Abstract
Porcine teschovirus (PTV) was isolated in cell culture and/or demonstrated by polymerase chain reaction in samples of brain and/or spinal cord in pigs in Indiana during the 2002–2007 period. Testing was initiated on pigs originating from populations exhibiting nervous clinical disease and/or pigs with microscopic lesions in central nervous tissues, indicating viral encephalitis and/or myelitis. Virus was demonstrated in pigs with and without lesions as well as with and without nervous clinical disease. Nucleotide sequence analysis of the 5′-nontranslated region of the viral genome revealed that these isolates had low-level genetic heterogeneity but were homologous to porcine PTV serotype 1 (PTV-1). These findings indicate that low-to-moderate virulence strains of PTV with some homology to PTV-1 are endemic in many swineherds of Indiana and are associated with subclinical and clinical nervous disease in weaned pigs.
Porcine teschovirus (PTV; order Picornavirales, family Picornaviridae, genus Teschovirus) is a nonenveloped, positive-sense, single-stranded RNA virus. It was previously recognized as group I Porcine enterovirus (PEV) of the genus Enterovirus but has recently been classified as a separate genus, Teschovirus, to include former PEVs 1–7 and 11–13. 1,2,10 In the new classification, serotypes 8–10 of PEV are retained as species PEV B under the genus Enterovirus. 2,11 Natural infections of PTV and PEV are limited to pigs. Infection is usually acquired through the oro-fecal route and is followed by virus replication in the tonsils and intestinal epithelium. Infection with virulent strains is usually followed by viremic spread of the virus to the central nervous system (CNS), resulting in neurological disease. 2 Porcine teschovirus serotypes 1–3 and 5 have been associated with polioencephalomyelitis in pigs. 8,9 Highly virulent strains of PTV-1 cause a severe and often fatal polioencephalomyelitis (Teschen disease), which is largely confined to central Europe and parts of Africa and has never been reported in North America. A milder CNS disease called Talfan disease, or benign enzootic paresis, associated with infection by less-virulent strains of PTV-1, is sporadically observed in Western Europe, North America, and Australia. 2 These less-virulent strains of PTV are widespread in conventional swineherds and are believed to be maintained predominantly in weaned pigs, which are infected shortly after weaning, when maternal antibodies wane and pigs from multiple groups are mixed. 2,7 Talfan disease is characterized by low morbidity and mortality. Nursery-age pigs are affected more commonly than older, growing pigs. Rarely, the disease progresses to overt clinical signs, complete paralysis, and death. The factors responsible for variable expression of the disease are not known. Marked differences in neurovirulence among various strains of each serotype have been reported.
Although high-virulence strains of PTV have not been reported in the Western hemisphere, wide variation in the neurovirulence of the reported PTV strains warrants further molecular analysis. These reported strains are difficult to distinguish by conventional serotyping using hyperimmune sera, primarily as a result of high levels of cross-reactivity among various strains. As a result of extensive cross-reactivity of polyclonal sera, serotyping gives little information regarding circulating genotypes of PTV. 10 In the current study, molecular genetic comparison of PTV strains from central nervous tissues of pigs isolated from several sporadic cases of porcine teschoviral polioencephalomyelitis in the state of Indiana from 2002 to 2007 is reported.
From 2002 to 2007, several commercial swineherds from different counties of Indiana reported a 3–5% incidence of neurological disease in pigs approximately 2–3 weeks after weaning. Clinical findings were reported in most pigs and included stiff legs, lateral recumbency, tremors, and paddling movements and in a few cases, paresis or paralysis (Table 1, case nos. 1–6). A few piglets with no reported clinical signs were also presented for routine diagnostic workup and were included in the present study because they belonged to herds with a history of similar neurologic disease. Antibiotic therapy and anti-tetanus toxin or other palliative therapies were attempted in a few earlier cases without much success. The clinical course of the disease was 1–6 days before the pigs were dead or euthanized. Other pigs (Table 1, case nos. 5 and 7) had no neurological signs and were submitted for respiratory or enteric disease investigation. Selected pigs from the affected groups were submitted live or dead for complete necropsy and follow-up diagnostics to the Purdue University Animal Disease Diagnostic Laboratory (West Lafayette, Indiana).
