Abstract
Beginning in 2002, a small number of pig farms in western Canada began reporting 4–7-week-old pigs with bilateral hind-end paresis or paralysis. Low numbers of pigs were affected, some died, most had to be euthanized, and those that survived had reduced weight gains and neurological deficits. Necropsies revealed no gross lesions, but microscopic lesions consisted of a nonsuppurative polioencephalomyelitis, most severe in the brain stem and spinal cord. The lesions were most consistent with a viral infection. Tests for circovirus, Porcine reproductive and respiratory syndrome virus, coronavirus, Rabies virus, and Pseudorabies virus were negative. Using immunohistochemistry, virus neutralization, fluorescent antibody test, and nested reverse transcription polymerase chain reaction, Porcine teschovirus was identified in tissues from affected individuals. To the authors' knowledge, this is the first report of teschovirus encephalitis in western Canada and the first reported case of polioencephalomyelitis in pigs in Canada, where teschovirus was confirmed as the cause.
Teschovirus encephalomyelitis, also known as Teschen and/or Talfan disease, was first reported in Teschen, Czechoslovakia, in 1929. Teschen disease was very severe, with high morbidity and mortality in pigs of all ages. The disease spread throughout Europe, causing devastating economic losses. 23 Twenty years later, a milder form of this disease was reported in Talfan, Wales. 11 Since then, a moderate form of the disease, with lower morbidity and mortality, has been described in the United States, 17 Canada (unconfirmed), 2 Europe, Africa, Australia, 7 China, and Japan. 20 Typically, these infections are asymptomatic, with virus being isolated from the feces of healthy pigs. 7 Teschovirus has also been associated with abortion, mummification, and stillbirth. 14 A severe outbreak of teschovirus encephalomyelitis with 30–40% morbidity and 40–50% mortality has been occurring in Haiti since January 2009 and is threatening to spread to neighboring countries such as the Dominican Republic (Food and Agriculture Organization of the United Nations: 2009, Teschovirus encephalomyelitis in the Republic of Haiti. Available at: ftp://ftp.fao.org/docrep/fao/012/ak137e/ak137e00.pdf. Accessed October 8, 2010).
Teschovirus was previously classified as Porcine enterovirus (PEV) of the Picornaviridae family and was divided into 3 groups: PEV−1 to −7 and PEV−11 to −13 (group I), PEV−8 (group II), and PEV−9 and −10 (group III). More recently, group I was classified as a separate genus, Teschovirus, of the Picornaviridae family. This genus consists of 11 types of the sole species Porcine teschovirus (PTV) serotypes 1–11. 14 All 11 serotypes, except for type 8, which is nonpathogenic, can be either pathogenic or nonpathogenic. Only PTV−1 has been found to be highly pathogenic (Teschen virus from 1929 outbreak up to mid-1950). Recent outbreaks, except for the one in Haiti, are moderate.
In the current study, the cause of polioencephalomyelitis outbreaks in pigs that occurred in Saskatchewan and Manitoba (Canada) between 2002 and 2008 was investigated. Outbreaks involved approximately 400 pigs from 6 different farms (3 different producers), varying in size from 4,000 to 24,000 animals. Affected pigs were 4–7 weeks old. According to the practitioner, clinical signs were similar in all farms, beginning with ataxia of the hind limbs, which progressed to paralysis in 24 hr. Animals were alert and maintained normal appetite, body condition, and temperature. Vomiting, diarrhea, respiratory distress, and reduced vocalization were occasionally present. Affected animals were frequently bullied by other healthy pigs and, as a result, went off their feed. According to owners, the morbidity varied from 0.1% to 2%, and case-fatality rates varied from 0.02% to 100% of the sick animals (Table 1); however, surviving animals remained unthrifty, and most were euthanized because of persistent neurological deficits and reduced weight gain. In one of the farms (farm 2), an outbreak of Porcine reproductive and respiratory syndrome virus (PRRSV) and Porcine circovirus-2 (PCV-2) occurred in one year.
