Abstract
Catecholamines can be used to evaluate neuroendocrine tumors, stress, and potentially pain, but catecholamines degrade rapidly. Their metabolites normetanephrine (NME) and metanephrine (ME) have better stability in urine. In cats, urine sampling in a home environment would be beneficial to reduce effects of clinical stress and simplify sampling. We evaluated a human urine ELISA for analysis of NME and ME in feline urine, and investigated the effects of acidification, cat tray pellets, and storage time at room temperature up to 8.5 h. In 26 feline urine samples, mean NME concentration was 192 ± 80 ng/mL, mean intra- and inter-assay CV was 6.5% and 4.2%, respectively, and spike recovery was 98–101%, but dilutional recovery was unsatisfactory. For ME, mean intra- and inter-assay CV was 10.2% and 4.1%, respectively. Mean urine ME concentration was 32.1 ± 18.3 ng/mL, close to the kit’s lowest standard, and spike recovery was 65–90%; the ELISA could not be validated for ME. The stability study, performed for NME on 12 urine samples, did not identify differences between acidified and non-acidified samples, cat tray pellets, or storage time, and no interaction effects. The ME ELISA was not suitable for feline urine; performance of the NME ELISA was acceptable, except for dilution recovery. For analysis of NME, feline urine can be sampled at home using cat tray pellets and stored at room temperature up to 8.5 h without acidification.
Introduction
The catecholamines, epinephrine and norepinephrine, are both produced in the adrenal medulla; norepinephrine is also produced in the postganglionic cells of the sympathetic nervous system.20,31 Catecholamines and their metabolites metanephrine and normetanephrine can be used as indicators of activation of the sympatho-adrenomedullary axis by various stimuli such as neuroendocrine tumors, stress, and potentially pain,16,20,26,31,42 and they have been shown to be useful for the diagnosis of pheochromocytoma in dogs and cats.26,27,42
The half-life of circulating catecholamines in humans is only a few minutes. 21 In human and canine plasma samples, catecholamines have been shown to be sensitive to oxidation; therefore, preservation by acidification and rapid processing of samples is needed to reduce degradation.3,7,11 In comparison, catecholamines have been shown to be relatively stable in human urine, with the highest stability in acidified urine.3,36
Epinephrine and norepinephrine are metabolized into metanephrine (ME) and normetanephrine (NME), respectively, by catechol-O-methyltransferase.16,29,31 These metabolites are collectively termed metanephrines. Compared to catecholamines, analysis of metanephrines may be preferable because the metabolites are more stable and can be found in higher concentrations in human plasma and urine as well as in canine urine samples.12,19,26,27,30
In people and dogs, concentrations of catecholamines and metanephrines can be measured in both plasma and urine samples.10,12,15,22,23,26,27,34,37,39 In cats, few studies are available on analysis of catecholamine and metanephrine concentrations in blood and urine.4,8,42 To the best of our knowledge, a study analyzing feline urine metanephrines using ELISA technology has not yet been published.
Blood sampling can be stressful for cats, especially in an animal hospital environment, 2 potentially leading to increased catecholamine concentrations. Urine sampling can be performed noninvasively in a home environment by natural voiding in a cat tray using special non-absorbent pellets. We were interested in investigating if this method of urine sampling could be used for analysis of metanephrines. A component of the pellets could potentially react with the urine and affect the analysis for metanephrines. Furthermore, it can be challenging for a cat owner to know precisely when the cat is voiding, and urine could remain at room temperature for extended periods of time before sample collection. Acidification could potentially be used for preservation 36 but might be difficult to perform by owners at home. Thus, the aims of our study were to validate a human urine ELISA for analysis of the metanephrines NME and ME in feline urine, and to investigate the potential impact of acidification, use of cat tray pellets, and room temperature storage on concentrations of urine metanephrines.
