Abstract
A Bulldog puppy that died at 1 day of age was presented for postmortem evaluation. Macroscopically, there was marked hypoplasia of the ascending, transverse, and proximal segments of the descending thoracic aorta and almost complete secondary thrombosis of the left ventricle causing a functional stenosis of the left atrioventricular valve. Separately, there was right atrioventricular valve dysplasia with secondary dilation of the right atrium. Microscopically, the left ventricular outflow tract was occluded by chondroid metaplasia, fibrosing recanalization of a left ventricular thrombus, and isolated Purkinje fiber degeneration and necrosis.
Multiple cardiac abnormalities in dogs are uncommon, only occurring in approximately 7–9% of dogs with a cardiac anomaly. 9 The most frequently reported cases of multiple cardiac anomalies involve either persistent right aortic arch or patent ductus arteriosus (PDA) accompanied by another malformation. 5,7,8,10 Aortic tubular hypoplasia in the dog has only been reported once and was associated with a PDA, 14 while focal aortic hypoplasia of the left aortic arch in a dog combined with a double aortic arch and PDA has also been described. 10 In addition, coarctation (interruption) of the aorta as a single entity has also been reported several times in the dog. 4,6,17
A 1-day-old male Bulldog from a multiple litter was found dead and was presented for necropsy to the University of Minnesota Veterinary Diagnostic Laboratory (St. Paul, Minnesota). Macroscopic examination revealed that the external circumference of the aorta was markedly reduced, originating at the root of the aorta and involving the ascending, transverse, and proximal portion of the descending thoracic aorta where the luminal diameter was consistently 2-2.5 mm (Fig. 1). To confirm visual judgment of the marked reduction in the size of the puppy's aorta, it was compared with the aorta (average aortic luminal diameter of 6-7 mm) and pulmonary arteries from a 1-day-old mixed breed puppy of similar size that was presented for routine necropsy with no cardiovascular lesions (Fig. 2). The ductus arteriosus was patent; however, the temporal proximity to birth made it difficult to predict whether the patency would have remained. Both left and right pulmonary arteries of the affected puppy were considered to be of normal size relative to the pulmonary arteries of the unaffected puppy. In addition, the right atrioventricular valve was thickened with fusion of chordae tendinae and leaflets causing a markedly narrowed opening (stenosis; Fig. 1). Secondary to this, the right atrium was markedly dilated (Fig. 3). There were no observable atrial or ventricular septal defects. The left ventricle contained a mature thrombus (Fig. 4) that occupied most of the left ventricular lumen and extended into the left ventricular outflow tract.
Histologically, the luminal diameter of the affected aorta varied from having small segments of patency occupied by low numbers of erythrocytes to that of being minimally detectable (Fig. 5). The latter observation may be an artifact of the orientation of the section. Where present, the tunica intima and media of the aorta appeared normal (Fig. 5). The left ventricular outflow tract, including the aortic root and the aortic valves, was extensively fibrosed and was accompanied by 2 predominant cell populations. The first cell population was of a myxomatous nature, arranged in clusters with the cells having abundant clear foamy cytoplasm and a small darkly basophilic nucleus (Fig. 5). The second cell population, admixed with the first, was composed of chondroid cells arranged in islands, poorly demarcated, and embedded in a lightly basophilic, homogeneous matrix, which was multifocally mineralized. This change was interpreted to be chondroid metaplasia and had most likely arisen from subendothelial stromal cells, which normally produce extracellular matrices such as collagen or elastin (Fig. 5). 9 The authors considered the possibility that these areas of abnormal tissue might have arisen primarily from chondrocytes sometimes resident in the fibrous base of the heart. However, because of the architecture of the tissue and its histopathologic variability and complexity, the possibility was not considered further. The atrioventricular node and bundle of His were adjacent and partially entrapped by the chondroid metaplasia and also abutted the left ventricular thrombus. Because of the architectural distortion of the outflow tract and the aortic valves by inflammation associated with thrombosis, a determination as to whether the aortic valves were primarily dysplastic or destroyed through inflammation could not be made. The left and right ventricular myocardium did not show any microscopic abnormalities; however, the endocardium of the left and right ventricle was diffusely thickened by fibrous connective tissue. Some subendocardial Purkinje fibers either appeared enlarged with clear cytoplasm devoid of content and containing a pyknotic nucleus (degenerate) or slightly shrunken with hypereosinophilic cytoplasm and karyorrhexis (necrotic).