Cases of porcine encephalomyelitis in Indiana (2002–2007).*
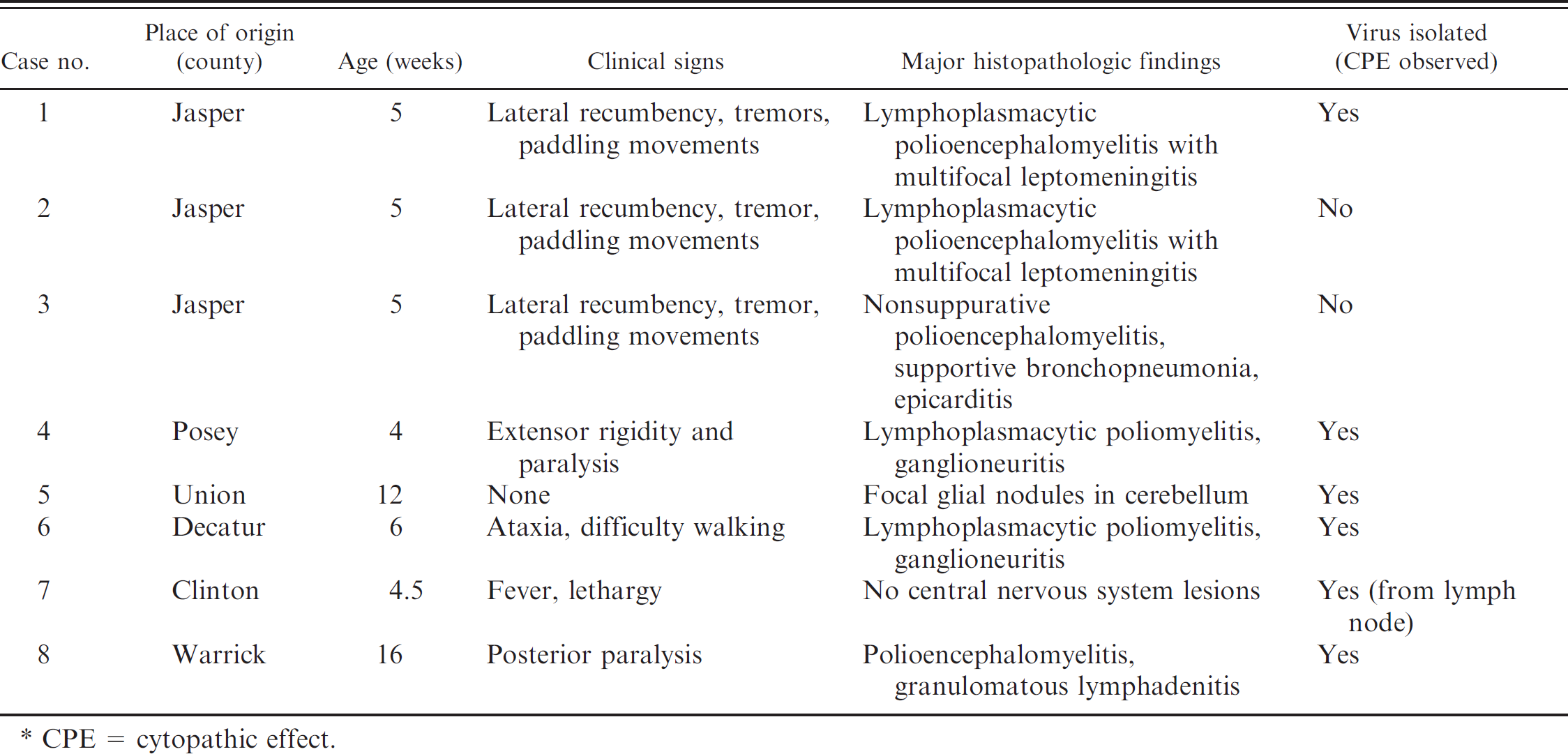
CPE = cytopathic effect.