Materials and methods
Animals and study design
Our study was performed at the Swedish University of Agricultural Sciences (Uppsala, Sweden) and at Evidensia Animal Clinic (Västerås, Sweden), using urine from privately owned cats. The study consisted of 2 parts. The first was validation of a human commercial reverse competitive urine ELISA kit for analysis of ME and NME in feline urine. Pending satisfactory results of the assay validation, we investigated the stability of metanephrines after acidification, when using cat tray pellets (Catrine, Kruuse, Langeskov, Denmark), and storage at room temperature on concentrations of ME and NME in feline urine.
The study was approved by the Uppsala Ethical Committee, Sweden (C117-12 and C51/15) and follows American Society for Veterinary Clinical Pathology quality assurance guidelines for urinalysis in veterinary laboratories. 18 Cats presented for routine neutering or dental treatment were included with owner consent. Healthy cats were also recruited from students and staff at the University and from clients at the Evidensia Animal Clinic. The owners were interviewed concerning the health status of their cats, and all cats underwent routine physical examination. To be included in the study, cats had to be clinically healthy based on history and physical examination (American Society of Anesthesiologists: ASA score 1). 1 Cats were not permitted to receive any medication during the preceding month to be included in our study. Urine specific gravity was measured using a digital refractometer (Pocket refractometer, Atago, Tokyo, Japan), and pH was measured with pH paper (MColorpHast, Merck, Darmstadt, Germany). For assessment of renal health status, urine protein-to-creatinine ratios were calculated. Urine concentrations of protein and creatinine were determined using a commercial multianalyzer (Architect c4000, Abbott Laboratories, Abbott Park, IL) with the manufacturer’s reagents. Urine protein was measured with a turbidimetric method using benzethonium chloride, and creatinine was measured with an enzymatic method.28,35,44 Both intra- and inter-assay CVs were <2% for creatinine. Intra-assay CV was <2%, and inter-assay CV was <5% for protein.
Sample collection for validation study
Samples were collected from untreated cats or from cats given anesthetic and analgesic substances in conjunction with routine neutering or dental treatment. Untreated cats were sampled by natural voiding at home or in the hospital, or by cystocentesis in the hospital. Samples from anesthetized cats were collected in association with routine emptying of the urinary bladder by manual compression prior to neutering or dental procedures. Substances used for neutering and dental procedures are detailed in Supplementary Table 1. All urine samples were acidified to a pH of 2–3 (checked by pH paper) by adding 3.2 M HCl, thereafter aliquoted and frozen. Acidification was performed within 20–30 min by a coauthor (T Srithunyarat or S Hanås), and all samples were aliquoted and frozen within 50 min. Samples were stored at −70°C until analysis, with a maximum storage time of 9 mo. Aliquots of one urine sample from a cat sampled at the hospital under anesthesia during a dental procedure was used as the feline urine control in all of the ELISA analyses.
Sample collection for stability study
For the stability study, cats were sampled at the animal hospital in conjunction with neutering or dental procedures by urinary bladder emptying as described previously, given the same anesthetic and analgesic substances (Supplementary Table 1), or sampled by natural voiding at home. Samples from cats sampled at home were brought to the animal hospital at ambient temperature within 30 min of sampling.
Each urine sample was divided into 3 tubes with 2 mL of urine per tube. Tube 1 was untreated (i.e., non-acidified sample). Tube 2 was acidified as described previously. Ten cat tray pellets were added into non-acidified urine for tube 3. All samples were kept at room temperature (18–23°C).
At 0, 30, 210, and 510 min after sampling, aliquots of 0.5 mL were taken from tube 1 and 2, respectively, transferred to cryotubes (Low temperature freezer vials, VWR International, Leuven, Belgium), and frozen at −70°C. The 0-min samples were omitted from the cats sampled at home because of the time needed for transportation of samples from home to the laboratory. At 210 and 510 min of sampling, aliquots of 0.5 mL were taken from tube 3, transferred to cryotubes, and frozen at −70°C. All samples were stored at −70°C until analysis, with a maximum storage time of 10 mo.