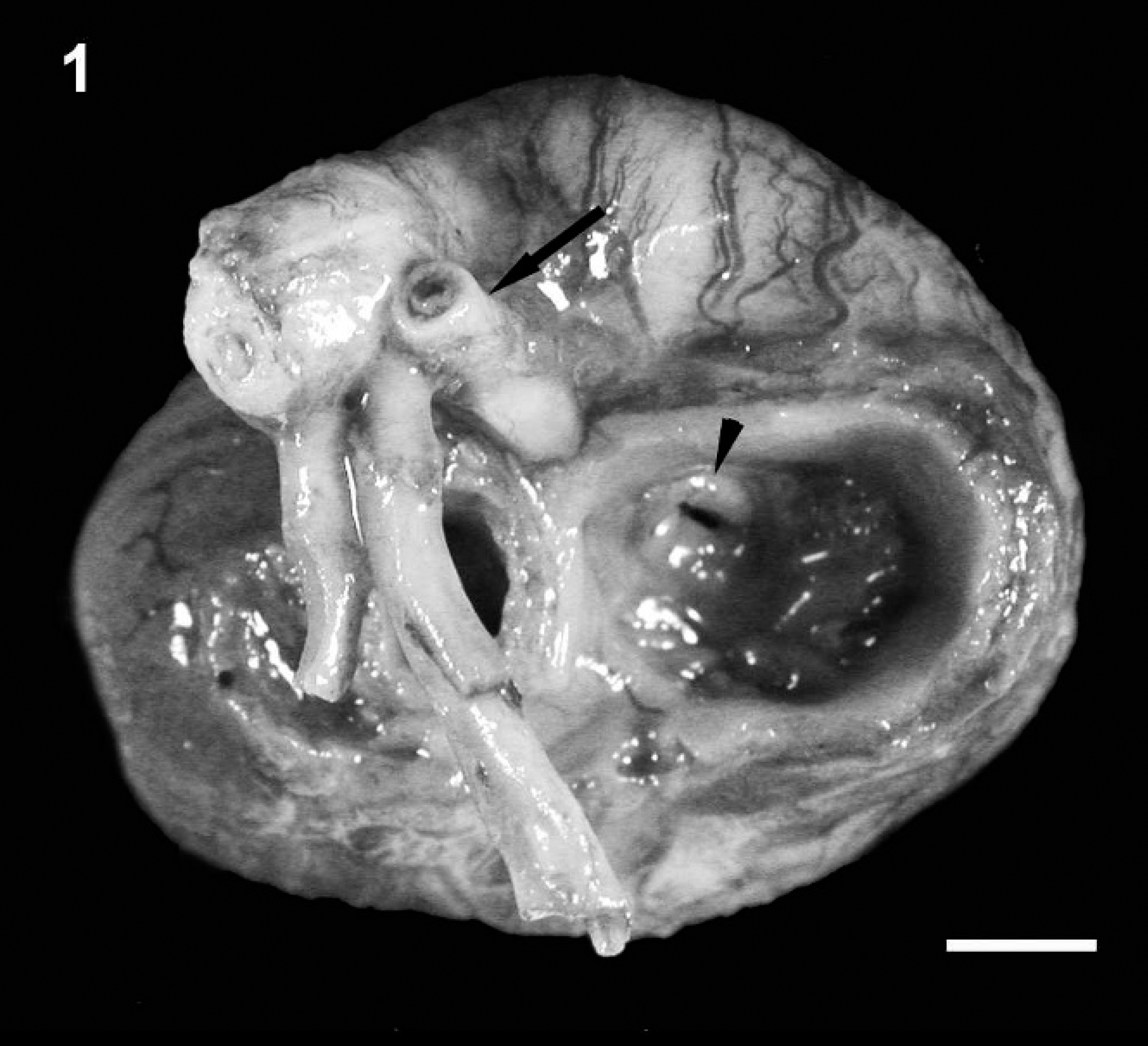
Aortic hypoplasia and right atrioventricular valve dysplasia. Macroscopic view of the heart base and great arteries showing marked hypoplasia of the aorta (arrow) and right atrioventricular valve dysplasia (arrowhead) with normal pulmonary arteries. The left atrioventricular valve is obscured by the 2 primary branches of the pulmonary artery. Bar = 5 mm.
