Abstract
Urinary markers for renal dysfunction are gaining interest, but effects of sampling method, storage conditions, and urinary tract inflammation or infection on these markers are unclear. Therefore, the objectives of the current study were to determine the difference in urinary albumin (uALB), urinary retinol-binding protein (uRBP), and urinary N-acetyl-β-D-glucosaminidase (uNAG) concentrations in cystocentesis and voided samples and to investigate concentration changes after storage at −20°C and at −80°C. Effects of a protease inhibitor were also assessed in samples stored at −80°C for 12 months. In a pilot experiment, influence of in vitro hematuria, pyuria, and bacteriuria on the urinary markers was evaluated. A mixed model was used to calculate mean differences and 95% confidence intervals. Urinary ALB, uNAG, and uRBP concentrations were similar in voided and cystocentesis samples. After storage for 4 months at −20°C, uALB concentration was not affected, and uRBP concentration showed a mild and clinically irrelevant decrease, whereas uNAG activity was significantly lower compared with fresh samples. After storage for 12 months at −80°C, uALB and uRBP concentrations did not differ from fresh samples, but uNAG activity was severely decreased. Protease inhibitor addition did not preserve uNAG activity. Experimental hematuria, pyuria, and bacteriuria did not seem to affect urinary markers, although further research is needed.
Introduction
Early diagnosis of decreased kidney function remains a challenge in veterinary medicine because of limitations of current diagnostic tests. Traditional diagnostic markers such as blood urea nitrogen and serum creatinine will only be increased when already 75% of functional renal mass is lost. 8 Measurement of glomerular filtration rate is a more sensitive technique but usually requires labor-intensive and time-consuming clearance tests. 32 Therefore, novel urinary markers that have demonstrated their potential for early and site-specific detection of renal dysfunction in human studies are gaining interest in companion animal medicine. 15,21,24,27 Among others, these urinary markers include intermediate weight proteins such as albumin (ALB), low molecular weight proteins such as retinol-binding protein (RBP), and urinary enzymes such as N-acetyl-β-D-glucosaminidase (NAG). Glomerular dysfunction causes increased filtration of albumin, whereas tubular dysfunction leads to excessive urinary excretion of RBP and NAG due to disturbed reabsorption or structural damage of tubule cells, respectively. 10 These markers may not only allow early diagnosis of primary renal disease, they are also able to detect subclinical renal dysfunction secondary to infectious or endocrine disorders (e.g., pyometra, leishmaniasis, hyperthyroidism, and Cushing's disease). 9,12,20,31
In order to perform reliable clinical studies using urinary markers, urine samples need to be collected and stored in optimal conditions to preserve protein integrity and enzyme activity. Studies evaluating the effect of sampling method and storage conditions on determination of urinary markers are lacking in veterinary medicine and therefore mandatory.
Hence, the first objective of the current study was to determine whether urinary (u)ALB, uRBP, and uNAG concentrations differ between samples collected by cystocentesis or by free catch. The second aim was to investigate whether urinary marker concentrations decrease over time depending on storage duration and freezing temperature and if a protease inhibitor (PI) cocktail preserves protein concentrations in samples stored at −80°C for 12 months. In a pilot experiment, effects of in vitro hematuria, pyuria, and bacteriuria on urinary marker concentrations were assessed.