Analysis of urine metanephrines
All ELISA analyses were performed by the same researcher (A Svensson) according to the manufacturer’s instructions including hydrolysis and acylation of samples before analysis and shaking during incubations. The reverse competitive human ELISA kit used in our study is available as a combination package for ME and NME (2-Met urine ELISA, BA E-8600, Labor Diagnostika Nord, Nordhorn, Germany) or for each metabolite separately (Normetanephrine urine ELISA BA E-8500, Metanephrine urine ELISA BA E-8400, Labor Diagnostika Nord). Validation analyses were performed using 3 kits with the same lot number of the combined 2-Met urine ELISA (BA E-8600). Each urine sample was analyzed in triplicate. Included ready-to-use standards were used. The standard concentrations were 0, 30, 90, 300, 900, and 3,000 ng/mL for NME. The standard with the highest concentration (3,000 ng/mL) was only used in the first run and excluded in the following runs because all samples had a concentration <900 ng/ml. For the ME assay, the concentrations of the standards were 0, 20, 60, 200, 600, and 2,000 ng/mL.
The feline urine control and the 2 human controls provided by the manufacturer were included on each ELISA plate and used to determine inter-assay coefficients of variation (CVs). Intra-assay CVs were calculated based on triplicate results from all urine samples. 25
For determination of dilution recovery, 2 feline urine samples as well as the high control included in the kit were diluted 1:2, 1:4, 1:6, 1:8, and 1:10 with standard A (calibrator 0, included in the kit). For the high control, the 1:4 dilution was omitted given limitation of space on the ELISA plate. Diluent for sample dilutions was not specified in the manufacturer’s instruction. For spike recovery, the feline urine control was spiked 1:1 with the low kit control and the high kit control, respectively.
Based on the results from the validation study, only the NME part of the assay (BA E-8500) was used in the stability study, and samples were analyzed in duplicate.
Statistical analysis
Statistical analyses were performed (SAS 2014, JMP Pro 11.2.0, SAS Institute, Cary, NC). ME and NME concentrations are presented as mean ± standard deviation (SD) and total range. CV data are presented as mean and total range.
To correct for urine flow rate when comparing NME between groups, urine NME-to-creatinine ratios were calculated. Urine NME-to-creatinine ratios were compared between cats given anesthetic and analgesic substances and the untreated group in the validation study using nonparametric Wilcoxon signed rank test. Urine NME-to-creatinine ratios are given in ng/mL/g/L.
In the stability study, which included several measurements on the same cats, analyses were based on mixed statistical models 32 using the mixed procedure of the SAS package. 38 The repeated-measures part of the data assumed a spatial power covariance structure between observations. 13 The basic model included storage time (0, 30, 210, or 510 min), additive (none, acidification, or cat tray pellets), and the additive × time interaction as fixed factors. The assumptions underlying the model were checked by preparing diagnostic plots. Deviations from normality or homoscedasticity were not detected; therefore, the mixed statistical model described above was used for the analyses. Pairwise comparisons were adjusted for multiplicity using the Tukey method.
Sampling site was assessed by Wilcoxon signed rank test by comparing urine NME-to-creatinine ratios between samples obtained at home and samples obtained at the animal hospital in the validation and stability studies combined. In the stability study, the NME-to-creatinine ratio from the acidified sample at time-point 30 min was used in the analysis. A value of p ≤ 0.05 was considered significant for all statistical analyses.
Results
Twenty-six urine samples from 23 cats were included in the validation study including assay precision, dilution recovery, and spike recovery (Tables 1, 2). Of these samples, 13 were collected from untreated cats and 13 from cats given anesthetic and analgesic substances in conjunction with routine neutering or dental treatment. In the sub-study on NME stability, urine samples from 12 cats were included. In the validation study, 8 of the untreated cats were sampled by natural voiding and 5 by cystocentesis. In the stability study, 7 cats were sampled at the animal hospital in conjunction with neutering or dental procedures by urinary bladder emptying as described previously, and 5 cats were sampled by natural voiding at home. Samples from 3 cats with sufficient urine volume were used in both the validation and stability study.
Characteristics of cats included in the validation study (n = 23) and in the stability study (n = 12) of urine metanephrines.
