Abstract
Measurements of serum fructosamine, glycated hemoglobin, and glycated albumin (GA) are increasingly used to complement serum glucose concentration for better management of diabetes mellitus. Fructosamine tests are currently not performed in veterinary medicine in Japan. As such, the measurement of GA may serve as a replacement test. Therefore, in the current study, serum GA and fructosamine were evaluated for a positive correlation in dogs, and, depending on the correlation, a reference range of GA percentage would also be determined from healthy control dogs. The degree of glycemic control in diabetic dogs was determined by fructosamine concentration. A positive correlation between GA and fructosamine was observed with both normal and diabetic animals. In addition, the reference interval of serum GA percentage in control dogs was determined to be 11.4–11.9% (95% confidence interval). Interestingly, no significant difference in serum GA percentages was observed between samples from diabetic dogs with excellent glycemic control and control dogs. However, good, fair, and poor glycemic control diabetic dogs resulted in a significant increase in serum GA percentages in comparison with control dogs. These results suggest that serum GA may be a useful diagnostic indicator, substituting for fructosamine, to monitor glycemic control in diabetic dogs.
Assays for glycated blood proteins have not enhanced the diagnosis of diabetes mellitus (DM) but rather allowed better management of DM. Measurements of serum fructosamine, glycated hemoglobin, and glycated albumin (GA) are increasingly used to complement serum glucose concentration in diagnosing DM. The serum fructosamine test has long been used as a tool for diagnosing and monitoring the effect of treatment of DM in dogs. 2 Serum fructosamine concentration reflects the degree of glycation of serum proteins and the mean serum glucose concentration from the previous 1–3 weeks in dogs. 4,8 Thus, it is not only used for determining whether glycemic control has been reached and maintained within a target range but also to estimate the risk of complications associated with chronic diabetes (e.g., cataract formation). 2 Currently, fructosamine tests are not being performed on dogs in Japan because commercial reagents required for testing have been discontinued. Therefore, the measurement of GA may act as a replacement test. Measurement of GA has been commonly advocated in human patients with diabetes and can also provide an index of glycemic control for 1–3 weeks. 3,9,10 The working hypothesis of the current study was that GA could be substituted for fructosamine in the diagnosis and treatment monitoring of diabetic dogs because GA is a main constituent of fructosamine. Therefore, the objectives of this study were 2-fold. First, GA and fructosamine levels were evaluated for correlation in control and diabetic dogs to validate the study's hypothesis. Second, a reference interval of GA percentage in healthy control dogs needed to be identified and created to evaluate glycemic control in diabetic dogs based on GA percentage.
A total of 159 dogs were used for the current study. Ninety-six healthy control dogs (Table 1) were kept and maintained by volunteer families around the Nippon Veterinary and Life Science University (Tokyo, Japan), the Japan Guide Dog Association, and the Japan Service Dog Association. The dogs were fed on various commercial diets. Random blood samples were collected from the control group to obtain serum. Serum biochemistry test values, including assays for glucose, total protein, albumin, cholesterol, triglyceride, serum urea nitrogen, creatinine, sodium, potassium, chloride, aspartate aminotransferase, alanine aminotransferase, and alkaline phosphatase were within reference intervals to be classified as nondiabetic. Sixty-three diabetic dogs were categorized into 4 groups based on glycemic control level (excellent, good, fair, poor), which was assessed by serum fructosamine concentration as previously described. 2,11 Inclusion criteria for diabetic dogs were the following: >250 mg/dl plasma glucose level, persistent glucosuria, polyuria, polydipsia, polyphagia, and insulin-treated or untreated. If diabetic dogs had some concurrent disease (e.g., dermatosis, heart failure, hyper-adrenocorticism, renal failure), they were not included in the study.
Serum samples were stored at −80°C until analyzed. Before use, serum samples were thawed at room temperature and immediately processed. Serum GA levels were measured as a percentage of total serum albumin by an enzymatic method with the Lucica GA-L a assay and were processed by an autoanalyzer b by using the following steps: 1) a glycated amino assay for GA by using proteinase, ketamine oxidase, and peroxidase; 2) an albumin assay by using bromocresol purple; and 3) calculation of the percentage of GA in total albumin.