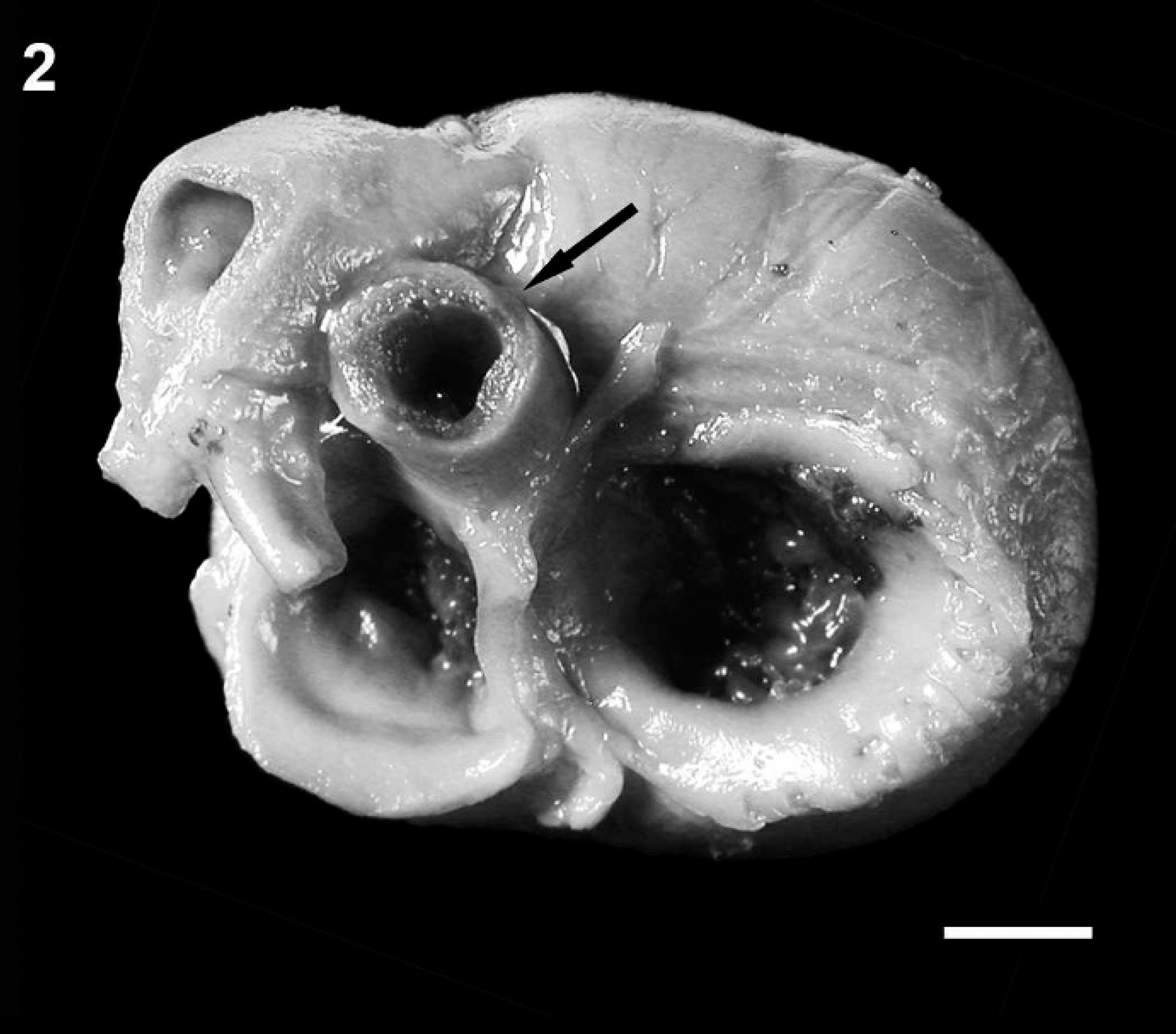
Heart, dorsal view of a similarly aged and sized mixed breed puppy. Note the diameter of the aorta (arrow). Bar = 5 mm.