At necropsy, multiple tissues, including the brain and spinal cord, were collected in 10% neutral buffered formalin. Formalin-fixed, paraffin-embedded tissue sections were routinely processed, sectioned at 4 μm, and stained with hematoxylin and eosin. Selected fresh tissue samples were submitted for virology and bacteriology. Fresh tissue samples were also frozen for molecular diagnostics.
Complete necropsy examinations revealed no specific gross lesions in the brain or spinal cord. In a few cases, respiratory tract and lymphoid tissues had lesions attributable to other non-CNS bacterial or viral infections. Microscopically, multifocal lymphoplasmacytic meningitis, encephalitis, and poliomyelitis were observed in all pigs with neurological signs and in 1 pig with only respiratory signs reported (Table 1, case no. 7). In the pigs with CNS lesions, histologic findings were those of multifocal nonsuppurative polioencephalitis, poliomyelitis, and leptomeningitis (Figs. 1, 2). Spinal cord lesions in pigs with neurological signs were those of a nonsuppurative poliomyelitis associated with neuronal degeneration, perivascular cuffing, and gliosis. Lesions were most severe in the ventral horn gray matter. There were prominent perivascular cuffs of mononuclear inflammatory cells, primarily lymphocytes with a few macrophages in the gray matter. Scattered lymphocytes and macrophages were in the surrounding neuroparenchyma. Intralesional capillaries were lined by swollen endothelial cells. Scattered neurons were degenerated, with central chromatolysis, densely eosinophilic cytoplasm, or vacuolation of the soma. Affected neurons were often surrounded by microglial cells (satellitosis and neuronophagia). Neuronal loss accompanied by nodular glial formation was conspicuous in the sections examined. There was moderate diffuse gliosis. Rare intralesional swollen axons were present. Lesions in the white matter, characterized by mononuclear perivascular cuffing and nodular gliosis, were mild and much less frequent. Lymphocytic infiltrate was also occasionally observed in dorsal and ventral spinal nerve roots from all animals.
Similar lesions were also observed in the brain. Multifocally, cerebral cortical gray matter, nuclei of the brain stem, deep cerebellar nuclei, and thalamus had perivascular cuffs of lymphocytes and randomly distributed gliosis and glial nodules. Mild and multifocal nonsuppurative lepto-meningitis, characterized by mild to moderate mononuclear infiltrate usually associated with vessels, was observed in multiple pigs. Significant histopathologic changes were not observed in 2 pigs (from Clinton and Union counties). Bacterial isolation on brain and meninges from the affected pigs was negative. Concomitant infection with Porcine respiratory and reproductive syndrome virus infection and Porcine circovirus-2 (PCV-2) in 1 case and Hemophilus parasuis infection in the pig with respiratory disease were also diagnosed.
