Abstract
Tissue samples from 2 juvenile ferrets (Mustela putorius furo) from a colony that was undergoing an outbreak of respiratory disease were submitted to the Iowa State University Veterinary Diagnostic Laboratory. Microscopic examination of lung samples revealed bronchointerstitial pneumonia with necrotizing bronchiolitis. Influenza A virus was detected in sections of formalin-fixed lung by immunohistochemistry and reverse transcription polymerase chain reaction assay. A field investigation of the premises and analysis of additional samples led to the confirmation and characterization of an influenza virus with high homology to contemporary reassortant H1N1 swine influenza viruses. Although ferrets have been used extensively to research the virulence and transmissibility of avian, human, and swine influenza virus strains, no published information exists on naturally occurring outbreaks of swine influenza in ferrets.
A routine diagnostic case that consisted of pooled tissue samples from 2 juvenile ferrets (Mustela putorius furo) with a history of clinical respiratory disease was submitted to the Iowa State University Veterinary Diagnostic Laboratory (Ames, IA). The ferrets represented a population of approximately 1,000, of which 8% of the animals were exhibiting respiratory signs, including severe dyspnea, sneezing, coughing, and crusting of the eyes and nose. Mortality at the time of the first submission was 0.6%. Field investigation of the farm undertaken 2.5 weeks after the initial submission revealed the presence of multiple avian species (including ducks, geese, and peacocks), small mammal species raised for purchase (raccoons, skunks, and fox), a cow and calf herd, horses, and llamas. The ferrets were housed in individual cages constructed with wood and galvanized wire. Ferrets were hand fed a ground turkey and chicken mixture and hand watered daily. The farm was not routinely tested for pathogens; however, 4 years before the above-described case, the farm experienced similar clinical signs. In both cases, clinical signs occurred in early spring. Clinical signs among the ferrets were still apparent, and additional samples were collected to characterize the influenza virus strain that was affecting the colony. Nasal swabs, bronchoalveolar lavage samples, serum, and tissues were collected from 4 ferrets that displayed acute clinical signs. Serum samples from 4 ferrets that were not showing acute clinical signs were also obtained for serologic analysis.
Microscopic examination of lung sections revealed bronchointerstitial pneumonia with necrosuppurative bronchiolitis. This was characterized by patchy areas of alveolar septal thickening; pulmonary congestion; coagula of debris in alveoli, with associated atelectasis; and a locally extensive region in which airways were dilated and filled with neutrophils, macrophages, degenerate cells, and debris. Scattered airways were characterized by attenuation of airway epithelium, bronchiolitis, and bronchiogenic abscessation (Fig. 1). No bacterial or mycoplasmal agents were isolated from the lung tissue. Canine distemper virus (CDV) was not detected by fluorescent antibody test 5 or real-time reverse transcription polymerase chain reaction (real-time RT-PCR) by using a set of primers and probe targeted to the nucleoprotein gene of CDV. 19 Influenza A virus (family Orthomyxoviridae, genus Influenzavirus A; H1N1 subtype) was detected in the lung tissue by immunohistochemistry 22 (IHC; Fig. 2) and by real-time RT-PCR. 11 Briefly, the IHC procedure used a monoclonal antibody, which targeted the nucleoprotein of Swine influenza virus (SIV), a a biotinylated goat anti-mouse antibody, b peroxidase-conjugated streptavidin, b and diaminobenzidine tetrahydrochloride solution. c The primers used in the RT-PCR assay are designed for the nucleoprotein, hemagglutinin (H1 and H3), and neuraminidase (N1 and N2) gene segments of the SIV genome.
The sequence analysis of the hemagglutinin (HA) gene (GenBank accession no. FJ50522) indicated high homology (98%) with that of viruses that belong to one of the major clades of triple reassortant H1N1 SIV represented by MN02 SwH1N1 (Fig. 3). Influenza A virus, which was subtyped H1N1, was isolated from 3 of 4 lung sample homogenates collected during the follow-up visit by using both the Madin–Darby canine kidney cell line and embryonated chicken eggs. 23,25 There was a 99.7% nucle-otide homology for the HA gene between the viruses recovered from the first and second submissions (Fig. 3). The sequence analysis for the matrix gene (GenBank accession no. GQ149509) of the isolates revealed that the virus was from a swine lineage in origin (Fig. 4).
