Abstract
The effect of para-JEM® BLUE on Mycobacterium avium subspecies paratuberculosis (MAP) inoculated into broth-based culture media was evaluated by using 84 fecal samples with known MAP status. Results showed that growth of the organism in samples inoculated into the broth without the para-JEM BLUE was detectable 1–35 days (average of 6 days) earlier in 35 of the samples (42%) compared with the same samples inoculated in broth with para-JEM BLUE. Four additional samples (5%) that were MAP positive in the culture broth that lacked the para-JEM BLUE gave negative results when the reagent was included. Of the remaining 45 samples, growth of MAP was detected 1–4 days (average of 3 days) earlier in 4 of the samples (5%) inoculated in the broth with para-JEM BLUE compared with the same samples inoculated in the broth without the para-JEM BLUE, whereas 41 samples (49%) yielded equivalent results with respect to time-to-growth detection and negative growth, regardless of whether para-JEM BLUE was present in the culture broth. However, exclusion of para-JEM BLUE from the broth increased the number of samples that produced false-positive instrument signals compared with the number that produced false-positive signals when the reagent was added. Modification of the sample processing step had no measurable effect. Observations indicated that, although elimination of para-JEM BLUE from the broth increased false-positive instrument signals, its inclusion has an adverse effect on the growth of certain MAP, which suggests that its elimination from broth cultures may increase sensitivity.
Mycobacterium avium subspecies paratuberculosis (MAP) is a major pathogen of cattle. Clinical signs include granulomatous enteritis, weight loss, fatigue, diarrhea, and decreased milk production, which can result in high mortality and substantial economic loss. MAP can also cause diseases in several other domestic and wild animals. 1,4 Recently, MAP was associated with Crohn's disease in humans, thus making it a pathogen of public health concern. 2
MAP-infected animals shed the organism in feces; as such, fecal material is the sample of choice for laboratory diagnosis. For many years, infected animals were identified by fecal culture on solid media, which takes up to 16 weeks to conclude because of the slow growth rate of the bacteria. Such a lengthy culture method is not ideal for on-time management of the disease. The need for a more-rapid culture method prompted the development of a broth-based nonradiometric method that enhances the growth of the organism, as well as an instrument that continuously monitors the bacterial growth and produces signals and characteristic graphs when the presence of MAP is detected. 1 Unlike solid media, the broth-based method takes a maximum of 42 days to conclude.
For the broth-based method, each fecal sample is processed by a double-incubation method described previously, 1 and 1 ml of the processed sample is added to a culture bottle that contain 11.5 ml of para-JEM® broth, a 1 ml of para-JEM GS, a 1 ml of para-JEM EYS, a 500 μl of para-JEM AS, a and 50 μl of para-JEM BLUE. a The bottles are then incubated until a positive signal is detected by the culture instrument or for a maximum of 42 days. b Thereafter, acid-fast staining is performed on all cultures, and the identity of MAP organisms is confirmed by the polymerase chain reaction (PCR) method by using specific primers. 3 During evaluation of the broth-based method and associated reagents between 2002 and 2003, high false-positive instrument signals were noted by participating laboratories (Okwumabua, unpublished observation, 2003). A false-positive instrument signal in such a case is not contamination (growth of an organism other than target organism) but disturbance of an established baseline of the culture instrument by factors other than organism growth. Such factors include temperature fluctuations, electrical noise location, instrument communication failures, or noisy transducer (hardware failure at an individual site), any of which can cause the instrument to signal positive. The observed rate of instrument high false-positive signals prompted the manufacturer to introduce the para-JEM BLUE reagent supplement to level baseline drift and prevent false signals. However, it was observed at the time and discussed in 2003 at the annual meeting of the American Association of Veterinary Laboratory Diagnosticians in San Diego, California, that addition of the para-JEM BLUE to the culture broth slowed down the growth of certain isolates of MAP up to 1 week compared with the broth lacking the culture reagent. Based on that observation, the Wisconsin Veterinary Diagnostic Laboratory (WVDL; Madison, Wisconsin) made a choice to extend the recommended 42 days incubation time to 49 days to compensate for this negative effect. Recently, several veterinary diagnostic laboratories that use the broth culture method reportedly performed poorly in the National Veterinary Services Laboratory (NVSL; Ames, Iowa) proficiency tests administered for laboratory certification because several positive samples were identified as negative because of the failure of MAP in those samples to grow in the broth-based culture system. Based on past experience, it was reasoned that the problem might be associated with the para-JEM BLUE culture reagent. Therefore, comparisons were performed in the present study by using culture broth with or without the para-JEM BLUE. Because the organism may be trapped in feces and sediment with fecal material during sample processing, thereby making it unavailable for inoculation into the broth, the sample-processing step was also slightly modified by shaking the fecal sample–water combination vigorously for 30 min before standing for 30 min at room temperature to enhance dislodging of large fecal particles.