List of healthy control dogs used in the current study.
Serum fructosamine concentrations were determined by the FOD (fructosyl amino acid oxidase)-TOOS (N-ethyl-N-(2-hydroxy-3-sulphopropyl)-m-toluidine) c method, an enzymatic assay for glycated proteins, and were processed by an autoanalyzer. b Fructosamine was measured directly in μmol. Intra-assay reproducibility of the Lucica GA-L assay was estimated by using 2 pooled serum samples with normal (level 1) and abnormal (level 2) levels of GA with 10 replicates. The same kinds of specimens were also used for evaluating the interassay imprecision, with 9–10 independent runs over 1 month. Linearity was assessed by serially diluting a high-level serum pool (approximately 24% GA up to 32-fold) to obtain various GA values.
For statistical analysis, values were expressed as 95% confidence interval (CI). To assess significance between groups, the Mann-Whitney U-test was used for data with non-normal distribution. Moreover, the D'Agostino-Pearson omnibus test was used for testing normality of distribution. Pearson's product moment correlation was used to identify significant correlations between measurements, and Pearson's product moment correlation coefficient, r, is displayed. All tests were conducted by using SigmaStat analysis software version 3.5.e
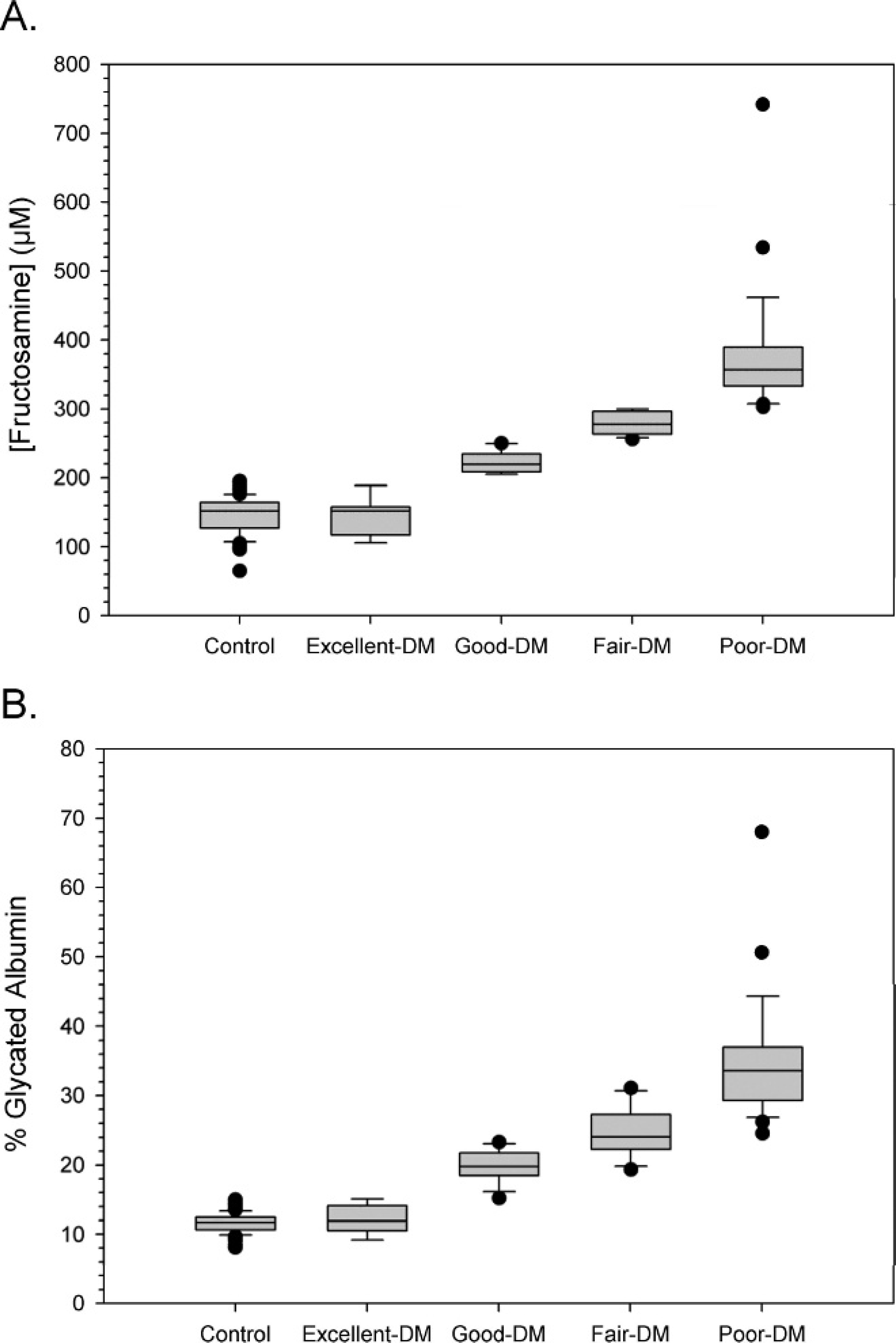
Diabetic dogs had significantly higher serum fructosamine and GA percentage levels compared with control animals (Fig. 1; P < 0.05, Mann-Whitney U-test). When fructosamine and GA levels were examined, a positive correlation (r = 0.979, P < 0.001) was observed for all animals (Fig. 2C). When each group was examined alone, a positive correlation between fructosamine and GA percentage was still observed (control group: r = 0.824, P < 0.001; DM group: r = 0.971, P < 0.001; Fig. 2A, 2B). A high level of correlation between GA and fructosamine was observed with control and diabetic dogs. Moreover, this correlation occurred with a wide range of serum concentrations regardless of breed and age of dogs, thus illustrating its strength.
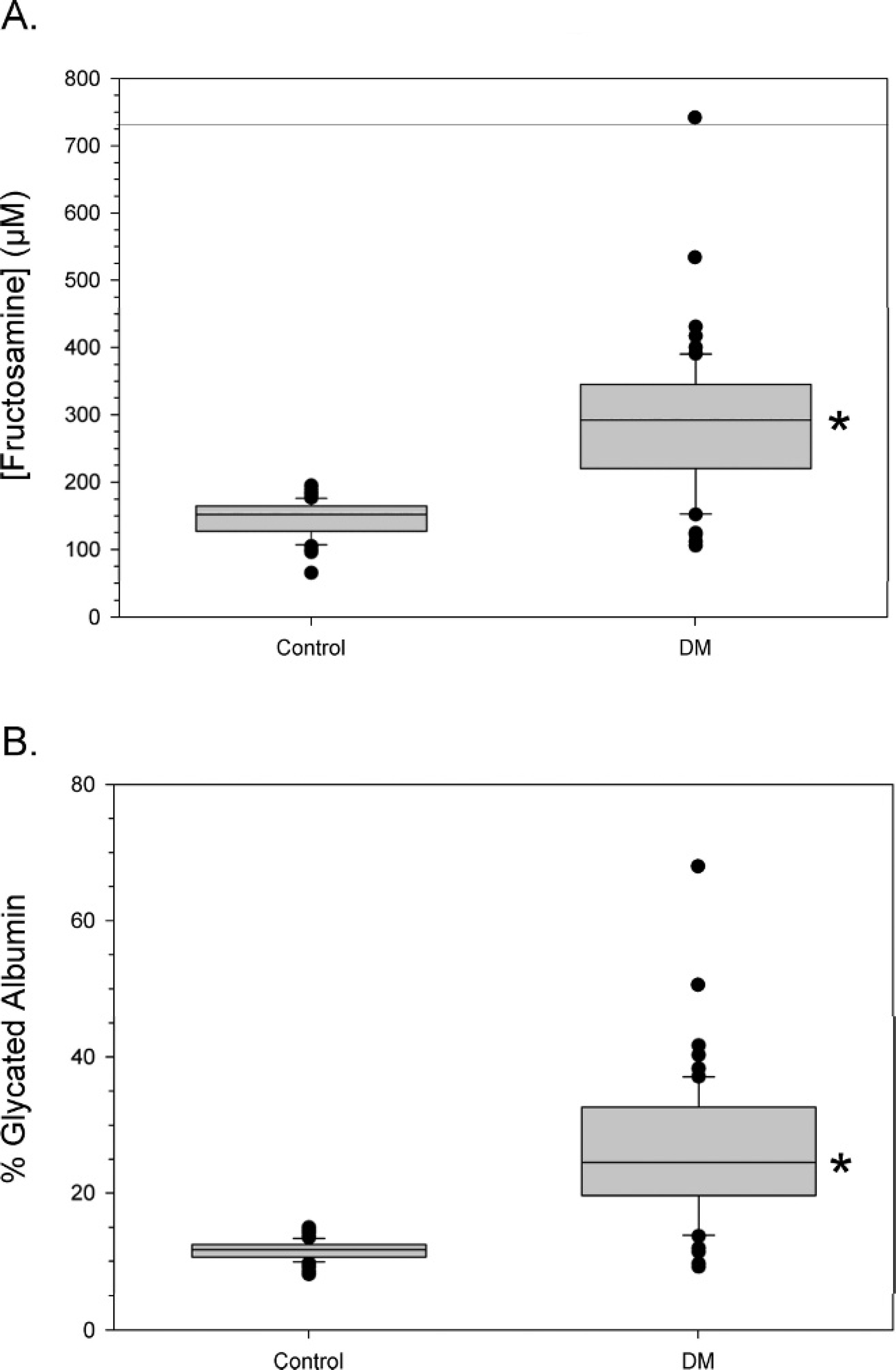
A, fructosamine levels in control and diabetic dogs.
Because there was a positive correlation between fructosamine and GA percentage, a reference range of GA percentage that originated from healthy normal dogs was established, which would be considered