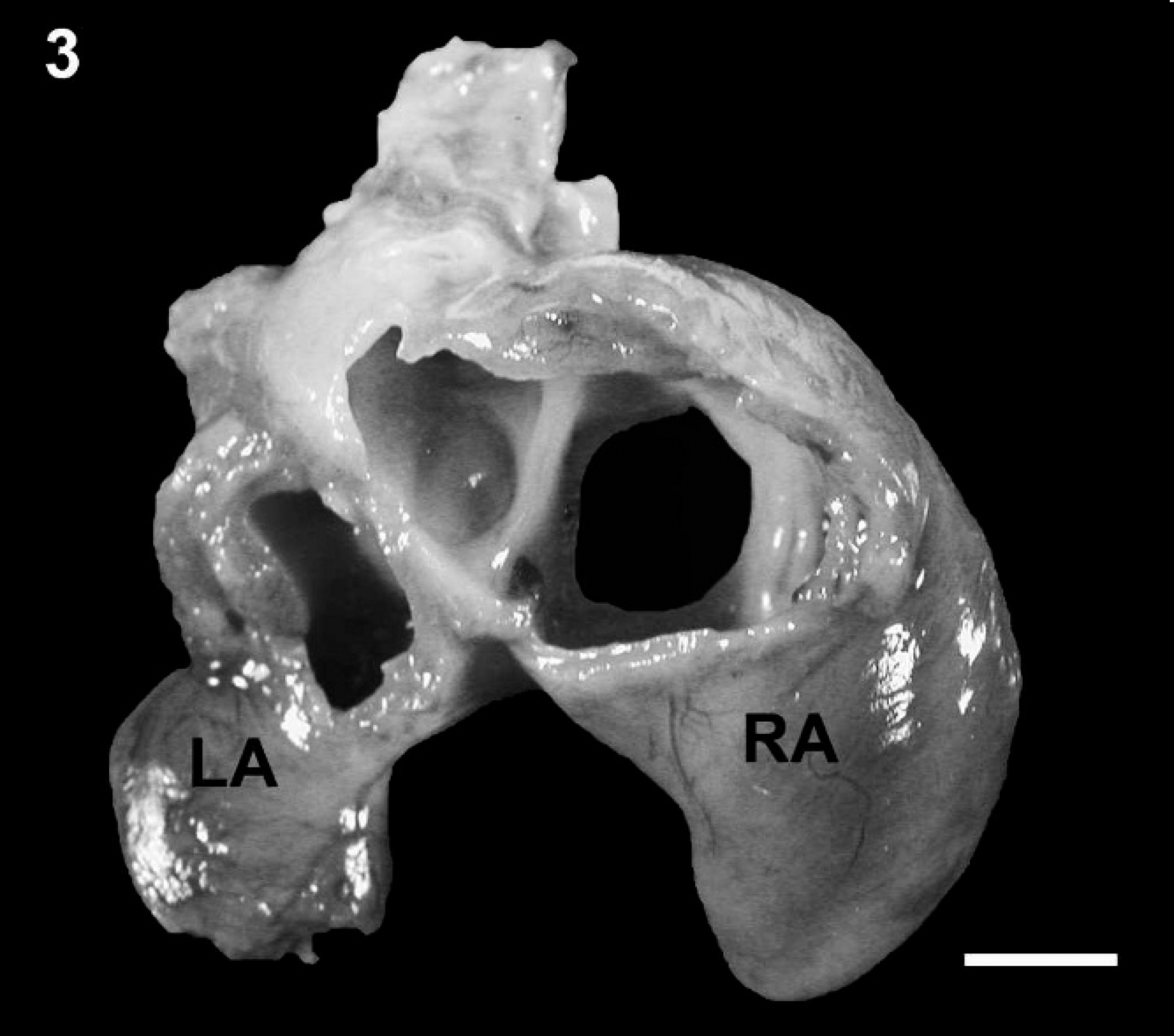
Left and right atria removed from the heart at the atrioventricular annulus: ventrodorsal view. The right atrium and auricle are markedly dilated compared with the left atrium and auricle. LA = left atrium; RA = right atrium. Bar = 5 mm.