Epidemiological data and tests performed in pigs with polioencephalomyelitis in Canada.*
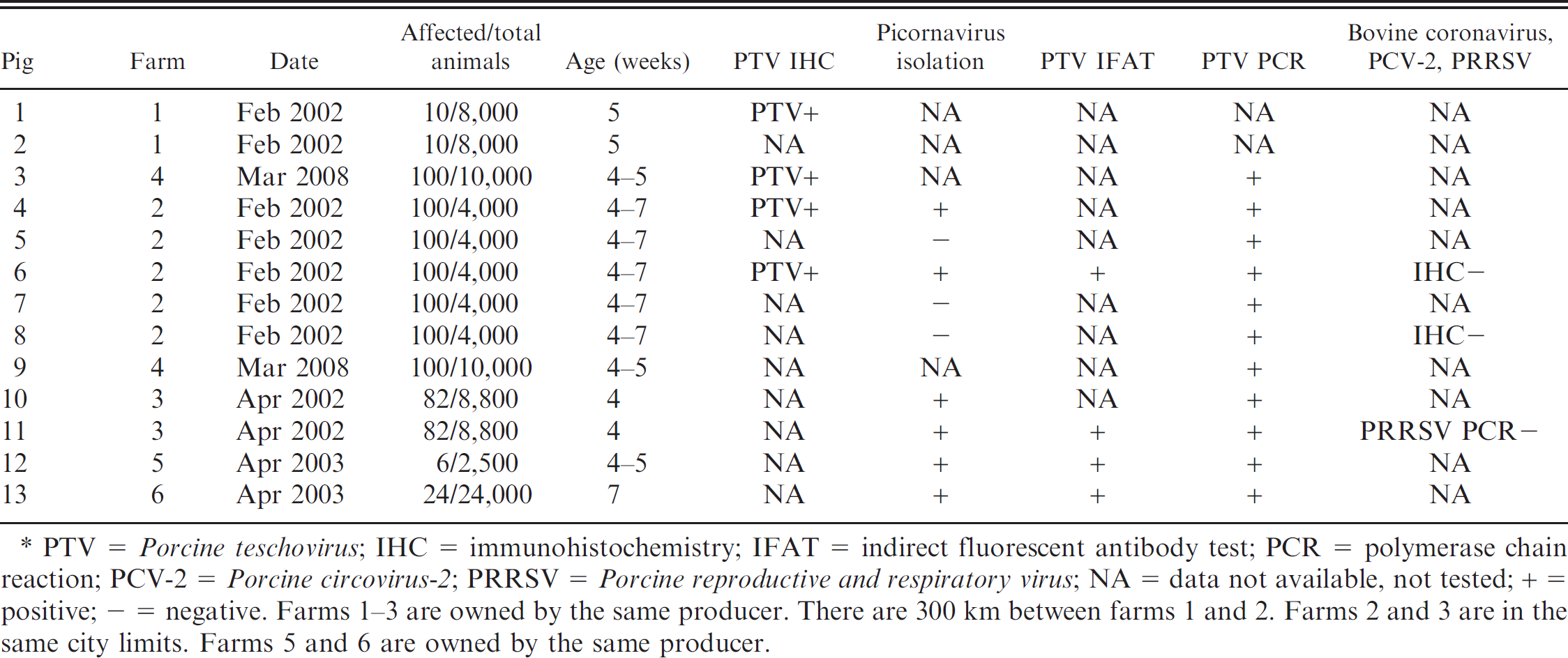
PTV = Porcine teschovirus; IHC = immunohistochemistry; IFAT = indirect fluorescent antibody test; PCR = polymerase chain reaction; PCV-2 = Porcine circovirus-2; PRRSV = Porcine reproductive and respiratory virus; NA = data not available, not tested; + = positive; - = negative. Farms 1–3 are owned by the same producer. There are 300 km between farms 1 and 2. Farms 2 and 3 are in the same city limits. Farms 5 and 6 are owned by the same producer.
Necropsies were performed on 13 animals at the Prairie Diagnostic Services at the University of Saskatchewan (Saskatoon, Saskatchewan, Canada), and tissues were processed routinely for histopathology. Grossly, the only findings were enlarged mesenteric lymph nodes and hyperkeratosis of the pars esophagea in a few animals. Microscopically, lesions were confined primarily to gray matter of the brain stem and spinal cord. Lesions consisted of multifocal gliosis and perivascular cuffs composed primarily of macrophages with fewer lymphocytes, plasma cells, and rare eosinophils; satellitosis, chromatolysis, neuronal necrosis, and neuronophagia were often observed (Fig. 1). In the spinal cord, lesions were bilateral, asymmetric, and most severe in the ventral horns. Infrequently, cuffs and gliotic foci were seen in the white matter along with occasional small foci of axonal degeneration and phagocytic vacuoles. Lesions were more common in the cerebellum than the cerebrum and typically involved the molecular layer. Microhemorrhages occurred sporadically. Meninges overlying the lesions were mildly infiltrated by macrophages and lymphocytes. In the few spinal ganglia examined, there were infiltrates of macrophages and lymphocytes around neurons.
Immunohistochemistry (IHC) for PRRSV (monoclonal antibody SDOW 17), a PCV-2, b and Bovine coronavirus c were performed on 2 pigs. Immunohistochemistry was not performed in the other pigs due to financial restrictions. The IHC technique was based on techniques described elsewhere. 10 The following tissues were used for PRRSV and Bovine coronavirus IHC: spinal cord, skeletal muscle, kidney, lung, lymph node, and tonsil. For PCV-2 IHC, spinal cord, kidney, tonsil, and lymph node were tested. All staining was negative. A cytopathic agent was isolated from PCV-free porcine kidney epithelial (PK-15) cells infected with suspensions of pooled tissues (from pigs 4, 6, 10, and 11) and brain alone (from pigs 12 and 13). The isolate was agglutinated with convalescent serum from a herd mate and was observed with electron microscopy. Viral particles with picornavirus morphology 14 measuring 29 nm were observed. Unfortunately, the serum also contained viral particles that compromised the interpretation of the results. In pig 11, polymerase chain reaction (PCR) using PRRSV primers 8 was performed in the cell culture (PK-15 cells) that was used for picornavirus isolation. The PCR was also negative. Pigs from the same farm as pigs 3 and 9 were necropsied and tested for rabies by IHC, which was negative. Brain samples from these same pigs were submitted to the Canadian Food Inspection Agency for pseudorabies testing, which was also negative. There were no significant findings on bacteriological examination.
