Abstract
Equid alphaherpesvirus 4 (EqAHV4; Orthoherpesviridae, Varicellovirus equidalpha4; equine rhinopneumonitis virus) has seldom been associated with complications such as abortion and myeloencephalopathy, given the low tendency of this virus to induce viremia. We investigated the frequency of EqAHV4 viremia in horses with fever and respiratory signs. Case selection included all equids with EqAHV4 quantitative real-time PCR (qPCR)–positive nasal secretions (defined as EqAHV4 qPCR–positive cases) submitted to a diagnostic laboratory. Controls consisted of each case submitted before and after each EqAHV4 qPCR–positive case. Purified nucleic acid from blood samples collected from EqAHV4 qPCR–positive horses and control cases was tested for EqAHV4 by qPCR. We selected 183 EqAHV4 qPCR–positive horses and 376 EqAHV4 qPCR–negative horses. In general, EqAHV4 qPCR–positive horses were younger, and had a lower rate of anorexia and a higher rate of nasal discharge compared to the EqAHV4 qPCR–negative horses. A total of 25 of 183 (13.7%) horses with EqAHV4 qPCR–positive nasal secretions tested qPCR-positive for EqAHV4 in blood. EqAHV4 viremic horses were significantly younger (p < 0.015 for group <1-y-old) and had a significantly higher occurrence of distal limb edema (p < 0.05) than EqAHV4 non-viremic horses. Our data support the observation that EqAHV4 viremia is rarely detected in EqAHV4-infected horses, which explains the low level of reported complications, such as abortion and myeloencephalopathy.
Equid alphaherpesvirus 4 (EqAHV4; Orthoherpesviridae, Varicellovirus equidalpha4; equine rhinopneumonitis virus) is considered one of the leading respiratory viruses associated with upper airway infections in horses. EqAHV4 is enzootic in most horse populations, and most horses have serologic evidence of exposure to this virus.4,23 Most clinically infected horses are <3-y-old, with most affected horses <1-y-old. 16 Following a short incubation period, infected horses develop fever, lethargy, anorexia, mandibular lymphadenopathy, and profuse serous nasal discharge that later becomes mucopurulent. A dry cough secondary to rhinotracheitis is occasionally observed but is not a consistent feature of the disease.
Although abortion and equine herpesviral myeloencephalopathy (EHM) are expected to occur with the closely related equid alphaherpesvirus 1 (EqAHV1; Orthoherpesviridae, Varicellovirus equidalpha1; equine abortion virus), such complications have seldom been reported for EqAHV4.1,9,10,18,22,24 The inefficient capacity of EqAHV4 to infect mononuclear cells explains in part the rarity of EqAHV4-induced viremia, and subsequently, the rarity of EqAHV4-induced abortion or EHM. 21 The diagnosis of EqAHV4 infection relies on the molecular detection of EqAHV4 in respiratory secretions during the acute disease stage. In contrast to EqAHV1, little-to-no attention has been directed toward the detection of EqAHV4 in whole blood. EqAHV4 viremia has rarely been reported following natural or experimental infections.11,14 Therefore, our aim was to document the frequency of EqAHV4 viremia in horses with acute onset of fever and respiratory signs and to determine if EqAHV4 viremia was associated with specific prevalence factors, such as demographic parameters and clinical signs.
We used nasal swabs and whole blood collected from horses with acute onset of fever and respiratory disease between January 2020 and December 2023. The samples were part of an ongoing equine respiratory biosurveillance program that enrolled 324 equine veterinary clinics across 45 states. The submission criteria for this respiratory biosurveillance program included fever (rectal temperature >38.6°C [101.5°F]) and one or more of the following clinical signs: nasal discharge, cough, limb edema, anorexia, lethargy. Samples were submitted along with a questionnaire to capture the signalment (age, breed, sex, use) and clinical signs for each case. Rayon-tipped nasal swabs (Puritan) were tested for Streptococcus equi subsp. equi (S. equi), equine influenza A virus (EIV; Orthomyxoviridae, Alphainfluenzavirus influenzae), EqAHV1, EqAHV4, equine rhinitis A virus (ERAV; Picornaviridae, Aphthovirus burrowsi), and equine rhinitis B virus (ERBV; Picornaviridae, Erbovirus aequirhi). Whole blood was tested for EqAHV1 only.
