Abstract
Between August 2008 and May 2009, 386 brain and serum samples from adult cattle (2–7 years old) showing a variety of clinical signs of downer cow syndrome were received by the National Veterinary Research and Quarantine Service. All brain samples were tested for the presence of Bovine viral diarrhea virus (BVDV) by reverse transcription polymerase chain reaction (RT-PCR), immunohistochemistry (IHC), and antigen capture ELISA (Ag-ELISA). The BVDV nucleic acid was detected in 54 of 386 (15.5%) brain samples tested by RT-PCR. Positive results were detected in 14 (3.67%) and 13 (3.4%) of samples tested by IHC and Ag-ELISA, respectively. Both BVDV nucleic acid and antigen were detected in 11 cattle (2.9%) by all 3 diagnostic tests; however, antibodies against BVDV were not detected in these 11 cattle. A molecular classification of the identified viral strains (n = 40) was also carried out. Neighbor-joining phylogenetic analysis revealed that most of the identified viruses belonged to BVDV genotype 1a (n = 10), 1b (n = 16), and 2a (n = 8). The remaining strains were subtypes 1c (n = 1), 1n (n = 4), and 1m (n = 1). Interestingly, most of the BVDV-1b strains (n = 9) identified in brain samples were confirmed by all 3 diagnostic tests. Further studies should be performed to determine why the BVDV-1b strain was found in brain samples that were positive using all 3 diagnostic tests.
Keywords
Introduction
The term downer has been used to describe cattle that are too injured, weak, or sick to stand and walk. Numerous reasons exist for cattle losing the ability to stand and walk. Causes can be divided into general categories, including injuries, metabolic imbalances, and infectious or toxic diseases. 10 It has long been known that the primary causes of nonambulatory cattle are noninfectious factors. However, some infectious diseases can cause recumbency in cattle. Viral, bacterial, and parasitic diseases may result in neurologic disorders that cause recumbency. Bovine viral diarrhea virus (BVDV; family Flaviviridae, genus Pestivirus) infection is one viral disease that causes neurologic disorders. Fetuses infected with noncytopathic BVDV between days 30 and 125 of gestation develop immune tolerance and will be born persistently infected (PI; virus-positive and antibody-negative). Bovine viral diarrhea virus may replicate in certain nonlymphoid and nonepithelial tissues, including the central nervous system (CNS). 5,8,9,18
Since its first description in 1946, 23 BVDV infection has occurred worldwide and has caused significant economic losses. Bovine viral diarrhea is an important disease in cattle and is associated with several conditions, including respiratory and gastrointestinal infections, and reproductive problems, such as infertility, abortion, stillbirth, and sickly offspring. It has also been reported that BVDV in the CNS of cattle is associated with neurologic symptoms in persistently infected calves 9,17,19 and 15-month-old heifers. 4 It is known that neurons are the most important target cells in the CNS of persistently infected young and adult cattle. 9
Various methods are available for the diagnosis of acute or persistent BVDV infections. These include immunohistochemistry (IHC), several antigen detection and antibody detection enzyme-linked immuno-sorbent assays (Ag-ELISA and Ab-ELISA, respectively), reverse transcription polymerase chain reaction (RT-PCR) assays, and virus isolation. 27 29
Based on their antigenic and genetic properties, BVDV isolates can be divided into 2 principal genotypes: BVDV-1 and BVDV-2. 3,12,24,26 The BVDV-1 genotype has been divided by various groups into 2–11 subgenotypes (subtypes). 25 The BVDV-2 genotype has been found primarily in the United States, Canada, South America, and Japan, 6,20,25,26 but has also been reported in several European countries. 1,33 Recent reports have described a new genotype, BVDV-11, 14 and 2 new BVDV-1 subtypes, 1m and 1n. 21
The distribution of BVDV in the brains of fetuses and calves has been reported. 9,17,19 Brain lesions and viral antigens with BVDV-2 infection have also been reported in a 15-month-old heifer. 4 However, very little is known about BVDV infection in the brains of adult cattle acquired congenitally or postnatally. In the current study, the characterization and prevalence of BVDV were investigated from downer cattle that were unable to stand after 24 hr of recumbency, but were able to maintain sternal recumbency.