Immunohistochemistry for PTV was performed on selected central nervous system sections from pigs 1, 3, 4, and 6. Following heat-mediated antigen retrieval in citrate buffer (pH 6.0) at 97°C for 20 min, a rabbit polyclonal antiserum raised against PTV-1 (VIR 2236/99) 25 was applied to the sections. Sites of primary antibody binding were detected using a polymer-based peroxidase system. d Specific labeling for PTV was observed in all 4 pigs examined. Intense diffuse to granular labeling of scattered neuronal cell bodies and processes was present in areas of nonsuppurative inflammation (Fig. 2A). Intense punctuate labeling associated with neuronophagic nodules was observed in all immunolabeled sections (Fig. 2B).
To characterize the viruses isolated from the pigs with polioencephalomyelitis, PTV serotype–specific hyperimmune sera against the 11 PTV reference strains were used in a neutralization assay according to a technique described elsewhere. 16 Ten-fold dilutions of PTV isolates from pigs 6, 11, 12, and 13 were incubated with hyperimmune serum followed by incubation in swine testis (ST) cells (CCLV RIE-606) and evaluation of cytopathic effects. A virus titer reduction of greater than 3 log10 was considered specific. Only pig 13 did not react with any of the 11 hyperimmune sera. The observed titers demonstrated antigenic differences between the 4 isolates but did not allow classification of the virus isolates to any of the 11 PTV strains (Table 2).
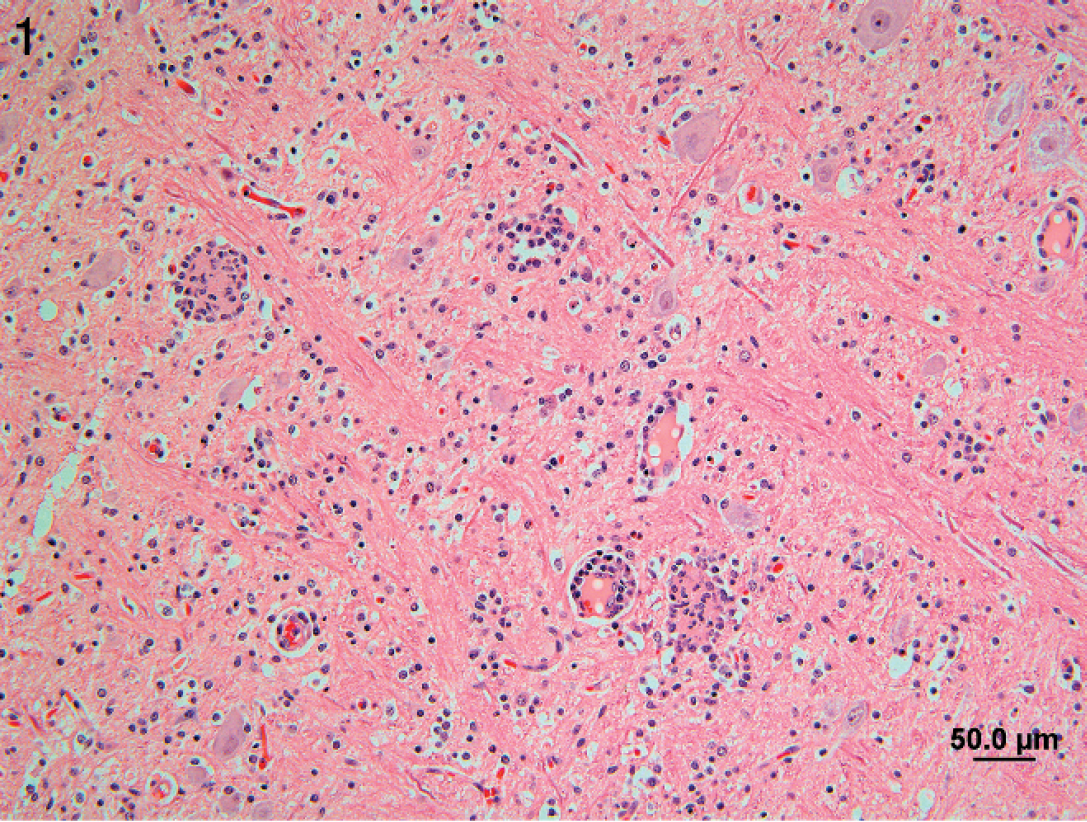
Brain stem of pig 12 with Porcine teschovirus polioencephalomyelitis showing neuronophagia and perivascular mononuclear cell cuffing. Hematoxylin and eosin. Bar = 50 μm.