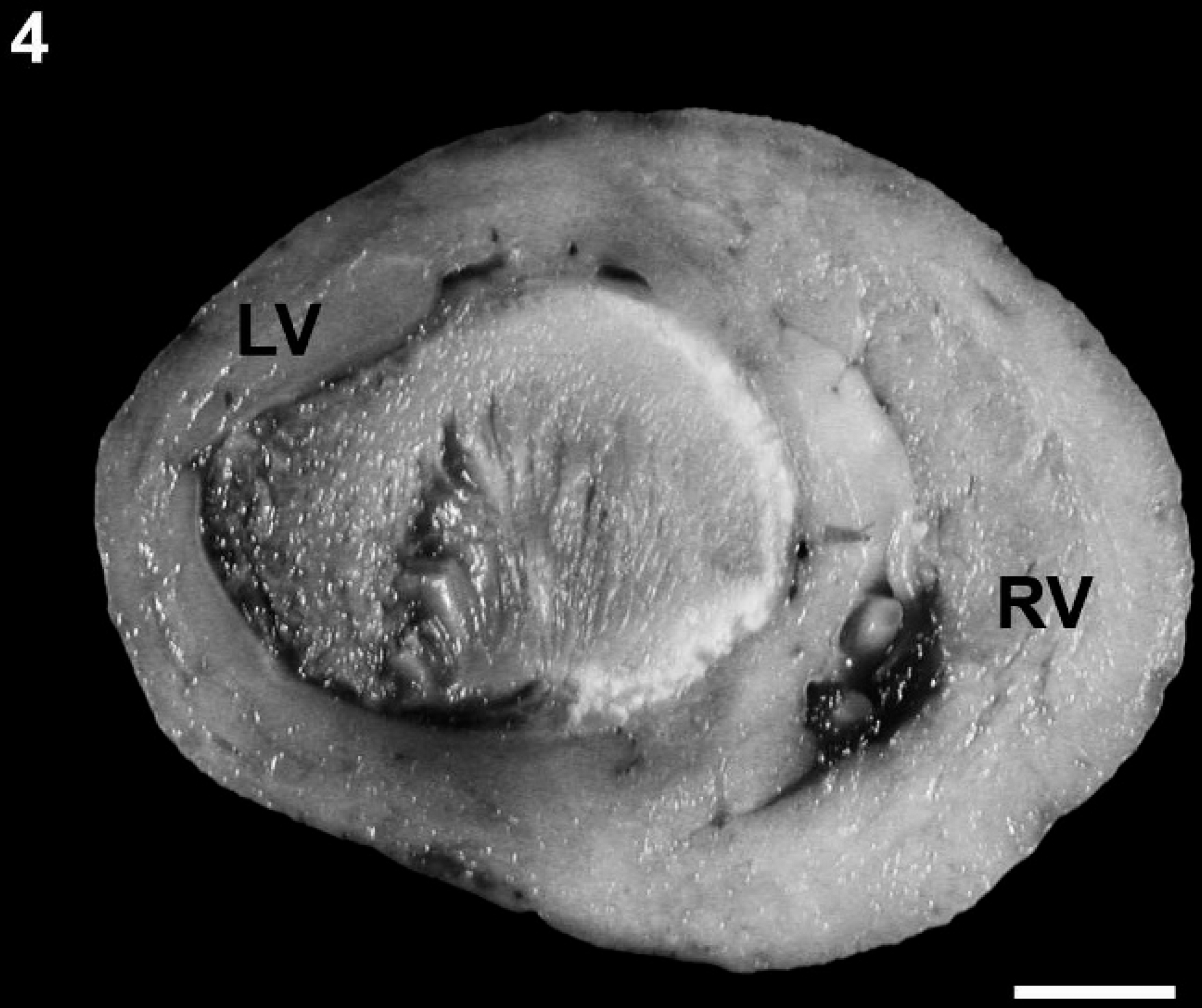
Left ventricular thrombus. Cross-section of the left and right ventricles showing the left ventricle filled with a mature thrombus occluding the lumen. RV = right ventricular free wall; LV = left ventricular free wall. Bar = 1 cm.
The left ventricular thrombus was attached to greater than 90% of the endocardium, and the periphery of the thrombus was mineralized and surrounded by lymphocytes, macrophages, multinucleated giant cells, and extensive fibrosis. The thrombus was extensively recanalized (Fig. 6).