useful for diagnosing and treatment monitoring of diabetic dogs for glycemic control. To the authors' knowledge, a normal GA percentage range in dogs has never previously been reported. The canine population that represented the control group passed the D'Agostino-Pearson omnibus normality test, which revealed a Gaussian distribution (P = 0.84). The reference interval of serum GA percentage in control dogs was determined to be 11.4–11.9% (95% CI). Because the reference interval was determined, sensitivity and specificity data for GA percentage were very important. As such, 96 healthy control dogs and 15 of 64 first-time visit diabetic dogs were evaluated. In the current study, the cutoff point of GA percentage > 11.9% was chosen. Because the GA percentage was between 32–68% in 15 first-time visit diabetic dogs, very high sensitivity (100%) was demonstrated. However, 38 of 96 healthy dogs were also over the GA percentage of 11.9%, thus demonstrating specificity to be approximately 60%.
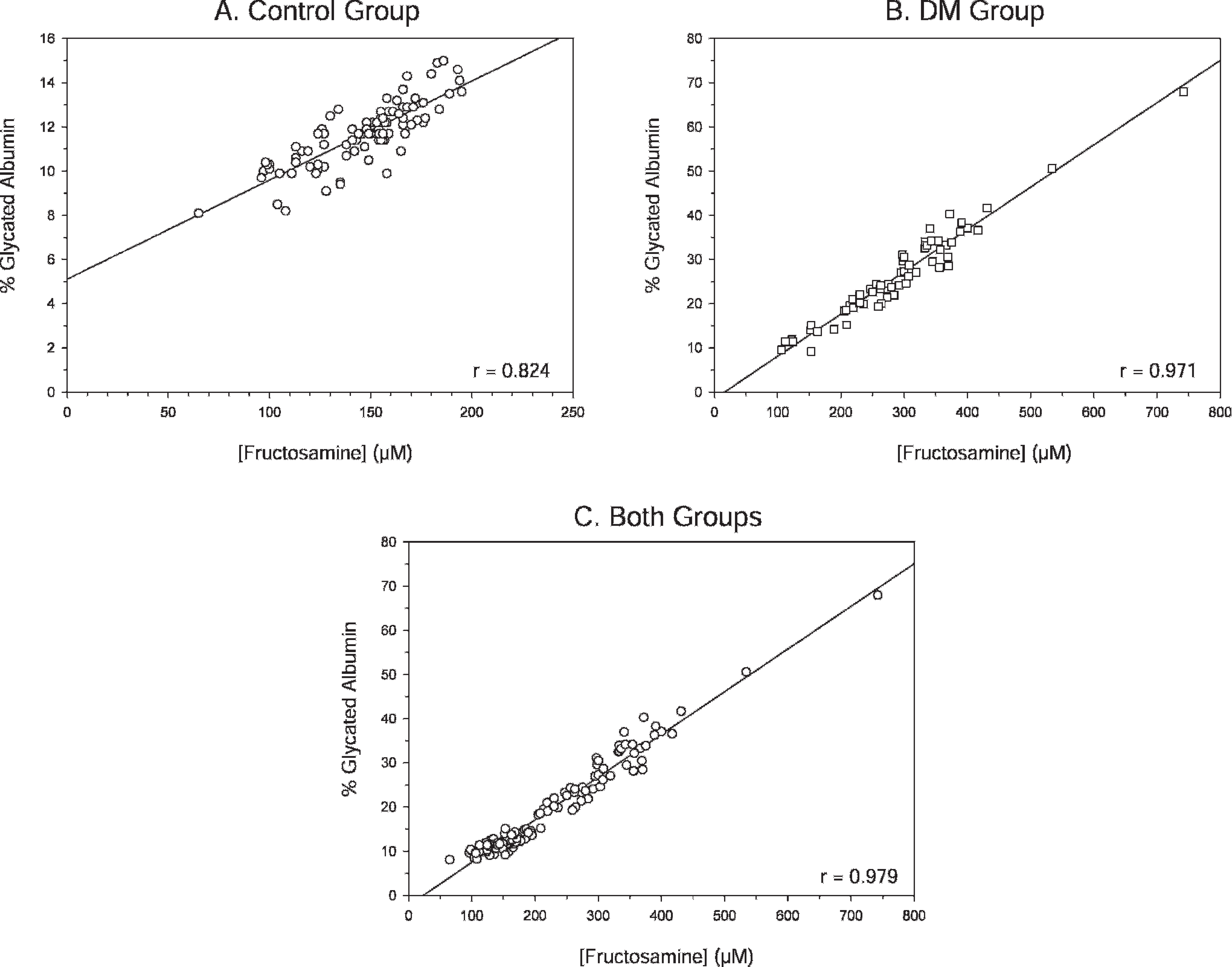
Correlation between serum glycated albumin and fructosamine levels in random diabetes mellitus (DM) samples for
To use GA percentage as a gauge, diabetic dogs were categorized based on glycemic control level (excellent, good, fair, poor) according to serum fructosamine concentration, to create a link between glycemic control level and GA percentage (Tables 2, 3). Interestingly, no significant difference in serum GA percentage and fructosamine was observed between diabetic dogs categorized with excellent glycemic control and control dogs. However, good, fair, and poor-control diabetic dogs resulted in significant increases in serum GA percentage and fructosamine compared with control dogs (Fig. 3). In fact, excellent-control-categorized dogs had their GA percentage range overlap with that of control dogs. However, diabetic dogs with excellent control may be at high risk for hypoglycemia, 1 which is the most serious condition seen with insulin-treated diabetic dogs. Therefore, the ideal level of glycemic control for diabetic dogs should correspond to a GA percentage of 14–21%. This interval is between excellent and good glycemic control in diabetic dogs.
Interpretation of glycemic control of diabetic dogs used in the current study.

Relationship between fructosamine, glycated albumin (GA) percentage, and glycemic control.