Samples were analyzed using quantitative real-time PCR (qPCR) testing as reported previously. 17 Briefly, on the day of sample arrival at the laboratory, nucleic acid was extracted from the submitted nasal swabs and whole blood using an automated nucleic acid extraction system (QIAcube HT; Qiagen). cDNA synthesis was performed (QuantiTect reverse transcription kit; Qiagen) following the manufacturer’s directions, with slight modifications as described previously. 3 All of the samples were tested for the presence of the reference gene eGAPDH, as described previously, to ensure sample quality and efficiency of nucleic acid extraction. 8
Case selection included all equids with EqAHV4 qPCR–positive nasal secretions for which purified nucleic acid from blood was available (defined as EqAHV4 qPCR–positive cases). Controls (defined as EqAHV4 qPCR–negative cases) consisted of each case submitted before and after each EqAHV4 qPCR–positive case. Purified nucleic acid from blood samples collected from EqAHV4 qPCR–positive horses and control cases had been saved at −80°C before EqAHV4 analysis by qPCR as reported previously. 17 Quantitative PCR results for EqAHV4 were expressed as the number of glycoprotein B (gB) target genes/106 cells, as reported previously. 14
Demographic and clinical factors were compared between EqAHV4 qPCR–negative and –positive horses. Further, EqAHV4 qPCR–positive nasal secretion and blood-positive horses were compared to EqAHV4 qPCR–positive nasal secretion and blood-negative horses. Demographic factors included breed, use, sex, and age (categorized into 5-y increments); clinical parameters included fever, lethargy, anorexia, nasal discharge, coughing, and distal limb edema. Parametric (chi-square and Student t-test) and nonparametric tests (Fisher exact and Mann–Whitney U tests) were used, as appropriate, to compare categorical and continuous factors.