Serum samples from the ferrets were tested by hemagglutination inhibition (HI) assay 3 against a variant swine reassortant H1N1 (SIV99), a classic swine H1N1 (IA76), a cluster I H3N2 (TX98) SIV, and the ferret H1N1 isolate. Inhibition titers exhibited high titers (particularly in convalescent sera) to the ferret isolate, moderate titers to the variant swine reassortant and the cluster I H3N2 isolate, and low titers to the classic swine H1N1 isolate (Table 1). Titers to the cluster I H3N2 isolate could be nonspecific or indicative of previous exposure to H3 virus of human, swine, equine, or canine. The HA gene of TX98 is human lineage in origin. A low degree of cross reactivity among swine H3, equine H3, and canine H3 can be expected.
Because of similarities in the clinical signs, pathogenesis of disease, and immunologic response induced in both humans and ferrets, 15 the ferret model has been extensively used to assess the pathogenicity of avian, swine, and human Influenza A virus isolates. 2,9,14,26 Clinical signs and lesions induced by swine, avian, and human Influenza A virus isolates in ferrets have been well characterized in previous studies and were similar to those observed in the naturally infected animals in the current study. 12,13,15 Natural infection of a ferret colony with a human influenza virus was reported in 1948 1 ; however, to the authors' knowledge, natural infections in colonies with a swine isolate have not previously been reported.
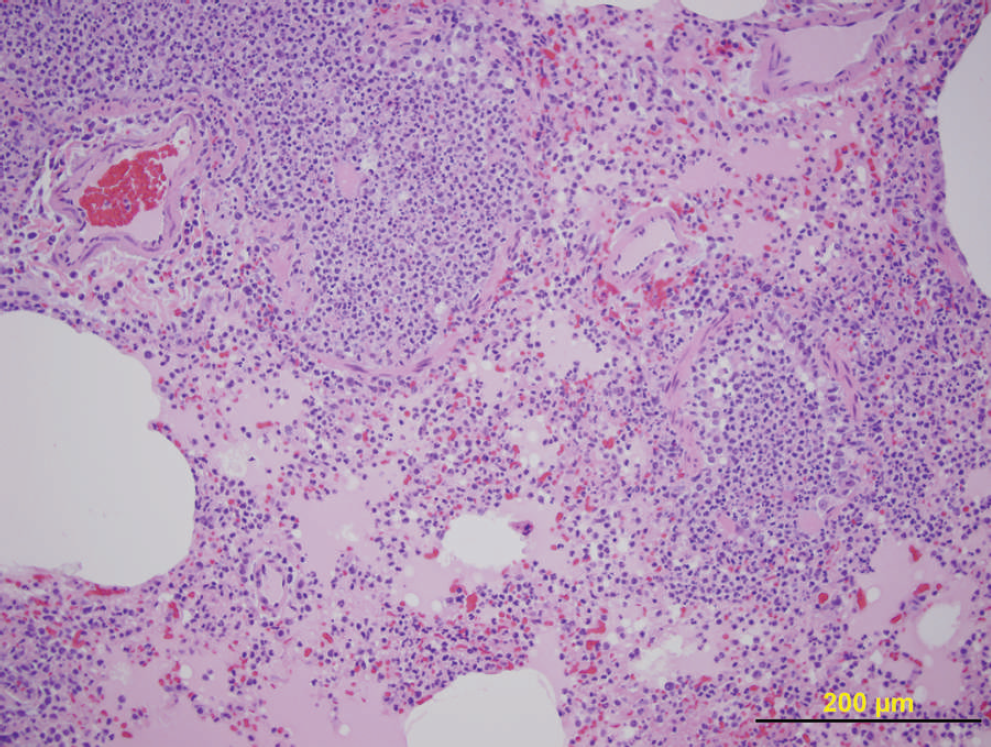
Necrosuppurative bronchiolitis in the lung of a ferret infected with Influenza A virus. Hematoxylin and eosin. Bar = 200 μm.