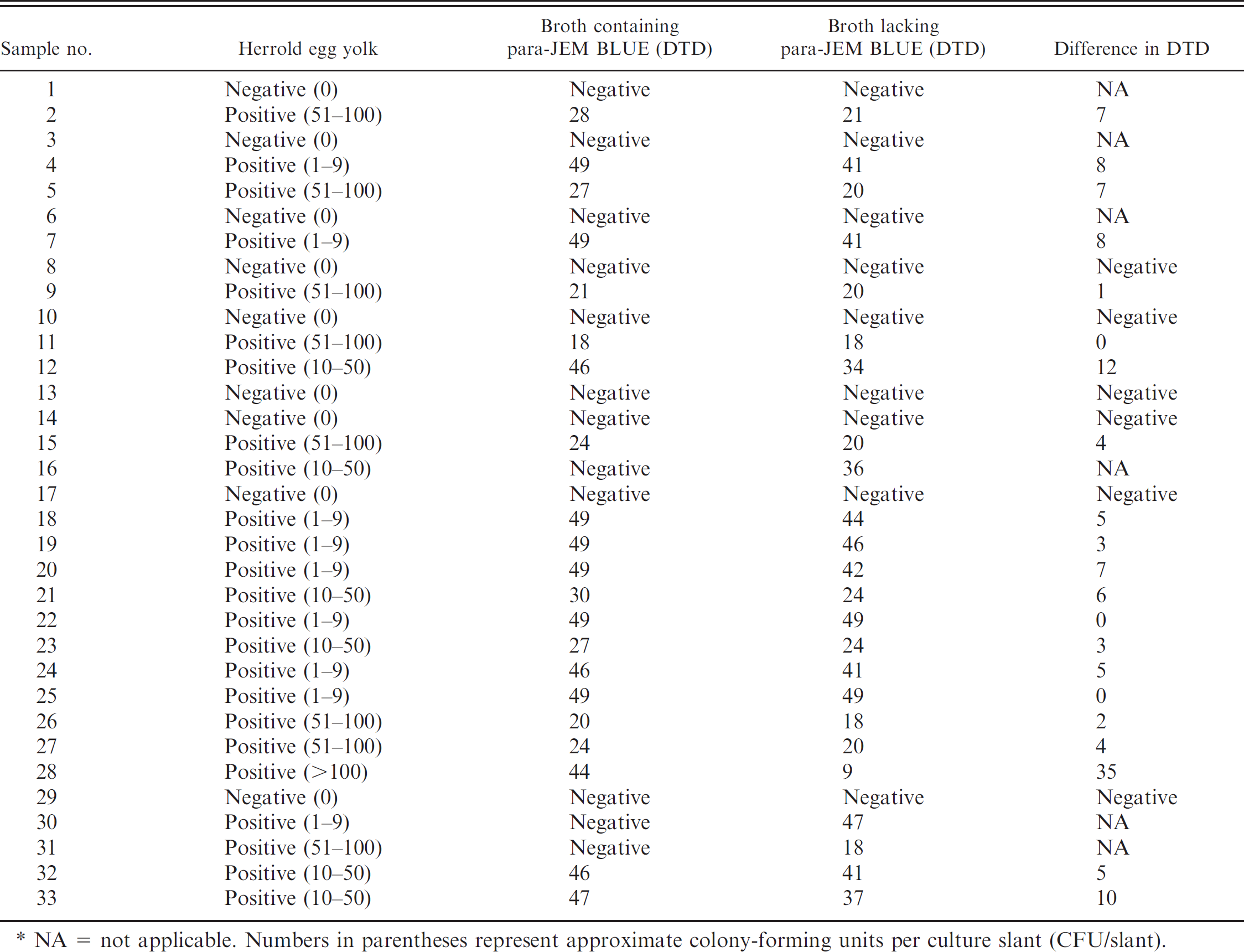
A total of 84 fecal samples with known MAP status were used in the evaluation. Twenty-five samples were obtained from the NVSL as part of the Johne's national annual proficiency test administered to veterinary diagnostic laboratories for certification. Nineteen of the 25 samples were confirmed positive, and 6 were confirmed negative for MAP by NVSL. Fifty-nine samples were collected from cattle with clinically proven or suspected MAP infection submitted to the WVDL from various geographical locations for culture. Forty of the 59 samples were confirmed to be positive and 19 samples were confirmed to be negative when using the Herrold egg yolk medium. c Each of the fecal samples tested was processed by using the standard1 and modified sample-processing methods described herein, and portions were used to inoculate the broth media that contained or lacked the para-JEM BLUE. Cultures were then incubated in an automated system b until the signal or for 49 days. Samples that produced false instrument signals were streaked and incubated aerobically on trypticase soy agar that contained 5% sheep blood and on Sabouraud dextrose agar to rule out bacterial or fungal contamination. All the signal-positive samples were acid-fast stained, and all the acid-fast–positive samples were confirmed by PCR by using MAP-specific primers. 3 All signal-negative samples were acid-fast stained after 49 days of incubation, followed by the MAP-specific PCR assay, regardless of acid-fast results. The median difference in days-to-positive between MAP-positive cultures incubated in broth with and without para-JEM BLUE were compared by using the Wilcoxon signed rank test. The McNemar chi-square test d was used to compare the proportions of MAP-positive cultures obtained by using broth with and without para-JEM BLUE.
Sample results are shown in Table 1. When samples signaled positive in the instrument, acid-fast positive, and PCR positive, they were denoted as MAP positive. Also, when samples were signal negative, acid-fast positive, or negative and PCR positive at the end of incubation, they were denoted as positive. Samples that did not meet these criteria were denoted as negative. Of the 59 confirmed positive samples, 55 were identified to be signal and/or PCR positive in the broth with para-JEM BLUE, whereas all 59 were identified to be positive in the broth without the para-JEM BLUE (McNemar chi-square = 4.00, df, P = 0.046). In the broth without para-JEM BLUE, 35 (42%) of the samples were positive for MAP 1–35 days (average of 6 days) earlier than the same samples