Over the 4-y study period, biological material from 2,893 equids with acute onset of fever and respiratory signs was submitted to the laboratory. A total of 183 horses with EqAHV4 qPCR–positive nasal secretions with stored whole blood were available. The EqAHV4 qPCR–negative control population consisted of 376 horses. For both study groups, additional respiratory pathogens were detected, including EIV, S. equi, ERBV, and EqAHV1 (Table 1).
Frequency of selected respiratory pathogens detected in nasal secretions and blood of horses with acute onset of fever and respiratory signs.
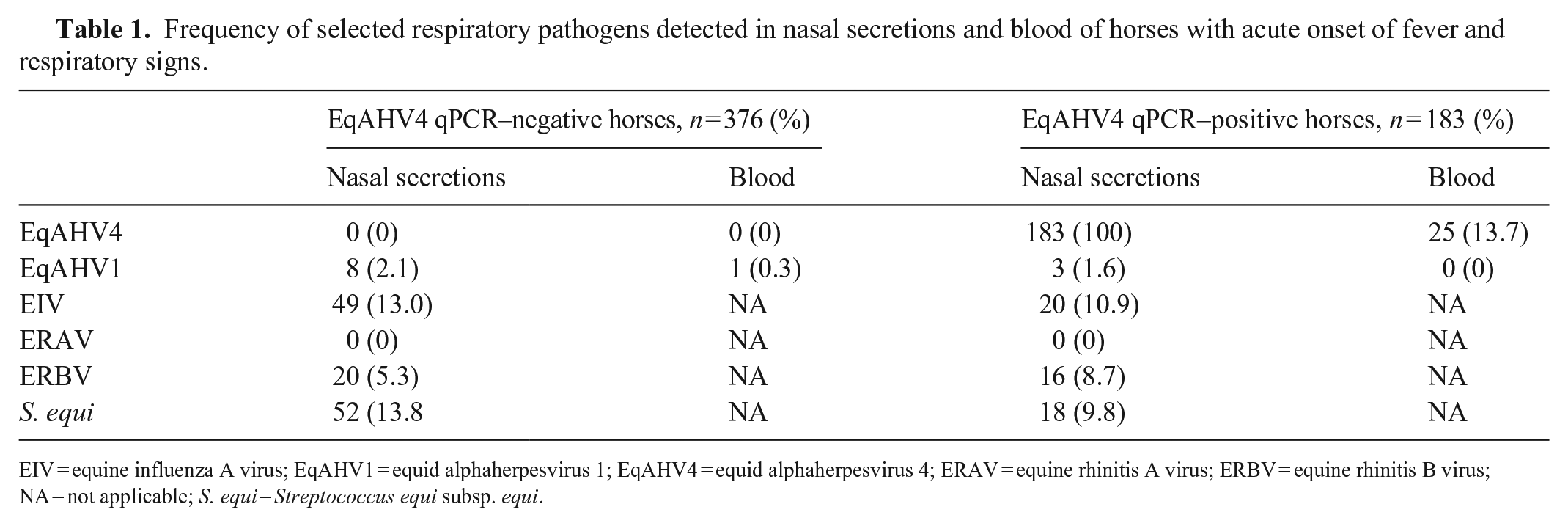
EIV = equine influenza A virus; EqAHV1 = equid alphaherpesvirus 1; EqAHV4 = equid alphaherpesvirus 4; ERAV = equine rhinitis A virus; ERBV = equine rhinitis B virus; NA = not applicable; S. equi = Streptococcus equi subsp. equi.
Detection of EqAHV4 qPCR–positive horses was significantly higher in the <1-y-old age group than in the EqAHV4 qPCR–negative horses (p < 0.001; Table 2). Further, lack of anorexia and presence of nasal discharge were significantly more frequently detected in the EqAHV4 qPCR–positive horses compared to the EqAHV4 qPCR–negative horses (p = 0.024 for lack of anorexia and p = 0.06 for the presence of nasal discharge). All of the other prevalence factors showed no statistical differences between the 2 EqAHV4 qPCR groups (Table 2).
Year and season of sample submission, demographic and clinical data from 183 horses with EqAHV4 qPCR–positive nasal swabs and 376 control horses with EqAHV4 qPCR–negative nasal swabs.
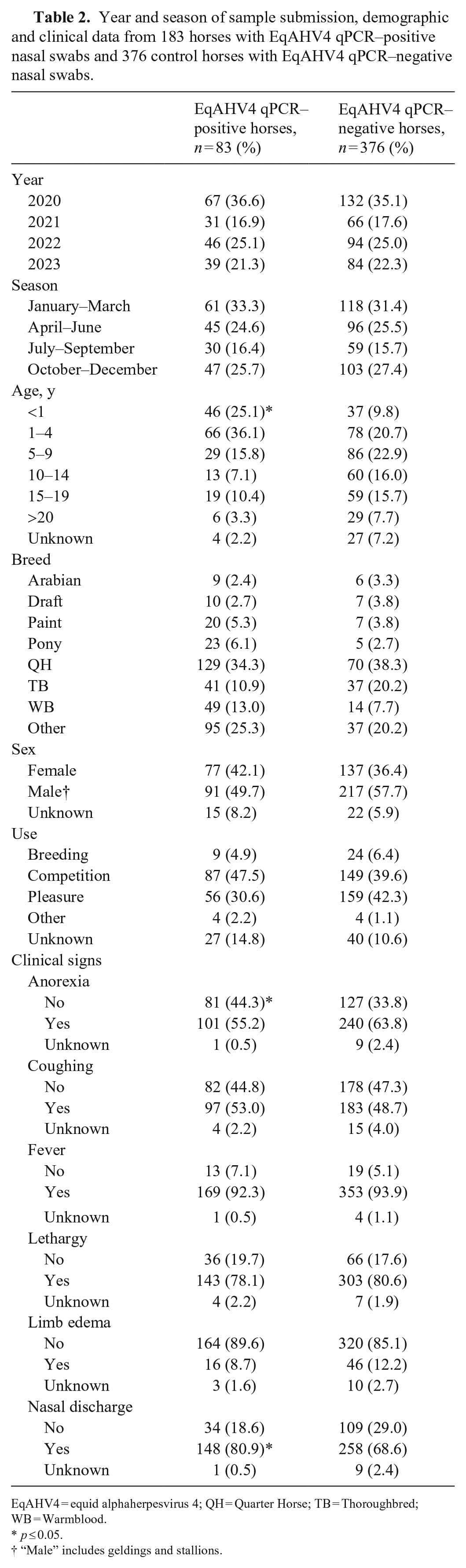
EqAHV4 = equid alphaherpesvirus 4; QH = Quarter Horse; TB = Thoroughbred; WB = Warmblood.