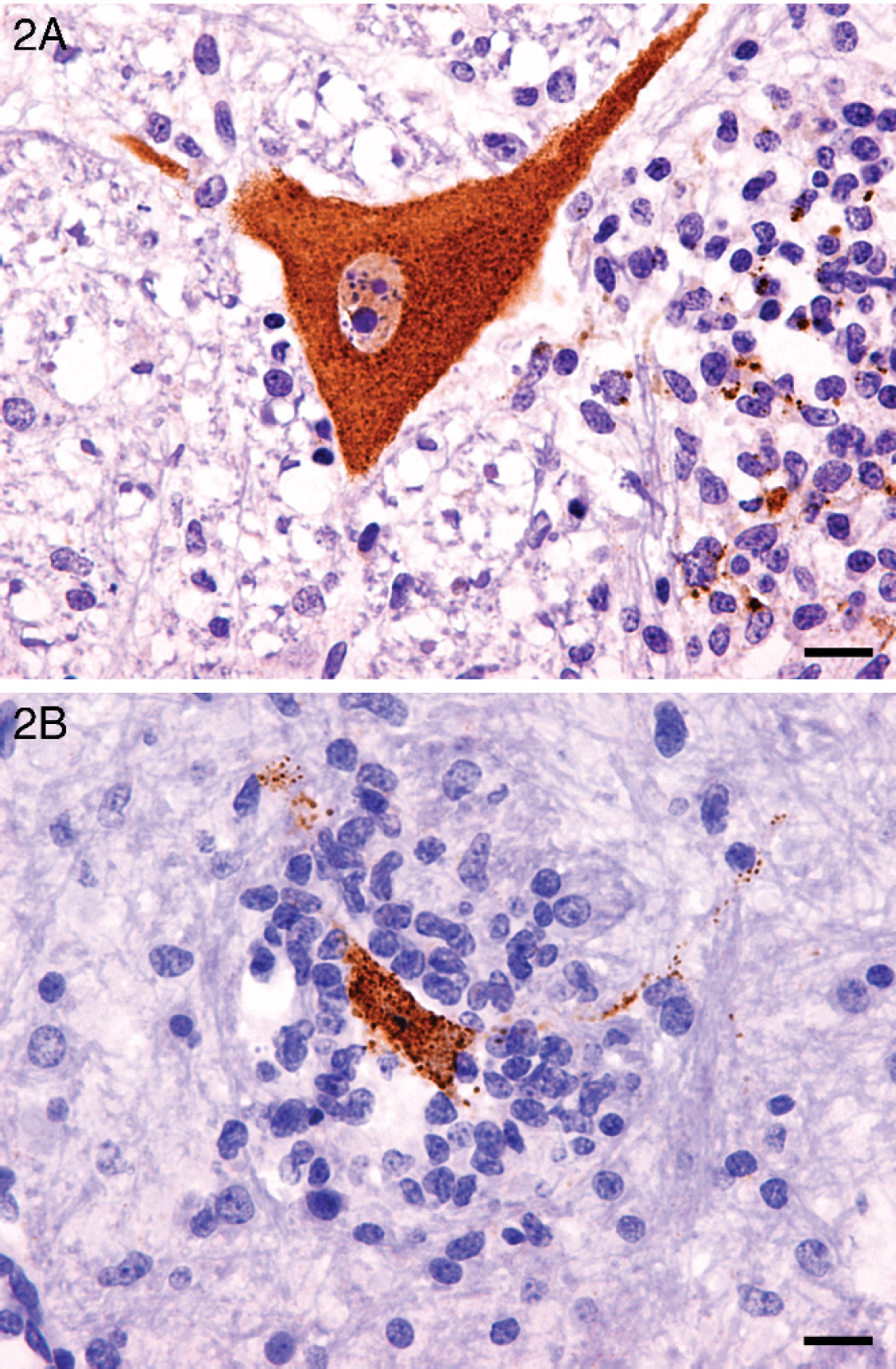
Rostral medulla, pig. A, diffuse labeling of cytoplasm of a large nerve cell body and processes, together with punctate labeling associated with an adjacent glial nodule. Peroxidase immunohistochemistry using rabbit antiserum raised against porcine teschovirus. Bar = 50 μm. B, punctuated labeling associated with a large glial nodule. Peroxidase immunohistochemistry using rabbit antiserum against porcine teschovirus. Bar = 25 μm.
Titer reduction by neutralizing hyperimmune sera (log10).
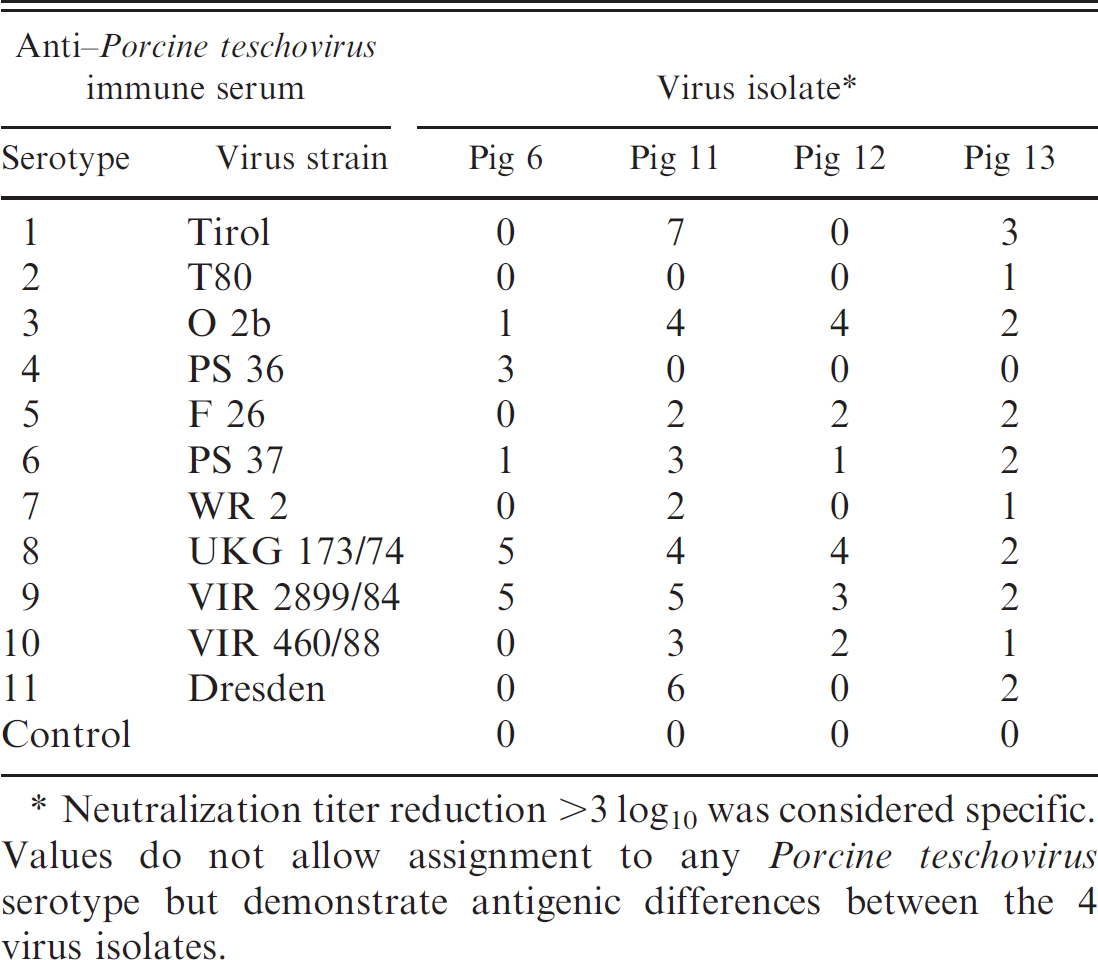
Neutralization titer reduction >3 log10 was considered specific. Values do not allow assignment to any Porcine teschovirus serotype but demonstrate antigenic differences between the 4 virus isolates.
Using monoclonal antibodies generated against the 11 PTV strains 25 as primary antibodies, indirect fluorescent antibody test (IFAT) was carried out in ST cells infected with virus isolates from pigs 6, 11, 12, and 13. Indocarbocyanine e or fluorescent dye f conjugated goat anti-mouse immunoglobulin G antibody was used as secondary antibody. All virus isolates from the 4 pigs reacted with anti-PTV species-specific monoclonal antibodies 040/4B1 and 156/1B9. Virus isolates from pigs 11 and 13 were additionally detected by PTV-1–specific monoclonal antibody 158/5D2 alone (Table 3).