inoculated in broth with para-JEM BLUE. The range of days-to-positive as determined by instrument signal and or MAP-specific PCR was 9–49 days in broth without and 18–49 days in broth with the reagent. Four additional samples (5%) that were MAP positive in the culture broth that lacked the para-JEM BLUE gave negative results when the reagent was included. Of the 4 samples, 3 contained MAP in low numbers (1–50 colony-forming units [CFU]/slant) and 1 contained the organism in large numbers (>100 CFU/slant). Of the remaining 45 samples, growth of MAP was detected 1–4 days (average of 3 days) earlier in 4 of the samples (5%) in the broth with para-JEM BLUE compared with the same samples inoculated in the broth without para-JEM BLUE, whereas 41 samples (49%) yielded equivalent results with respect to time-to-growth detection and negative growth, regardless of whether para-JEM BLUE was present in the culture broth. Modification of the sample-processing step yielded similar results as the standard protocol. Comparison of the 55 matched pairs of days-to-positive indicated that the use of para-JEM BLUE significantly increased the median culture time by 3 days to detect the presence of MAP (Wilcoxon signed rank S = 346, P < 0.0001). It was observed that, when para-JEM BLUE was excluded from the broth, 49 of the total 84 samples (58%) tested produced false-positive signals in the culture instrument compared with 23 of the samples (27%) that gave false-positive signals when para-JEM BLUE was added, as evidenced by negative bacterial and fungal growth of the broth samples on plating media and negative MAP DNA detection when using specific primers in a PCR assay with the broth samples. These results indicated that, although the addition of para-JEM BLUE to the broth-based culture media decreased the instrument false-positive rate, the reagent had a negative effect on some isolates of MAP, such as growth retardation and inhibition. Consequently, this can lead to the inability of laboratories that use the broth-based method to culture certain MAP, thus leading to the reporting of false results. The high rate of false-positive signals by the instrument in the absence of para-JEM BLUE could be tolerated until a better solution is available. This is because the culture system generates unique graph characteristics of true MAP growth, which can be differentiated from the type of graph produced in response to a false-positive signal. For the graph produced in response to true-positive growth, gas production, or consumption because of microbial growth causes the pressure readings of the culture system to drop