Materials and methods
Samples from 386 cattle showing a variety of clinical signs of downer cow syndrome were sent to 9 veterinary service laboratories (located in 9 provinces, Republic of Korea) for bovine spongiform encephalopathy (BSE) testing between August 2008 and May 2009. All cattle tested were BSE-negative (data not shown). All brain and serum samples were received at the National Veterinary Research and Quarantine Service (Anyang, Republic of Korea) for further testing. The brain and serum samples from cattle at least 2 years old were collected from different geographic regions representing all areas of Korea. All samples from both noninfectious and infectious types of cattle were tested.
BVDV antigen and antibody detection
Brain samples were analyzed for the presence of BVDV using RT-PCR, Ag-ELISA, a and IHC, as described later. Viral nucleic acids were extracted from brain samples using a commercial kit b and stored at −80°C until used for analysis. The RT-PCR reactions were performed in a onetube system using the pan-pestivirus primer pair V324/V326. This system amplifies a portion of the 5'-untranslated region (UTR) of pestivirus genomes. 15,31 The BVDV antigens in brain samples were identified by Ag-ELISA, according to the manufacturer's protocol. a All brain samples were fixed in 10% formalin. Representative portions of neuroanatomical regions were processed for histopathological examination. At least 5 regions of the brain, including major neuroanatomical areas, such as the hippocampus, were immunostained for pestivirus antigen using anti-BVDV antibody 15C5, c which is suitable for use on formalin-fixed, paraffin-embedded tissues. 11 All BVDV antigens were detected in the middle of the cerebrum, including the hippocampus (data not shown). Antibodies to BVDV in serum were also detected by Ab-ELISA, d according to the manufacturer's protocol. An antibody detection test in serum was performed in cases of RT-PCR-positive samples.
Sequence determination
The amplified 297-bp DNA fragment was purified using an agarose gel DNA extraction kit e and subcloned into the vector pGEM-T, f according to the manufacturer's instructions. Automated nucleotide sequencing of the 5′-UTR gene, inserted into the vector, was performed using a genetic analyzer g with a cycle sequencing kit. g All nucleotide positions were confirmed by 3 or more independent sequencing reactions in both directions.
Sequence comparison and phylogenetic analysis
Nucleotide sequence alignments were generated using the BioEdit computer program. h For phylogenetic analyses, partial 5′-UTR sequences of 40 BVDV strains from cattle brains were aligned with 34 reference BVDV sequences retrieved from GenBank. The phylogenetic tree was rooted with Classical swine fever virus (CW2002S) as an outgroup. A phylogenetic analysis based on the 5′-UTR sequences was also conducted using BioEdit with the Molecular Evolutionary Genetics Analysis (MEGA) 3.1 program with bootstrap values based on 1,000 replicates. 13 The cutoff point for bootstrap replication was more than 70%.
Results and Discussion
In the current study, BSE-negative brain and serum samples were used to investigate the prevalence and characterization of BVDV. The results are summarized in Table 1. All brain samples were tested for the presence of BVDV by RT-PCR. The BVDV genome was detected in 54 of 386 (15.5%) brain samples tested by RT-PCR. In addition, IHC and Ag-ELISA were performed. Bovine viral diarrhea virus was detected in 14 (3.67%) and 13 (3.4%) of 386 brain samples tested using IHC and Ag-ELISA, respectively. Eleven (2.9%) samples were positive by all 3 diagnostic tests (Table. 1). Possible explanations for the disagreement among the 3 tests in the 386 brain samples are as follows: RT-PCR is capable of detecting both acutely and persistently infected animals; Ag-ELISA rarely detects acutely infected animals as it was designed to detect persistently infected animals; and IHC can detect both acutely and persistently infected animals.
Eleven brain samples (2.9%) were positive by all 3 tests. The sera from these 11 cattle were also tested by Ab-ELISA. Antibodies against BVDV were not detected in any of the 11 samples. However, 17 of the 39 cattle positive by RT-PCR but negative by IHC or Ag-ELISA were positive by Ab-ELISA. The presence of pestivirus antigen in brain neurons indicated congenital persistent infection. 9,17,19 Generally, BVDV antigen is not found in the brain tissue of acutely infected animals. Therefore, these 11 cattle, in which the brain samples were positive in all 3 tests and antibodies were absent in their sera samples, could be persistently infected. However, in the case of acute BVDV infections, specific antibodies are usually not detectable during the first 1–2 weeks after infection. Although additional studies are needed, it was impossible to perform because all of the cattle studied were slaughtered. The results of the present study indicated that BVDV infection associated with nonambulatory cattle was more common than could be expected.
