Abstract
A straightforward, reliable technique for postcollection processing and evaluation of cytologic specimens for antigen detection using an automated immunostainer was developed. Visual assessment of cell suspension turbidity was used in parallel with light microscopic examination of concentrated cytospin preparations to verify the diagnostic utility of samples for immunocytochemical staining. Fine-needle lymph node biopsies from 81 dogs with lymphadenomegally and a cytologic or histologic diagnosis of lymphoma were introduced into ethylenediamine tetra-acetic acid tubes containing standardized storage media. Cell suspension turbidity was assessed to estimate cell concentration and resultant volume required for cytospin preparations with optimal cellularity. Preliminary cytospin preparations (using estimated volumes based upon turbidity) were stained using modified Wright stain and examined microscopically for intact neoplastic cell concentration. Once an optimal volume for cytospin preparations was established, additional concentrated slides were prepared for immunophenotyping, using an automated immunostainer and antibodies specific for cluster of differentiation (CD)79a and CD3e. All cell suspension samples with adequate gross turbidity had ample intact neoplastic cell concentration for immunocytochemical staining. Based on CD79a and CD3e expression, 51 (63%) B cell, 19 (23%) T cell, 3 mixed T and B cells (4%), and 3 non-T- and non-B-cell lymphomas (4%), as well as 5 (6%) nondiagnostic samples were identified. Three out of 5 of the nondiagnostic samples were submitted early in the investigation prior to the establishment of gross specimen turbidity guidelines. Immunocytochemical staining results were in complete agreement with all 6 available immunohistochemical correlates. The ability to visually assess sample adequacy prior to sample submission may encourage more widespread use of immunocytochemical techniques.
Keywords
Introduction
Cytology is commonly employed to obtain a diagnosis of lymphoma because of ease and accuracy as well as low morbidity, time, and technical costs. 4,13 Immunophenotypic characterization of these disorders in dogs has become an integral part of a minimal database, carrying significant prognostic relevance. 9,10,16,24 Immunocytochemistry provides an attractive alternative to more conventional immunophenotyping techniques because of its relatively rapid, simple, and noninvasive nature.
A variety of approaches have been in use over the past several decades for phenotypic identification of neoplastic cells, including cytochemical staining, immunohistochemistry, flow cytometry, immuno-cytochemistry (ICC), and molecular analysis. 1,7,8,15,17,19,20,24 Diagnostic ICC has historically been underutilized in veterinary medicine as a result of difficulties in consistent acquisition of adequate cytologic samples; this remains a significant obstacle. Traditionally prepared air-dried fine-needle biopsy preparations hinder consistent, reliable immunocytochemical staining results because of inherent sample variability and sensitivity to nonspecific and/or background staining interferences. A high degree of variability in neoplastic cell concentration, hemodilution, and degree of cell disruption exists within individual fine-needle biopsy specimens, making it difficult to estimate specimen adequacy prior to submission. These intrinsic factors significantly increase the likelihood of unrewarding, costly, and inconclusive test results.
Utilization of concentrated cytospin preparations for ICC in humans 5,15,21 and animals has been reported. 3,6 Immunocytochemical staining of cytospin preparations has been associated with fewer complications (e.g., background staining, cell distortion, poorly cellular specimens, etc.) and is generally preferred when compared with traditional fine-needle biopsy techniques. 3,12,21,23 Several investigations have described the use of various standardization medias and/or cell fixatives to attempt to enhance the accuracy and consistency of immunocytologic results. 10,12,21–23 The purpose of the current investigation was to develop a simple, reliable, cost-effective technique for postcollection handling, assessment, and processing of fine-needle biopsy specimens prior to automated immunocytochemical staining. The specific goal was consistent acquisition of high-quality diagnostic samples.
Materials and methods
Fine-needle biopsy specimens were obtained from 81 dogs presenting to the Michigan State University Veterinary Teaching Hospital (MSU-VTH; East Lansing, MI) for lymphadenomegally and suspected lymphoma. None of the subjects had undergone any form of chemotherapy prior to sample collection. A diagnosis of lymphoma was obtained through routine cytologic and/or histologic evaluation for all subjects. Fine-needle biopsy samples were obtained percutaneously from a palpable lymph node using a 21-gauge needle attached to a syringe (e.g., 6 ml or 12 ml). Multiple redirected passes (3–5) through the lymph node were performed without applying negative pressure to the syringe. The number of needle passes beyond 3 did not appear to affect sample adequacy to any appreciable degree. Specimens were introduced into a 3-ml vacutainer vial containing prepared standardized storage media. Vials of storage media were created prior to sample collection by adding 1 ml of 0.9% phosphate buffered saline (PBS) and 40 μl of 22% bovine serum albumin a to a 3-ml K3-ethylenediamine tetra-acetic acid vacutainer tube. Groups of 10 vials of storage media were prepared and stored for up to 1 month at 3–8°C prior to specimen collection.