below the established baseline, which produces a reverse-sigmoid curve, whereas, that of a false-positive signal is linear and waving in character. Because reliable detection of gas consumption and production depends on the closed system provided by the culture bottle, comparing data from the baseline can assist a user in determining a cause for false positives. This feature, therefore, allows trained microbiologists to make appropriate decisions before removing the samples from the instrument for follow up. For example, to improve workflow and to make labor less intensive because of the high-volume workload at the WVDL, the false-positive signals are left in the instrument until time permits for the samples to be reset. Resetting of samples is accomplished by removing them from the instrument and placing them at room temperature for 30 min to cool, followed by re-incubation in the instrument. Thereafter, any sample that gives a false-positive signal the second time is left untouched until the end of the 42-day incubation period and then analyzed. Because acid-fast stain is performed on all samples, and identity of positive samples are confirmed by using MAP-specific primers in a PCR format, confidence in identification is increased. It is worth mentioning that, in a recent release of the liquid media results for the 2009 Johne's disease fecal proficiency test by the NVSL, samples in the MGIT a system reportedly signaled positive earlier than the same samples in the TREK b machines. However, it remains to be determined whether this observation is related to the para-JEM BLUE reagent or to other factors.
NA = not applicable. Numbers in parentheses represent approximate colony-forming units per culture slant (CFU/slant).
In conclusion, the results presented herein suggest that elimination of the para-JEM BLUE from broth cultures may increase sensitivity of MAP isolation and expedite the rate of positive signal in the bottles. Since the conclusion of the present study, the WVDL has eliminated the addition of para-JEM BLUE reagent to the culture broth and reduced the incubation time to 42 days.
Acknowledgements
This work was supported in part by TREK Diagnostics Inc., Sun Prairie, WI. The authors thank Eileen Shull and Erik Twaroski for technical assistance.
Footnotes
a.
ESP para-JEM broth, -GS, -EYS, -AS, para-JEM-BLUE; TREK Diagnostic Systems Inc., Cleveland, OH.
b.
ESP® Culture System II, VersaTREK™, TREK Diagnostic Systems Inc., Cleveland, OH.
c.
Herrold egg yolk agar (HEY) with and without mycobactin J, BD Biosciences, Sparks, MD.
d.
SAS version 9.1, SAS Institute Inc., Cary, NC.