Material and methods
Urine samples
The present study was performed at the Faculty of Veterinary Medicine, Ghent University (Merelbeke, Belgium), after approval by the Local Ethical Committee. Eleven privately owned dogs were included (2 neutered males, 5 intact females, and 4 spayed females), diseased as well as healthy, to ensure a broad range of urinary marker concentrations. The study group consisted of 3 healthy dogs, 1 dog with diabetes mellitus, 1 dog with combined diabetes mellitus and Cushing's disease, 1 dog with Cushing's disease, and 5 dogs with chronic kidney disease. Immediately after morning urine samples were obtained by ultrasound-guided cystocentesis, dogs were walked, and a voided urine sample was collected from each dog. During cystocentesis, care was taken to avoid peripheral blood contamination, and none of the samples showed macroscopic discoloration possibly leading to falsely increased urinary albumin concentrations. 29 After centrifugation (3 min, 447 × g), a complete urinalysis (dipstick, a urine specific gravity, urine protein-to-creatinine ratio, sediment analysis, and bacterial culture) was done on cystocentesis samples. Dipstick and sediment analyses of the voided samples were also performed. The remaining supernatant was divided into aliquots of 0.5 ml and frozen at −20°C and −80°C. Sufficient aliquots were frozen to avoid multiple freeze—thaw cycles during future analyses. Fifty microliters of PI cocktail b was added to 1 ml of cystocentesis and voided urine, and 4 aliquots of each were frozen at −80°C. One aliquot of both cystocentesis and voided samples was used immediately for measurement of uALB, uRBP, and uNAG in fresh samples.
Analysis of urinary markers
Urinary ALB, uRBP, and uNAG concentrations were measured in cystocentesis and voided samples within 3 hr after collection (time 0: T0) and after 4 months of storage at −20°C (time 4 months: T4 months). Furthermore, urinary markers were determined in cystocentesis samples after 12 months of storage at −20°C and at −80°C (time 12 months: T12 months) and in PI samples stored at −80°C. Urinary ALB concentration was determined with a commercial canine-specific sandwich enzyme-linked immunosorbent assay (ELISA) kit, c uRBP with a human ELISA kit, d and uNAG activity using a colorimetric assay, e as previously validated and described. 15,27 Mean interassay coefficient of variation was 12.0% for uALB analysis, 8.5% for uRBP, and 8.1% for uNAG. 15,27
Effect of hematuria, pyuria, and bacteriuria
Cystocentesis urine samples of 2 healthy dogs were used to evaluate the effect of experimental hematuria, pyuria, and bacteriuria on uALB, uRBP, and uNAG concentrations. A heparinized blood sample of 1 healthy dog was used to mimic hematuria and to isolate white blood cells. Whole blood was diluted 4,000- and 16,000-fold in the urine samples, yielding a 3+ and 1+ hematuria on dipstick analysis, f respectively. Furthermore, white blood cells were isolated after gradient centrifugation on Percoll. g , 28 Briefly, 2 ml of blood was layered carefully on Percoll with a density of 1.077. The tube was centrifuged at 340 × g for 30 min at room temperature. Then plasma was discarded, and the opaque band containing the white blood cells at the interface between plasma and the Percoll layer was aspirated with a Pasteur pipette and transferred into a conical tube. After 3 washing steps in a phosphate buffered saline solution, the white blood cells were counted. h Cytospin preparations i were stained j and showed isolated white blood cells to consist mainly of neutrophils (74%) and further of lymphocytes (21%) and monocytes (5%). White blood cells were added to the urine samples until a final concentration of approximately 80 white blood cells per μl was obtained, corresponding with 1+ pyuria on dipstick analysis.
For the bacteriuria experiment, Escherichia coli bacteria were grown overnight, and bacterial concentration was determined using a standard curve plotting the colony forming units (CFUs) as a function of the absorbance at 550 nm. Bacteria were diluted in the urine samples until a final concentration of 105 CFU per ml was obtained.
All urine samples (i.e., 1+ and 3+ hematuria, 1+ pyuria, and bacteriuria) and blank urine samples were incubated at 38°C for 2 hr to simulate a retention period in the bladder at body temperature. After centrifugation (3 min at 447 × g), the supernatant was divided into aliquots of 1 ml and stored at −80°C until analysis within 2 weeks. Additionally, blank urine samples without incubation at 38°C were also centrifuged and stored at −80°C as negative controls. Urinary ALB, uRBP, and uNAG concentrations were compared between blank samples and hematuria, pyuria, and bacteriuria samples of the 2 dogs to assess the effect of red blood cell, white blood cell, and bacterial contamination.