For visual cell suspension assessment, vials containing resuspended cell material were examined for overall turbidity and degree of red discoloration by holding them against 10–12-point font printed on white paper. Cell suspensions that prevented reading of print through the vial were deemed adequately cellular. Specimens that did not meet this requirement were rebiopsied until adequate turbidity was achieved. During the preliminary phases of this investigation, gross cell suspension turbidity was compared with the microscopic appearance of modified Wright-stained cytospin preparations to establish general guidelines for this collection protocol. Based on several initial samples, red discoloration did not preclude a conclusion of sample adequacy; only samples with a noticeable lack of gross turbidity were deemed inadequate. The degree of red discoloration in cell suspensions was generally mild overall and did not interfere with gross or microscopic interpretation.
Preliminary concentrated cytospin survey slide preparations were usually made within 24 hr of specimen collection by adding 2 different volumes (10–50 μl, based on visual turbidity in order to select a concentration with optimal neoplastic cellularity) of gently mixed cell suspension plus 0.9% PBS for a total volume of 100 μl, into 2 cytospin cups, centrifuging at 25 X g b for 10 min, and staining with modified Wright stain. The remaining cell suspension was stored at 3–8°C. Survey slides were evaluated for sample adequacy, neoplastic cell concentration, and confirmation of the cytologic diagnosis prior to submission for automated ICC. Once an adequate diagnostic cellularity dilution was established, additional unstained cytospin preparations (6–10) were made for immunocytochemical staining. All concentrated preparations used for both survey slides and for immunocytochemical staining were generally prepared within 24 hr of specimen collection.
Cytospin preparations were stained using an automated immunostainer c and a polymer detection kit d according to manufacturers' instructions. Monoclonal antibodies against cluster of differentiation (CD)3e e and CD79a f were used to distinguish T- versus B-cell lymphoma subtypes, respectively. These antibodies have been used extensively for this purpose in veterinary species. 6,7,19,24 A negative control (no primary antibody) was run in parallel for each case. All immunocytologic preparations were examined microscopically to evaluate stain uptake pattern, quality, and background staining. For the purposes of the current study, B-cell lymphomas were diagnosed based on predominantly CD79a-positive-staining neoplastic cells (≥80%) with low numbers of residual CD3e-staining cells; T-cell lymphoma was diagnosed based on predominantly CD3e-positive-staining neoplastic cells (≥80%) with low numbers of residual CD79a-staining cells. Although not a focus of this investigation, histologic lymph node sections were obtained from 6 cases early in the study and stained using the automated immunostainer and the same antibodies for correlation purposes; beyond these 6 cases, individual positive control samples were not used during the course of this investigation.
Results
During the present investigation, all cell suspensions with sufficient gross visual turbidity (as described) yielded cytospin-concentrated slide preparations of excellent diagnostic utility for immunophenotypic characterization using an automated immunostainer. Cell suspensions initially ranged from clear to moderately turbid and from colorless-to-white to pink-to-red coloration. Microscopic examination of stained survey slides revealed that cell suspension turbidity and red coloration were directly proportional to neoplastic cellularity and hemodilution, respectively (Fig. 1). These characteristics were independent of each other; the degree of hemodilution (typically mild) had no effect on turbidity. All specimens with sufficient turbidity to obscure reading print through the vial provided adequate neoplastic cellularity for immunophenotypic evaluation regardless of the degree of hemodilution present. Hemodilution did not interfere with immunostaining quality. Automated cell counts g performed on 4 samples with adequate turbidity had cell concentrations ranging from 2,000 to 6,000 cells/μl. All cell suspensions required some degree of dilution with 0.9% PBS to obtain optimal concentrated preparation results.