Nested reverse transcription (RT)-PCR was performed to verify and characterize the PTV genome. 25,26 RNA was extracted from spinal cord, brain, or infected PK-15 cell cultures of 12 animals, including a negative probe of a healthy pig using a commercial kit. g Reverse transcription PCR reactions were performed with a commercial kit g and the described primers pev-1a and pev-1e. 14 The reverse transcription reaction was carried out at 50°C for 30 min, followed by an initial PCR activation step at 95°C for 15 min. The PCR reactions were amplified through 35 cycles. Denaturation was carried out at 94°C for 50 sec, followed by primer annealing at 55°C for 50 sec and elongation 72°C for 1 min. Nested PCR was performed using pev-c and pev-d primers. 14 An initial activating step was performed at 94°C for 5 min. Reactions were amplified through 35 cycles. Denaturation was performed at 94°C for 50 sec, primer annealing at 55°C for 50 sec, and elongation at 72°C for 1 min.
The nested RT-PCR as described amplifies sequences of a highly conserved 5'-nontranslated region. Positive reactions confirmed the existence of teschovirus-specific genome in probes of all 11 pigs with polioencephalomyelitis (Fig. 3). The product obtained from pigs 3 and 10 was isolated and sequenced. The sequences were compared with different types of teschovirus using the search engine BLAST (http://www.ncbi.nlm.nih.gov/blast/Blast.cgi). The sequence obtained from pig 3 had 99% alignment with PTV serotype 1 (Talfan strain) and 97% alignment with the Teschen-Konratice strain. The oligonucleotide sequence from pig 10 had 96% alignment with the Talfan strain and 97% alignment with the Teschen-Konratice strain.