Statistical analysis
Analyses were performed with a commercial software program k using a mixed model with dog as random effect, and with collection method, storage time, freezing temperature, and addition of PI as categorical fixed effects. Urinary ALB, uRBP, and uNAG concentrations were compared between cystocentesis and voided samples at T0 and at T4 months. Urinary marker concentrations at T4 months and T12 months were compared with concentrations at T0. At T12 months, uALB, uRBP, and uNAG concentrations in samples stored at −80°C with and without PI were compared with each other and with T0 samples. All tests were performed at 5% significance level.
Mean differences between sampling techniques, storage time, and temperature with 95% confidence intervals (CIs) were derived. Equivalence between sampling techniques or between storage conditions can be claimed when the CI is contained in a region of differences, which is considered to be clinically irrelevant. To calculate the mean percentage change of uALB, uRBP, and uNAG over time, a simple regression analysis was performed with difference at 2 time points as response variable and the value at earliest time point as covariate.
Results
There was no statistically significant difference in uALB, uRBP, and uNAG concentrations between cystocentesis and voided urine samples at T0 or T4 months (P > 0.05). Mean differences between cystocentesis and voided samples at T0 and T4 months are presented in Figure 1.
Differences in uALB, uRBP, and uNAG concentrations after storage at −20°C and −80°C are also shown in Figure 1. At T4 months after storage at −20°C, uALB concentration was not significantly different from T0 (P > 0.05), with a mean difference of 29.96 mg/l (95% CI: [−18.55; 51.48]). Urinary RBP concentration was significantly but mildly decreased (P = 0.02), with a mean difference of 40.11 μg/l (95% CI: [12.51; 67.71]). Urinary NAG activity was significantly lower compared with T0, with a mean decrease of 0.61 U/l (95% CI: [0.27; 0.95]; P = 0.005). Mean percentage decrease ± standard error (SE) was 15% ± 3% for uRBP and 20% ± 11% for uNAG, respectively.
At T12 months after storage at −20°C, uALB and uNAG concentrations were significantly lower than at T0 (P = 0.006 and P < 0.0001, respectively). Mean difference was 99.83 mg/l (95% CI: [44.25; 155.4]) and 2.87 U/l (95% CI: [2.13; 3.6]) for uALB and uNAG, respectively. For uALB, mean percentage decrease ± SE was 20% ± 6% and for uNAG 92% ± 4%. Urinary RBP was not significantly different compared with T0 (P > 0.05), with a mean difference of 37.92 μg/l (95% CI: [−2.13; 77.97]).
At T12 months after storage at −80°C, only uNAG activity showed a significant decrease of 2.7 U/l (95% CI: [2.09; 3.3]) compared with T0 (P < 0.0001), with a mean decrease of 89% ± 5%. Urinary ALB and uRBP concentrations did not differ significantly from T0 (P > 0.05), with a difference of 42.89 mg/l (95% CI: [−15.33; 101.1]) and 7.3 μg/l (95% CI: [−4.84; 19.44]), respectively.
Samples with addition of PI were analyzed only at T12 months after storage at −80°C. Urinary ALB, uRBP, and uNAG concentrations were not statistically different in samples with PI compared with samples stored without PI (P > 0.05). Urinary NAG activity remained significantly lower compared with T0 (P < 0.0001), regardless of PI addition.
As presented in Table 1, uALB, uRBP, and uNAG concentrations did not differ between blank samples and urine samples with 1+ hematuria, 1+ pyuria, and bacteriuria. Urinary ALB and uRBP, but not uNAG concentration, showed a very mild increase in samples with 3+ hematuria.
