Abstract
A 6-year-old ferret (
Malignant mesenchymoma is a rare malignant tumor reported in animals as well as humans. Typically, mesen-chymoma is a tumor composed of 2 or more distinct cell types having unrelated malignant mesenchymal differentiation (e.g., chondrosarcoma, fibrosarcoma, leiomyosar-coma, liposarcoma, osteosarcoma, or rhabdomyosarcoma). 12 Immunohistochemistry is used to provide a definitive identification of the malignant cellular populations. Malignant mesenchymoma can be located in different anatomic sites and is usually considered a high-grade sarcoma with poor prognosis; however, low-grade neoplasms have also been reported. The primary tumor may be infiltrative, and potential metastases may be expected during clinical and postmortem evaluations.
A 6-year-old, spayed, female, albino ferret (
On physical examination, the ferret appeared to be in good body condition. A large, round-to-oval, firm mass was detected in the abdominal cavity by palpation. No other abnormal findings were apparent during physical examination. Diagnostic evaluation included an abdominal survey radiograph, complete blood cell count, and serum biochemical profile. The complete blood cell count revealed a moderate, normocytic, normochromic, nonregenerative anemia. Marginal hyperalbuminemia of 3.9 g/dl (reference interval: 2.6–3.8 g/dl) was present, suggesting dehydration. Because of simultaneous dehydration, the grade of anemia was considered to be worse than implied by the hemogram results and was likely due to chronic disease. Abdominal latero-lateral radiographs demonstrated the presence of a sharply marginated, oval mass with an estimated diameter of 5 cm and multifocal radiopaque foci, suggestive of calcification or extraskeletal bone formation.