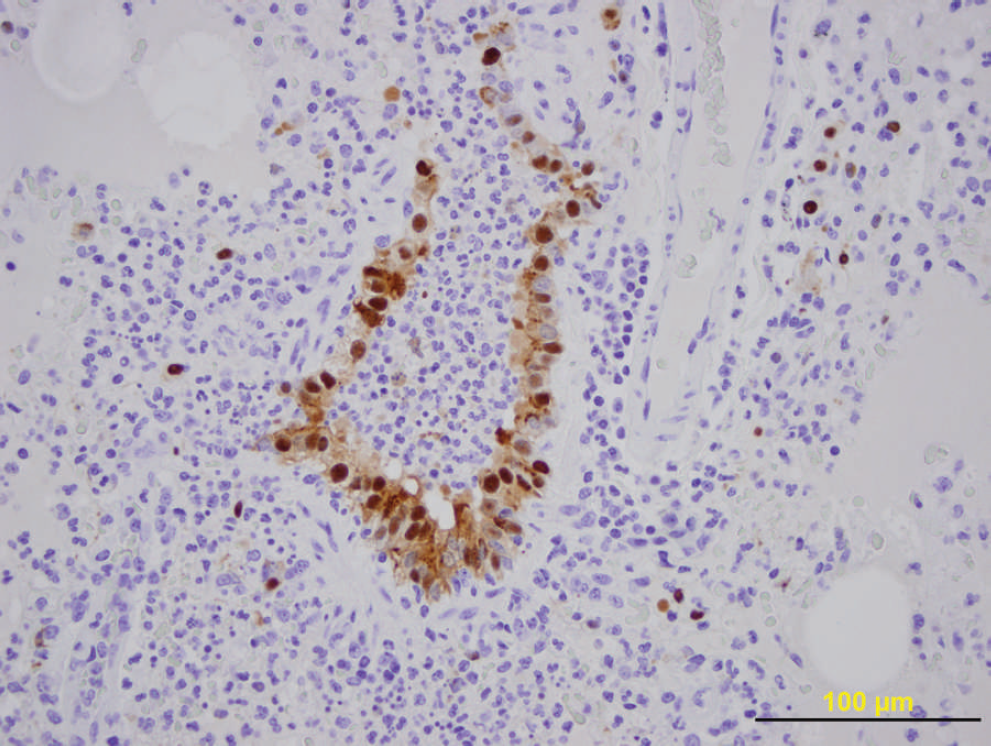
Immunohistochemical stain of the ferret lung, illustrating the presence of influenza virus antigen within bronchiolar epithelial cells. Bar = 100 μm.
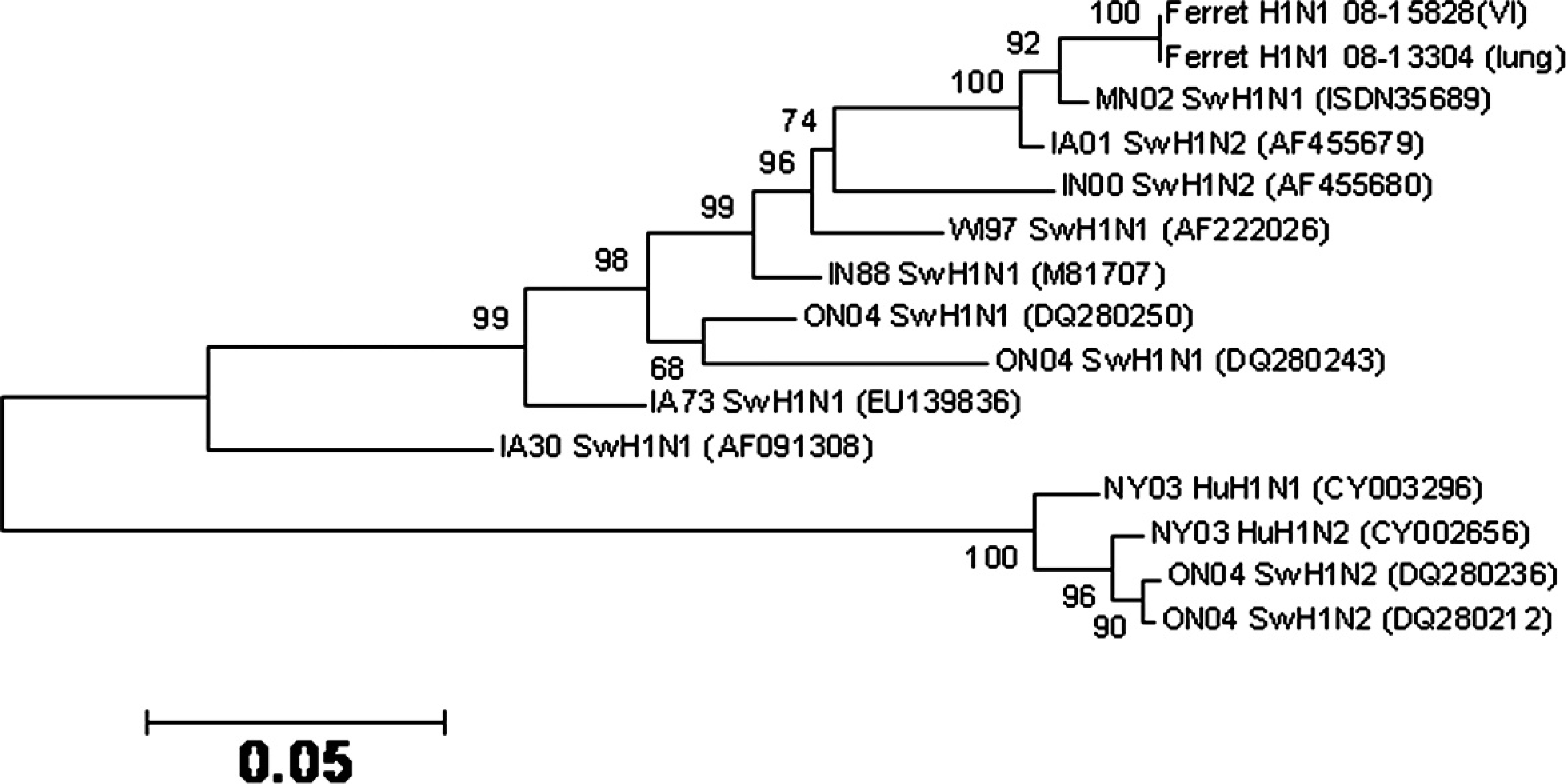
Phylogenetic relationship of ferret isolate (08-15828) of influenza A virus to representative H1 swine (Sw) and human (Hu) influenza viruses and the influenza virus in lung tissue from a clinically ill ferret (08-13304) based on nucleotide sequence of the hemagglutinin RNA segment. Reliability of the tree was assessed by bootstrap analysis with 1,000 replications. The GenBank accession number of each reference virus is given within the parentheses.