Discussion
Results of the present study demonstrate that uALB, uRBP, and uNAG concentrations do not differ significantly between cystocentesis and voided urine samples. In contrast, storage conditions may affect uALB and uRBP concentrations and uNAG enzyme activity. Results also suggest that the addition of a PI cocktail does not preserve uNAG activity after long-term storage at −80°C. Effects of experimental microscopic hematuria, pyuria, and bacteriuria on these urinary marker concentrations seem to be minimal, although these in vitro results do not reflect the complexity of an inflammatory process in vivo, and further research is needed.
In human medicine, there are many reports on urine sample storage conditions, their effect on protein concentrations and on subsequent data interpretation. 4,11,16,17,26 To the authors' knowledge, the effect of sampling method on urinary marker concentrations has never been reported in dogs or cats. In reports on uALB, uRBP, or uNAG in canine or feline urine, cystocentesis as well as catheterization and midstream voided urine have been used. 12,19,30 In contrast to voided samples, cystocentesis avoids contamination with lower urinary tract epithelial cells, proteases, and bacteria that may affect urinary markers. 22 When performing cystocentesis, care must be taken to avoid peripheral blood contamination leading to grossly discolored urine samples, since this may cause a falsely increased urinary albumin concentration. 29 However, results of the present study did not indicate a statistically significant difference in uALB, uRBP, or uNAG concentrations between cystocentesis and voided samples at T0 and at T4 months after storage at −20°C. Despite this general finding, 1 dog with combined diabetes mellitus and Cushing's disease, and a negative urine culture, had increased uALB, uRBP, and uNAG concentrations in the voided sample (voided: uALB, 183.17 mg/l; uRBP, 71.1 μg/l; uNAG, 5.86 U/l; cystocentesis: uALB, 33 mg/l; uRBP, 29.8 μg/l; uNAG, 1.51 U/l). In this male castrated dog, macroscopic hematuria was not present in either the cystocentesis or voided sample. Sediment analysis of the cystocentesis sample did not reveal pyuria or inflammation. However, in the sediment of the voided sample, increased amounts of red blood cells, white blood cells, and epithelial cells were detected. Therefore, one hypothesis might be that urethral inflammation or preputial disease caused severe contamination of the urine sample, which affected the urinary markers. This finding suggests that other components of the inflammatory response, not mimicked by the in vitro experiment, may also play a role. Although, in general, a significant difference between cystocentesis and voided samples was not detected, possible inter-individual variations should be considered.