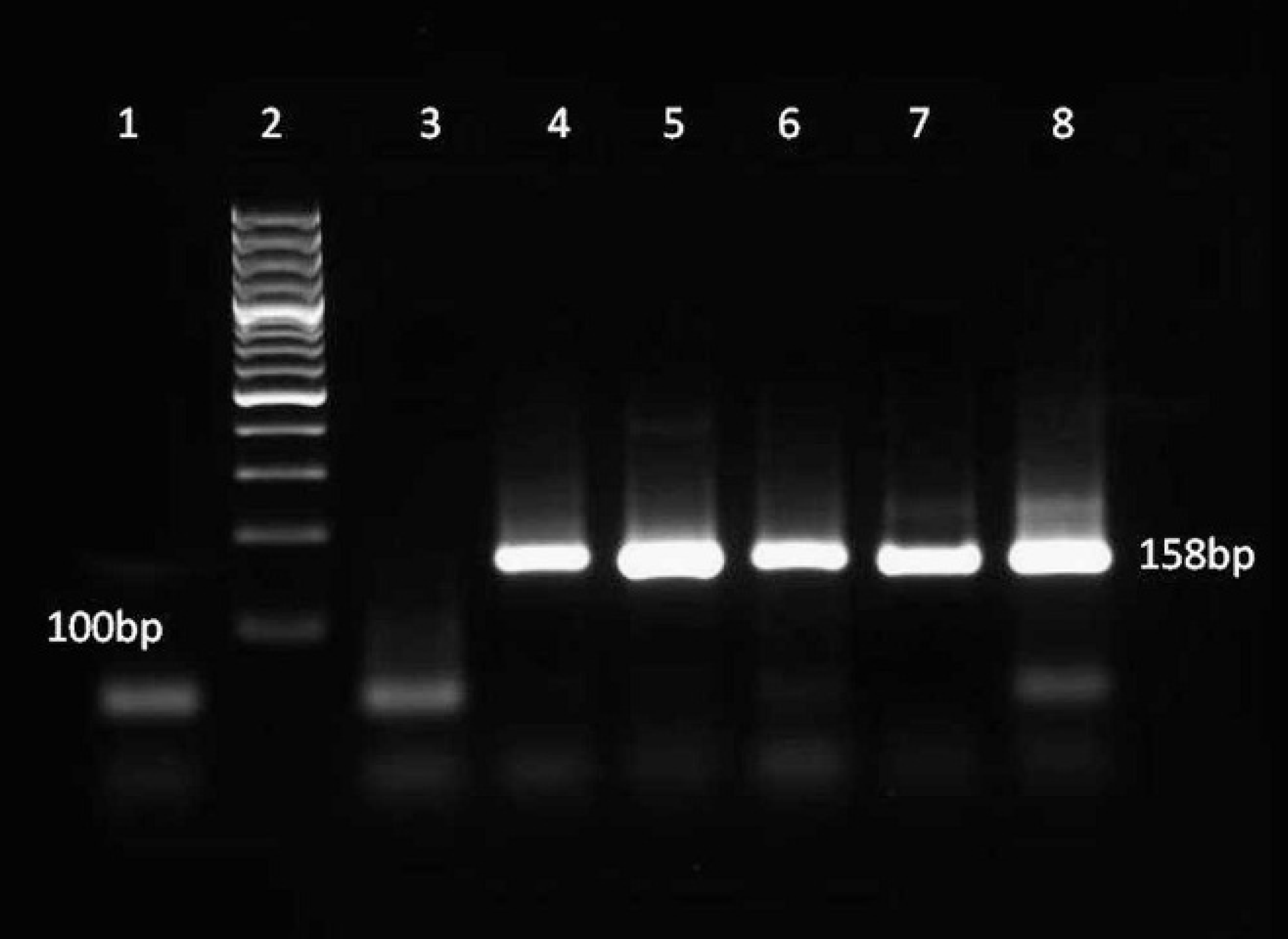
Nested reverse transcription polymerase chain reaction using Porcine teschovirus–specific primers on tissue (lanes 4, 7, 8) or cell culture (lanes 5, 6) templates from 5 pigs with polioencephalomyelitis and 1 healthy pig as negative control (lane 3). Lane 1: negative template; lane 2: 100-bp ladder. All the pigs with polioencephalomyelitis have the expected DNA product of 158 bp, which corresponds to the size of teschovirus sequences when using pev-b and pev-d primers.
Indirect fluorescent antibody reaction of Porcine teschovirus (PTV)-specific monoclonal antibodies with virus isolates from pigs 6, 11, 12, and 13.*
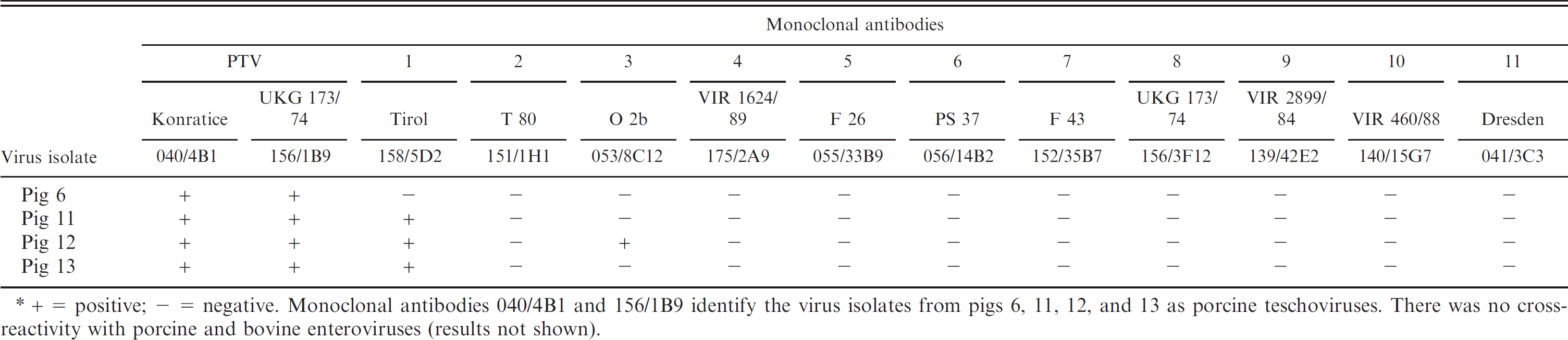
+ = positive; - = negative. Monoclonal antibodies 040/4B1 and 156/1B9 identify the virus isolates from pigs 6, 11, 12, and 13 as porcine teschoviruses. There was no cross-reactivity with porcine and bovine enteroviruses (results not shown).