Abdominal laparotomy revealed a voluminous mass in the right retroperitoneal space that was adherent to the dorsal abdominal wall. The mass protruded into the abdominal cavity and partially enveloped the caudal pole of the right kidney. The mass and the right kidney were surgically excised. The mass measured 5.0 cm × 3.9 cm × 3.6 cm and was well separated from, and did not infiltrate, the kidney. The cut surface of the mass appeared white to gray to yellow and was composed of multilacunar spaces and a mixture of fatty to hard fibrous tissue with small, multifocal calcifications. Small, rounded fragments of green material, compatible with suture material, were seen near the center of the lesion. Cytologic examination of Romanowsky-stained tissue imprints were moderately cellular with a pleomorphic population of round to plump to fusiform cells. Anisocytosis, anisokaryosis, and an increased nuclear to cytoplasmic ratio were observed. Individual cells had a round to oval nucleus with a granular chromatin pattern and occasional prominent nucleoli. The cytoplasm appeared basophilic and occasionally contained small pink granules. Cytoplasmic borders were indistinct. Few mitotic figures and inflammatory cells were seen. The sparse inflammatory cell population included nondegen-erative neutrophils, macrophages, lymphocytes, and rare giant cells that were scattered throughout the slide. Eosinophilic material resembling osteoid matrix was also observed. The cytologic interpretation was mesenchymal cell neoplasia with bony differentiation.