p ≤ 0.05.
“Male” includes geldings and stallions.
When both EqAHV4 qPCR–positive groups with and without viremia were compared, only age and limb edema were determined significant. EqAHV4 viremic horses were significantly younger (p < 0.015 for <1-y-old age group) and had a significantly higher occurrence of limb edema (p < 0.05) compared to the EqAHV4 non-viremic horses (Table 3). The EqAHV4 viral load in nasal secretions from horses with and without EqAHV4 viremia was 383–978,296,072 gB genes/106 cells (median: 5,217,812 gB genes/106 cells) and 11–491,952,203 gB genes/106 cells (median: 1,311 gB genes/106 cells), respectively (Fig. 1). EqAHV4 qPCR–positive horses with viremia had significantly higher EqAHV4 viral loads in nasal secretions compared to EqAHV4 qPCR–positive horses without viremia (p < 0.0001).
Year and season of sample submission, demographic and clinical data from EqAHV4 qPCR–positive horses with and without viremia.
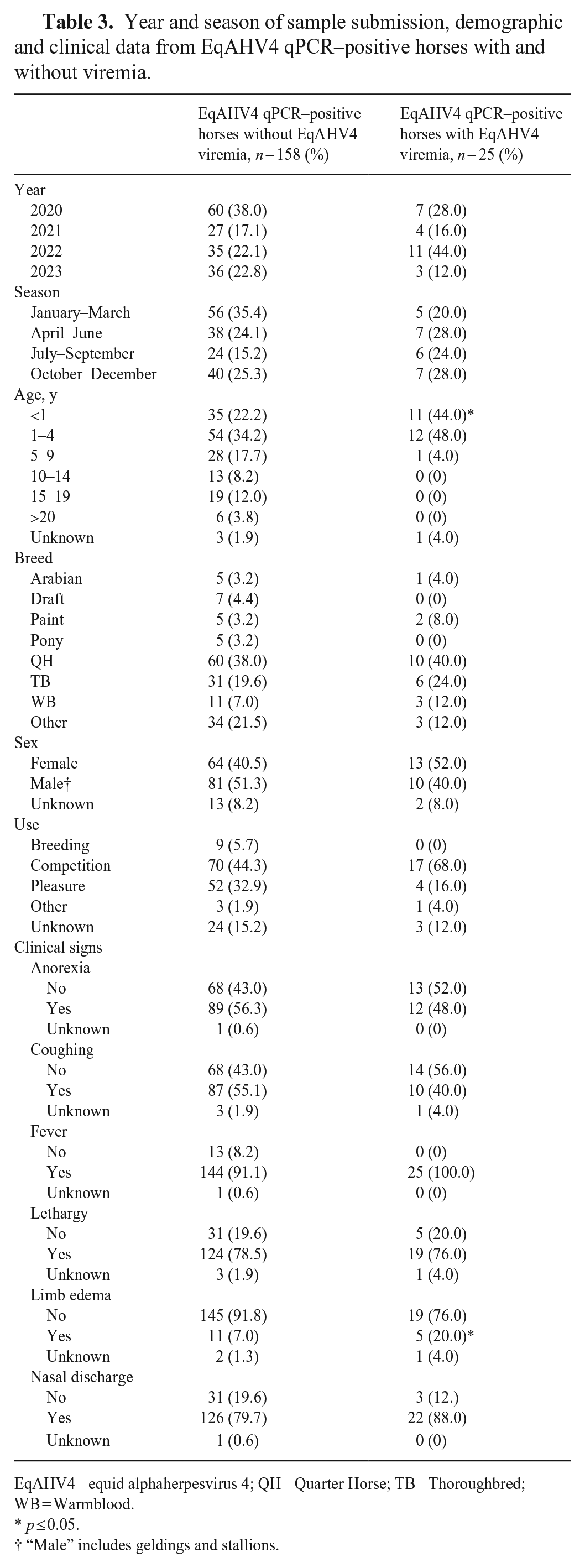
EqAHV4 = equid alphaherpesvirus 4; QH = Quarter Horse; TB = Thoroughbred; WB = Warmblood.