Sections of the heart were exposed to 3% hydrogen peroxide to quench endogenous peroxidases before being incubated with a primary mouse anti-Canine parvovirus 1 and 2 antibody, a followed by incubation with a secondary goat anti-mouse antibody conjugated to horseradish peroxidase. b Sites of primary antibody binding were localized with diaminobenzidine, and the tissue sections were counterstained with hematoxylin. Neither tissue section showed any positive immunostaining for either virus. Electron microscopic evaluation of a multiple tissue homogenate (heart, spleen, kidney, brain) did not reveal any viral particles. Additionally, polymerase chain reaction on formalin-fixed, paraffin-embedded heart samples was also negative for Canine parvovirus 1 and 2. c Aerobic bacteriological culture of heart, lungs, liver, and spleen produced no significant growth. A tissue homogenate (heart, spleen, and kidney) was incubated in tissue culture for virus isolation (optimized for Canid herpesvirus 1), but the culture did not yield any virus growth.
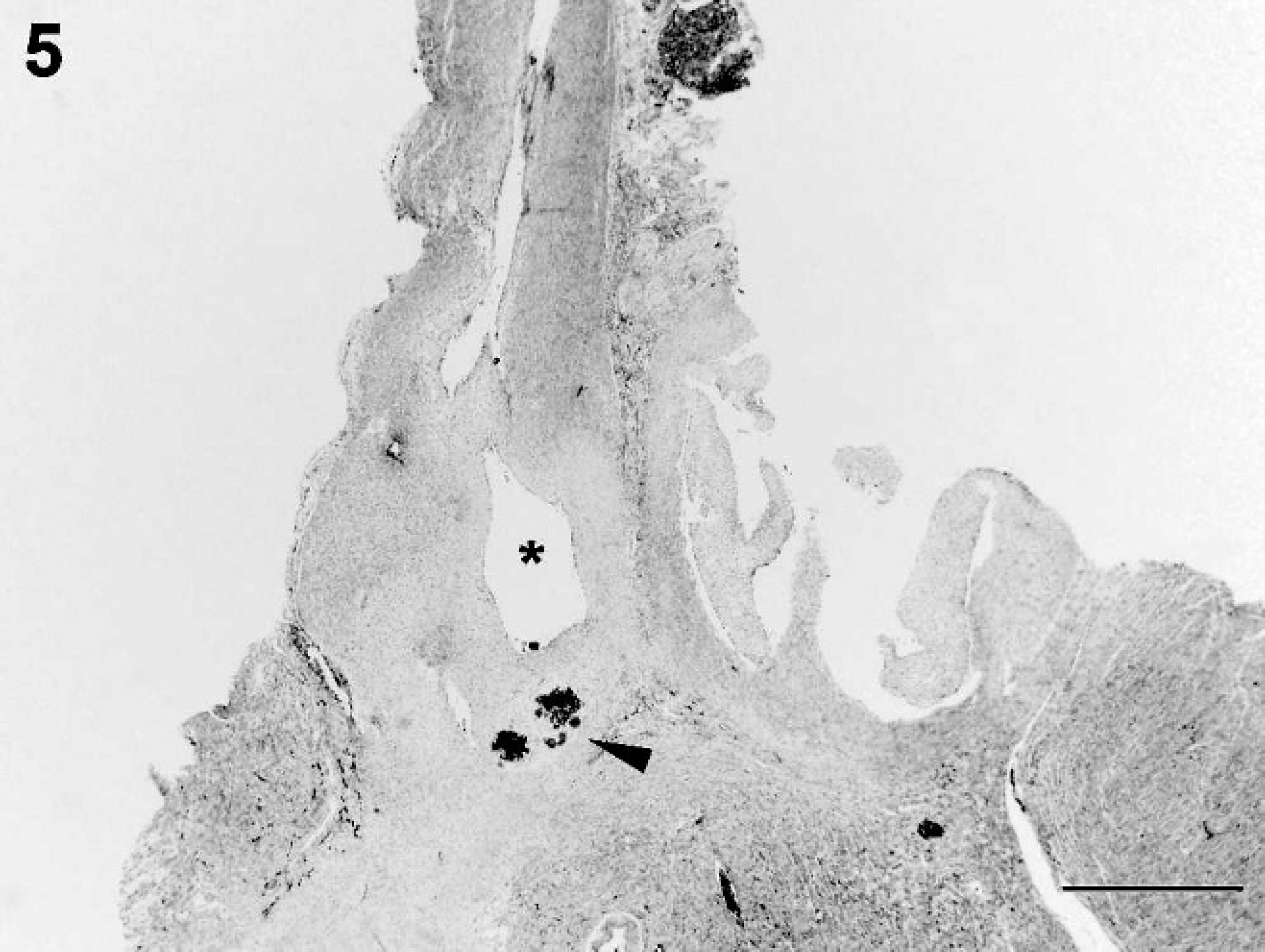
Aortic outflow tract. The aorta is shown in the left outflow tract with a markedly narrowed lumen, absence of the aortic semilunar valves (asterisk), and obstruction by chondroid metaplasia (arrowhead) at the level of the aortic semilunar valves with multifocal mineralized areas. Hematoxylin and eosin. Bar = 1 mm.