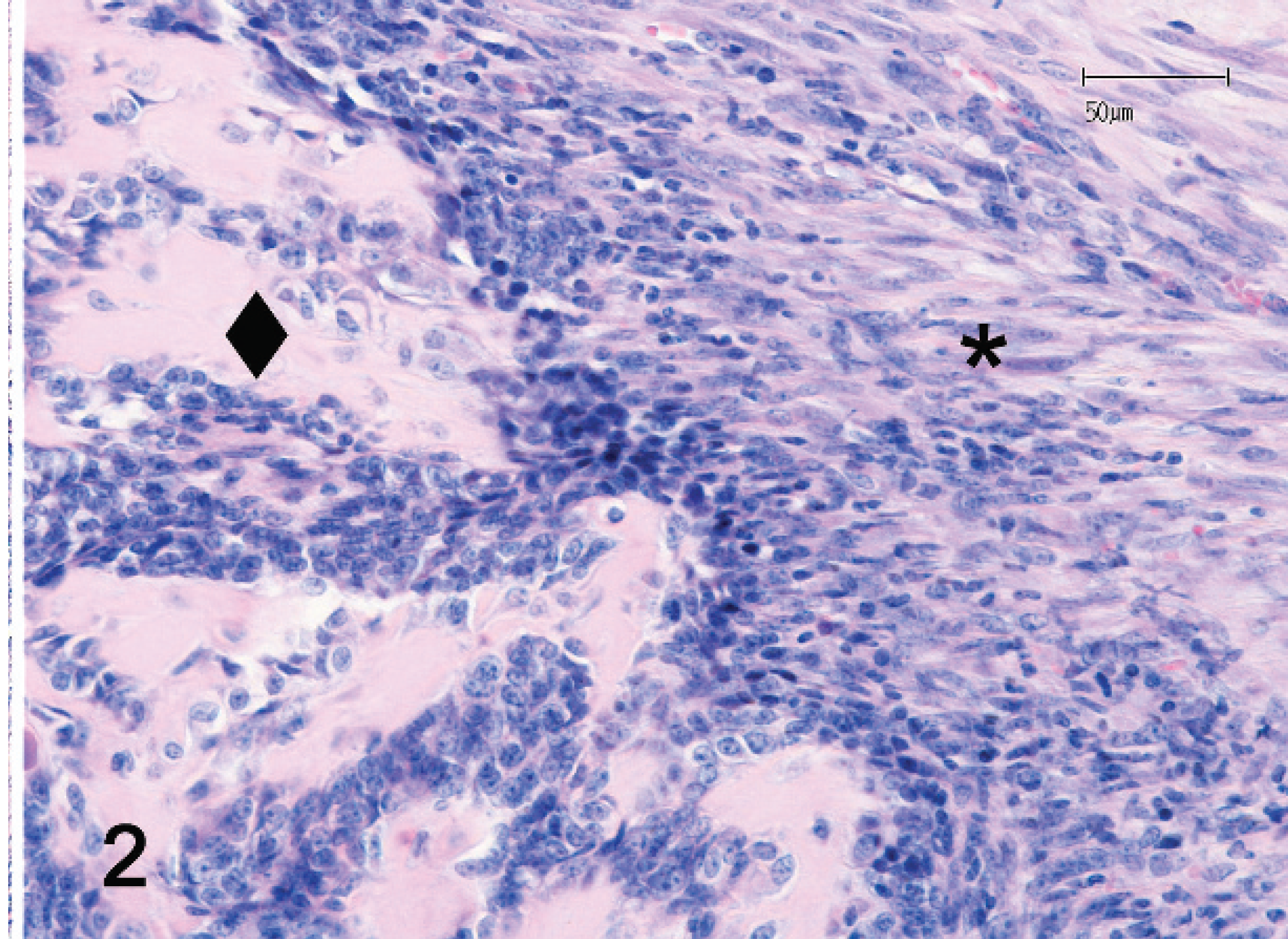
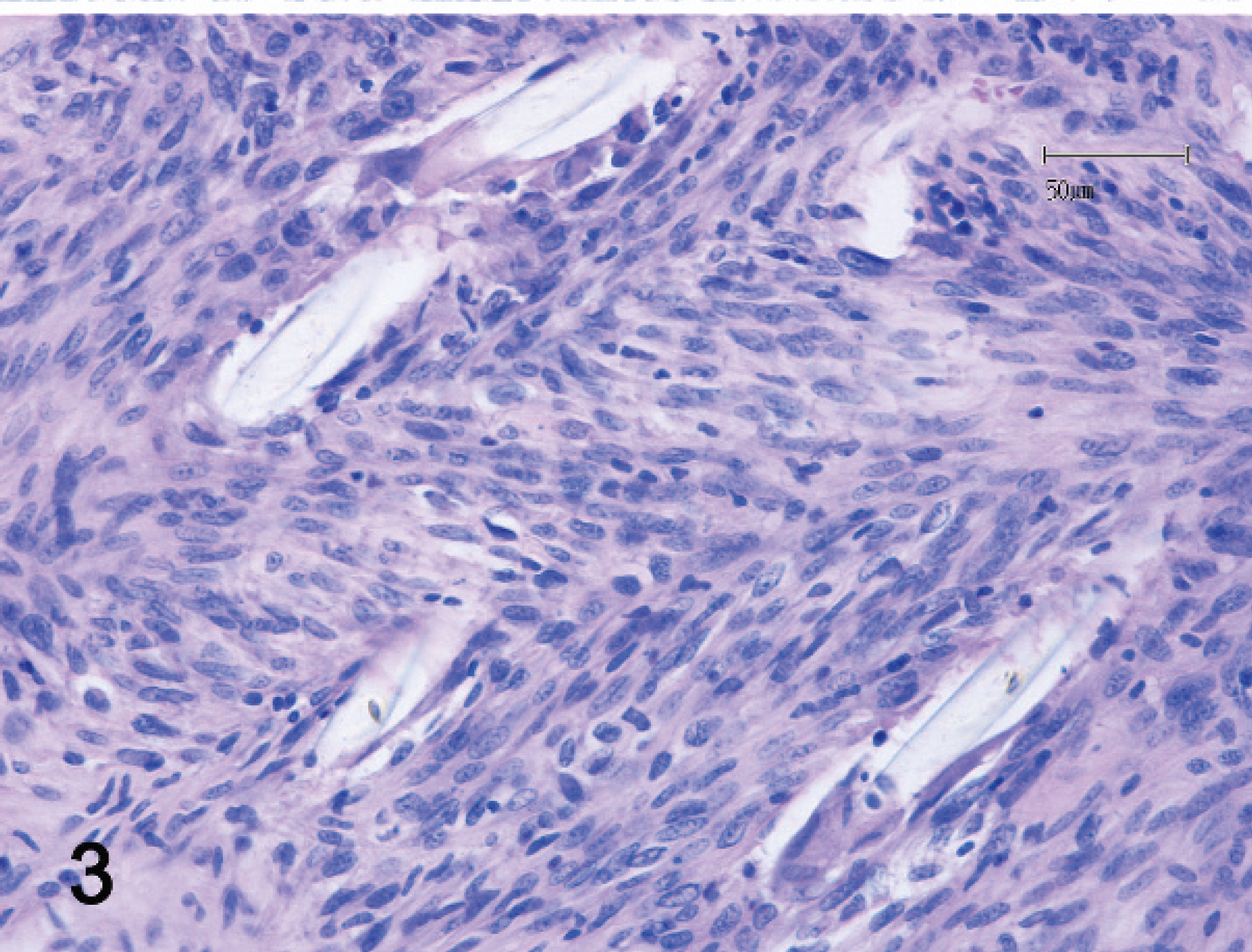
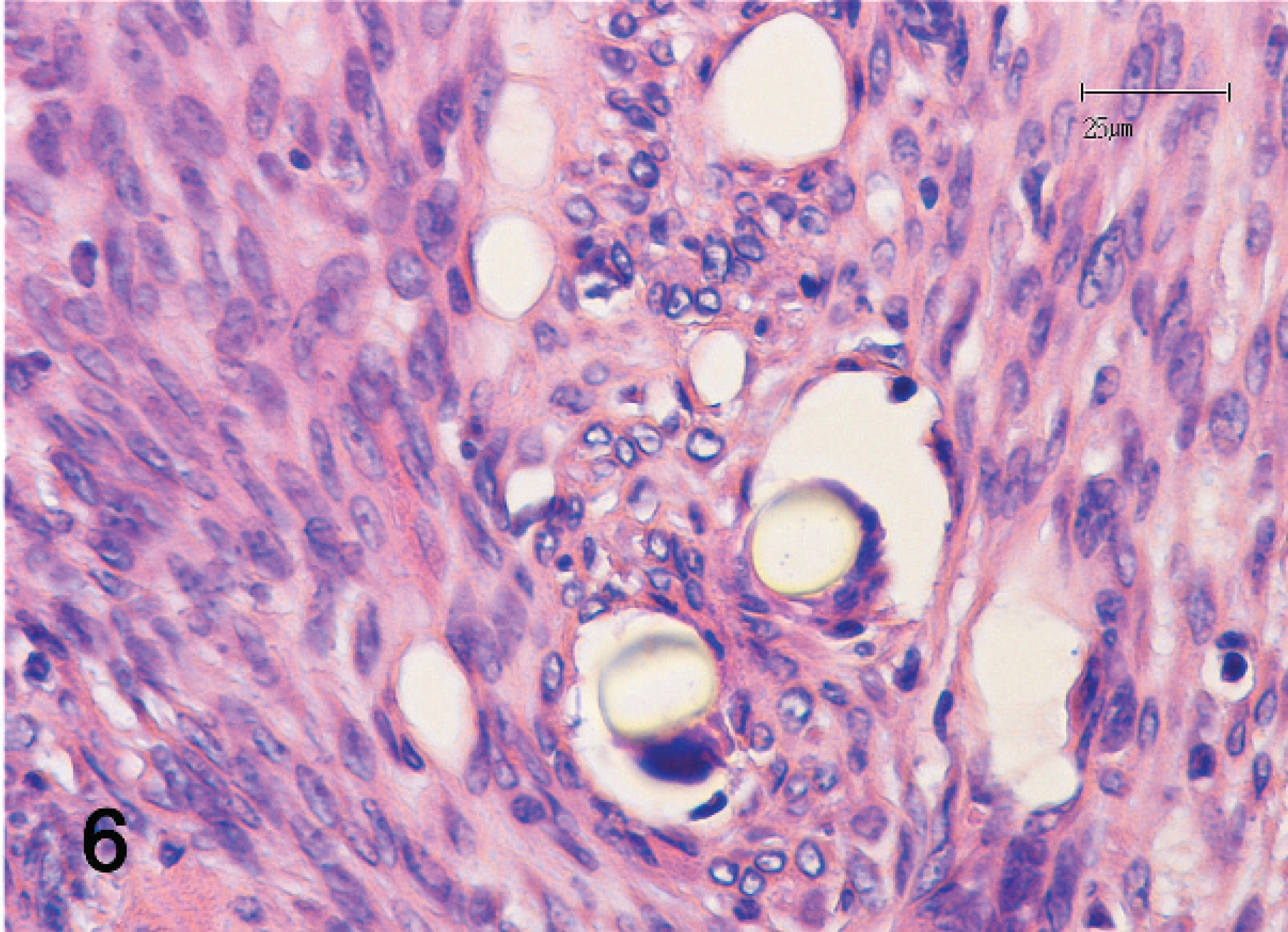
The mass was fixed in 10% neutral buffered formalin, routinely processed, embedded in paraffin, sectioned, stained with hematoxylin and eosin, and examined microscopically. The neoplasm was composed of a pleo-morphic population of mesenchymal cells with 2 distinct developmental components (Figs. 1, 2). The first component consisted of spindloid cells organized in irregularly oriented fascicles. Individual cells had a moderate amount of eosinophilic cytoplasm with indistinct cell borders (Fig. 3). Individual nuclei were oval to pleomorphic in shape, varied in size, and had vesiculated and irregularly arranged chromatin. Prominent nucleoli were occasionally seen. The mitotic rate ranged from 0 to 3 mitoses per high-power field of view (HPF, 400x). The second component consisted of round to spindloid cells containing round to pleomorphic nuclei with a granular chromatin pattern and prominent nucleoli. The cytoplasm appeared lightly baso-philic. The mitotic rate ranged from 2 to 6 mitoses per HPF (400x). Multifocal intercellular deposits of eosinophilic matrix, consistent with osteoid production (Fig. 2), were also observed. Scattered, multiple, round to polygonal to fusiform, unstained fragments of foreign