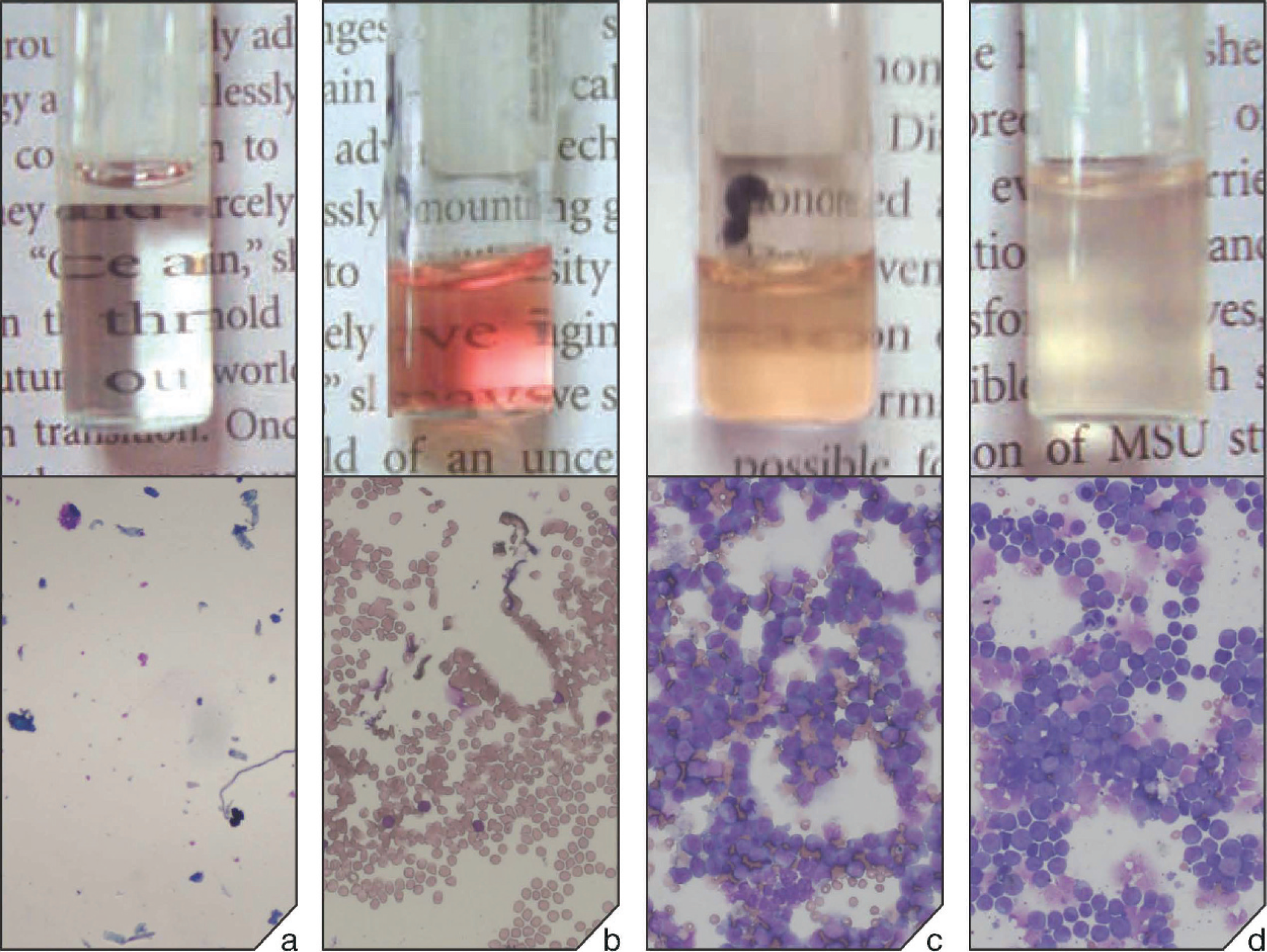
Several cases (5), most collected early in the investigation, were poorly cellular based on turbidity and subsequent microscopic examination of survey preparations (Fig. 1a, b). Cell suspensions from these cases had poor gross turbidity with varying degrees of red coloration resulting in clear recognition of printed letters through the specimen vial. Microscopically, these specimens had low to moderate numbers of erythrocytes and occasional leukocytes, consistent with the amount of blood. Cellularity was deemed inadequate and immunocytochemical staining was not pursued. This problem was subsequently circumvented by immediate lymph node recollection until adequate turbidity was obtained. This modification of the collection protocol, used for the remainder of the study, resulted in nearly 100% proficiency in obtaining diagnostic specimens.