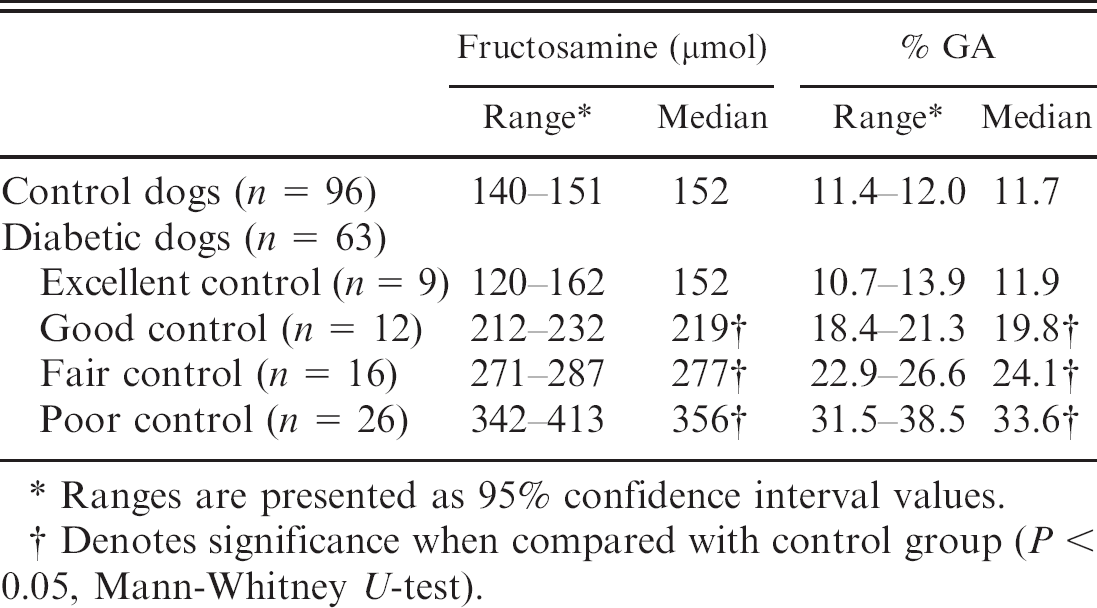
Ranges are presented as 95% confidence interval values.
Denotes significance when compared with control group (P < 0.05, Mann-Whitney U-test).
Assessing intra- and interassay coefficients of variation of the glycated albumin (GA) assay.*
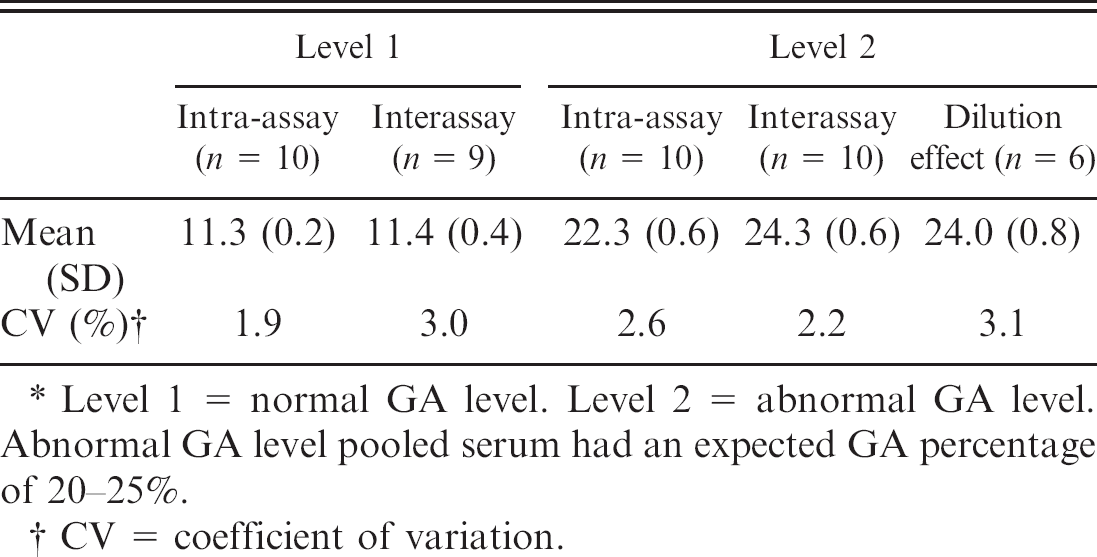
Level 1 = normal GA level. Level 2 = abnormal GA level. Abnormal GA level pooled serum had an expected GA percentage of 20–25%.
CV = coefficient of variation.
Because the GA percentage was determined by using the Lucica GA-L assay kit, it was important to assess its reproducibility and imprecision. Analytical evaluation of the Lucica GA-L assay indicated acceptable analytical imprecision, with intra- and interassay coefficients of variation (CV) not exceeding the commonly accepted 5% limit (Table 4). Intra- and interassay CVs for GA percentage with normal (level 1) levels of GA were <2% and <3%, respectively. When abnormal (level 2) levels of GA were tested, intra- and interassay CVs for GA percentage were <3.0%. Moreover, excellent linearity was demonstrated with all of the dilutions tested, up to 32-fold dilution (Table 5), with an intra-assay CV of 3.1%, which indicates that the commercial kit is reliable and that its results are reproducible and accurate.