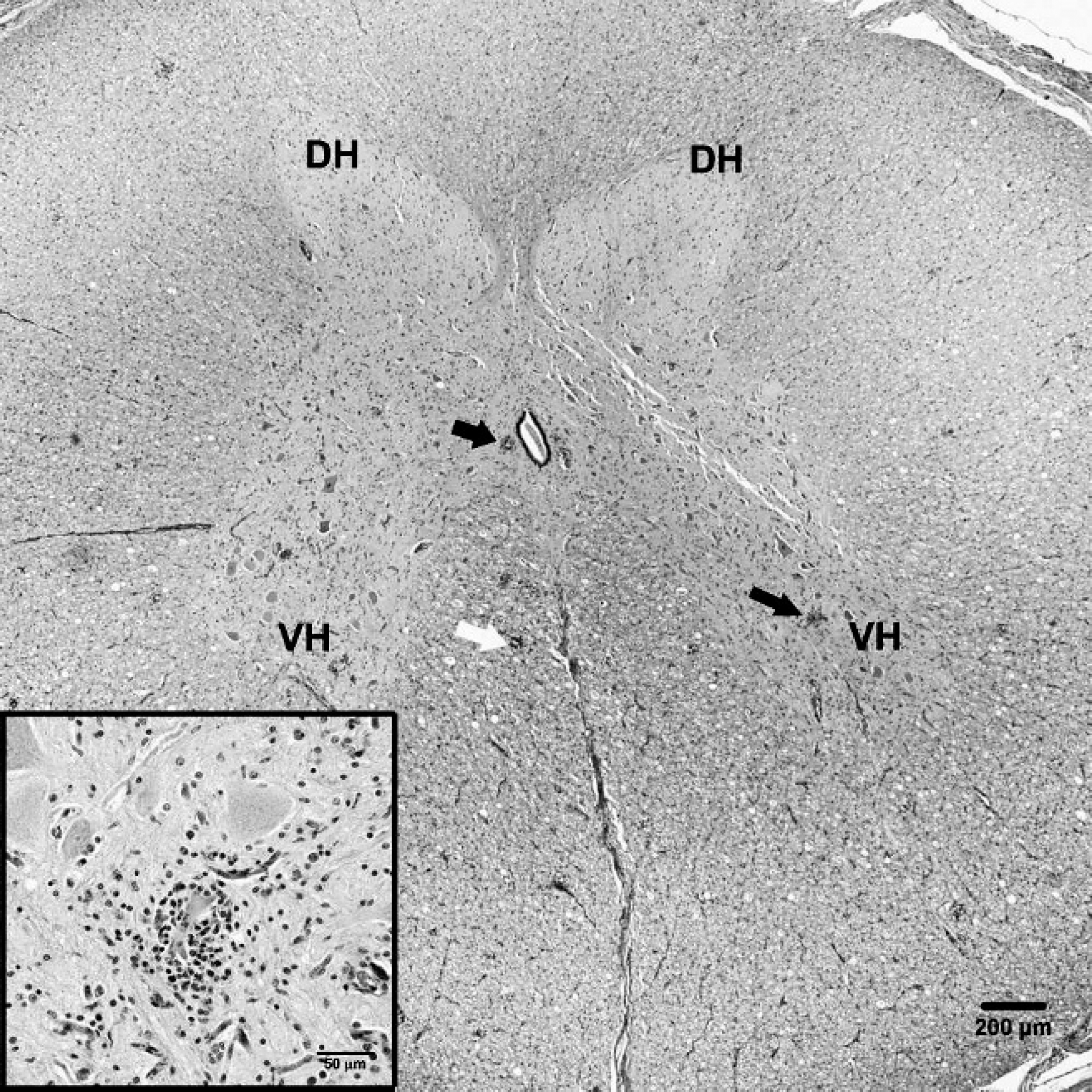
Spinal cord, pig (pig P4.Posey.06). Section of spinal cord showing glial nodules (dark arrows) in the gray matter, most prominent in the ventral horns (VH), with relative sparing of the dorsal horns (DH). Also note the mild gliosis and occasional glial nodules in the white matter (white arrow). Bar = 200 μm. Inset: Higher magnification of gray matter lesion showing neuronophagia and satellitosis. Hematoxylin and eosin. Bar = 50 μm.
