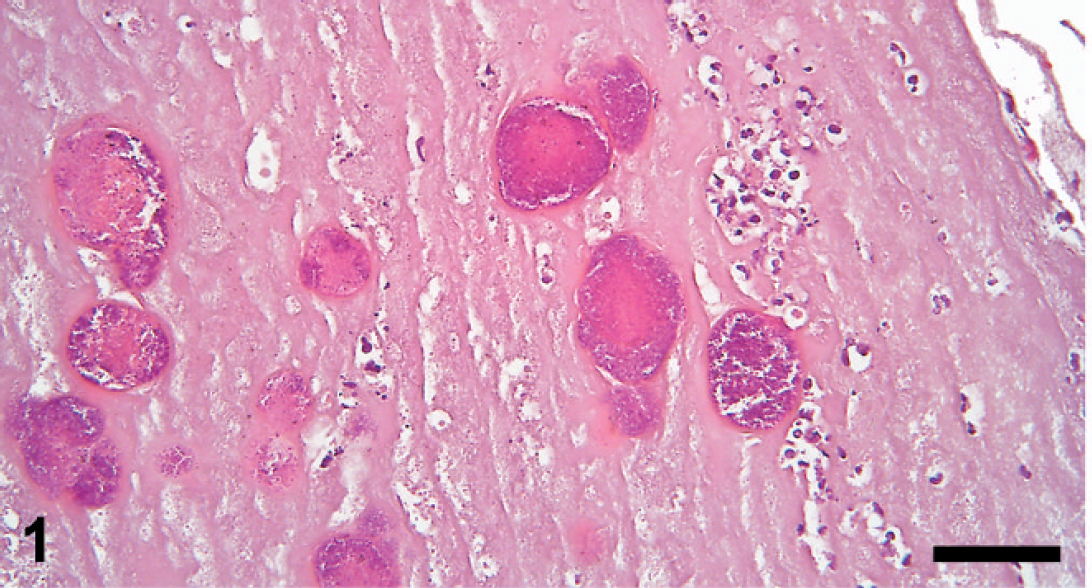
Cusp of the left atrioventricular valve of a pig with endocarditis. Within the thick layer of fibrin, multiple colonies of Streptococcus suis are present. The colonies are enclosed by a capsule-like structure. Hematoxylin and eosin. Bar = 50 μm.

Cusp of the left atrioventricular valve of a pig with endocarditis. The colonies containing streptococci are mineralized and enclosed by a capsule-like structure. Hematoxylin and eosin. Bar = 30 μm.

Cusp of the left atrioventricular valve of a pig with endocarditis. Beneath the diffusely Erysipelothrix rhusiopathiae–infected layer of fibrin, a heavy infiltration with neutrophils is present. Hematoxylin and eosin. Bar = 80 μm.

Cusp of the left atrioventricular valve of a pig with endocarditis. In Erysipelothrix rhusiopathiae–caused endocarditis lesions, a massive infiltration of neutrophils (N) is situated beneath the layer of fibrin (F). The bacterial colonies are located diffusely in a rim (

Cusp of the right atrioventricular valve of a pig with endocarditis. With the Streptococcus suis–specific probe, individual coccoid bacteria are seen within the focal colonies. Fluorescence in situ hybridization. Bar = 60 μm.

Cusp of the left atrioventricular valve of a pig with endocarditis. Using the Erysipelothrix rhusiopathiae–specific probe, the bacterial colonies are present in a diffusely arranged manner. Fluorescence in situ hybridization. Bar = 60 μm.

Cusp of the left atrioventricular valve of a pig with endocarditis. Next to an area with mineralization (M) in a Streptococcus suis–infected endocarditis lesion, a granulomatous reaction with formation of giant cells of the foreign body type (G) is present. Hematoxylin and eosin. Bar = 60 μm.