Hypoplasia of the aorta in dogs is extremely rare and has only been described twice in dogs. 10,14 Coarctation of the aorta in dogs has also been described twice. 8,11 The 2 case reports with hypoplasia of the aorta were part of multiple abnormalities occurring in conjunction with a PDA 14 or with persistent right aortic arch and PDA. 10
The present case report describes tubular aortic hypoplasia, left ventricular thrombosis, and right atrioventricular valve dysplasia in a Bulldog puppy, and although this breed is predisposed to congenital cardiac malformations such as tetralogy of Fallot, subaortic stenosis, and pulmonic stenosis, 1,2,12 hypoplasia of the aorta has never been reported in this breed to the authors' knowledge. Hypoplasia of the aorta in humans is a rare condition that has been recognized since the mid-18th century. 3 Aortic hypoplasia in humans usually occurs in combination with other cardiac anomalies and usually manifests clinical disease in infants presumably because of the severity of the anomalies. The nomenclature associated with this condition has varied, with one author suggesting the term “aortic hypoplasia” be restricted to cases where the entire length of the thoracic and abdominal aorta is hypoplastic. 3 However, modern terminology generally classifies hypoplasia into 3 categories: localized stenosis, which can manifest as either aortic valvular or annular stenosis 13 ; coarctation of the aorta, in which the ascending and transverse aorta are affected with a localized short segment of hypoplasia 15 ; and tubular hypoplasia, which can range from complete hypoplasia of the ascending, transverse, and descending aorta to the more commonly described tubular hypoplasia of the abdominal aorta. 3 The hypoplasia is sometimes followed by aneurysm of the aorta either within or adjacent to the hypoplastic segment. The underlying cause for this range of congenital aortic hypoplastic conditions is unknown in both humans and dogs.
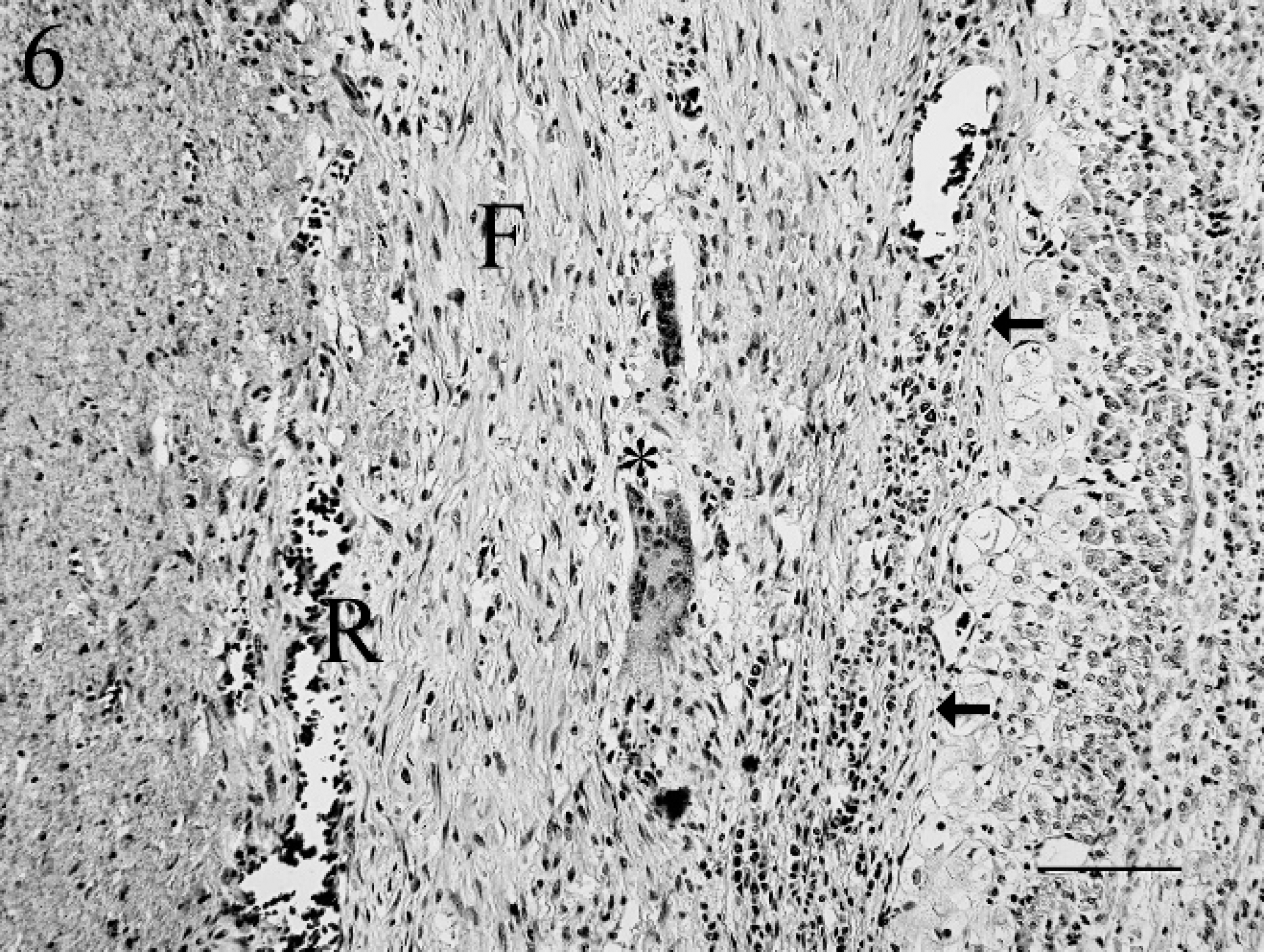