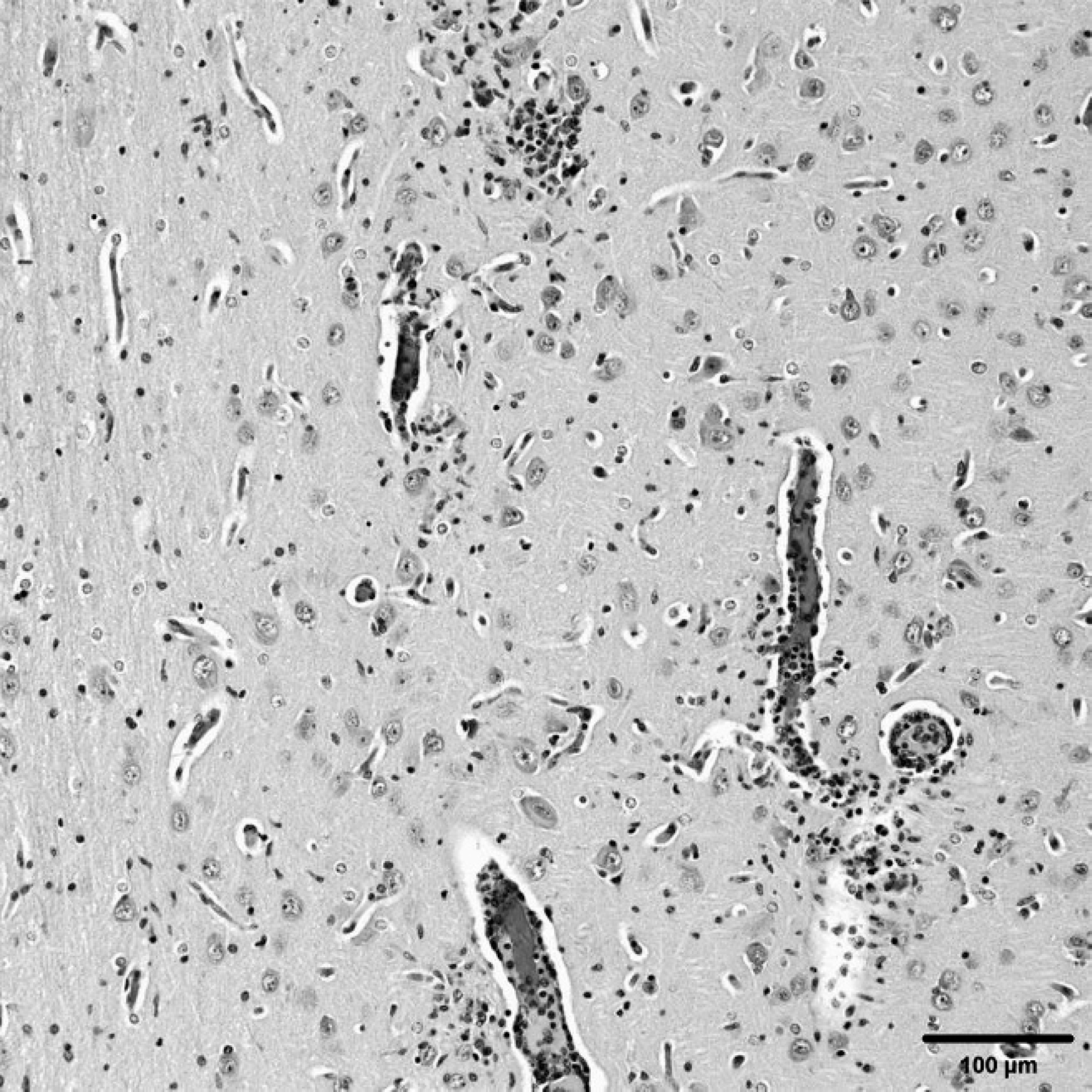
Brain, pig (pig P4.Posey.06). Cerebral cortex showing perivascular infiltration of lymphocytes and plasma cells. Increased numbers of glial cells are in the neuroparenchyma. Hematoxylin and eosin. Bar = 100 μm.