Reverse transcription PCR for the teschovirus VP-2 region was performed on isolates from pigs 11–13, according to techniques previously described. 13 The PCR product obtained was isolated and sequenced. The DNA sequences were used to construct a phylogenetic tree according to methods described elsewhere (Fig. 4). 21 All 3 isolates were classified as PTV serotype 1. Clustering the RNA products from pigs 11–13 indicated that isolates from pigs 12 and 13 were identical and were different from pig 11 isolate.
The diagnosis of PTV-induced polioencephalomyelitis was based on the location and pattern of lesions in the central nervous tissue, IHC, observation by electron microscopy of a virus with morphology and size compatible with picornavirus, viral neutralization, IFAT, and RT-PCR. To the authors' knowledge, this is the first report of teschovirus polioencephalomyelitis in western Canada and is the first confirmed case of this disease in Canada. In 1959, in Ontario, outbreaks of encephalomyelitis of unknown cause in pigs were described, and Talfan and/or Teschen disease was suggested as an etiology. 2 Lesions were similar to those described in the current study. In 1984, another outbreak of encephalomyelitis was described in Ontario, and viral particles consistent with enterovirus were observed by electron microscopy. 15 Teschovirus encephalomyelitis is diagnosed occasionally at other diagnostic facilities in the United States (R. M. Pogranichniy, personal communication, 2009). There are many viral encephalitides in pigs. Canada is free of classical and African swine fevers and pseudorabies (Canada Food Inspection Agency: 2010, Federally reportable diseases in Canada—2010. Available at: http://www.inspection.gc.ca/english/anima/disemala/rep/repe.shtml. Accessed October 8, 2010). In 1971, in Canada, a paramyxovirus was isolated from the brain of a pig with neurological clinical signs, but since then, there have been no other reports. A paramyxovirus resembling Bovine parainfluenza virus 3 has been rarely associated with encephalitis in pigs in the United States. 12 Other paramyxoviruses, such as rubulavirus and henipavirus, are exotic to Canada. PRRSV, PCV-2, malignant catarrhal fever, West Nile virus, Hemagglutinating encephalomyelitis virus, Encephalomyocarditis virus, and Rabies virus are all present in Canada and are potential causes of encephalitis in pigs.
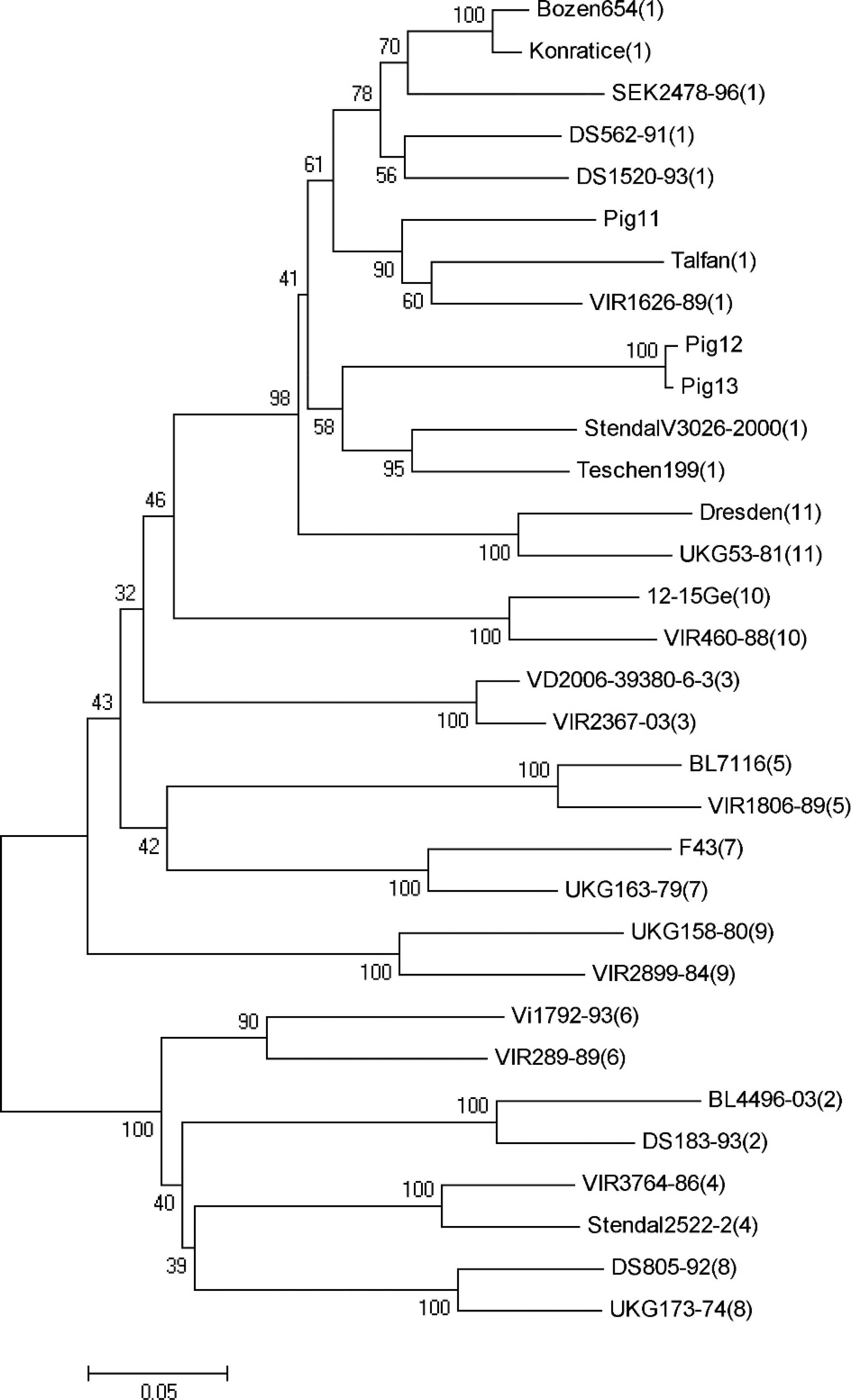
Phylogenetic tree derived using the maximum composite likelihood method (MEGA software version 4.0) shows the relationship of western Canadian teschovirus isolates to other isolates belonging to each of the 11 Porcine teschovirus (PTV) strains. (Strain type is indicated by the number in brackets following the isolate name.) Pigs 11–13 are clustered as PTV serotype 1.