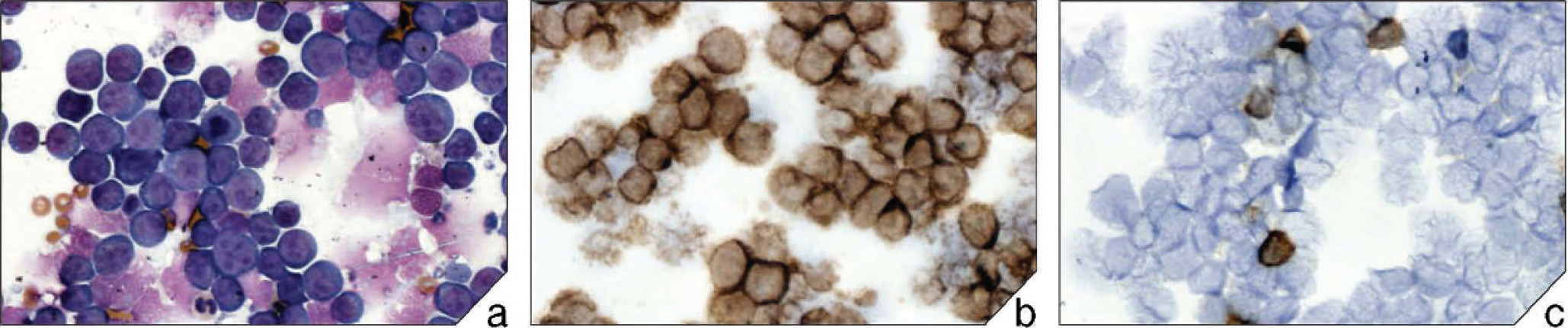
Representative case of B-cell lymphoma. Cytospin preparation stained with (
Immunocytochemistry was performed on all samples with adequately turbid cell suspensions (76); of these, 51 patients had B-cell, 19 had T-cell, 3 had mixed T- and B-cell, and 3 had non-T-, non-B-cell lymphomas. Ideally, concentrated preparations were produced to obtain a monolayer of neoplastic cells that filled approximately 40–90% of the cytospin area. Modified Wright-stained survey preparations typically had >90% neoplastic lymphocytes (% total cells) with greater than 50% intact cells (Fig. 2). Overall cell morphology after processing remained adequate to excellent, allowing recognition of multiple cell types. Small to moderate numbers of erythrocytes in the background were proportional to grossly observed red color changes. Occasional macrophages, nonneoplastic lymphocytes, and neutrophils also were observed in low numbers in many cases. Immunocytochemical results revealed that the majority of neoplastic cells stained positive for either CD79a (with a minority of residual cells staining positive for CD3) or CD3 (with rare residual CD79a-positive cells; Fig. 2). Antibody uptake was generally intracytoplasmic, with some membrane-associated staining around the periphery of cells. Negative control slides had minimal nonspecific background staining. Immunohistochemical correlates were obtained for 6 cases with immunocytochemical staining results, and all were in complete agreement (5 B-cell and 1 T-cell lymphoma).
Discussion
Although immunocytology is desirable for immunophenotyping lymphoma in veterinary medicine, inherent variability of traditionally prepared fine-needle aspirated samples precludes consistent diagnostic results. Carefully prepared cytospin preparations are generally required to obtain diagnostically useful immunostaining results. Several publications 3,6,10 describe use of concentrated cytospin preparations for immunocytostaining in animals, mainly for research applications. Moreover, most of these reports employed manual immunostaining methods associated with increased labor, turnaround time, technical expertise, and individual variation. To the authors' knowledge, no study has utilized a standardized processing and a gross sample evaluation technique combined with an automated immunostainer for commercial diagnostic application.
Starting with a morphologically well-preserved, adequately cellular, minimally contaminated cytologic specimen remains one of the most important factors in obtaining diagnostic immunocytochemical results. Previous studies have documented various difficulties in consistently obtaining adequate samples for immunocytochemical procedures 3,6,21 utilizing traditional fine-needle biopsy collection techniques. It was hypothesized in the current study that much of the inconsistency resulted from sample variations involving insufficient neoplastic cellularity, cellular disruption and/or lysis, and/or the presence of interfering substances such as blood or proteinaceous material. Clinicians often attempt to compensate for these problems by increasing the number of slides collected or repetitive sampling of the site. These obstacles are further amplified when nondiagnostic specimens are submitted and processed, resulting in significant economic and time losses and discouraging use of immunocytology. Visual evaluation of cell suspension turbidity as described in the current report provides a repeatable guideline for estimation of specimen adequacy prior to submission, helping to minimize inconclusive, discrepant, and nondiagnostic results. Samples with insufficient turbidity are immediately recollected until adequate turbidity is obtained. In addition, utilization of standardized high-protein collection media aids in maintaining sample preservation, which facilitates the best possible diagnostic results.