Left ventricular thrombus (to the left of the arrows) showing recanalization (R), fibrosis (F), and multinucleated giant cells (asterisk). Hematoxylin and eosin. Bar = 100 μm.
The death of the puppy in the current report within the first 24 hr of life likely relates to profound changes in blood flow and pressures that occur at birth. The ductus arteriosus permits flow of oxygenated blood from the pulmonary artery to the aorta in utero, which normally reverses shortly after birth, stimulating the closure of the ductus arteriosus and thereby placing a much higher pressure and volume load on the left ventricle. This, accompanied by the almost complete occlusion of the left ventricular outflow tract including the aortic root and aortic valves by the thrombus and accompanying chondroid metaplasia, is likely to have caused further impedance of flow, turbulence, and stagnation of blood. Although not actually demonstrable, death was probably the result of minimal systemic circulation and hypoxia because of the continuation of a right to left shunt through the ductus arteriosus.
Although the dilation of the right atrium was likely secondary to the stenotic right atrioventricular valve, it is unclear to what extent the stenotic right atrioventricular valve contributed to restriction of blood flow to the lungs after birth (this is presuming prior to birth that blood flow was primarily diverted from the right atrium to the left via the foramen ovale, bypassing the stenotic right atrioventricular valve).
The maturity of the left ventricular thrombus indicates that it formed days to weeks prior to birth and was probably the result of prenatal turbulent blood flow associated with the aortic hypoplasia. 16 Right atrioventricular valve stenosis is not an uncommon cardiac malformation in dogs 9 ; however, any possible relationship to aortic hypoplasia in dogs is unknown.
Acknowledgements
The authors would like to thank Prof. Wayne Robinson for his critical review of the manuscript.
Footnotes
a.
Custom Monoclonals International, W. Sacramento, CA.
b.
Lucy Whittier Molecular and Diagnostic Core Facility, Davis, CA.
c.
Dako North America Inc., Carpinteria, CA.