Virus isolation was performed using porcine kidney epithelial (PK-15) cells cultured in 48-well tissue culture plates, as previously described. 7 When cytopathic effect (CPE) was evident, cells were stained with PEV-specific polyclonal antibody from the National Veterinary Service Laboratories (Ames, Iowa). Virus-induced CPE was identified in 4 cases. Subsequently, the presence of PTV was confirmed by diagnostic polymerase chain reaction (PCR) using primers specific to the 5′-nontranslated region (NTR) of PTV genotypes I–III, as described previously. 11 The PTV-1 genome was detected in tissue homogenate from all 8 cases and in 6 cases from cell culture supernatant when the presence of CPE was evident (Table. 1). Within the PTV genome, 5′ and 3′ NTR are among the most conserved regions and hence have been utilized for molecular phylogenetic studies. 3,10 Real-time PCR assays based on 5′ NTR have previously been described 4,5,6,11 for molecular detection and phylogenetic analysis of PTVs. In the present study, amplified PCR products were sequenced using PTV-1–specific reverse transcription PCR (RT-PCR), as previously described. 11 In brief, 140 μl of the fluid were used for RNA extraction using a commercial viral RNA kit, according to the manufacturer's instructions. a Reverse transcription PCR was carried out using a commercial 1-step RT-PCR kit. a Five microliters of the RNA template were added to 45 μl of the RT-PCR master mix. Polymerase chain reaction was performed using a set of primers corresponding to the conserved region at 5′ NTR and cycling conditions, as previously described. 11
The amplified PCR products incorporating the 321–base pair region of 5′ NTR were purified using a commercial purification kit, according to the manufacturer's protocol. a The products were sequenced using an automated sequencer at the Purdue University Genomic Core Facility (West Lafayette, IN), analyzed, and their homology to other PTV strains determined based on published sequence information of reference viruses. a Analysis was done using MegAlign ClustalW method applications of Lasergene software. b A phylogenetic tree was generated (Fig. 3) to demonstrate relative homology among the PTVs isolated in the present study and other published PTV sequences, including reference strains of PTV of different genotypes. Even though all teschoviruses that were sequenced belonged to group 1, with small variations on 5′ NTR of viral genome, it was clear that clinical signs, outcomes of disease, and lesions were similar.
The cases of porcine teschoviral polioencephalomyelitis present in the current study were unrelated to each other and indicate the enzootic presence of PTV in conventional and commercial swineherds. Clinical signs related to the PTV infection were observed in most cases, while PTV identification in a few cases was unrelated to the clinical history and was not associated with gross or histopathologic alteration. For example, for case no. 7 (Clinton County), PTV was isolated from lymph nodes, but the pigs had no neurological clinical signs. Although the brain was the only nervous tissue examined, and although it was found to have no significant histologic changes, fecal contamination of tissue samples collected for virus isolation cannot be completely ruled out. Nonetheless, positive virus identification in such cases indicates the endemic presence of PTV. Molecular genetic analysis (Fig. 3) of virus isolates indicated a low level of genetic heterogeneity, typical of picornaviruses. Whether these genetic differences lead to serotypic and/or antigenic variation requires further investigation. Phylogenetic comparisons in the current study were performed based on the nucleotide sequence of the 5′ NTR region of PTV. It remains to be investigated if molecular genetic analysis based on the nucleotide sequence from other regions of the genome or the entire PTV genome would indicate similar or different phylogenetic relationships.
The enzootic presence of PTV strains in commercial swineherds may have important implications, especially in herds in which PCV-associated disease and other immuno-suppressive virus infections are prevalent. Humoral defense mechanisms are important in preventing PTV infection, and it has recently been suggested 2,8 that immunosuppression resulting from PCV-2 may lead to clinical porcine teschoviral CNS disease. While most PTV strains are mildly pathogenic and rarely cause CNS disease, endemic PCV-2 infections in Indiana swineherds may predispose piglets to clinical PTV infections. Commercial vaccination is not available for PTV. On-farm vaccination with autogenous vaccine is currently practiced at a few farms but has met with variable results. This may be due to multiple concurrent strains of PTV infecting herds. The molecular genetic information reported herein will be helpful in further understanding the molecular epidemiologic attributes of PTV in swineherds. Such information will be vital to devising effective prevention and control strategies to prevent introduction and/or circulation of PTV strains within and among different herds.