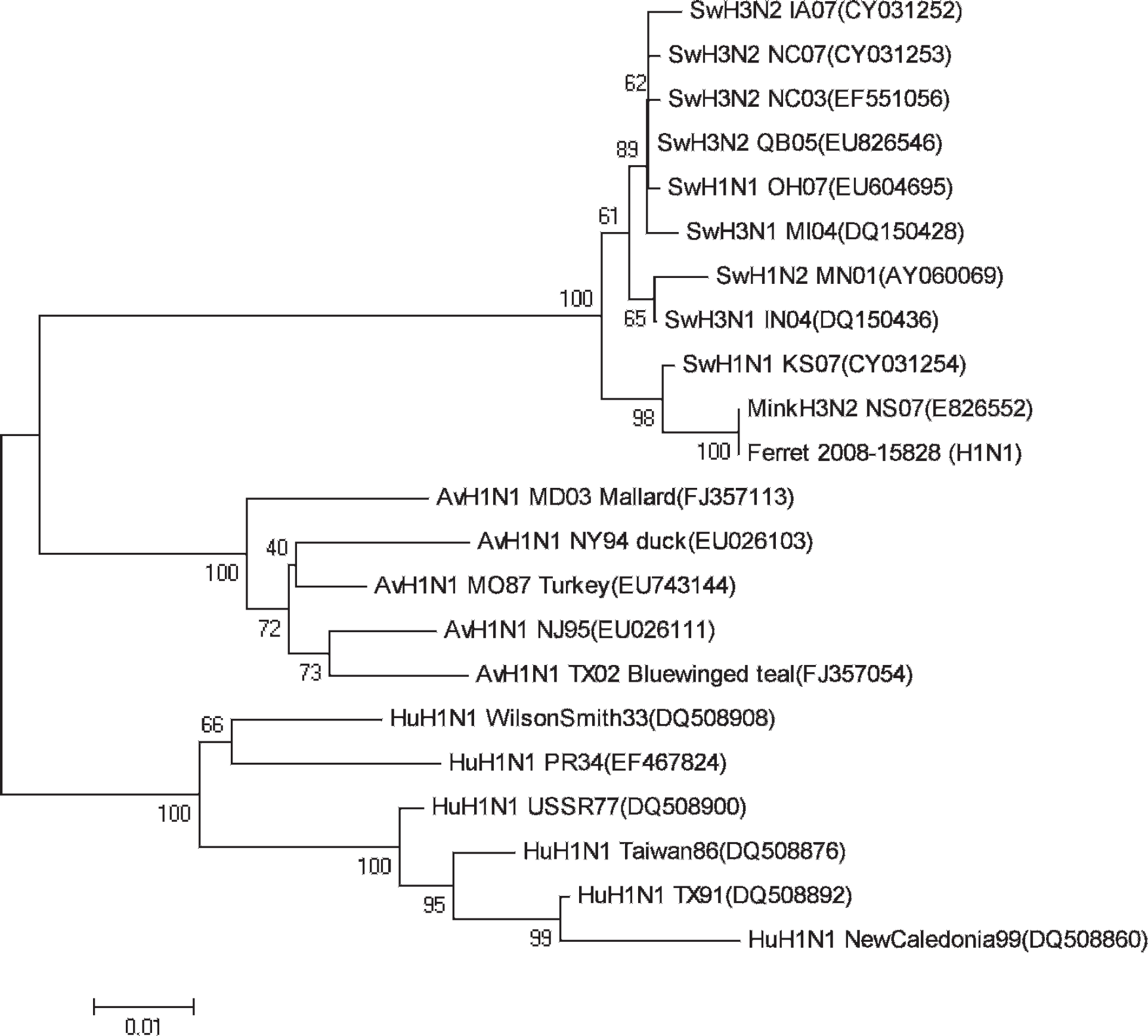
Phylogenetic relationship of ferret isolate (08-15828) of Influenza A virus to representative swine (Sw), human (Hu), and avian (Av) influenza viruses based on nucleotide sequence of the matrix RNA segment. Phylogenetic and molecular evolutionary analyses were conducted by using MEGA version 4. The tree was generated by using the neighbor-joining method. Reliability of the tree was assessed by bootstrap analysis with 1,000 replications. GenBank accession numbers are in parentheses.