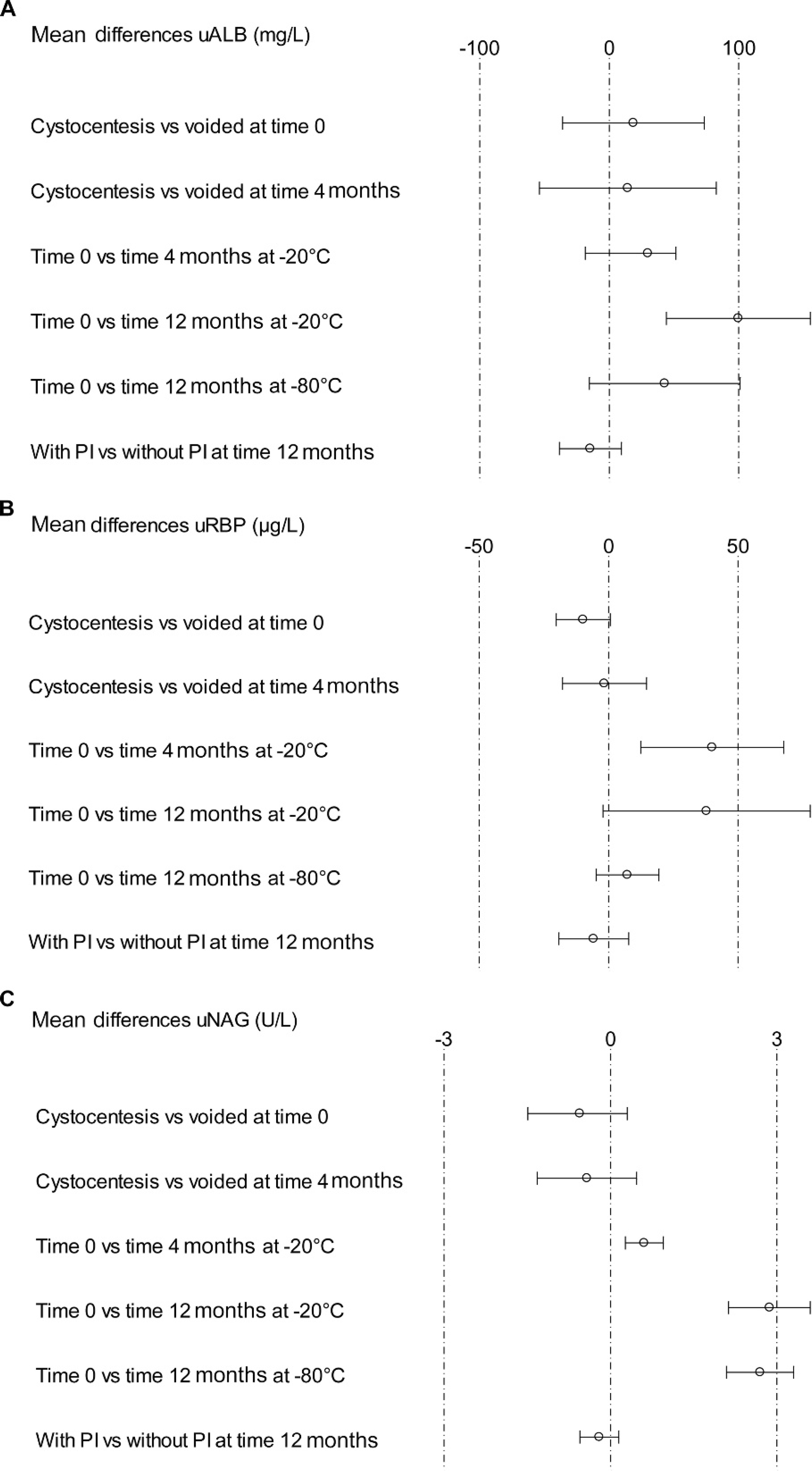
Mean differences in (
Urinary marker concentrations in blank urine samples and urine samples with experimental hematuria, pyuria, and bacteriuria. *
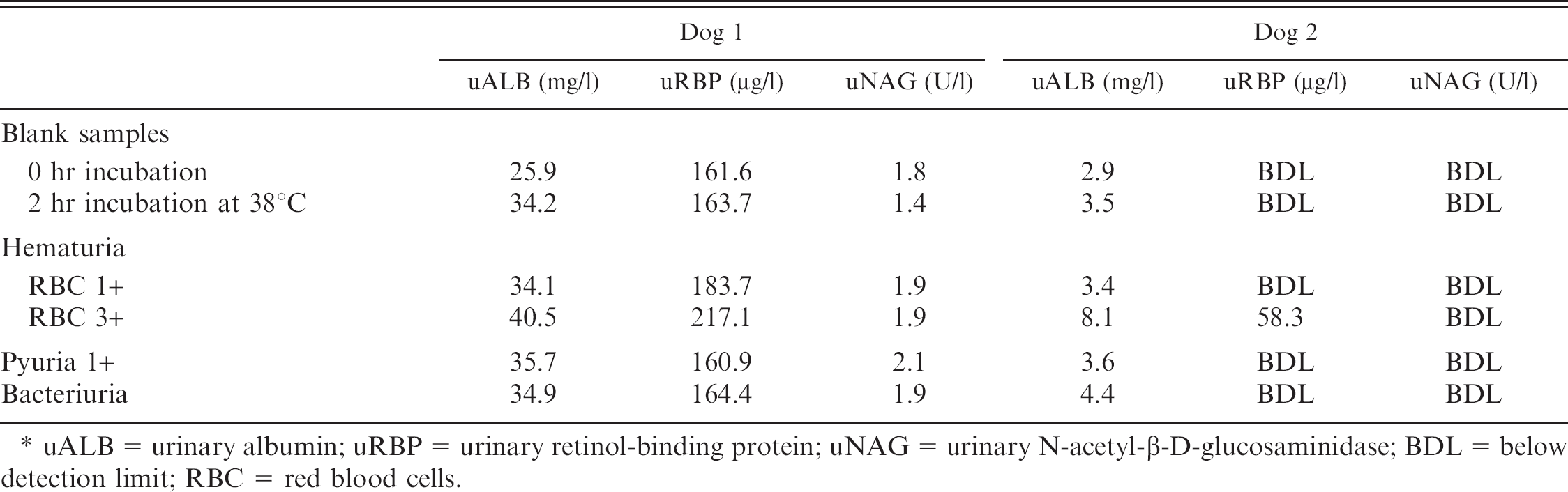
uALB = urinary albumin; uRBP = urinary retinol-binding protein; uNAG = urinary N-acetyl-β-D-glucosaminidase; BDL = below detection limit; RBC = red blood cells.
To the authors' knowledge, there are no reports in the veterinary literature on stability of uRBP, uALB, and uNAG after frozen storage. At T4 months after storage at −20°C, uALB concentration was comparable with T0. In human medicine, there are contradictory reports on uALB stability. Results of the present study are in agreement with previous studies that describe no significant change in uALB concentration after storage at −20°C for up to 6 months. 3,7 However, others detected a decrease of 20% in uALB concentration after 6 months at −20°C. 26 In the present study, uRBP was significantly lower at T4 months compared with T0. Nevertheless, this decrease was mild (40.11 μg/l), and the entire 95% CI was within a clinically acceptable range. Moreover, the RBP ELISA range of analytical variation for canine urine is 15.6% (mean interassay coefficient of variation + 2× standard deviation) and mean percentage decrease of uRBP reported herein was 15%. 15 Therefore, the decrease in uRBP may be due to assay variation. Urinary NAG was significantly lower at T4 months compared with T0, with a mean percentage decrease of 20%. This finding corresponds to results of studies in human medicine, which demonstrated a 25.6% decrease in uNAG after 2 months of storage at −20°C. 2
While both uALB concentration and uNAG activity were significantly decreased at T12 months after storage at −20°C, only uNAG was significantly lower compared with T0 after storage at −80°C. For uALB, these findings are similar to previous results demonstrating a significant decline in human uALB after 12 months at −20°C, but not at −80°C. 11 In the present study, uNAG was dramatically decreased at T12 months with losses of 92% at −20°C and of 89% at −80°C. Addition of a PI cocktail had no effect on the preservation of uNAG. The decrease at −80°C was unexpected because studies in human medicine indicated a largely preserved uNAG activity (84.6%) after storage for 12 months at −70°C. 17 Still, components of canine urine differ from human urine, and enzyme activities are influenced by many factors, such as temperature, pH, specific inhibitors, creatinine, urea, and electrolytes. 