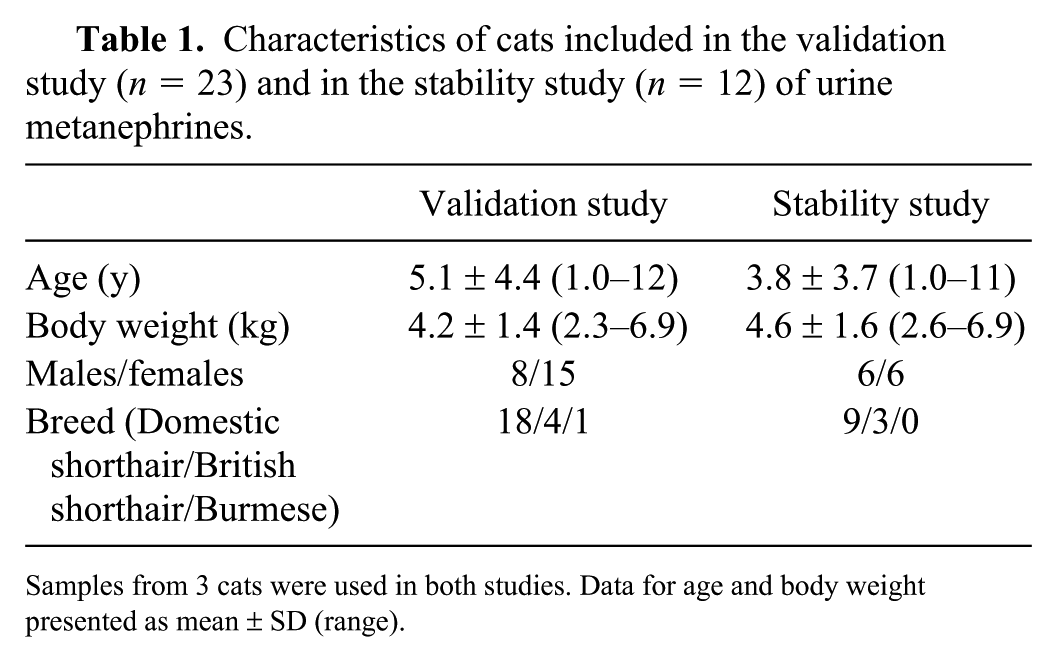
Samples from 3 cats were used in both studies. Data for age and body weight presented as mean ± SD (range).
Urine specific gravity, pH, protein, and creatinine concentrations of feline urine used to evaluate a commercial ELISA for metanephrines.
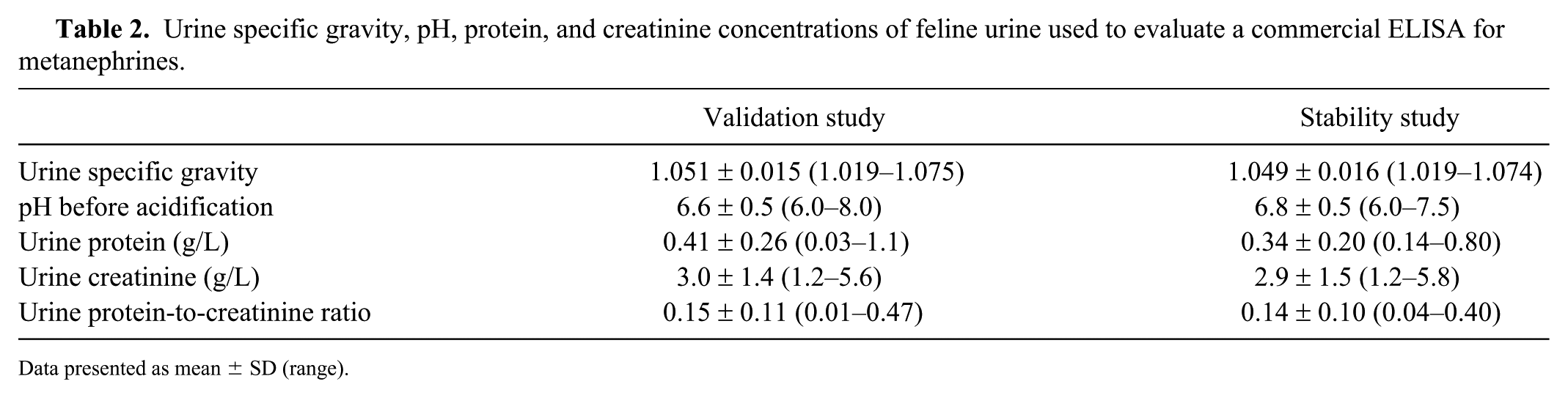
Data presented as mean ± SD (range).