material (non-absorbed sutures) had well-defined margins and exhibited birefringence under polarized light. These suture fragments were admixed with both sarcomatous components of the neoplasm (Figs. 3–6) and were often surrounded by 1 or 2 giant multinucleated cells (Fig. 6). Infiltrates of nondegenerative neutrophils, macrophages, a few giant cells, and rare lymphocytes were scattered throughout the tissue sections. In 1 tissue section, a normal-appearing tubular structure was observed that represented the terminal portion of an oviduct. Because of the dual cellular differentiation observed, further immunohisto-chemical investigations were done to elucidate the cellular origins.
Immunohistochemistry was performed g using primary antibodies for smooth muscle actin, a muscle-specific actin, b desmin, c myoglobin, d S100, e and vimentin f using an automatic immunostainer g as well as a commercial detection system. h Positive and negative control tissues were processed in parallel with sections of the neoplasm. Immunohistochemical examination demonstrated diffuse positive staining for vimentin in both cellular components (Figs. 7, 8). In contrast, no immunohistochemical staining was observed for smooth- or striated-muscle actin, desmin, myoglobin, or S100. The immunohistochemical staining results were indicative of a malignant mesenchymal neoplasm of osteosarcomatous and fibrosarcomatous origin with chronic inflammation and fragments of non-absorbed suture material. The definitive diagnosis of the neoplasm was malignant mesenchymoma.