13 Alkaline pH (>8) causes a rapid decrease of uNAG, but urinary pH values in the present study ranged from 5.5 to 8, and only 1 sample had a pH of 8. 13,18
At T12 months after storage at −20°C, uRBP was not significantly lower compared with T0. This may seem contradictory because uRBP was mildly decreased at T4 months. However, the former decline is probably due to assay variability, and none of the observed differences is so large as to have clinical relevance. In the current study, storage at −20°C for 4–12 months seems to have only minimal effects on uRBP concentration. This finding is in contradiction with results obtained from human samples demonstrating decreased uRBP concentrations after storage at −20°C for 6–8 months. However, it must be noted that only a minority of samples in the latter study showed substantial underestimation. 26 At T12 months after storage at −80°C, the present study indicated no significant difference in uRBP compared with T0. This is in accordance with data from human medicine demonstrating no significant change in uRBP when stored at −70°C for 8 months. 26
Guidelines advise diagnosticians to exclude urinary tract inflammation or infection (UTI) before identifying the origin of proteinuria because it influences urinary total protein-to-creatinine ratio. 1,14 However, the effect on other urinary markers is currently unclear. In human medicine, there is no evidence that asymptomatic UTI causes proteinuria or albuminuria. 6 Results of the present pilot experiment suggest that experimental pyuria, 1+ and 2+ hematuria, and bacteriuria do not affect uALB, uNAG, or uRBP concentrations. However, these findings have to be interpreted with caution because in vitro addition of leucocytes or bacteria to urine does not fully reflect an in vivo inflammatory process, with its complex interactions between stimulated leucocytes and bacteria involving inflammatory cytokines, bacterial products, and urinary molecules released from various sources. Samples with 3+ hematuria had a negligible increase in uALB and uRBP, probably caused by contamination of plasma albumin and RBP through addition of whole blood to the urine samples. These results are in agreement with previous data from canine urine samples showing no increased uALB in the majority of pyuric samples and a uALB above normal range only in macroscopically hematuric samples. 