Hemagglutination titers from acutely affected ferrets (A–D) and previously exposed but clinically normal ferrets (E–H) by using a variant H1N1 (SIV99), a classic H1N1 (IA76), a cluster I H3N2 (TX98) SIV, or the virus isolated from the affected ferrets (Ferret H1N1).*
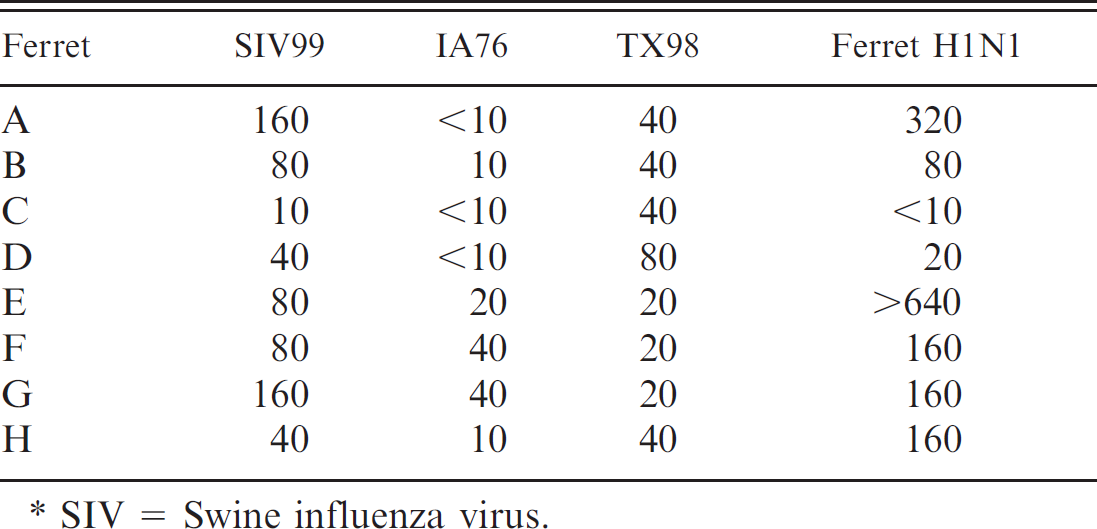
SIV = Swine influenza virus.
The specific source of infection was not identified in the present case, but the close association with other birds and mammals offered several possible sources of transmission. No swine were housed on the farm, but a possible source of infection was aerosol transmission from a swine operation located approximately 0.25 miles from the ferret colony. Influenza viruses are nearly ubiquitous in swine, 14 and it is not uncommon for swine operations to be endemically infected. 17 In addition, aerosol transmission between adjacent swine operations was reported as a potential means of transmission. 6 The fact that the virus isolated from the ferret colony had an HA gene with 98% nucleotide homology to that of a SIV isolate (MN02 SwH1N1) and also contained the matrix gene of SIV origin suggests likely transmission from the adjacent swine operation. Unfortu- nately, because of the inability to test surrounding swine operations, this hypothesis could not be confirmed. No evidence of horizontal transmission through uncooked swine byproducts, as previously reported in a mink population, 8 was identified in this colony. However, uncooked turkey was included in the diet. This poses a risk, because turkeys are known to carry SIVs. 4 Because numerous species, such as horses, 24 dogs, 18 minks, 7 cats, 16 raccoons, 10 and marine mammals, 21 can act as potential reservoirs for maintenance and transmission to natural hosts, the potential for horizontal transmission from one of the other species on the farm cannot be ruled out.
Another possible source of infection would include interaction with infected human workers that results in transmission of a swine H1N1 strain to the colony. The ability of the ferret to transmit influenza virus to humans was reported in the 1930s. 20 Subsequent studies confirmed the infectious nature of human influenza isolates in ferrets. However, workers on the farm with the ferret colony had no known contact with swine, and influenza was not diagnosed by a physician in any of the workers at the time of the outbreak.
Although the specific source of infection was not identified, based on nucleotide sequence and HI analysis, the most likely source of infection was from an adjacent swine operation. To the author's knowledge, the present case represents the first report of a natural swine influenza infection in a ferret colony. In the course of the investigation, the colony was treated with injectable antibiotics to prevent secondary bacterial infections. The outbreak ended without further intervention; vaccination of the herd was discussed but not implemented.
Acknowledgements. The authors would like to thank Joe Bender, Amy Chriswell, Fanghong Zhou, and Suzanne Block for technical assistance; Dr. Nathan Peterson for the initial case submission; and Pfizer Animal Health for financial support.
Footnotes
a.
VMRI, Ames, IA, not commercially available.
b.
DAKO® Labeled Streptavidin-Biotin 2 System kit, Dako North America Inc., Carpinteria, CA.
c.
DAKO® Liquid DAB (diaminobenzidine) Substrate-Chromogen System, Dako North America Inc., Carpinteria, CA.