Normetanephrine
The mean intra-assay CV for urine NME in all feline urine samples was 6.5% (range: 4.7–9.2%). The inter-assay CV was 3.9% (0.3–8.0%) for the low control sample (mean concentration validation plates: 184 ng/mL), 3.0% (0.5–6.1%) for the high control sample (mean concentration validation plates: 568 ng/mL), and 4.2% (0.7–11.6%) for the feline urine control sample (mean concentration validation plates: 132 ng/mL). The coefficients of correlation (R2) for all calibration curves were >0.997.
The mean urine NME concentration in the feline urine samples in the initial validation study was 192 ± 80 (96–392) ng/mL. Spike recovery of urine NME was 101% when the feline urine control sample was spiked with the low control sample (205 ng/mL) and 98% when spiked with the high control sample (520 ng/mL). Dilution recovery of the high control sample was acceptable with recovery of 111–122%; dilution recoveries for the 2 feline samples were 139–208% and 136–197%, respectively (Fig. 1).
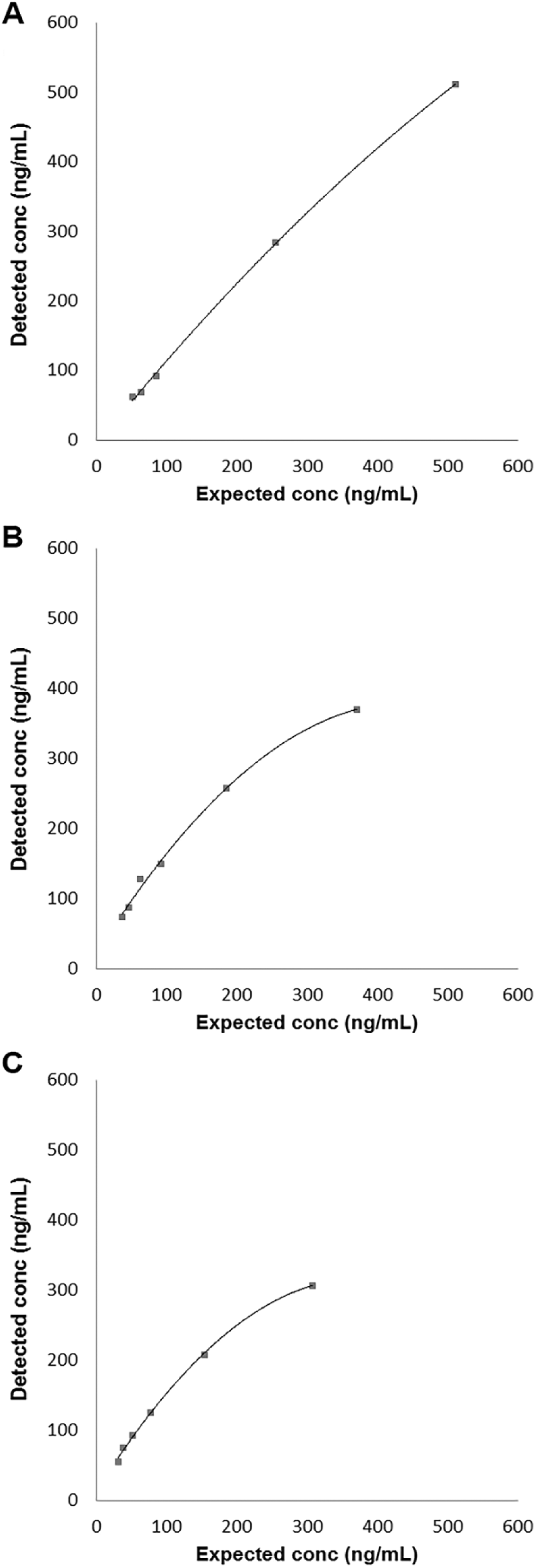
Dilution recovery for normetanephrine (NME) in the feline validation study:
Urine NME-to-creatinine ratios in the validation study did not differ between the untreated cats and the group of cats administered anesthetic and analgesic substances (p = 0.12).