Summary of Bovine viral diarrhea virus (BVDV) antigen and antibody detection tests in adult cattle with downer cow syndrome.*
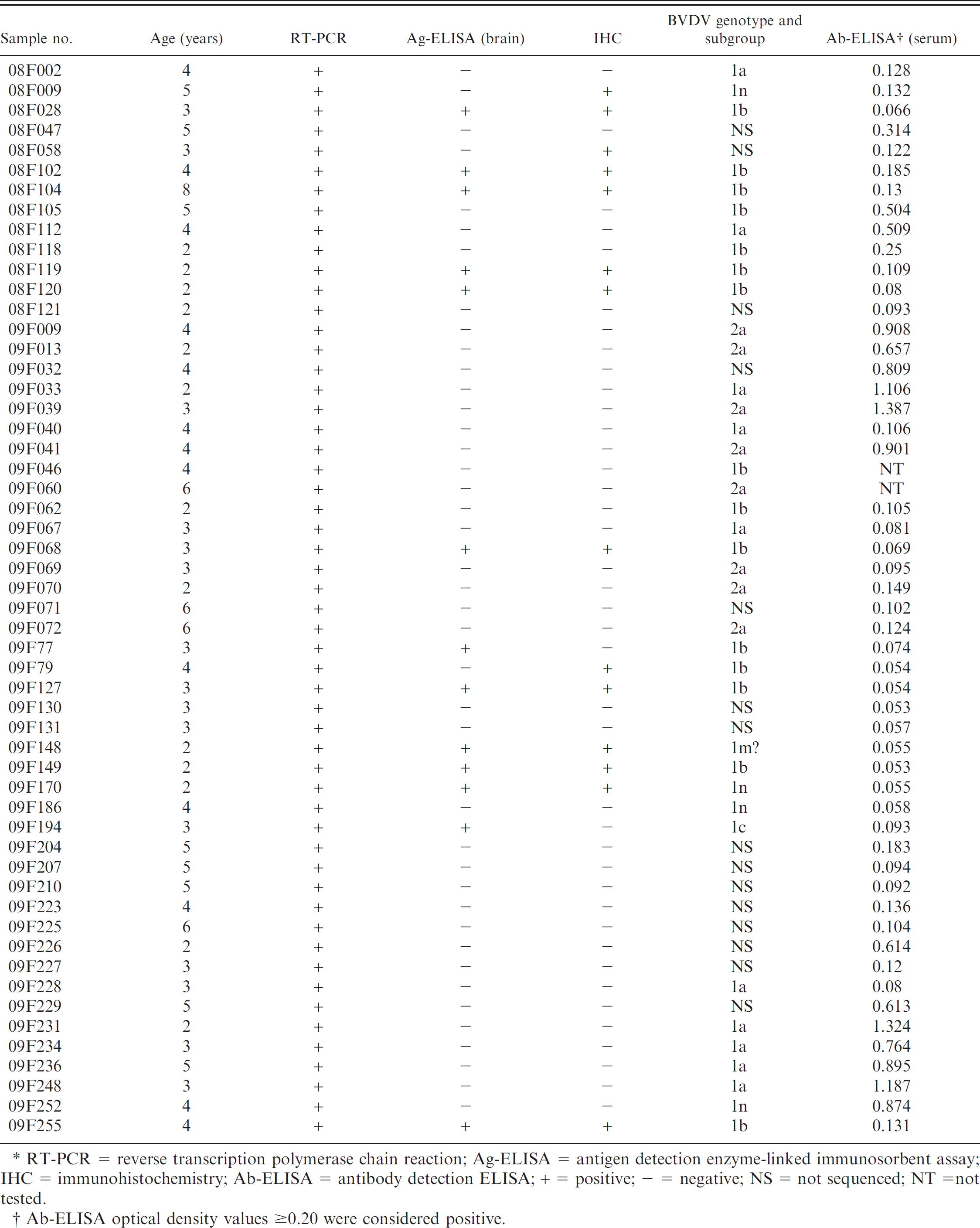
RT-PCR = reverse transcription polymerase chain reaction; Ag-ELISA = antigen detection enzyme-linked immunosorbent assay; IHC = immunohistochemistry; Ab-ELISA = antibody detection ELISA; + = positive; - = negative; NS = not sequenced; NT =not tested.