The current study describes a malignant mesenchymoma in a female ferret that developed at the site of surgical suture placement near the right oviduct 60 months after ovariectomy. Malignant mesenchymoma is considered to be a mesenchymal neoplasm with at least 2 different cell lines of differentiation. 1,18 To the authors' knowledge, only 1 case of mesenchymoma in a ferret has been previously reported. 6 In dogs, malignant mesenchymoma has been reported in the abdominal cavity, 22 heart, 13 liver, 14 thigh, 17 and mandible. 18
The 2 major differential diagnoses for the neoplasm in the ferret of the current report were an extraskeletal compound osteosarcoma and a poorly differentiated soft tissue sarcoma with osseous metaplasia. The diagnostic criteria for extraskeletal osteosarcoma usually include the production of bone, presence of osteoid and possibly chondroid matrix, and a neoplasm that is located in soft tissue without any attachment to the skeleton. 28 A compound extraskeletal osteosarcoma should be differentiated from an undifferentiated sarcoma with osteoid metaplasia and chondrosarcoma. 28 Soft tissue sarcoma was excluded because the primary tumor had 2 distinct populations of neoplastic mesenchymal cells (fibrosarco-matous and osteosarcomatous cells). Immunohistochemi-cal staining for actin, desmin, and myoglobin excluded the presence of a muscle component. Fibroplasia with osseous metaplasia was an unlikely diagnosis because of the typical features of malignancy (e.g., irregularly oriented fascicles and bundles of fibroblasts, cellular and nuclear pleomorphism, and high mitotic index) within the mass.

Malignant mesenchymoma; ferret (
Malignant mesenchymoma; ferret (
Malignant mesenchymoma; ferret (

Malignant mesenchymoma; ferret (

Malignant mesenchymoma; ferret (
Malignant mesenchymoma; ferret (

Malignant mesenchymoma; ferret (

Malignant mesenchymoma; ferret (
Malignant mesenchymoma is rarely reported in humans and animals, 8,21 and its histogenesis is still uncertain. 25 It has been postulated that this tumor could arise from primitive and uncommitted mesenchymal cells that differentiate along multiple cell lines. 8,16
Various authors have postulated a relationship between trauma and scar formation in the development of mesenchymoma in humans 24 and dogs. 20 A radiation-induced malignant mesenchymoma has been considered in human medicine. 23 In the case described herein, chronic inflammation associated with foreign material could have played a role in tumor development. A previous case of neoplastic mesenchymal proliferation has been reported at the site of deep nonabsorbable suture placement in a cat. 4 In addition, malignant mesenchymal tumors have been described in dogs at sites of microchip implantation. 26,27 Chronic inflammation may depend not only on the nature of the material (e.g., absorbable vs. nonabsorbable, chemical composition, biocompatibility) but also on the morphology of the implanted material and the trauma induced by the needle. 2 A typical finding is the presence of giant cells that may be stimulated by the polymer and could induce the synthesis of tumor necrosis factor. 2 Giant cell activation may also depend on the surface morphology of the biomaterial involved. 10,11 Chronic reactions and persistent extracellular matrix remodeling occurring in tissues as a reaction against synthetic polymers may also depend on the role played by other factors, such as heat shock protein 70, 10 metalloproteinases and their inhibitors, 9 tenascin, and fibronectin. 7 However, a subjective and genetic predisposition also may play a role in the pathogenesis of these malignancies.
The occurrence of malignancy associated with retained surgical sponges in domestic animals and humans is rare. Retained surgical sponges could induce inflammation, leading to the formation of a sterile granuloma that may be asymptomatic for years.
16
These reactions are commonly called gossypiboma or textiloma, and their malignant transformation has been related to the microenvironment created by the fibrous capsule of the tumor and cotton fibers.
5,16
In domestic animals, neoplastic transformation of gossypiboma or textiloma is rare, and only 3 cases of mesenchymal tumors (osteosarcomas) have been re-ported.
3,16,19
The International Agency for Research on Cancer (IARC) reported that many polymeric materials have been tested for carcinogenicity in mice and rats.
15
Some of those materials (i.e., cellophane, ε-caprolactone–lactide copolymer, polyamide [nylon], polyethylene, poly-
Footnotes
a.
Actin (smooth muscle, clone 1A4), Dako North America Inc., Carpinteria, CA.
b.
Actin (muscle-specific, clone HHF35), Dako North America Inc., Carpinteria, CA.
c.
Desmin, Dako North America Inc., Carpinteria, CA.
d.
Myoglobin, Ventana Medical System SA, Illkirch, France.
e.
S100, Dako North America Inc., Carpinteria, CA.
f.
Vimentin (V9), Dako North America Inc., Carpinteria, CA.
g.
BONDTM immunostainer, A. Menarini Diagnostics srl, Flor ence, Italy.
h.
EnVisionTM+ Dual Link System-HRP (K4063), Dako North America Inc., Carpinteria, CA.