In the stability study, mean urine NME concentration was 204 ± 82 (85–521) ng/mL, and mean urine NME-to-creatinine ratio was 84 ± 36 (32–180). Acidifying the urine, adding the cat tray pellets, or storage up to 510 min did not significantly affect urine NME-to-creatinine ratios (p ⩾ 0.39; Table 3). There was no interaction between additives (acidification or cat tray pellets) and storage time on the urine NME-to-creatinine ratio (p = 0.96).
Normetanephrine-to-creatinine ratio in urine from 12 cats collected in hospital (n = 7) and at home (n = 5) stored under different conditions over time.
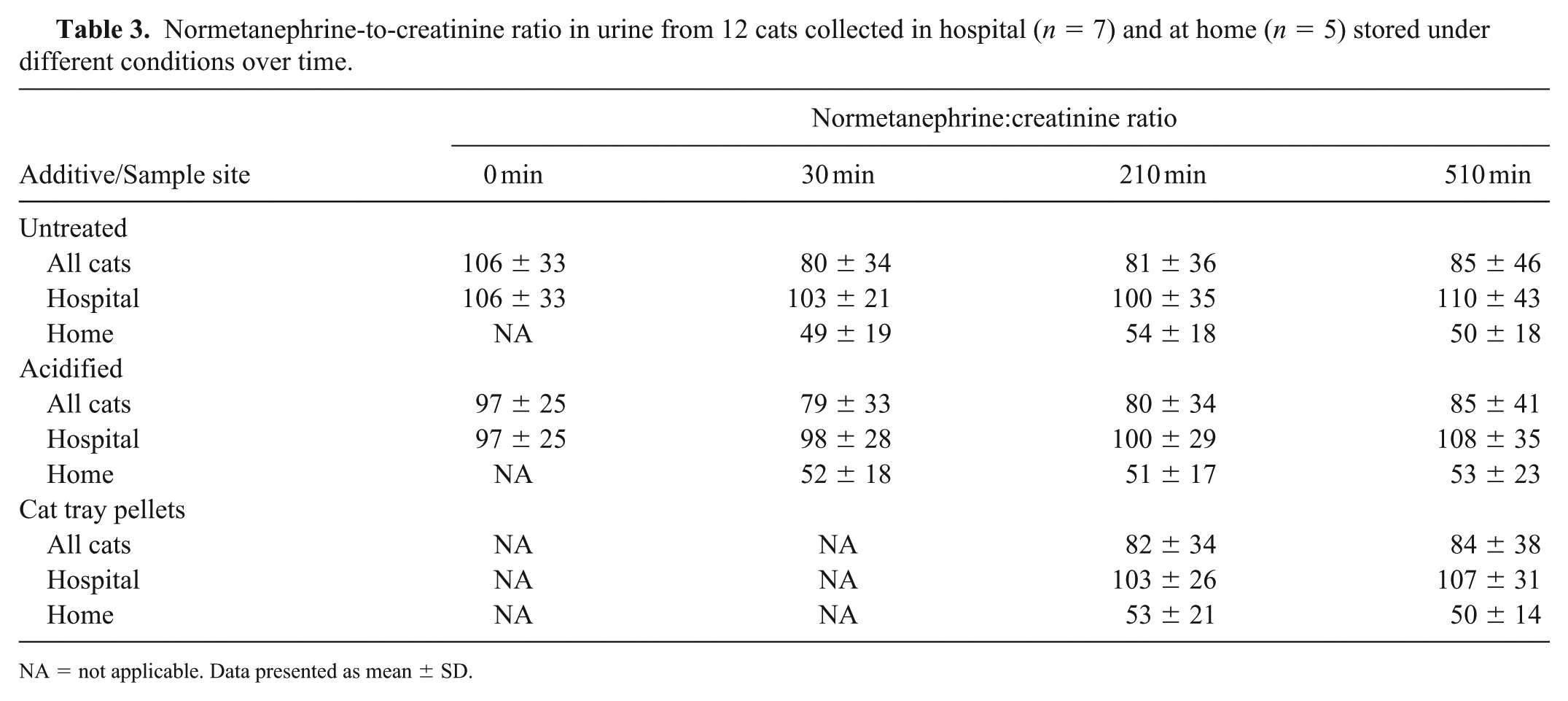
NA = not applicable. Data presented as mean ± SD.