p ≤ 0.05.
“Male” includes geldings and stallions.
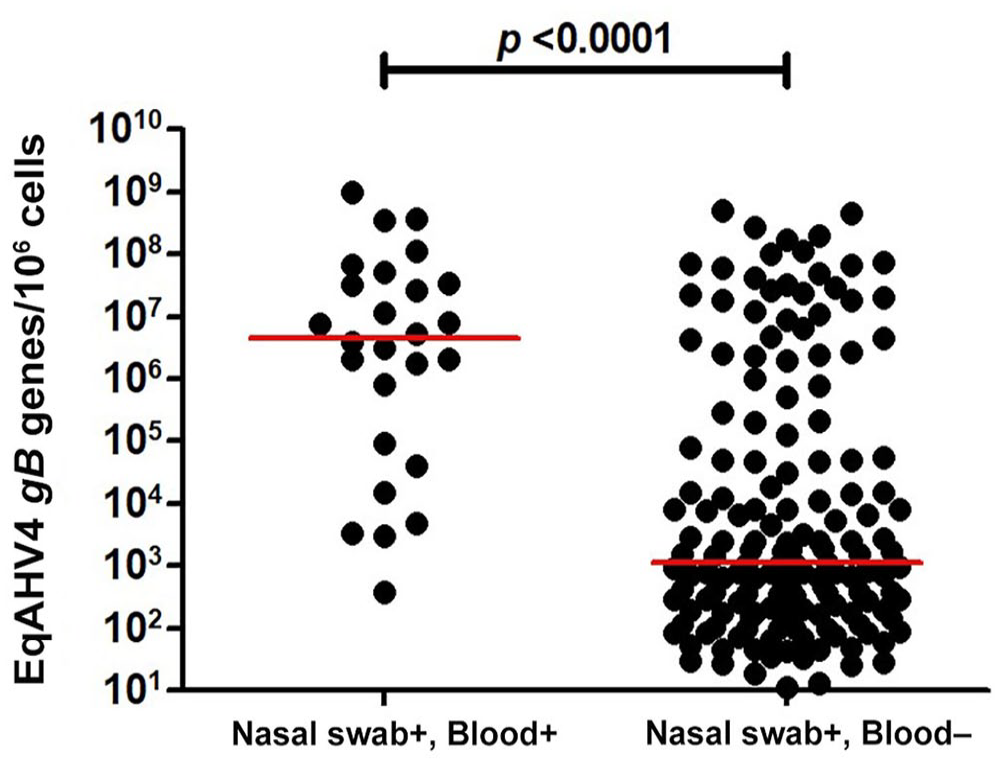
Absolute quantification of equid alphaherpesvirus 4 (EqAHV4) qPCR–positive results in nasal secretions for horses positive and negative for EqAHV4 in blood. The results are expressed as number of EqAHV4 gB target genes/106 cells, and the horizontal bars are the median viral load for each disease category.