Ab-ELISA optical density values ≥0.20 were considered positive.
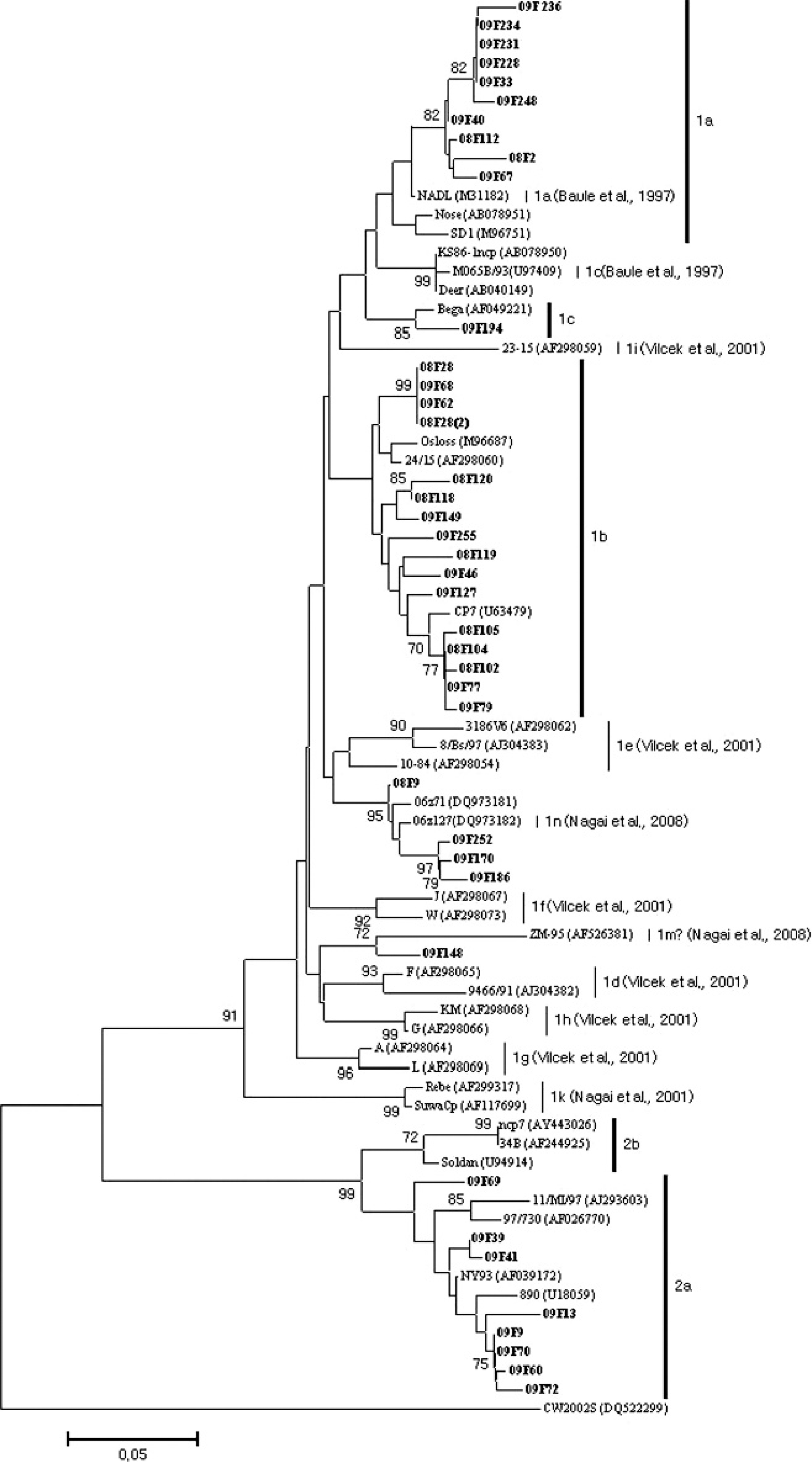
Phylogenetic analysis of 5′-untranslated region nucleotide sequences among diverse strains of Bovine viral diarrhea virus (BVDV). The nucleotide sequences of 40 BVDV strains were aligned using the BioEdit and Megna 3 computer programs. One thousand bootstrap replicates were subjected to nucleotide sequence distance and neighbor-joining methods. The consensus phylogenetic tree is shown with bootstrap values >70% displayed above the tree branches.