The NME-to-creatinine ratio was significantly higher in samples obtained at the hospital (79 ± 29) compared to samples taken at home (54 ± 18, p = 0.013).
Metanephrine
The intra-assay CV for urine ME in the validation study was 10.2% (8.5–12.0%). The inter-assay CVs for the low control sample (mean concentration: 119 ng/mL), the high control sample (mean concentration: 374 ng/mL), and the feline urine control sample (mean concentration: 21.9 ng/mL) were 3.9% (1.6–7.7%), 9.0% (1.9–16%), and 4.1% (0.6–6.2%), respectively. The R2 for all calibration curves was >0.998.
The urine ME concentrations in the feline samples were generally low, with a mean of 32.1 ± 18.3 (13.6–100) ng/mL. Dilution recovery was therefore not performed for ME.
Spike recovery of urine ME was 90% when the feline urine control sample was spiked with the low control (127 ng/mL) but CV was high (45%). Spike recovery was 65% when the feline urine control sample was spiked with the high control (400 ng/mL).
Because validation results suggested that the ME assay in the human 2-Met urine ELISA was not suitable for measuring ME concentration in feline urine under these experimental conditions, the stability study was not performed for urine ME.
Discussion
For NME, precision was good with mean intra-assay and inter-assay CVs <7%. Spike recovery of NME was very good, with values of 98–101%. It should be noted that spiking of the feline urine control was performed using the 2 human controls provided with the kit. If a feline control had been available for spiking, the recovery results might have been different.
Measured concentrations of NME in the feline samples were 96–392 ng/mL, which is low compared to the original measuring range of the assay of 30–3,000 ng/mL. The low concentrations complicated evaluation of dilution recovery. Two cat samples as well as the high kit control (672 ng/mL) were diluted 1:1, 1:2, 1:4, 1:6, 1:8, and 1:10. The feline samples had initial concentration of NME of 307–370 ng/mL because no feline sample with very high concentration was available for the dilution study. Dilution recovery of the high kit control was acceptable, but the 2 feline urine samples showed poor dilution recovery. The start concentrations were too low and the dilutions steps were too few to properly evaluate the linear range of the NME assay. The manufacturer did not specify any diluents for sample dilutions; therefore, samples were diluted with standard A (calibrator 0) included in the kit. Dilution with other diluents may have given better recovery. Results from diluted and undiluted samples should not be compared until more thorough dilution studies have been performed.
Our validation results for ME were not satisfactory. Many feline samples had very low ME concentrations compared to the measuring range of the assay, with high CV values. No feline samples with sufficiently high concentration were available to reliably perform recovery upon dilution studies, and spike recovery was unsatisfactory. For these reasons, the human 2-Met urine ELISA did not prove reliable for measuring ME in feline urine samples under the conditions in our study. The second part, concerning treatment of the urine sample before analysis of metanephrines, was therefore performed for urine NME, but not for urine ME.
The sampling procedure is important because of rapid degradation of catecholamines. 21 To enhance the stability of catecholamines in human urine, acidification of samples to pH 2–3 is recommended, whereas metanephrines have been shown stable.36,41 In cats, noninvasive urine sampling can be performed in the home environment by letting the cat urinate in a cat tray, thus avoiding the risk of elevated catecholamine concentrations because of a stressful clinical environment. 26 However, because time from voiding until urine is sampled by the owner may vary, there might be risk for degradation. When testing the impact of storage for 0, 30, and 210 min (4.5 h) on rapid degradation, and 510 min (8.5 h, estimated average overnight time) on longer term degradation, we found no significant differences in NME-to-creatinine ratios between the different times, either in acidified or in non-acidified urine. Furthermore, ratios did not differ significantly between acidified and non-acidified samples at any time, and there was no interaction with sample site. Given the time needed for transportation of the samples from home to the hospital, samples taken at home could not be included at time 0.