With regard to the dilution effect, the starting concentration was serially diluted up to 32-fold and gave satisfactory results by confirming good linearity in the assay. The sensitivity of the assay was as low as 20 mg/dl GA, which was tested in the current study. However, the assay has a theoretical limit of sensitivity of 3.2% GA. Results indicated that the GA enzymatic test proved to be reproducible and robust, as reported in previous studies. 5,7 Moreover, the enzymatic method for GA measurement used in the current study showed high accuracy and reproducibility compared with conventional high-performance liquid chromatography methods. 5 Because this method can use an autoanalyzer, rapid and multiple sample determinations can be performed with ease and convenience.
Linearity and sensitivity of the glycated albumin (GA) assay.*
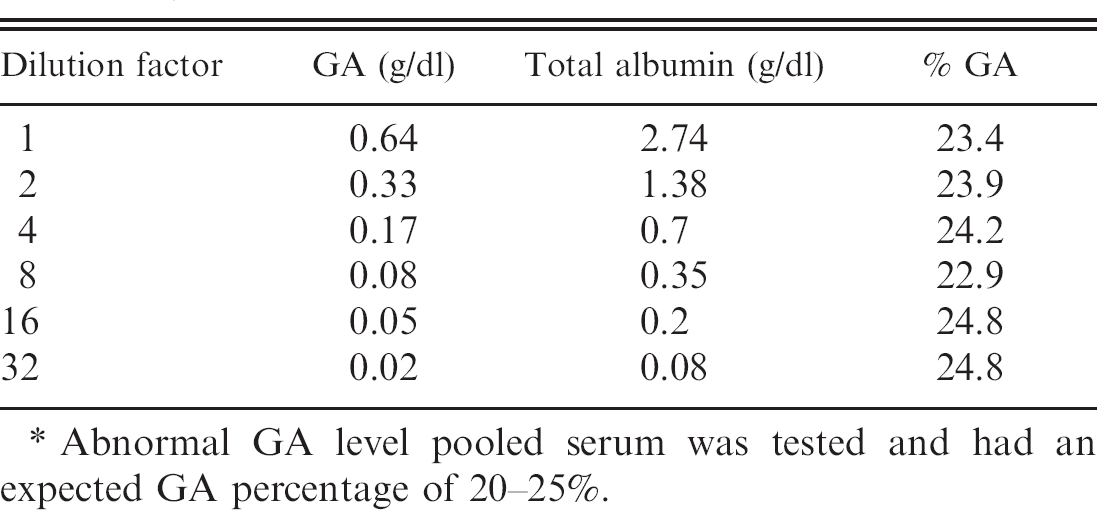
Abnormal GA level pooled serum was tested and had an expected GA percentage of 20–25%.
In conclusion, serum fructosamine concentration can reflect long-term changes in blood glucose concentration and the effect of injected insulin in diabetic dogs. 2 Fructosamine and GA levels in healthy human patients and human patients with diabetes have been reported to show strong correlation. 6,7 As such, the current study sought to determine whether this correlation would also hold true for dogs and, if so, whether GA could be used as a substitute for fructosamine in dogs. The data indicated that a strong positive correlation exists between fructosamine and GA in dogs. In addition, serum GA may be a useful diagnostic indicator to monitor for glycemic control in diabetic dogs as an alternative to fructosamine.
Acknowledgements. The authors wish to thank the Japan Guide Dog Association and the Japan Service Dog Association for their help in collecting blood samples. This work was supported, in part, by the Academic Frontier Project Grant awarded to private universities from the Ministry of Education, Culture, Sports, Science and Technology of Japan (MEXT), 2005–2009.
Footnotes
a.
LucicaTM GA-L, Asahi Kasei Pharma Corp., Tokyo, Japan.
b.
JCA-BM8, JEOL Ltd., Tokyo, Japan.
c.
Auto Wako Fructosamine, Wako Pure Chemical Industries Ltd., Osaka, Japan.
d.
SigmaStatR analysis software version 3.5, Systat Software Inc., San Jose, CA.