Although sample storage length was not a focus of the current investigation, several cell suspensions were reprocessed at subsequent intervals (i.e., after 7 and 14 days of storage at 3–8°C) to evaluate the preservation of cells using this storage media formulation. Cell morphology deteriorated over time and was considered poor by the day 7 postcollection interval; however, concentrated preparations could be stained and successfully interpreted for immunophenotypic purposes after 7 days of storage at 3–8°C. All cell suspensions from the current investigation were processed no later than 48 hr after collection, with most samples being processed within 24 hr of collection. Another report utilizing a similar storage media formulation for lymph node and bone marrow samples indicated successful immunostaining results for mail-in samples with less than 3-day sample collection-to-processing turnaround times. 23 Additional studies have also described long- and short-term storage of concentrated cytospin preparations for use in immunostaining procedures. 15,21
The 5 samples that were inadequately cellular were among the first cases obtained during this investigation and ultimately were helpful in determining a minimum magnitude of visual turbidity necessary for a diagnostic specimen. Improved sample adequacy submission rates were observed over time, approaching nearly 100% as clinicians became increasingly proficient at estimating cell suspension turbidity using these principles. Although the majority of specimens had adequate turbidity, slight volume adjustments were made because of individual variability in order to obtain optimal cytospin field cellularity. In the present study, neoplastic lymphocytes as B-cell (51), T-cell (19), mixed (3), or non-T-cell, non-B-cell (3) phenotypes based upon CD79a and CD3 expression were consistently immunophenotyped. All cases with available histologic correlates had an identical immunophenotype using both diagnostic modalities. Comparable results between immunocytologic and immunohistologic samples have been reported. 6,7 Techniques described herein have successfully been instituted for routine diagnostic use at MSU-VTH, illustrating their practical utility. In the present study, only lymph node samples and antibody markers for CD79a and CD3e were used. Additional unpublished data demonstrated that other solid tissues, including soft tissue masses, spleen, liver, and bone marrow, also may be processed and stained similarly. Moreover, other antibody markers (e.g., CD45, vimentin, cytokeratin, calretinin) were successfully tested during the present investigation, illustrating the versatility and wide range of potential applications for these techniques. Previous veterinary 11 and human 12,15 studies have also reported successful staining with a variety of these markers.
In conclusion, a simple, consistent technique for postcollection handling, assessment, and processing of fine-needle biopsy lymph node samples for immunophenotyping lymphomas using an automated immunostainer was developed. Seventy-six cases of lymphoma as B-cell, T-cell, mixed T-cell and B-cell, or non-T-cell, non-B-cell were characterized using these techniques. Standardized storage media was reliable for collection and short-term storage of high-quality specimens for immunophenotypic evaluation. Assessment of cell suspension turbidity using print significantly increased the proficiency of obtaining diagnostically adequate samples. Reliability of these methods increased the confidence level among practitioners for obtaining accurate, diagnostic immunocytochemical results. These practices are versatile and may be applied to a variety of other tissues and/or antibody markers for characterization of other neoplastic entities.
Acknowledgements
The authors are grateful to Tom Wood from the Diagnostic Center for Population and Animal Health for his technical and intellectual expertise on all immunochemical-staining procedures. Additionally, the authors thank Craig Lyons for his assistance in the graphic design of Figs. 1 and 2. These studies were supported by the Diagnostic Center for Population and Animal Health, College of Veterinary Medicine, Michigan State University. Abstract and poster presented at 49th Annual AAVLD Meeting, Minneapolis, MN, October 12–19, 2006.
Footnotes
a.
Sigma-Aldrich, St. Louis, MO.
b.
Shandon Cytospin® 3 Cytocentrifuge, Shandon Scientific Ltd., Astmoor, England.
c.
Bond™-max automated immunostainer, Vision BioSystems, Victoria, Australia.
d.
Bond™ polymer detection kit (DS9713), Vision Biosystems, Victoria, Australia.
e.
Clone CD3–12, P. F. Moore, University of California-Davis, Davis, CA.
f.
Clone HM57, Dako North America Inc., Carpinteria, CA.
g.
Advia® 120, Siemens Healthcare Diagnostics Inc., Deerfield, IL.