Nonabsorbent pellets are necessary for urine sampling in a cat tray. To evaluate whether any component of the pellets would react with the urine and affect analysis of NME, non-acidified urine was left at room temperature with cat tray pellets for 210 and 510 min. There was no difference in NME-to-creatinine ratios compared to samples without pellets at the same times. Our findings therefore indicate that commercial cat tray pellets can be used for sampling at home, and urine can be kept at room temperature for up to 510 min (8.5 h), without affecting results of a subsequent NME analysis.
The NME-to-creatinine ratios were significantly higher in samples obtained at the hospital compared to samples obtained at home. Handling and examination at a hospital might be stressful for cats. 2 In dogs, norepinephrine-to-creatinine ratios in naturally voided urine have been shown to increase substantially after transportation to and examination at a hospital compared to samples obtained at home. 22
Potential usage of an NME ELISA for cats in clinical practice would be for evaluation of sympathetic activation by stress, and possibly by pain as well, and for diagnosis of pheochromocytoma.31,42 Individuals with pheochromocytoma would be expected to have markedly elevated NME concentrations.12,42
Concentrations of metanephrines can be measured using high-performance liquid chromatography (HPLC), spectrophotometry, mass spectrometry, and radioimmunoassay in human samples5,19,24,33,39,41 as well as in canine samples.6,14,17,26,27,34,37 In feline samples, chromatographic methods have been used to measure metanephrines in plasma 42 and urine. 8 The above methods are often costly, time-consuming, and require special equipment and specially trained technicians. 5 In our study, metanephrines were analyzed by ELISA, a technique that is relatively simple, can be executed in many laboratories, and therefore is relatively easily accessible.40,43 ELISA has been validated against mass spectrometry for measurement of metanephrines and against HPLC for catecholamines in human urine with good results.40,43 In our study, a competitive ELISA was used. A competitive ELISA is less sensitive than a sandwich ELISA and is more likely to be affected by serum components, 9 thus may give higher CVs than non-competitive assays.
A limitation of our study is the lack of samples with high concentrations of NME, which made evaluation of dilution recovery for the whole measuring range difficult. No cat with pheochromocytoma was included, which is one explanation for the lack of samples with high NME concentrations. The utility of the assay in the diagnosis of pheochromocytoma needs further investigation. An additional limitation is the lack of complete urinalysis. Some component in urine or sediment could have affected metanephrine concentrations. Finally, in the sub-study on effect of storage time, few samples were available at time 0, which might have affected statistical analysis. However, because NME-to-creatinine ratios were stable across all times in urine from the cats sampled at the hospital, this is not expected to have significantly impacted the results.
Supplemental Material
DS1_JVDI_10.1177_10406387187931681 – Supplemental material for Evaluation of an ELISA for metanephrines in feline urine
Supplemental material, DS1_JVDI_10.1177_10406387187931681 for Evaluation of an ELISA for metanephrines in feline urine by Thanikul Srithunyarat, Anna Svensson, Sofia Hanås, Odd V. Höglund, Ragnvi Hagman, Inger Lilliehöök, Ulf Olsson, Ingrid Ljungvall, Jens Häggström, Bodil Ström Holst, Ann Pettersson and Katja Höglund in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank the Department of Clinical Sciences and the University Animal Hospital, Faculty of Veterinary Medicine and Animal Science, Swedish University of Agricultural Sciences, Uppsala, Sweden for allowing the use of facility and equipment. We are also grateful to all cat owners for their collaboration.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was financially supported by The Royal Thai Government Scholarship, the Agria Insurance Company’s and Swedish Kennel Club’s Research Foundation, Sweden, and The Research Foundation on Companion Animals (Sällskapsdjurens forskningsfond), Swedish University of Agricultural Sciences, Sweden.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