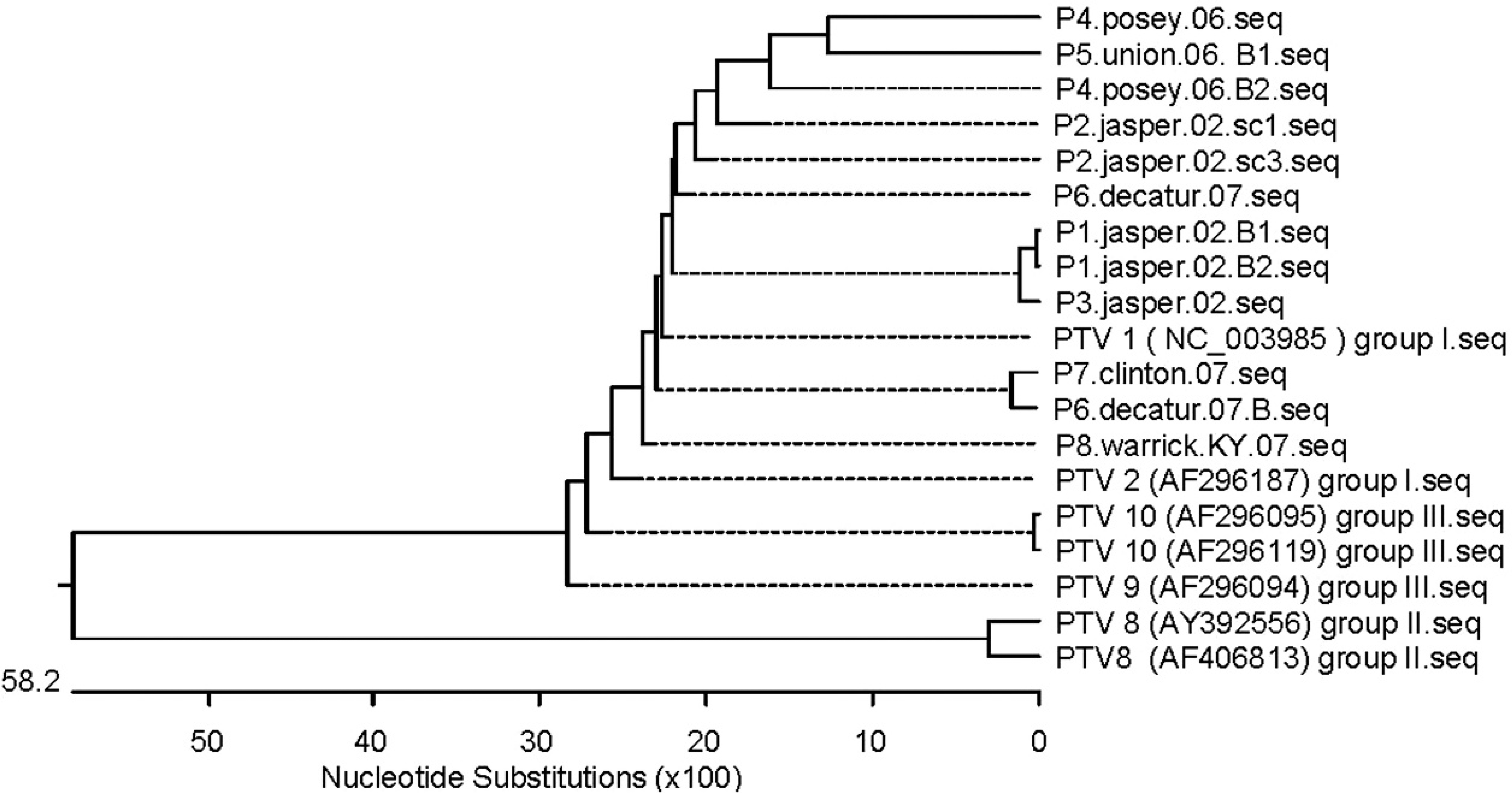
Phylogenetic tree of Indiana isolates of Porcine teschovirus (PTV) based on the sequence of 5′-nontranslated region.
Acknowledgements
The authors wish to thank all veterinary practitioners who submitted these cases and provided additional information. The authors also would like to thank the staff members of the virology and histology laboratories of the Purdue University Animal Disease Diagnostic Laboratory for their assistance.
Footnotes
a.
Qiagen Inc., Valencia, CA.
b.
DNASTAR Inc., Madison, WI.