Cusp of the left atrioventricular valve of a pig without endocarditis (control group). Focally, an area of proliferating endothelial cells is present on the cusp, which appeared normal at gross inspection. Masson trichrome. Bar = 50 μm.
The inflammatory reaction in lesions caused by E. rhusiopathiae was accompanied by a moderate-to-massive infiltration of neutrophils, characteristically situated in a distinct zone between the layer of fibrin and the granulation tissue (Fig. 3). The bacterial colonies were typically observed as diffusely arranged, deep in the layer of fibrin (Fig. 4). The bacterial colonies had a cloudy appearance, lacking a pronounced boundary, and separate bacterial cells were not visible. Mineralization was observed in 28% (10) of the E. rhusiopathiae endocarditis cases. This characteristic pattern of bacterial colonies was also apparent in the FISH-stained sections with streptococci (Fig. 5) and E. rhusiopathiae (Fig. 6).
Granulomatous inflammation was seen in 27 (23%) of the endocarditis cases without a significant relation to a specific bacterium. Fourteen of the cases were associated with streptococcal infection, whereas 3 were present in E. rhusiopathiae–infected vegetations. In the remaining 10 cases, an etiologic diagnosis was not established. Eight of these cases (80%) contained areas with a heavy mineralization. Therefore, a total of 62 cases (53%) were mineralized. The occurrence of granulomatous reactions was significantly related to the presence of mineralization (P < 0.0001), and within lesions, the granulomatous reaction typically surrounded mineralized material (Fig. 7). A varying number of multinucleated foreign body giant cells were observed adjacent to the areas of mineralization. These areas were surrounded by a concentrically arranged zone of macrophages and epithelioid cells. In a few instances, fibroblasts and connective tissue composed the outermost lining of the granulomatous foci. Occasionally, a few giant cells and epithelioid cells were scattered within the layer of granulation tissue.
Among the 90 control specimens, lesions were observed in 22 cases and were of 2 different types of cellular manifestations. In one of these, lymphocytelike cells were localized subendothelially or centrally within the stroma of cusps (n = 13). The other type (n = 9) was seen as local proliferation of superficial endothelial cells (Fig. 8). Regarding the topographic localization, 14 of the 22 lesions were situated in the left side of the heart (12 on the AV valve and 2 on the aortic valve, compared with 8 on the AV valve in the right side).
Discussion
The endocarditis lesions of slaughter-age pigs were predominantly located in the mitral valves, which is in accordance with previous observations. 8,11 Also, streptococci (especially S. suis) and E. rhusiopathiae being the predominant causes of infection is in line with previous reports. However, in some studies, 11,16 streptococci were most prevalent, whereas in others, E. rhusiopathiae was most frequently isolated. 8,20 From a previous study it seems that the etiology might reflect the age groups examined, as streptococci were more frequently observed in younger animals, whereas E. rhusiopathiae dominated in endocardial lesions of older animals. 11 However, in a former Danish survey of the bacteriology of endocarditis in slaughter pigs, E. rhusiopathiae was isolated 6 times as often as streptococci. 20 The dominance of different bacteria being the main cause of porcine endocarditis as observed in several studies may be due to differences in vaccination and treatment programs specifically directed toward E. rhusiopathiae and S. suis, respectively. It is tempting to speculate that an intensive vaccination program (e.g., E. rhusiopathiae of sows) might lower the frequency of E. rhusiopathiae–associated endocarditis in the offspring. However, natural fluctuation and predisposing factors like environmental and stress factors may affect the level of susceptibility to the bacteria and thereby influence the patterns of causes. The difference between streptococci and E. rhusiopathiae regarding localization on more than 1 valve may be related to the magnitude and length of infection with S. suis, which is a more frequent cause of systemic spread in piglets compared with E. rhusiopathiae. 18