We found that viremia was a relatively rare event in horses with acute onset of fever and respiratory disease, as only 25 of 183 (13.7%) horses with EqAHV4 qPCR–positive nasal secretions were also EqAHV4 qPCR–positive in blood. This is in sharp contrast to the detection of EqAHV1 in nasal secretions and whole blood in horses with similar clinical signs, reporting up to 100% of horses EqAHV1 qPCR–positive in both biological samples. 15 Although both equine alphaherpesviruses have been shown under natural and experimental conditions to induce viremia, the difference in frequency of viremia is likely related to their respective affinity for mononuclear cells.11,14,21 Further, even in the presence of a high frequency of EqAHV1 viremia in febrile horses, the complication rate remains limited to sporadic cases and occasional outbreaks of abortion or EHM. 19
The horses with EqAHV4 qPCR–positive nasal secretions had demographic data and clinical signs similar to the control population, with the exception of age and specific clinical signs. Our results are in line with work documenting that EqAHV4 qPCR–positive equids with acute onset of fever and respiratory signs are generally younger and have a higher rectal temperature and higher rate of nasal and ocular discharge compared to EqAHV4 qPCR–negative horses. 16 Of interest was the observation that 50 of 183 (27.3%) EqAHV4 qPCR–positive horses had coinfections with other respiratory pathogens, including EIV, EqAHV1, ERBV, and S. equi, which is higher than in other studies.17,23 The higher coinfection rate in EqAHV4 qPCR–positive horses compared to previous studies might be related to the demographic of our study population. It is also interesting to note that coinfection was higher in EqAHV4 qPCR–positive horses without viremia (30.4%) compared to EqAHV4 qPCR–positive horses with viremia (8.0%). One would expect EqAHV4 and EqAHV1 to be detected with greater frequency with other respiratory pathogens, given that the 2 equine alphaherpesviruses establish latent infection in the horse and can be reactivated during periods of stress. However, reactivation is often weak, of short duration, and generally not associated with viremia. 12 Previous work has shown that coinfected equids often have more severe clinical signs, highlighting the cumulative effect of various respiratory pathogens. 17 In calves, clinical signs, especially fever, are often more severe and of longer duration when multiple viruses are involved in respiratory infections. 6 It is therefore possible that some of the EqAHV4 qPCR–positive nasal secretions from horses with coinfection were the result of reactivation of latency rather than reflective of a primary infection. Independent of the role of EqAHV4 in coinfection (i.e., reactivation of latent infection, subclinical infection, or true clinical infection), horses with EqAHV4 qPCR–positive nasal secretions must be separated and considered infectious unless proven otherwise.
EqAHV4 viremia was restricted to horses with EqAHV4 viral shedding given that none of the control horses tested EqAHV4 qPCR–positive in blood. Among all selected prevalence factors, age and limb edema were significantly different between EqAHV4 qPCR–positive horses with and without viremia. EqAHV4 viremia has been reported in 100% of weanling foals with natural EqAHV4 infection and in 50% of young adult horses experimentally infected with EqAHV4.11,14 Viremia is apparently associated with young age and might be related to the naïve status of susceptible animals. It would have been interesting to capture and compare the EqAHV4 vaccination history between EqAHV4 viremic and non-viremic horses. Another interesting observation was the higher frequency of distal limb edema in the EqAHV4 viremic horses compared to the non-viremic horses. The mechanism by which edema was triggered in the EqAHV4 viremic horses is purely speculative but might be related to the replication of EqAHV4 in endothelial cells. 2
Disease severity of viral respiratory infections is generally modulated by viral and host factors, with high viral loads in respiratory secretions often linked to greater disease severity, longer clinical course, and slower recovery.5,13,20 In our study, EqAHV4 viral loads in nasal secretions of horses with viremia were significantly higher than in horses without viremia. It is also interesting to note that, with other respiratory viruses, such as SARS-CoV-2 in humans, there is a strong association between viral levels in anterior nasal secretions and in plasma, both of which impact disease severity. 7 Based on our data from nasal secretions and blood, it appears that a similar observation applies to EqAHV4.
Footnotes
Acknowledgements
We thank the participating veterinarians who collected the information and biological samples for our study.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Our study was funded internally by the Equine Infectious Disease Research Laboratory, School of Veterinary Medicine, University of California, Davis, CA, USA.