Central nervous system lesions have been described with PRRSV 19 and PCV-2 3 infections. Involvement of brain stem nuclei and neuronal necrosis, which were frequent findings in the present outbreaks, are not described in PRRSV encephalitis. 19 Lymphohistiocytic vasculitis and fibrinoid degeneration of blood vessels with associated hemorrhages are described in brains with PCV-2–associated neurological disease. 3 Vasculitis was not observed in the present outbreaks. More importantly, IHC staining of spinal cord for these 2 viruses was negative.
Malignant catarrhal fever is uncommon in pigs but can occur when they are housed with sheep, 1 which was not the case in the pigs in the current study. In addition, the lesions described herein are not compatible with malignant catarrhal fever. Although present in Canada, there are no reports of West Nile virus infection in pigs. Experimental infection of pigs with West Nile has been described; infection caused viremia but not disease. 22 The first worldwide description of encephalomyelitis due to Hem-agglutinating encephalomyelitis virus was in Canada in 1962. 9 The coronavirus causing this disease is antigenically very similar to Bovine coronavirus, and the antibody to Bovine coronavirus is known to cross-react with Porcine coronavirus in immunoelectron microscopy technique. 5 Pigs were tested for Bovine coronavirus by IHC but were negative. In addition, the size of the porcine coronavirus is 120 nm, 5 which is much larger than the viral particle described in the current report (29 nm). Encephalomyocarditis virus is present in Canada, but the disease usually affects preweaning pigs, and lesions are often present in the myocardium, with pigs dying suddenly of heart failure. 4 Myocardial lesions were not observed in the pigs from the outbreaks in the present study. Although rare, rabies has been described in pigs in Canada. 24 The pigs of the outbreaks in the current study were tested for rabies by IHC and were negative.
Recently, a new virus was isolated in the United States associated with a disease named porcine reproductive and neurological syndrome (PRNS). 18 This syndrome has not yet been described in Canada, but it is very unlikely that the outbreaks reported in the current study were due to this new virus. Neuronal pathology is not reported in PRNS, and reproductive failure was not reported on the farms associated with the polioencephalomyelitis outbreaks in the present study.
Porcine teschovirus polioencephalomyelitis in pigs is an immediately notifiable disease in Canada and can cause a negative impact in the international market if a highly pathogenic strain is identified. Less virulent forms of this disease are likely underreported and underdiagnosed; however, it is very important that veterinarians are aware of this disease as there is potential for the virus to mutate into more pathogenic strains. In 2008, a case of this disease in association with PCV-2 infection was reported. 20 Immunosuppressive diseases, such as PRRSV and PCV-2, which are common throughout the swine industry, may contribute to the occurrence of PTV polioencephalomyelitis.
Acknowledgements.
The authors acknowledge Drs. Farshid Shahriar, Brendan O'Connor, Choon Yong, Helene Philibert, and Edwin Waters for contributing to the investigation of this disease; Drs. Dale Godson, John Campbell, and John Harding for advice and assistance; and the excellent technical support of M. Algar, M. McFarlane, and J. Shannan. This study was supported by the Saskatchewan Agriculture and Food Disease Investigation Fund and the Interprovincial Graduate Fellowship Fund, Western College of Veterinary Medicine.
Footnotes
a.
Rural Technologies Inc., Brookings, SD.
b.
Kindly donated by Dr. G. Allan, Department of Agriculture for Northern Ireland, Veterinary Sciences Division, Belfast, United Kingdom.
c.
Kindly donated by Dr. G. Cox, VIDO–University of Saskatchewan, Saskatoon, Canada.
d.
EnVision+ Detection System, Dako UK Ltd., Ely, Cambridge-shire, United Kingdom.
e.
Jackson ImmunoResearch Laboratories, Inv: dianova, Hamburg, Germany.
f.
Alexa Fluor®, Invitrogen GmbH, Frankfurter, Germany.
g.
RNeasy®, One Step RT-PCR kit, Qiagen Inc., Mississauga, Ontario, Canada.