29 Two studies report similar canine uNAG activity in samples with or without hematuria, asymptomatic bacteriuria, or positive urine cultures. 5,12 One report showed increased uNAG concentration in dogs with pyelonephritis but not in dogs with lower UTI. 25 Effects of UTI on uRBP have not been reported in veterinary medicine. Data from human medicine, however, showed increased uRBP in patients with acute pyelonephritis, but not in patients with cystitis or asymptomatic bacteriuria. 23
Limitations of the present study are that data were obtained at only 3 time points (0, 4, and 12 months) in a relatively small sample group and that the effect of a PI was examined after 12 months at −80°C, but not at −20°C. The pilot experiment to assess effects of hematuria, pyuria, and bacteriuria is limited because it does not fully mimic the complexity of an in vivo cystitis. Therefore, further work is needed to evaluate the true influence of UTI on these urinary markers. The study was not designed to clarify underlying mechanisms of storage effects on the markers but provides essential information regarding storage conditions for further research in the domain of urinary markers.
In conclusion, samples for analysis of uALB and uRBP can be stored at −20°C for up to 4 months, whereas −80°C is preferred for storage up to 12 months. Urinary NAG enzyme activity is less stable at −20°C as well as −80°C, with already substantial losses after 4 months at −20°C and a more severe decline after 12 months at −80°C.
Acknowledgements
This work was funded by a grant from the “Fonds voor Wetenschappelijk Onderzoek (FWO) Vlaanderen.” The authors thank Kristel Demeyere for her excellent technical assistance.
Footnotes
a.
Combur stick, Roche Diagnostics, Mannheim, Germany.
b.
Calbiochem protease inhibitor cocktail set n° III, Merck KGaA, Darmstadt, Germany.
c.
Canine albumin ELISA, Immunology Consultants Laboratory, Newberg, OR.
d.
Retinol binding protein ELISA, Immundiagnostik AG, Bensheim, Germany.
e.
β-N-Acetylglucosaminidase Assay Kit, Sigma-Aldrich, St. Louis, MO.
f.
Multistix 8 SG, Bayer AG, Leverkusen, Germany.
g.
Percoll, Sigma-Aldrich, St. Louis, MO.
h.
Coulter Counter, Coulter Electronics Ltd., Buckinghamshire, England, United Kingdom.
i.
Shandon Cytospin cytocentrifuge and Shandon Cytoclip slide clips, Thermo Scientific, Runcorn, Cheshire, United Kingdom.
j.
Hemacolor, Merck KGaA, Darmstadt, Germany.
k.
SAS version 9.1, SAS Institute Inc., Cary, NC.