Genetic typing of pestiviruses is based primarily on the 5′-UTR, Npro, and E2 genomic regions. 2,26,32,33 Some reports suggest that the 5′-UTR region might not serve as an appropriate target sequence for phylogenetic analysis because of its highly conserved nature. 3 However, several investigators demonstrated that the 5′-UTR region is suitable for subtyping BVDV isolates. 25,26,32 The 5′-UTR region is efficiently amplified by RT-PCR and is the most frequently analyzed portion of the genome. Knowledge of the genetic diversity of BVDV isolates is important not only for taxonomic purposes, but also for laboratory diagnoses (RT-PCR or DNA chip).
Phylogenetic analyses were carried out to determine the genetic relationships among the identified viruses based on the nucleotide sequences of the 5′-UTR sequence (Fig. 1). Genetic analysis of the 5′-UTR sequences of 40 BVDV revealed that 32 were of genotype BVDV-1, and the rest were grouped within a cluster of BVDV-2 strains. The overall phylogenetic branching pattern of the BVDV-1 strains showed that they were of subtypes 1a (n = 10), 1b (n = 16), 1c (n = 1), 1n (n = 4), and 1m (n = 1), according to the nomenclature previously proposed. 21 Ten of the Korean strains clustered together with the NADL (National Animal Disease Laboratory) strain (M31182). This result is consistent with previous work showing that BVDV-1a is closely related to the NADL strain in Korea. 22 In a previous study, 22 36 samples (18 from aborted fetuses, 16 from diarrheal stools, and 2 from brains) were positive for BVDV. Only 1 strain (KA07) was classified as BVDV-1b. Interestingly, 16 BVDV-1b strains were identified in the present study, and most of the BVDV-1b strains were identified in the brain samples that were positive by all 3 diagnostic tests. All 46 BVDV isolates reported from persistently infected alpacas in the United States were BVDV-1b subtypes. 29 As inactivated BVDV-1a strain has been used in Korea, BVDV vaccines may not influence the predominance of BVDV-1b in persistently infected cattle in Korea. Additional studies are needed to reveal why Korean cattle are predominantly infected with BVDV-1b subtypes.
The 5′-UTR sequence of 4 BVDV strains were placed in a cluster with strains 06z71 (DQ973181) and 06z127 (DQ973182), which were identified in Korea between 2004 and 2006, 21 and were thus classified as BVDV subtype 1n. One BVDV strain (08F148) was also clustered as BVDV subtype 1m. 21 When the 5′-UTRs of 8 BVDV-2 strains were compared with those of other BVDV-2 sequences available from GenBank, all 8 were of subtype BVDV-2a.
In the present study, BVDV nucleic acid was detected using RT-PCR in 54 of 386 (15.5%) brain samples of downer cattle, infected either acutely or persistently. There is no evidence that BVDV is related to the neurologic signs in downer cows.
However, the prevalence (15.5%) of BVDV in downer cows was noticeably high and BVDV infection may influence the neurologic signs in downer cattle. In the molecular classification of the identified virus strains, most of the BVDV-1b strains were identified in brain samples that were positive by all 3 diagnostic tests.
Acknowledgements
This study was supported by a grant from the National Veterinary Research and Quarantine Service, Ministry of Agriculture, Anyang, Republic of Korea.
Footnotes
a.
HerdChek BVDV Ag/Serum Plus, HerdChek Bovine Diarrhea Virus Test Kit; IDEXX Laboratories Inc., Westbrook, ME.
b.
RNeasy™ Mini Kit, Qiagen Inc., Valencia, CA.
c.
Anti-BVDV antibody 15C5, Syracuse Bioanalytical, Syracuse, NY.
d.
SVANOVIR™ BVDV-Ab ELISA, Svanovar Biotech AB, Uppsala, Sweden.
e.
Agarose Gel DNA Extraction Kit, iNtRON Biotechnology Inc., Daejeon, Korea.
f.
pGEM-T Vector, Promega Corp., Madison, WI.
g.
ABI 3130xl Genetic Analyzer, Big Dye™ Terminator Cycle Sequencing Kit; Applied Biosystems, Foster City, CA.
h.
BioEdit computer program, Ibis Biosciences Inc., Carlsbad, CA.