Although lesions caused by E. rhusiopathiae and S. suis cannot be distinguished at gross examination, very characteristic histologic differences among the 2 etiologies were present with relation to both pathology and the shape and localization of bacterial colonies. This is in contrast to 2 previous reports, in which no morphological difference was seen. 8,11 However, in these 2 studies, only a limited number of hearts (21 and 30 cases, respectively) were examined compared with the more than 100 in the present series. In these former studies, 10 cases due to E. rhusiopathiae were compared with 4 cases due to streptococci, 8 and 6 cases due to E. rhusiopathiae with 23 cases caused by streptococci, 11 respectively.
Mineralization, especially in areas with disintegrating bacteria, has previously only been described to occur in a few cases of endocarditis lesions in pigs. 11,22 However, the high frequency (53%) of this manifestation in the present material and its significant association with streptococcal infections may be related to a longer period of infection because S. suis infections are typically established at a young age (i.e., in piglets). 17 Also, the significant correlation between mineralization and the occurrence of a granulomatous reaction, a type of inflammation that has hitherto not been described in endocarditis of pigs, is interesting. Due to the strong association between these 2 manifestations, it is most likely that the granulomatous reaction is a foreign body reaction established as a reaction to the dystrophic mineralization. A comparable pattern of reactivity has recently been described in human beings, in which microorganisms causing endocarditis may disappear, leaving the area with granulomatous inflammation including giant cells and calcified deposits. 23 However, in human beings, nonmineralized endocarditis lesions caused by streptococci may also evoke a granulomatous reaction. 7 Observation of multiple mineralized areas in 8 of the endocarditis lesions from which bacteria could not be cultured nor be demonstrated by FISH using the general bacterial probe supports the view that completely calcified bacteria are nonviable. 21
The majority of nonvegetative lesions within the 90 control hearts were found in the left side of the hearts, which is in accordance with the observations in a previous study in which 500 apparently normal hearts from slaughter pigs were examined. 12 The occurrence of intravalvular infiltration with inflammatory cells has also previously been observed in apparently normal porcine hearts, and it was speculated that they might represent a state of a healed endocarditis lesion. 6,12 The presence of local proliferation of valvular endothelial cells, which has not previously been observed in apparently normal hearts, might predispose to the susceptibility for thrombosing endocarditis as endothelial surface changes enhances adherence of bacteria. 4 The initiation of these lesions, however, remains obscure.
In conclusion, a high number of endocarditis lesions with mineralization was detected, and this phenomenon was significantly related to the occurrence of a granulomatous inflammation of the foreign body type. With respect to the etiology of lesions, specific and discriminating features with respect to histopathology and colony characterization were present. Finally, in macroscopically apparently normal hearts, the occurrence of local endothelial proliferation is thought to predispose for the initiation of thrombosing endocarditis in pigs.
Acknowledgements. This work was supported by grant 271-07-0417 from the Danish Medical Research Council.
Footnotes
a.
Oxoid Ltd., Cambridge, United Kingdom.
b.
TagCopenhagen A/S, Copenhagen, Denmark.
c.
bNucleoSpin® Extract II, Machery-Nagel GmbH & Co. KG, Düren, Germany.
d.
Macrogen Inc., Seoul, South Korea.
e.
Superfrost® Plus, Menzel-Gläser, Braunschweig, Germany.
f.
Dako Denmark A/S, Glostrup, Denmark.
g.
Sigma-Aldrich Denmark A/S, Vallensbæk Strand, Denmark.
h.
Lab Vision Corp., Fremont, CA.
i.
Eurofins MWG Operon, Ebersberg, Germany.
j.
Centre for Environmental Biotechnology, Lawrence Berkeley National Laboratory, Berkeley, CA.
k.
National Centre for Biotechnology Information, U.S. National Library of Medicine, Bethesda, MD.
l.
Sequenza™, Thermo Shandon, Cheshire, United Kingdom.
m.
VECTASHIELD®, Vector Laboratories Inc., Burlingame, CA.
n.
Axio Imager M1, HBO lamp; Carl Zeiss MicroImaging GmbH, Oberkochen, Germany.
o.
GenePix 4200AL, Molecular Devices, Silicon Valley, CA.
